Abstract
The objective of this study was to investigate the age-dependence of the prevalence and characteristics of migraine headache and migraine visual aura. A neurologist interviewed 728 women attending a mammography screening programme. International Headache Society (IHS) criteria were used. The lifetime prevalence of migraine headache was 31.5% and the 1-year prevalence 18.0%. The magnitude of the decline of the prevalence of active (one or more attacks in the previous year) migraine headache was estimated to 50% per decade. The prevalence of active migraine visual aura was 3.8%. This did not vary by age. Except for the pain intensity and the presence of nausea, other characteristics and concomitant symptoms did not change with age. Active migraine headache and migraine visual aura in middle-aged and older women are common and modified differently by age. We suggest that the decline of prevalence of active migraine headache with age is caused by a decrease in pain intensity.
Introduction
Active migraine affects 4–8% of men and 11–25% of women (1–5). The incidence is large in children and adolescence. Bille reported in his 40-year follow-up study that 29% of adults who experienced migraine attacks in their childhood continued to have an attack at least once a year and that another 22% still had attacks but with some migraine-free periods (6). Although there is substantial evidence from cross-sectional studies (7) that the prevalence of migraine headache peaks around the age of 40 years and declines thereafter, only little systematic information is available on how this happens and even less information on why (8). Case histories tell us that headache following migraine aura may disappear with advancing age, leaving only the aura as a late-life migraine accompaniment. It is not known if this observation translates into an observable decrease in pain intensity of headache following migraine aura in the general population.
In a previous study we found that the prevalence of migraine headache in middle-aged and older women attending a mammography screening programme (MSP) declined with age (unpublished data). The main aim of this cross-sectional study was to evaluate how the prevalence and characteristics of migraine headache and migraine visual aura change with age in middle-aged and elderly women from the general population. We speculated that the age-dependent efficacy or use of hormonal replacement therapy (HRT) and drugs with potential anti-migraine prophylactic properties, i.e. beta-blockers, aspirin, and non-steroidal anti-inflammatory drugs (NSAIDs), might contribute to changes in the prevalence of active migraine headache with age. A further aim was to test hypotheses regarding the impact of these drugs and the educational level on active migraine headache and the association between migraine headache and age.
Study population and methods
The mammography screening programme
Östhammar is located in Uppsala County in central Sweden. It has 22 120 residents. All women aged 40–74 years in Östhammar are invited regularly to a MSP: women aged 40–54 years every 18 months and women aged 55–74 years every 24 months. Women living in Östhammar are invited to visit the local mammography unit (MUT). After contact with the staff of the MSP they are free to attend the central MUT in Uppsala. The MSP is characterized by a high attendance rate; during the first 6 months of 1998 79.1% of all invited women (n = 1337) attended the local MUT, 7.3% attended the central MUT, and 13.6% did not respond to the invitation. In this study, attenders of the MSP are labelled as attenders. Those who participated in our study are labelled as participants.
Study population
The migraine study was conducted at the local MUT on 32 working days from November 1997 to October 1998. We asked women who attended the local MUT to participate in our study. The staff of the MSP sent an up-dated invitation list to the local MUT every working day. The invitation list did not contain the names of the women who chose to attend the central MUT. The names of 996 women were on the invitation lists (Fig. 1). Of the 996 women, 158 did not attend the MSP. Of the remaining 838 women, 686 women agreed to participate. In November 1998, we mailed a short questionnaire to women who visited the local MUT, but did not participate in the study (n = 152). Forty-two women returned the questionnaire. They were interviewed by telephone and were subsequently included in the study. Thus, 728 women (86.9% of 838) participated in the study.
