Abstract
Objective and design: This open‐label study was conducted to evaluate the tolerability and efficacy of the 5HT1 agonist naratriptan with repeated use in the acute treatment of migraine attacks for 1 year. Four hundred and seventeen (417) migraine patients treated 15 301 migraine attacks over the course of the study. Results: The results show that 84% of attacks treated with a single 2.5 mg dose of naratriptan were not associated with the occurrence of an adverse event. The percentage of attacks associated with an adverse event did not increase with number of doses used to treat a given attack (1 vs. 2) or duration of use (0–6 months vs. > 6–12 months). The only adverse events experienced in > 2% of attacks throughout the 1‐year study were nausea (3% of attacks), hyposalivation (2% of attacks), and drowsiness/sleepiness (2% of attacks). Headache relief 4 h post‐dose was reported in a median 70% of moderate or severe attacks and a median 86% of mild attacks treated with naratriptan tablets 2.5 mg. The percentages of patients reporting headache relief did not diminish as a function of increased duration of treatment (0–6 months vs. > 6–12 months) or frequency of use (for > 36 vs. < 36 attacks). The mean number of tablets taken per attack was 1.2. A second naratriptan 2.5 mg tablet was taken for headache recurrence in a mean 16% (median 8%) of attacks. Conclusion: The results of this study demonstrate that naratriptan tablets 2.5 mg taken for acute migraine attacks over a 1‐year period are well‐tolerated and effective.
Until recently, sumatriptan was the only selective 5HT1 agonist available for migraine. Available in four formulations (injection, tablet, nasal spray, suppository), sumatriptan allows clinicians to tailor therapy to meet the needs of the individual patient. The introduction of additional 5HT1 agonist tablets such as naratriptan further expands the range of therapeutic options for acute treatment of migraine. Although trials directly comparing the newer 5HT1 agonists have not been conducted, results of placebo‐controlled, non‐comparator studies have begun to reveal therapeutic features that distinguish these newer 5HT1 agonists from one another.
Data from single‐attack, placebo‐controlled studies suggest that naratriptan at the therapeutic dose of 2.5 mg may have a particularly good tolerability profile (1–3). The overall incidence of adverse events as well as the incidence of specific adverse events (e.g. neurological, cardiovascular) after treatment with naratriptan tablets 2.5 mg does not differ from the incidence with placebo in controlled clinical trials (1, 2). Approximately 65% of patients treated with naratriptan tablets 2.5 mg compared with approximately 30% of placebo‐treated patients reported relief of headache and associated symptoms such as nausea and photophobia 4 h post‐dose in these studies (1–3).
This open‐label study was conducted to determine if the tolerability and efficacy of naratriptan observed in single‐attack studies is maintained with repeated use in the acute treatment of migraine attacks for 1 year.
Methods
Patients
Male and female patients were included in the study if they were 18–65 years of age and had a 1‐year history of migraine with or without aura diagnosed according to International Headache Society criteria (4) with 1–6 moderate or severe migraine attacks per month in each of the 6 months prior to screening for the study. Reasons for exclusion from the study included: pregnancy or lactation; uncontrolled hypertension; screening supine diastolic blood pressure greater than 95 mmHg or supine systolic blood pressure greater than 160 mmHg; history of cardiovascular disease, atherosclerotic disease, epilepsy, basilar migraine, or hemiplegic migraine; current or historical abuse during the past year of ergotamine or opiate analgesics; current lithium use; and known hypersensitivity to, intolerance of, or contraindications to, the use of sumatriptan, naratriptan or other 5HT1 agonists.
Procedures
The procedures for this open‐label, 12‐month study have been described elsewhere (5). Ethics committees at each of the 29 study sites in Australia, France and The Netherlands approved the study protocol. The study was conducted in accordance with the Declaration of Helsinki, as modified by the 41st World Medical Assembly, Hong Kong, September 1989. Written, informed consent was obtained from all patients participating in the study.
Clinic visits occurred at screening and quarterly thereafter through the 1‐year duration of the study. A supply of study medication was provided to patients at each clinic visit.
