Abstract
The objective of this study was to evaluate economic and health effects of sumatriptan relative to customary therapy in Canada. The relationship between treatment and functionality was established based on analysis of existing data from a multinational study. A Monte Carlo model was developed to simulate 1 year for each of customary therapy and six sumatriptan formulations. Costs are expressed in 1998 Canadian dollars. Sumatriptan is expected to reduce the time spent with migraine symptoms and resulting time lost. Under customary therapy, the annual cost of lost time is estimated at £908 ($1973). With sumatriptan, these costs ranged from £406 ($882) with subcutaneous sumatriptan to £577 ($1254) with nasal sumatriptan 10 mg, saving £331–502 ($719–1091) in the annual cost of time lost. All these benefits are expected to be obtained at an additional drug cost ranging from £869 ($1889) for subcutaneous sumatriptan to £278 ($605) for sumatriptan suppository. The cost of sumatriptan treatment is significantly offset by a substantial reduction of costs associated with time lost due to migraine symptoms.
Introduction
Migraine is a debilitating chronic disease that affects approximately 23 million Americans (1) and more than three million Canadians (2). Despite advances in migraine treatment, a cure has not been found. Instead, the aim of available therapies is to reduce the intensity of the symptoms and the consequent disability. These treatments differ not only in efficacy and side-effects, but also in terms of cost. Moreover, the difference in efficacy may have significant implications given that migraine imposes an economic burden on society because of the associated costs of medical care and lost productivity. In a population survey conducted in Canada, half of interviewed migraine sufferers discontinued normal activities because of their headaches, nearly one-third needed bed rest, and in three-quarters of the subjects with migraine there was at least a limiting degree of disability, which led to approximately 7 million lost working days per year (2). Others have found that migraine sufferers from a managed health-care plan in the USA generated nearly twice as many medical claims as a comparable group of members without migraine, and nearly 2.5 times as many pharmacy claims (3). Thus, it is important to evaluate both the economic and health consequences of each option in order to establish properly the optimal treatment strategy.
Sumatriptan, is a 5-HT1-agonist that acts by constricting cranial blood vessels and inhibiting the release of vasoactive neuropeptides from perivascular trigeminal axons in the dura mater. Sumatriptan is highly effective in aborting migraine attack and providing symptom relief, thereby increasing the individual's ability to continue with activities and reduce productivity losses. This was demonstrated in a double-blind, placebo-controlled study, where sumatriptan reduced migraine-associated productivity loss during a minimum 8-h work shift by approximately 50% (4). In addition, in two open-label studies sumatriptan treatment was associated with a 30–38% reduction in lost workplace productivity (5, 6). In a more recent study, healthcare resource use and time lost from workplace productivity and non-workplace activity were reduced after 6 months of sumatriptan therapy (7).
Although these studies determined the effectiveness of sumatriptan in reducing lost workplace productivity, no costs were assigned to these benefits. Furthermore, the assessments did not weigh the clinical benefits of sumatriptan against its acquisition cost. Several studies have also assessed medical resource use, but the costs of the resources involved were not considered (8). More detailed cost-effectiveness studies, incorporating both direct and indirect costs, have been conducted, but these have only compared sumatriptan with specific alternative migraine therapies (subcutaneous sumatriptan vs. subcutaneous DHE (9) and oral sumatriptan vs. oral cafergot/ergotamine (10)).
The purpose of this study was to evaluate the incremental economic and health benefits of sumatriptan relative to customary therapy, with emphasis placed on Canadian direct medical costs and indirect costs. The results are presented primarily as cost-effectiveness ratios, although cost benefit ratios are also provided.
Methods
Model structure
An economic model was created based on the concept that the impact of migraine symptoms on the patient may translate into absenteeism from work and decreased productivity, as well as an increase in the use of medical services and other health care resources.