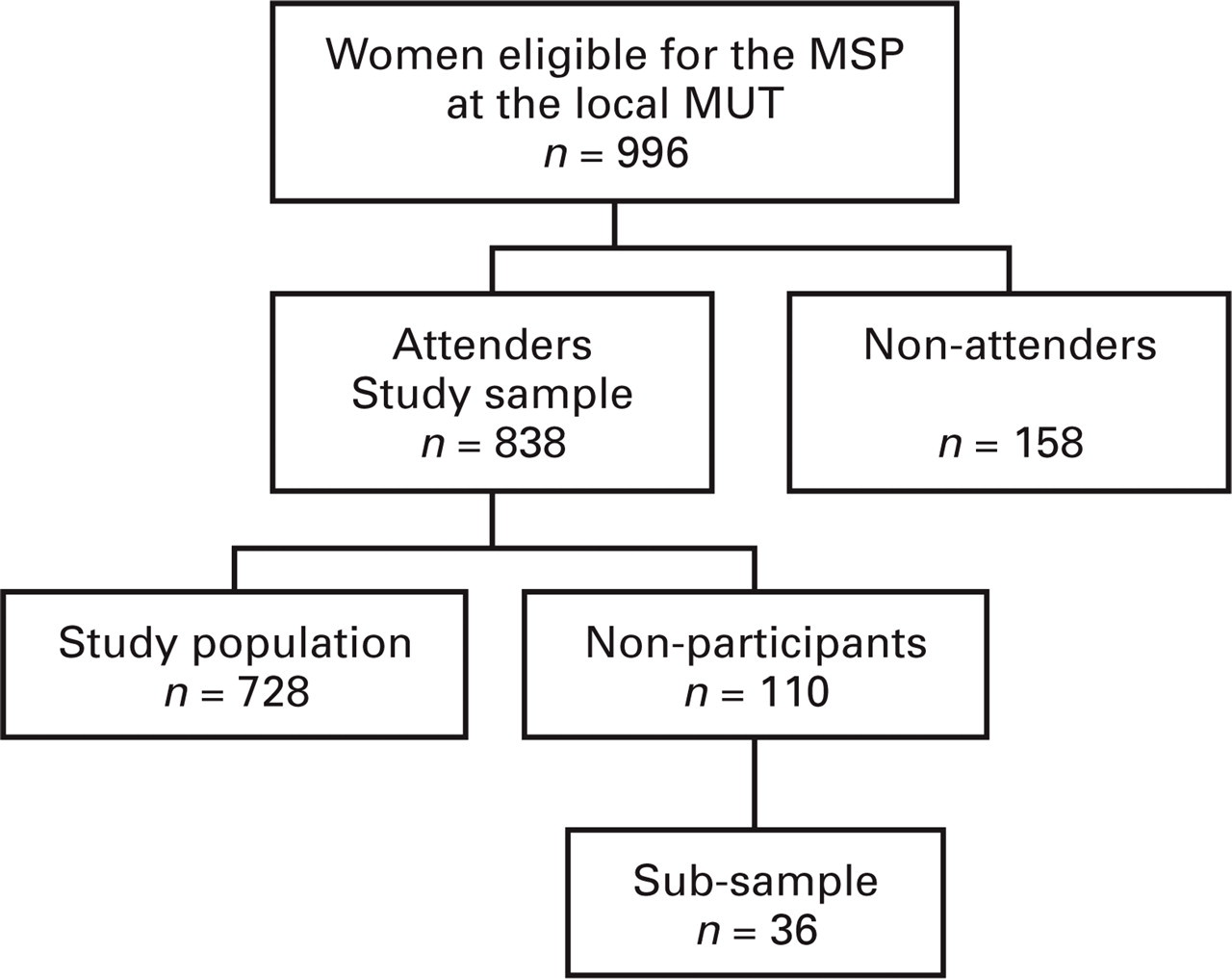
Study design.
Interview, measures, and definitions
Participants filled out a questionnaire and were interviewed face-to-face by a board-certified neurologist (P.M.). Physical and neurological examinations were not performed. The questionnaire included the question ‘Do you have or have you ever had migraine?’ (self-reported migraine). The interview was completed over the telephone if the time for interview was insufficient (n = 92).
The first and main part of the interview focused on the 1-year prevalence of migraine headache and on migraine visual aura. The criteria for migraine headache corresponded to migraine without aura in the International Headache Society (IHS) classification (9) with two exceptions. First, the disability criterion was mandatory (at least inhibition or prohibition of daily activities during headache). Second, participants fulfilled the criteria irrespective of whether they experienced a transient visual disturbance in conjunction with the onset of a migraine headache attack or not.
