Abstract
Little is known about the long-term effects of homeopathic treatment. Following a double-blind, placebo-controlled trial of classical homeopathy in chronic headaches, we conducted a complete follow-up study of all trial participants. All patients enrolled in the double-blind study were sent a 6-week headache diary, a follow-up questionnaire. Eighty-seven of the original 98 patients enrolled returned questionnaires, 81 returned diaries. There was no additional change from the end of the trial to the 1-year follow up. The improvement seen at the end of the 12-week trial was stable after 1 year. No differential effects according to treatment after the trial could be seen. Patients with no treatment following the trial had the most improvement after 1 year. Approximately 30% of patients in homeopathic treatment will benefit after 1 year of treatment. There is no indication of a specific, or of a delayed effect of homeopathy.
Keywords
Introduction
Clinical studies of homeopathy yield an equivocal picture of the clinical efficacy and effectiveness of homeopathy. One of the problems of randomized controlled trials (RCTs), not only when studying homeopathy, is that for practical, ethical and organizational reasons they have to be kept as short as possible and only as long as necessary to draw conclusions. Often these conclusions are preliminary and do not warrant any decisions about feasibility of a treatment and its acceptability, since observation periods are too short. When we planned our RCT on the efficacy of homeopathy in chronic headaches (1, 2), which was a 12-week double-blind trial of classical, individualized homeopathy in chronic headaches, we were well aware of the fact that 3 months of treatment cannot capture the therapeutic process of homeopathic therapy, let alone long-term effects. So we planned the trial so that initial effects would be reflected, and decided to conduct a follow-up survey of all participants after a year. We report here the results of this follow-up survey of all patients 1 year after the end of the trial.
Patients and methods
The trial has been reported in detail elsewhere in this journal (2). Briefly, patients suffering from chronic headaches (migraine, tension-type headaches, cluster headache, as diagnosed according to International Headache Society (IHS) criteria) were eligible for the study. They were recruited via a publicity campaign and treated by one of six homeopathic doctors working together in a homeopathic clinic. The patients had to fulfil standard inclusion criteria commonly used for headache and migraine trials, and had to be deemed treatable by the physicians. After screening for eligibility, written informed consent was obtained. Patients then entered a baseline phase of at least 6 weeks, and were subsequently randomized to receive either classical, individualized homeopathic treatment, or placebo. A 12-week double-blind treatment phase ensued, and another prescription could be given after 6 weeks.
One year after the end of the treatment, all patients were sent another 6-week headache diary and an ad hoc constructed follow-up questionnaire. They were reminded in a letter that the 1-year follow up was part of the original trial and asked to return the material. Up to two reminders were sent. In four patients who had withdrawn their consent we refrained from sending reminders. Since patients were free after the trial to either persist with homeopathic treatment in the same or another clinic, to go back to their original treatment, or have no treatment at all, and all treatment was open, this follow-up questionnaire and headache-diary data give a segment of the headache patients after 1 year.
Statistical analysis
All data were analysed descriptively. Headache diary data were averaged over treatment periods. Exploratory statistical tests were non-parametric, Mann–Whitney U-test for independent and Wilcoxon test for dependent data. All analyses were done using STATISTICA for Windows, version 5.
Results
Of the original 98 patients, four had withdrawn consent. Of the remaining 94 patients, 87 (88.7%) returned the questionnaires. Altogether 54 patients had been in homeopathic treatment subsequent to the trial, either within a long-term observation (n = 18; data to be reported) or paid privately (n = 36). There was no difference between these patients in original group allocation. Thus, in roughly half of the patients the homeopathic treatment was carried on. Three of those have changed the homeopathic practitioner, 27 are still under treatment in the clinic. One third of the patients have taken up conventional treatment again, the other patients are trying out other approaches like psychotherapy, acupuncture, or physical therapy. All but one patient still suffer from headaches, and report only a very small improvement (median 3.5 on a 5-point Likert scale with 3 meaning ‘unchanged’ and 5 ‘much better’).