The model compares each one of the sumatriptan formulations with customary therapy in a simulated population. Figure 1 illustrates the basic structure of the model. The model has been described in detail elsewhere (11). Briefly, it uses Monte Carlo techniques to simulate a typical year of life for any given number of migraineurs under different treatment strategies: customary therapy, subcutaneous sumatriptan, oral sumatriptan 100 mg, oral sumatriptan 50 mg, sumatriptan 10 mg nasal spray, sumatriptan 20 mg nasal spray and sumatriptan suppository 25 mg. A Monte Carlo simulation estimates outcomes of a treatment by simulating a population with a given set of characteristics which affect outcomes based on known or estimated frequency distributions. The economic model is successively recalculated for a series of hypothetical migraineurs chosen to correspond to their frequency distributions. In the principal analysis, a patient population of 10 000 was simulated.

Schematic overview of the migraine model.
The model begins by randomly assigning an age and gender to each simulated migraineur according to the demographic distribution of migraineurs that are being considered (12). Based on the age and gender of the migraineur, employment status and wage are assigned. For each patient, probability distributions for the occurrence of migraine are sampled in order to assign a daily risk of the occurrence of migraine symptoms. After the risk is assigned to the simulated patient, severity is allocated (mild, moderate, severe) on any day that symptoms are present, as is the duration of symptoms, each based on its own probability distribution. Similarly, time lost due to migraine symptoms is estimated by constructing probability distributions for the number of hours lost per migraine day. These distributions were calculated according to employment status, severity and treatment. The total cost of time lost due to migraine is then estimated as: Σpaid work time lost × value of paid work time + Σnon-paid work time lost × value of non-paid work time.
Non-paid work time lost is defined as all work activities outside of paid employment (i.e. meal preparation, cleaning, clothing care, repairs and maintenance, other domestic work, help and care, management and shopping, and transportation and travel).
Pharmacological treatment, doctors visits, emergency room visits and Hospitalizations are also calculated for each migraine day, according to the severity of symptoms.
No specific value was assigned to side-effects, as these were implicitly incorporated in other areas where outcomes were measured. It was assumed that side-effects would manifest themselves either as time lost, increased duration of symptoms and/or additional medical resource use.
Probability distributions
Demographic characteristics of the migraine population were derived from the Canadian Migraine Prevalence Survey (12), details of which have been published elsewhere (13), and the demographic profile of all Canadians.
Efficacy and resource use data were derived primarily from a prospective sequential multinational study (Q24), which used patient-recorded diaries to collect migraine-specific data during 12 weeks of customary therapy and a subsequent 24 weeks of migraine treatment with subcutaneous sumatriptan (6 mg) (14). Probability distributions for the occurrence of migraine were taken from the Q24 study. The mean annual number of days with migraine was estimated at 82.5 days for males and 77.4 for females, while on customary therapy. Migraine days with sumatriptan was estimated to be lower, at 61.0 for males and 55.3 for females. The distributions of severity of symptoms were also assigned based on Q24-derived probability distributions (Table 1), as were the effects of sumatriptan on the duration of symptoms, analysed according to gender and severity.
Severity of migraine days

A reduction in doctor visits while on sumatriptan was observed in the Q24 data and other published results (15–17). Consequently, the same rate of reduction was incorporated into this analysis. The probability of doctor visits was further adjusted according to the severity of migraine symptoms (moderate or severe). This adjustment was made based on the frequency of doctor visits for severe relative to moderate migraines, reported in the Angus Reid Migraine Diary Study, conducted on behalf of Glaxo Wellcome in 158 randomly selected migraine sufferers who provided data on a total of 3800 headaches (18). The probability of emergency room (ER) visits was calculated in the same manner as for physician visits, with the exception that the probability of a visit was calculated for severe migraine days only. Annual ER visits for Canadian migraineurs were based on results from the Canadian Migraine Prevalence Survey (12), while the reduced number of visits on sumatriptan were derived from published findings (15, 17, 19) and Q24 results. Hospitalization rates for all patients, regardless of treatment, were based on the relative rates of Hospitalizations and ER visits in the Canadian Migraine Prevalence Survey.