Information on migraine visual aura was obtained independently of the presence or absence of migraine headache. The definition of migraine visual aura was equivalent to the definition of migraine with aura (MA) in the IHS classification. Since a typical headache phase is a prerequisite for migraine with acute onset aura (code 1.2.6 in the IHS classification), this form of migraine aura was excluded. Only visual auras with scintillations or zigzag lines were included, because purely negative scotoma may be difficult to recognize as migraine phenomena.
Participants were considered to have active disease if they fulfilled all criteria in at least one attack of migraine headache in the previous year. Measures of untreated or unsuccessfully treated attacks were used in the analyses with the exception of frequency, which was based on all attacks including successfully treated attacks. Frequency was measured in 10 predetermined categories as the number of days in the previous year during which the women had headache or visual aura. Duration of headache attacks was measured in 10 predetermined categories as the number of hours during which an attack lasted. Duration of visual aura was measured in minutes.
In the IHS classification, moderately intense headache corresponds to inhibition of daily activities and headache of severe intensity to prohibition of daily activities. In this study the term disability was used to describe this variable to distinguish from pain intensity, which was measured on a verbal analogue scale ranging from ‘No migraine pain’ (= 0) to ‘Migraine pain as intense as you can imagine’ (= 10).
Data on education (five levels: elementary, comprehensive, secondary, upper secondary school, or university), HRT (yes vs. no) (ATC codes G03C, G03D, and G03F), beta-blockers (yes vs. no) (C07A A05, C07A B02, and C07A B03), and drugs used for the treatment of migraine headache attacks (no treatment, analgesics (N02A and M01A), ergots (N02CA), or triptans (N02C C) were obtained in the interview. Women who attended the MUT and participated there (n = 686) were asked to fill in a questionnaire at home. In an open question women were asked to report regular use of drugs and the number of tablets, suppositories, etc. Six hundred and fifty-nine women returned it. NSAIDs included aspirin (≥ 500 mg per day, but not low-dose aspirin) (N02B A01 and N02B A51) and other NSAIDs (M01A) used for the treatment of pain. Data on NSAIDs were extracted from the questionnaire.
All women gave their informed consent for participation, and the Ethics Committee of the Medical Faculty, University of Uppsala, approved the study.
Non-participants, non-attenders, and attenders of the central MUT
In March 1999, 36 of the remaining 110 non-participants were randomly sampled for a telephone interview. Twenty-five women agreed to be interviewed. Non-attenders were not contacted for ethical reasons, but there was information on the previous attendance status of 674 participants in the invitation lists at the local MUT. The prevalence of self-reported migraine was estimated in women who attended the central MUT. The names of the women who attended the central MUT were identified through the same sample that was used to characterize the MSP (n = 98, see first paragraph of this section). We mailed a questionnaire to all 98 women. Eighty-six (87.7%) of them returned it and gave information on their own experience of migraine.
Statistical analysis
The statistical program package JMP (version 3.2.2) was used. The choice of a particular method was based mainly on the type of data to be analysed. Differences in proportions were analysed by the likelihood ratio (L-R) χ2 method. Discrete variables were regressed on age using nominal logistic regression. Confidence intervals (CIs) in logistic regression were calculated via maximum likelihood estimates. Kendall's non-parametric method was used to test for correlation. Linear regression was used to analyse the relation between age and non-categorized continuous variables. Ordinal logistic regression was used to test the effect of age on pain intensity when additional variables were taken into account. P<0.05 was considered significant. Ninety-five percent confidence intervals were computed using parametric methods.
A monotonous decline of active migraine was found from the late 40s onward (Table 1 and Fig. 2). Prevalence ratios were regressed on the median age in each 5-year age class (46, 52, 57, 63, 67, and 72 years). The straight-line regression was significant (R2 = 0.92, DF (1,4), F = 43.3, P = 0.003). Analysis of residuals and significant squared and cubic (censored) age terms pointed to a curvilinear relationship. To account for possible heteroscedasticity, we used an approximate solution with empirical weights (10). The weights were calculated as prevalence∗number/(1−prevalence). After log-transformation and the introduction of weights, R2 increased to 0.99 (DF (1,4), F = 323.8, P < 0.0001). The estimate of the slope can be translated into and interpreted as half-life (T1/2=natural logarithm of 0.5 divided by the estimate).