Patients were instructed to use one naratriptan tablet (2.5 mg) at the onset of each moderate or severe migraine. Patients were asked to take a second naratriptan tablet (2.5 mg) in the event of headache recurrence (return of moderate or severe pain within 24 h of initial dosing, where moderate or severe pre‐dose pain had been reduced to mild or none 4 h after initial dosing), but not for inadequate relief after initial dosing. The 2.5 mg starting dose could be reduced to 1 mg if the patient experienced intolerable adverse events on the 2.5 mg dose. Ergot‐containing medications and sumatriptan were prohibited within 24 h before or after use of study medication; analgesics, antiemetics, or other acute migraine medications, were prohibited within 6 h before use of study medication. Patients could take rescue medication (with the exceptions of sumatriptan or ergot‐containing medications) beginning 4 h after dosing with study medication.
For each attack treated with study medication, patients recorded on diary cards: (1) the occurrence of adverse events (any untoward medical occurrence regardless of its suspected cause); (2) headache severity (no, mild, moderate, or severe pain) pre‐dose and 2 and 4 h post‐dose; (3) date and time of rescue medication use; and (4) overall rating of the study medication (excellent, good, reasonable, poor, very poor).
The primary measure of tolerability was the incidence of adverse events, calculated as percentage of attacks with adverse events. The primary measure of efficacy was the percentage of moderate or severe attacks in which patients reported headache relief (reduction or moderate or severe pre‐dose pain to mild or no pain) 4 h post‐dose. Other efficacy measures included: (1) the percentage of attacks requiring rescue medication 4–24 h post‐dose; (2) the percentage of attacks treated with a second dose of naratriptan for recurrence; and (3) the percentage of patients rating study medication as good or excellent. The percentage of mild attacks with relief and the mean and median numbers of naratriptan 2.5 mg tablets taken per attack were also examined in post hoc analyses. Descriptive statistics, but no formal statistical tests, on the tolerability and efficacy data were calculated.
Results
Patients
Of the 451 patients enrolled in the study, 417 treated at least one migraine attack with study medication and were included in the tolerability and efficacy analyses. Thirty‐four of the 451 patients withdrew from the study prior to taking any study medication.
The 417 patients treated 15 218 attacks with naratriptan tablets 2.5 mg during the study. An additional 83 attacks among 10 patients were treated with naratriptan tablets 1 mg. These numbers include 266 attacks treated beyond the 12‐month period defined for the study.
Twenty‐eight per cent of the 417 patients treating at least one migraine attack with study medication withdrew from the study over the course of the year for reasons such as lack of efficacy (17%), adverse events (3%), failure to return to the clinic (3%), and other (including protocol violations, 5%).
Patient demographics and migraine histories are depicted in Table 1. The majority of patients were Caucasian women, most of whom had a history of migraine without aura only. Most patients typically experienced severe headaches, which occurred at a frequency of 1–3 per month.
Patient demographics and migraine histories

Adverse events
Eighty‐four per cent of attacks treated with a single 2.5 mg dose of naratriptan were not associated with the occurrence of an adverse event. The percentage of attacks associated with an adverse event did not increase with number of doses used to treat a given attack (1 vs. 2) or duration of use (0–6 months vs. > 6–12 months; Table 2). The only adverse events experienced in > 2% of attacks throughout the 1‐year study were nausea (3% of attacks), hyposalivation (2% of attacks), and drowsiness/sleepiness (2% of attacks). Adverse events of special interest with the 5HT1 class of compounds (including chest pressure/heaviness, chest pain, chest tightness, warm/hot sensation, numbness, tingling, pressure) were reported very infrequently (each event < 1% of attacks treated with naratriptan tablets 2.5 mg).
Percentage of attacks with adverse events after a 2.5‐mg dose

Adverse events precipitated 15 patients' (3%) withdrawal from the study over the course of the year. Of these adverse events, the only adverse events that were considered by the clinician to be probably related to administration of study medication were high blood pressure in one patient and dry mouth/flushed face/swollen tongue, nausea and vomiting, and worsening of headache in a second patient.
The patient with the high blood pressure considered to be probably related to study medication experienced a 30‐mmHg increase in systolic blood pressure (from 150 mmHg to 180 mmHg) and a 15‐mmHg increase in diastolic blood pressure after treating attack number 17 with one dose of naratriptan tablets 2.5 mg.
Chest pain leading to withdrawal in one patient was considered by the clinician to be possibly related to administration of study medication. This patient, a 46‐year‐old woman, experienced moderate chest pain beginning after she treated her third attack with naratriptan tablets 2.5 mg (+ 2.5 mg) and lasting for 3 h. ECGs and cardiac enzymes analyses performed after the adverse event were normal.