As mentioned, the main effectiveness data used in the model were taken from the Q24 study, which compared patients' customary therapy with subcutaneous sumatriptan treatment. Data from a suitable crossover study were not available for the other sumatriptan formulations. In order to estimate outcomes for the oral, nasal and suppository formulations therefore, a number of factors were assumed to be equal between the subcutaneous and other formulations, including medical services usage, the frequency of migraine days, the severity of migraine days and any adverse events with sumatriptan.
For the duration of symptoms and time lost, equivalence among formulations was considered unlikely given differences in relief rates between the various sumatriptan formulations. In order to estimate these parameters for other sumatriptan formulations, data on the efficacy of pain relief were taken from the Q24 study and other published and unpublished sumatriptan clinical trials (14, 20–26) and a detailed diary-based survey on general migraine treatments and their effects (18). Data from as many trials as possible were used in order to minimize the impact of potential measurement differences between the Q24 study and other sumatriptan trials. Based on these data, migraine symptoms were estimated to last 14.5% longer with oral sumatriptan 100 mg than with subcutaneous sumatriptan, 30% longer with oral sumatriptan 50 mg, 49% longer with nasal sumatriptan 10 mg, 31% longer with nasal sumatriptan 20 mg and 35% longer with sumatriptan suppository 25 mg. Similarly, time lost for other sumatriptan formulations was estimated as a direct function of subcutaneous sumatriptan estimates, using the same proportional differences as estimated for duration of symptoms.
Costs
The costs of physician visits, ER visits and Hospitalizations were assigned according to the fees listed in the Canadian physician fee schedules, the Ontario Case Costing Project (27) and the Alberta Case Cost Study (28), respectively. Table 2 presents the probabilities and the costs associated with each medical visit.
Medical resource utilization: probabilities and related costs
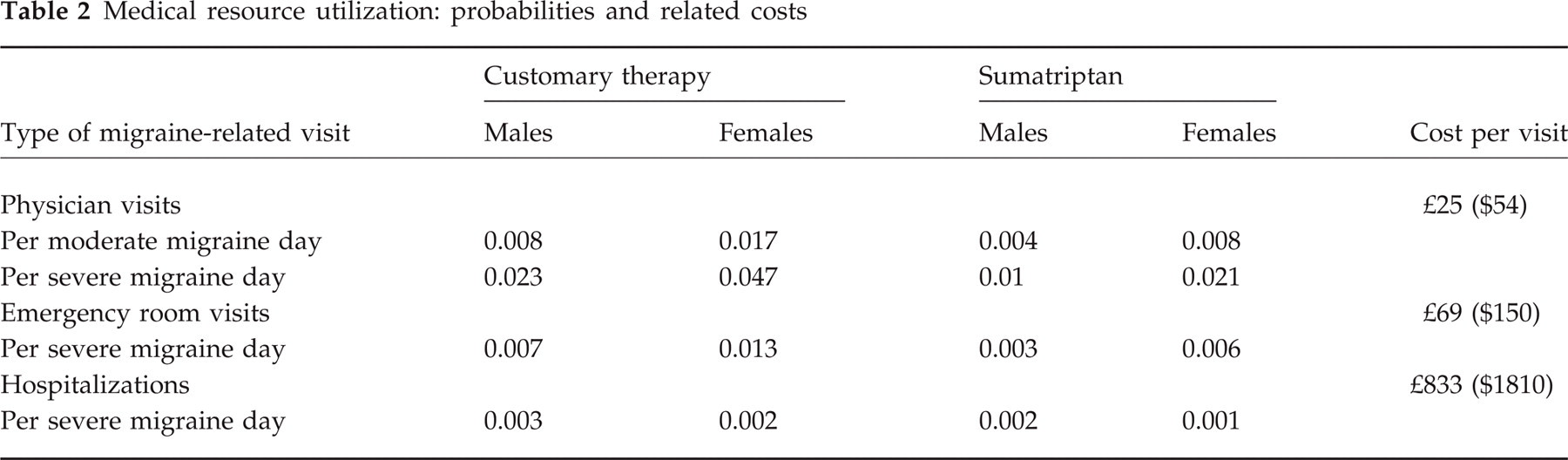
Drug costs were calculated using the average price listed in the provincial drug formularies. Since nasal sumatriptan 10 mg and suppository sumatriptan 25 mg are not available in Canada, proxy costs were used. The cost of nasal 10 mg was taken as the average of the nasal 5 mg and 20 mg formulations, both of which are available in Canada. The cost of sumatriptan suppository was estimated by determining the relative cost of the suppository formulation compared with oral sumatriptan 50 mg in eight European countries (the suppository formulation had an average price 5% lower than that of oral sumatriptan 50 mg), and then adjusting the Canadian cost of the 50 mg oral formulation. The unit cost of customary therapy depended on the drug mix used for mild, moderate, or severe migraine days, and was calculated as a weighted average of these drugs. Medication costs for the treatment of migraine days, by the initial severity of symptoms are presented in Table 3.