Model-based and observed 1-year prevalence of active migraine headache in women aged 40–74 years. Note: the squares represent the observed prevalence rates and the line represents the exponential model based on the monotonous decline of prevalence in women aged 45–74 years.
Age-specific distribution of non-participants and participants with and without migraine headache

Active migraine headache=at least one attack of migraine headache in the last year. Inactive migraine headache=migraine headache, but no attack in the previous year.
Headache characteristics and concomitant symptoms were not independent because they were part of the headache diagnosis. These and the prevalence of migraine visual aura were separately analysed in three age classes (40–49, 50–59, and 60–74 years) using the χ2 test. If there was evidence of a monotonous change in proportions, logistic regression analysis was applied.
Results
Prevalence of migraine headache
The lifetime prevalence of migraine headache (Table 1) was 31.5% (CI 28.0–34.8) and the 1-year prevalence was 18.0% (CI 15.2–20.8). The half-life of the prevalence of active migraine headache (Fig. 2) in women aged 45–74 years was 9.3 years (CI 8.0–10.9). In a logistic model including education, NSAIDs, HRT, and age (continuous) as independent variables and active migraine as the dependent variable, only age was significantly associated with active migraine. Two interaction terms were considered: one between age and HRT and another between age and NSAIDs. None of them was significant. This analysis was based on data from 659 participants (69 missing values). HRT was used by 19 of 266 (7.1%) women aged 40–49 years, 74 of 196 (37.8%) women aged 50–59 years, and 41 of 197 (20.8%) of women aged 60–74 years. NSAIDs were used on a daily basis by six of 266 (2.3%) women aged 40–49 years, seven of 196 (3.6%) women aged 50–59 years, and 12 of 197 (6.1%) of women aged 60–74 years. The half-life for active migraine as estimated from logistic regression changed only marginally.
In the analysis of the association between use of beta-blockers and migraine confounding by indication was anticipated. The analysis was therefore restricted to women who were on treatment with beta-blockers because of hypertension. Despite this manoeuvre beta-blockers turned out to be a strong risk factor (odds ratio (OR) = 4.3, 95% CI 1.0–21.4) for migraine headache in women aged 40–49 years, probably because they are the preferred anti-hypertensive treatment in middle-aged migraineurs. As a consequence of this, beta-blockers were not included in the final analysis.
Relation of headache characteristics and coexisting symptoms to age
Neither frequency nor duration of migraine headache attacks was associated with age. Intensity declined with age (n = 131, Kendall's τ = 0.26, P < 0.0001), but disability did not (Fig. 3). The decline in intensity was significant both in women aged 40–49 years (n = 83, Kendall's τ = 0.24, P = 0.005) and in women aged 50–74 years (n = 48, Kendall's τ = 0.34, P = 0.002). Among unilaterality, pulsating character, aggravation by daily activities, phono- and photophobia, nausea, and vomiting, only nausea was associated with age (logistic regression, 1 DF, L-R χ2=7.6, P = 0.006). Taking HRT and NSAIDs into account, the effect of age on pain intensity was still significant (1 DF, Wald χ2=11.6, P = 0.0007). After taking into account the treatment of migraine attacks, the effect of age on pain intensity remained significant (1 DF, Wald χ2=14.7, P = 0.0001).