Eighty-one patients (86% of all those who had finished the study) sent a follow-up diary. There was no significant difference in baseline variables between the patients who sent or did not send follow-up diaries.
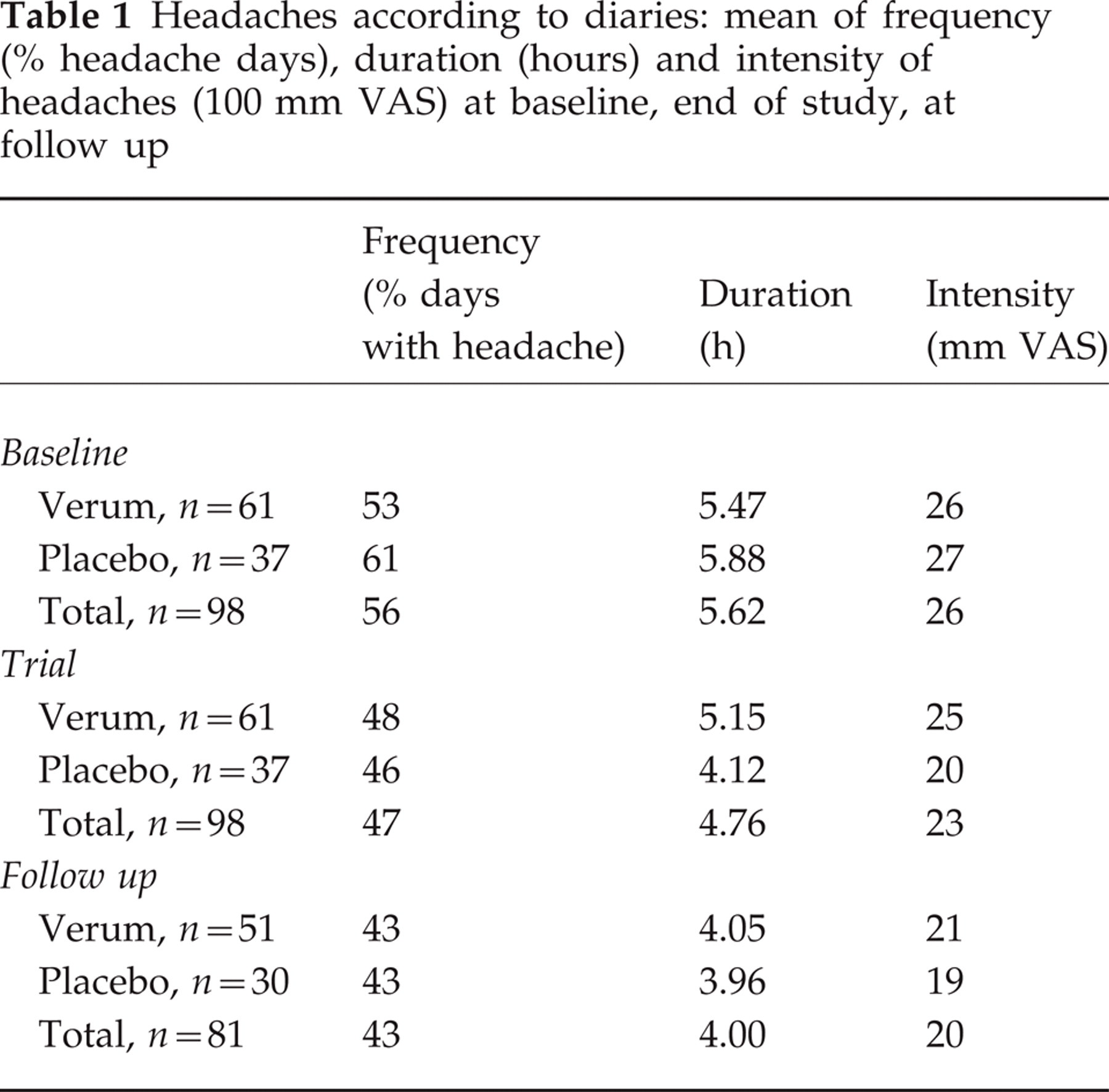
Table 1 gives mean values for the three outcome parameters of the study (frequency, intensity, and duration of headaches) as measured by the diary, for the last 4 weeks of baseline, the last 4 weeks of the double-blind trial phase, and the last 4 weeks of follow up. Improvement was visible in all parameters, without a notable difference between groups, mainly from baseline to the end of the double-blind trial. Improvement was stable at the end of the follow up. Changes from the end of the trial to follow up are not significant, those from the beginning of the trial to follow up are highly significant (Wilcoxon).