Cost of pharmaceutical treatment ∗

Sumatriptan costs per severity of migraine day include the costs of non-sumatriptan medications.
Analyses
Three types of analyses were conducted. The simplest involved calculating the net costs according to various definitions of ‘cost’ ranging from medical care only, through all aspects of time lost. Thus, the perspective varied from that of the health care system to societal. Where the economic effect of using sumatriptan was a net expense, a second set of analyses involved relating the net cost to the change in duration of migraine symptoms—a cost-effectiveness calculation. If sumatriptan use resulted in a net saving, then cost effectiveness ratios were not derived. Regardless of the direction of the net costs, a third set of analysis involved derivation of the cost–benefit ratio relating the increase in drug costs to the decrease in the costs of time lost from activities and other medical costs.
The results of each set of analyses were subjected to extensive sensitivity analyses.
All of the analyses were carried out for a time horizon of 1 year and thus no discounting of costs or of benefits was undertaken. The results are reported in 1998 Canadian dollars. No adjustments for charges were made as the estimates are taken to reflect true unit costs. Finally, the model was run under the assumption that only days with moderate to severe migraine would be treated with sumatriptan. All days with mild migraine would be treated with patients' customary therapies.
Results
Under customary therapy, patients are estimated to spend an average of 78 days (632 h) per year with migraine symptoms. The resulting loss of time for these patients is estimated to be over 243 h, 84 of which are lost from paid work activities, and a further 48 of which are lost from unpaid work activities.
Sumatriptan is expected to reduce substantially the time spent with migraine symptoms and resulting time lost. The drop in the duration of symptoms ranged from 315 h with subcutaneous sumatriptan 6 mg to 188 h with nasal sumatriptan 10 mg (Fig. 2).

Annual duration of symptoms under each treatment strategy.
Under customary therapy, the annual cost of lost time is estimated at £908 ($1973). With sumatriptan, these costs ranged from £406 ($882) with subcutaneous sumatriptan to £577 ($1254) with nasal sumatriptan 10 mg, saving £331–502 ($719–1091) in the annual cost of time lost.
All these benefits are expected to be obtained at an additional drug cost ranging from £869 ($1889) for subcutaneous sumatriptan to £278 ($605) for sumatriptan suppository (Table 4).
Average cost and time lost due to migraine under each treatment strategy

Cost effectiveness
Cost effectiveness calculations from the societal perspective were carried out only for subcutaneous sumatriptan, the formulation that led to a net expense. For this formulation, the cost per reduced hour of symptoms was 98p ($2.13). If only health care costs are considered all formulations lead to incremental costs with the cost per reduced hour of symptoms at £2.57 ($5.59) for subcutaneous sumatriptan and between 94p ($2.04) and £1.18 ($2.56) for the other formulations.