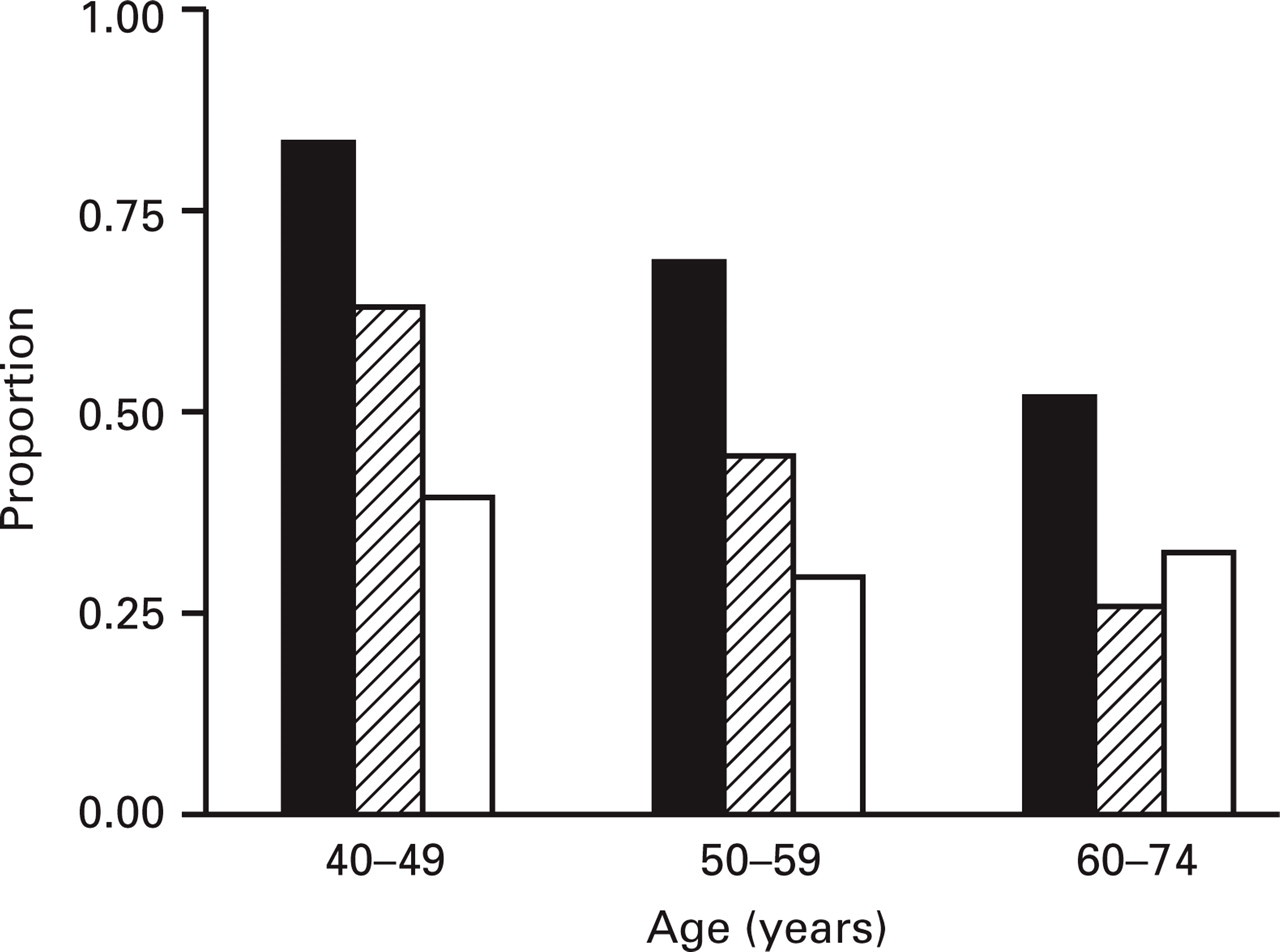
Proportion of women in each age category who experienced nausea (▪), high pain intensity (hatched), and severe disability (□) during migraine attacks. All women (n = 131) had active migraine headache. High intensity of pain was defined as a pain score equal to or exceeding the median. Disability could be rated as either moderate or severe. Age was significantly associated with nausea and intensity, but not with disability.
Migraine visual aura
Prevalence (Table 2) and duration or frequency of migraine visual aura were not related to age. Pain intensity of headache following a migraine visual disturbance (Table 2) declined significantly with age (n = 28, Kendall's τ = 0.33, P = 0.02). Six of 28 (21%) women did not experience any headache in conjunction with a migraine visual aura. Ten (36%) women experienced migraine headache, five (18%) migrainous headache (one criterion for migraine headache was missing), and seven (25%) other headache (at least two criteria were missing). The median pain intensity rating of migraine headache was 7, of migrainous headache 7, and of other headache 4.