Headaches according to diaries: mean of frequency (% headache days), duration (hours) and intensity of headaches (100 mm VAS) at baseline, end of study, at follow up
Responder analysis
In order to carry out a responder analysis, we formed three groups: those patients who had improved > 20% from baseline to follow up (25 patients or 30%), those who had only improved marginally (57 patients or 54%), and those who had deteriorated (15 patients or 18%). We used all baseline data (sociodemographic data, disease history, knowledge of homeopathy, attribution of and expectation of effects, personality inventory and complaint list). The groups were not different in any of the complaint list or personality variables. The only difference was that responder had a significantly longer duration of illness than non-responder: 25.9 years vs. 16.5 years. Responders at follow up thus had a disease history which was 10 years longer than that of non-responders (P = 0.04). All other variables showed no differences.
Discussion
One of the main arguments against results of RCTs in homeopathy is that they cannot capture true homeopathic treatment effects, because the blinded treatment invalidates some of the strengths of homeopathic treatment by introducing an element of uncertainty (3). That was the reason why we decided to conduct a 1-year follow up, in order to catch some possibly delayed or long-term effects. The main result was that most of the clinically important changes had happened during the 12 weeks of the double-blind study. Although half of all patients who responded had carried on with the homeopathic treatment, results were only slightly improved after 1 year. By our very relaxed standards of at least 20% improvement as opposed to IHS standards (4), 30% of all patients can be counted as responders. This result can hardly be criticized on methodological grounds. Our return rate of both questionnaires and follow-up diaries was high, and it can be surmised that those patients who have not returned questionnaires or diaries had no success and thus would rather have darkened the picture than otherwise.
It is interesting to see that no clear-cut prognostic variables were discovered by our exploratory analysis. However, we are now in the position to dispel some myths which have been argued against our results and against trials in homeopathy in general (5). Certainly disease history, and complexity of disease are not negative prognostic factors, since the patients who had the most improvement suffered from both tension-type and migraine headaches and had a disease history of 25 years on average, comparable to the rest of the sample.
In conclusion, there is no evidence that the lack of specific efficacy of homeopathy in headache as brought out by a previous RCT (2), and indeed two other trials (6, 7), is due to the shortness of duration of the study or the severity of the disease. Further homeopathic treatment adds only a slight advantage to the effects of the 12-week double-blind trial in this study. Improvements remain stable after 1 year.
Footnotes
Acknowledgements
This study was funded by the Robert-Bosch-Foundation, Stuttgart. Jürgen Firnkorn of the Foundation helped a great deal in bringing this study under way. We are grateful to Professor M. Eichelbaum, Professor U. Klotz and various other referees for improving the design of the study. We are especially grateful for logistic help with data entry to Professor W. Gaus (Department of Biometrics, Ulm). Several research assistants helped with study organization and data monitoring, among them Simone Eiche and especially Claudia Haager. Cornelia Dehm and Axel Schweikhardt helped with checking and preparing data for evaluation. Homeoden (Gent, Belgium) and Gudjons (Augsburg, Germany) provided the remedies.