Cost benefit
In this model, the cost–benefit ratio indicates the increase in pharmaceutical treatment costs associated with sumatriptan therapy in relation to the corresponding reduction in all other costs (medical visits and time lost). For five of the six formulations less than 46p ($1) in incremental drug costs (29p to 33p ($0.63 to $0.72)) would need to be spent to achieve a 46p ($1) saving in all other migraine-related costs. For subcutaneous sumatriptan, an additional 71p ($1.55) would need to be spent to achieve the same savings (46p ($1)).
Sensitivity analyses
Key inputs, including the efficacy of sumatriptan to reduce time lost due to migraine symptoms, frequency of migraine days, acquisition price of sumatriptan, non-prescription medical costs, costs of time lost, proportion of wages lost, value of unpaid work and value of leisure time, were varied over broad ranges for each one of the sumatriptan formulations.
Figure 3, which presents net costs varying the amount of time lost due to migraine with sumatriptan therapy, indicates that with subcutaneous sumatriptan 6 mg, net total costs remain positive even when time lost is assumed to be 50% lower than the base model estimate. At a 50% decrease in time lost from the baseline estimate, net annual costs would still be almost £138 ($300). On the other hand, the other formulations achieve net savings even when time lost is assumed to be 50% higher than the base time lost estimate for subcutaneous sumatriptan. The range of cost savings at this increased level of time lost spans from £71 ($154) in savings for oral sumatriptan 100 mg to £113 ($245) for sumatriptan suppository 25 mg.

Net total costs varying time lost with sumatriptan. Note that the abscissa refers to decrease in time lost relative to the base estimate for subcutaneous sumatriptan. In this figure, effectiveness is constant for all formulations at each point on the abscissa.
The hourly value of unpaid work was estimated at £5.28 ($11.48)/h. Figure 4 presents net cost outcomes under different valuations of lost unpaid work time. As the chart indicates, with the exception of subcutaneous sumatriptan, sumatriptan would maintain cost savings even if unpaid work were to have no value. On the other hand, the value of unpaid work would have to approach £17 ($36)/h for subcutaneous sumatriptan to reach a cost neutral position.
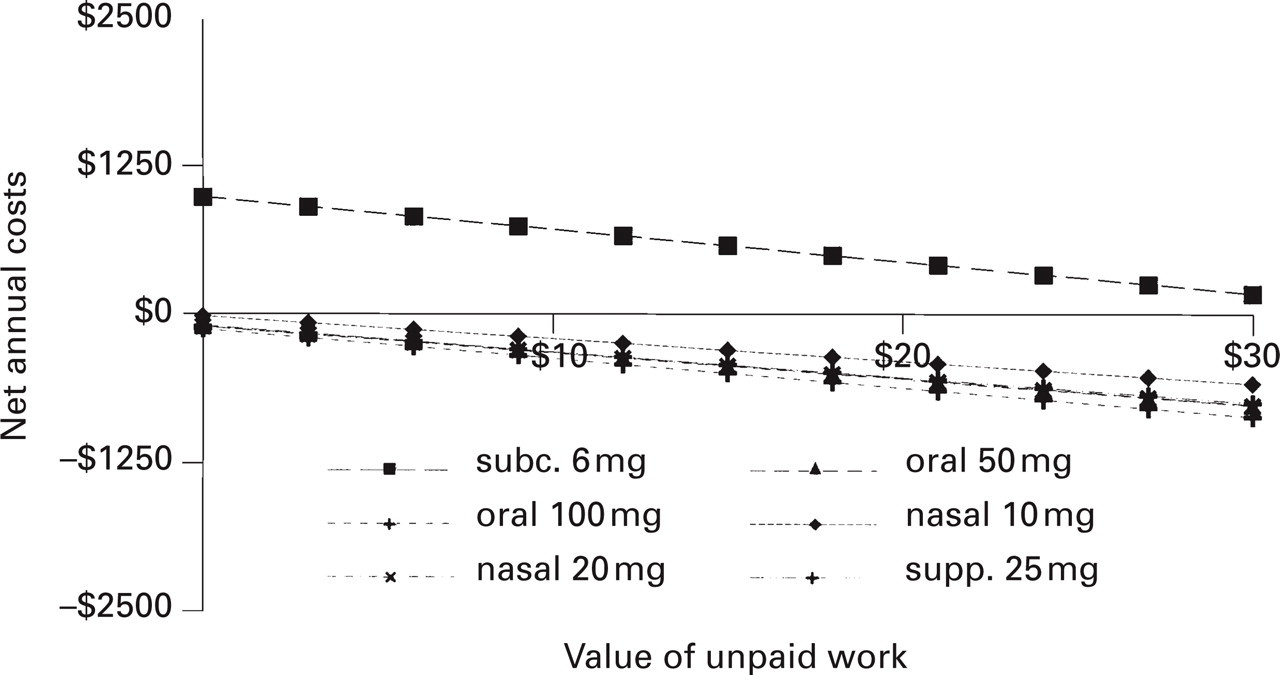
Net total costs varying the value of unpaid work.