Age-specific 1-year prevalence of migraine visual aura and the age-specific intensity of headache following migraine visual aura
Headache intensity was measured on a verbal analogue scale ranging from ‘No headache’ (= 0) to ‘Headache as intense as you can imagine’ (= 10).
Non-participants, non-attenders, and attenders of the central MUT
Two of 25 non-participants (8%) had a lifetime history of migraine headache. This was statistically different from the prevalence in participants (1 DF, χ2=7.3, P = 0.007, adjusted for age). Based on the invitation lists at the local MUT both attendance rate of the MSP (77.1%) and participation rate in the study (70.2%) were significantly lower among women aged 70–74 years than for all other age groups. In this regard, no other differences were detected (data not shown). There was no difference between the age distribution of the study population and all women aged 40–74 years living in Östhammar (data not shown). There was no difference in proportions of self-reported migraine between women attending the local (258/726 = 35.5%) and central (32/86 = 37.7%) MUT. Based on the interviews at the local MUT the positive predictive value of self-reported migraine for migraine headache was calculated as 184/258 = 71.3% and the negative predictive value was 423/468 = 90.4% (two missing values). Forty-eight (7%) of 674 women did not attend the MSP 2 years earlier. Controlling for age, the present migraine status did not differ between women who attended the MSP 2 years earlier and women who did not.
Discussion
Prevalence rates, methodology, and validity
We found that 31.5% of the women aged 40–74 years experienced migraine headache during their lifetime and 18% experienced it in the previous year. To eliminate inter-rater variation, the same neurologist interviewed all participants. The study population was large (n = 728). Both the attendance rate of the MSP and the participation rate were high, approximately 87%. We did not perform physical or neurological examinations, which are required by the IHS classification, but probably of limited value in epidemiological research (11). The results of this study are limited to the concept of migraine based on the IHS classification and to women aged 40–74 years living in a small region of Sweden.
There was no evidence that prior attendance status of the MSP or choice of MUT would significantly influence the lifetime prevalence of migraine headache. The invitation letter to the MSP did not contain information about the migraine study. The study sample was representative of the corresponding age and sex segment with regard to age. Two of 25 non-participants had migraine headache, which probably reflected an interest-related bias. If we assume a prevalence rate of 8% in non-participants, the lifetime prevalence of migraine headache would be 28.4%. We may assume the lifetime prevalence of migraine headache in non-attenders of the MSP to be not less than 2/3 and not more than 4/3 of 28.4%. This means that we would expect the lifetime prevalence to be between 27.1% ((182∗0.667∗0.284 + 1155∗0.284)/1337) and 29.7% ((182∗1.333∗0.284 + 1155∗0.284)/1337) in women aged 40–74 years living in Östhammar.
Comparison with previous studies
The lifetime prevalence of migraine headache (31.5%) in this study is comparable to prevalence estimates of lifetime migraine obtained in Danish women aged 45–65 years of 27% (2), in Canadian women > 35 years of 31% (4), and in Dutch women aged 20–65 years of 33% (5). The high lifetime prevalence of migraine in women aged 40–44 years in this study may be explained by interest-related selection bias and random sampling error. The prevalence of active migraine headache was 18% in this study, similar to the prevalence rate of 18% in women aged 40–69 years in the USA (1, 7). However, 1-year prevalence estimates vary considerably between studies. The 1-year prevalence of migraine was 14% in Danish women aged 45–65 years and 25% in Dutch women aged 20–65 years. These differences may relate to differences in interpretation and modification of IHS criteria and in sampling techniques. In conclusion, the prevalence estimates of migraine headache in this study are within the upper range of other comparable studies.
Prevalence (about 4%) and duration or frequency of migraine visual aura experienced in the last year did not change with age. We did not include descriptions of pure negative scotoma or acute onset aura or any other neurological symptoms. This circumstance may imply that we underestimated the prevalence of migraine aura. However, Rasmussen et al. (2), using the IHS classification, reported that the 1-year prevalence of migraine with aura was 5% in women, i.e. a finding similar to ours, and that there were no age-related differences. The prevalence of headache with aura peaks during middle age in women according to some studies (12, 13), but not all (5, 14).