Leisure time was valued at $0 in the main analysis. Attaching an hourly value of £2.76 ($6) to leisure time, annual net costs would drop to £134 ($292) for subcutaneous sumatriptan and savings would increase to between £229 ($497) and £347 ($754) for other sumatriptan formulations. Leisure time would have to be valued at nearly £5 ($11) for subcutaneous sumatriptan to achieve cost neutrality. At this valuation of leisure time, other sumatriptan formulations would result in cost savings of £321–469 ($698–1019) annually.
Discussion
Based on the results of this economic analysis, sumatriptan is expected to reduce substantially the impact of migraine episodes on patients' work and non-work activities. These benefits translate into significant savings from costs associated with lost productivity. Under customary therapy the annual cost of lost time was estimated at nearly £920 ($2000). With sumatriptan, these costs dropped by more than £460 ($1000) with subcutaneous sumatriptan, while the other sumatriptan formulations saved between £331 and £450 ($719 and $978).
The model assumes that migraineurs treated all moderate and severe migraine days with sumatriptan. If mild migraine days were also treated with sumatriptan, results may have been less favourable for sumatriptan, since the scope for potential reductions in time lost may be smaller on these days. If, however, more selective treatment of migraines occurs (e.g. treatment of more severe migraines with subcutaneous sumatriptan), the potential for reducing time lost per treated day relative to treatment cost could increase, thereby leading to greater savings.
The appropriate method for estimating the costs associated with lost time in an economic evaluation poses a dilemma. It has been indicated that a patient's time should be given a monetary value regardless of how it is used (29). In this study, however, we took a conservative (in the sense that it does not favour sumatriptan) approach by not giving a monetary value to leisure time. Although it is difficult to evaluate the economic impact of lost leisure time, qualitatively one may consider the following potential implications within the spectrum of quality of life: higher stress, which could lead to less productivity at work and further expenses in relaxation therapies, psychotherapy, massages, etc., and cancellation of social and/or family events. In addition, the reduced time the patient has to be able to function at a normal level, both at work and socially, may have major emotional implications.
Finally, it is worth noting that while this model was structured using Monte Carlo techniques, the probability distributions constructed for the analysis were based on documented data (30).
Although the higher costs of new medications are sometimes perceived as an unjustified additional burden, the results of this economic study provide evidence that use of sumatriptan in the treatment of migraine is economically sound.
Footnotes
Acknowledgements
Data from the Migraine Prevalence Survey were provided by the Institute for Social Research, York University. The survey was completed for Dr Bernie O'Brien and Mr Ron Goeree of the Centre for Health Economics and Policy Analysis at McMaster University. Neither the Institute for Social Research nor the Centre for Health Economics and Policy Analysis are responsible for the analysis and interpretations here. Financial support for this study was provided by Glaxo Wellcome.