The decline of active migraine headache prevalence with age seems to be somewhat more pronounced in this study than in two other studies (1, 3, 7). Launer et al. (5) presented a high 1-year prevalence rate of migraine in women aged 55–65 years and, thus, the decline with age was not large.
There was evidence from the data that intensity and the presence of nausea of unsuccessfully treated migraine headache attacks decline with age, whereas disability, frequency, duration, or the presence of symptoms related to migraine headache do not. This agrees with previous findings that nausea (14), but not frequency (1, 5) and disability (1), change with age. Waters (15) measured on a scale ranging from 0 to 14 ‘severity of headache’ in women aged 20–65 years from the general population. This scale combined severity and disability. There was an association with age when all women were included, but not within headache sufferers. Although a comparison is confined by differences in methodology, our findings seem to agree with his.
Magnitude and mechanism of the decline of prevalence of active migraine headache with age
In the age interval 45–74 years there was a remarkably good fit (R2 = 0.99) to an exponential decline model. Several other biological phenomena can be approximated to an exponential function. The T1/2 of active migraine headache prevalence, derived from this model, was close to 10 years. This is a straightforward and understandable description that may be used when discussing prognosis with patients. Headache intensity in conjunction with a migraine visual disturbance also declined with age. Educational level or the use of NSAIDs or HRT did not confound the magnitude of the age-related decline of prevalence. The effect of beta-blockers could not be analysed because of confounding by indication. Other agents, such as calcium-channel blockers, were not considered because of their low prescription rates or, alternatively, lack of an anti-migraine prophylactic property.
We suggest that the age-related decline in pain intensity causes the decline in active migraine prevalence. Since the severity criterion was mandatory for a diagnosis of migraine headache, we did not recognize women with mild migraine headache without aura. However, using the IHS classification, Rasmussen et al. (2) did not recognize any participant with mild headache among 58 participants with migraine without aura. Since frequency and duration of attacks did not change with age in our study, we suggest that there is a threshold of pain intensity for IHS-defined migraine headache. Below a certain pain level the headache attacks will no longer fulfil the criteria for migraine headache. Such a mechanism would lead to an age-related decline of criterion-based prevalence of migraine headache. The pain ratings of headache following migraine visual aura indicate that this level on a verbal analogue scale would be between 4 and 7.
A relationship between migraine pain intensity and migraine headache prevalence may result from variation in the migraine process itself, from factors affecting it such as medication or concurrent disease, or from factors affecting pain perception. There was no evidence in this study that NSAIDs, HRT, or differences in the treatment of attacks would affect the association between age and migraine headache prevalence or between age and migraine pain intensity. The apparent discrepancy that pain intensity and nausea declined with age, but not disability, may be explained by age-related factors that are part of the multidimensional experience of pain. Reduction of the ability to cope with challenges (16) is regarded as a hallmark of ageing. We are currently analysing factors with the potential to reduce the ability to cope with migraine pain and further hormonal aspects (to be published).
To conclude, the lifetime prevalence of migraine headache in middle-aged and older women was 31.5%. The prevalence of active migraine headache declined with age, a finding previously reported by other authors, whereas the prevalence of active migraine visual aura did not. We are not aware of any previous attempt to describe mathematically and estimate the magnitude of the decline, which was 50% per decade in this study. The decline was accompanied by a decrease in pain intensity, but not by changes in frequency or duration. We suggest that the decrease in pain intensity causes the decline in prevalence. Because of the cross-sectional setting of this study, cohort effects cannot be excluded. The results need to be confirmed in a longitudinal study. We plan to make a follow-up study of this cohort.
Footnotes
Acknowledgements
We are indebted to Dr Erik Thurfjell, Department of Diagnostic Radiology, and the staff at the Östhammar Mammography Unit for their co-operation. We wish to thank Professor Bengt Rosén, Stockholm University, for valuable statistical advice and Mrs Marita Jonsson for skilful assistance in data management. This study was supported by grants from the Erik, Karin and Gösta Selanders Foundation, the Research Foundation of the Uppsala University Hospital, the Foundation for Psychiatric and Neurological Research of Uppsala University, and the Swedish Migraine Society.