Efficacy data
Headache relief
In the 1‐year period defined for the study, 14 953 attacks (12 930 of which were moderate or severe) were treated with naratriptan 2.5 mg + 2.5 mg. (An additional 266 attacks, not included in this analysis, were treated after the end of the predefined study period.) Headache relief 2 h post‐dose was reported in a median 49% of moderate or severe attacks treated with naratriptan 2.5 mg ± 2.5 mg. Headache relief 4 h post‐dose (primary efficacy parameter) was reported in a median 70% of moderate or severe attacks treated with naratriptan tablets 2.5 mg (Fig. 1). Headache relief rates did not diminish as a function of increased duration of treatment (Fig. 1) or frequency of use (Fig. 2).
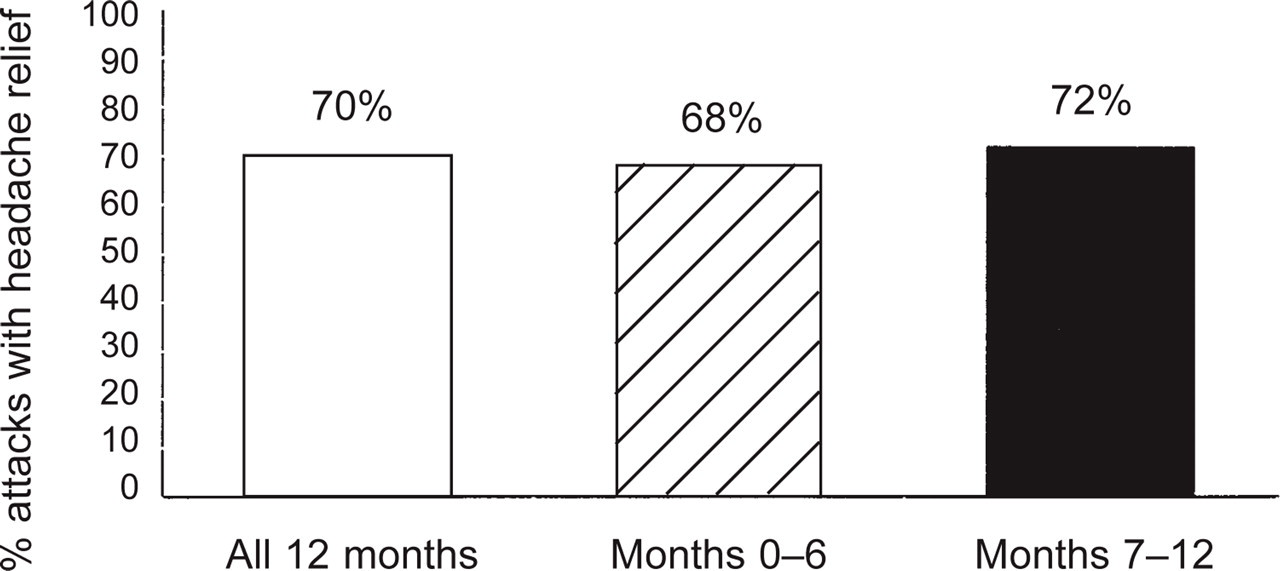
Percentage of attacks treated with naratriptan 2.5 mg in which moderate or severe pre‐dose pain was reduced to mild or no pain 4 h post‐dose across the 1‐year study and during the first vs. second 6 months of the study.

Percentage of attacks treated with naratriptan 2.5 mg in which moderate or severe pain was reduced to mild or no pain 4 h post‐dose in patients treating < 36 vs. > 36 migraine attacks over the 1‐year study period.
Naratriptan 2.5 mg was taken for a mild headache for 1744 attacks in violation of the study protocol, which specified that patients were to treat moderate or severe attacks. A post hoc analysis of data from these patients treating mild attacks revealed that no pain 4 h post‐dose was reported in a median 73% of mild attacks treated with naratriptan tablets 2.5 mg.
Numbers of tablets per attack
The mean and median numbers of naratriptan 2.5 mg tablets taken per attack were calculated in a post hoc analysis. This analysis included patients taking a second dose of naratriptan for recurrence, but not those violating the study protocol by taking a second dose of naratriptan as rescue medication. The mean number of tablets taken per attack was 1.2. The median number of tablets taken per attack was 1.0.
Second dose of naratriptan or other medication
A second naratriptan 2.5 mg tablet was taken for headache recurrence in a mean 16% (median 8%) of attacks. A second naratriptan 2.5 mg tablet was taken in violation of the study protocol as rescue medication in a mean 15% (median 9%) of attacks. Rescue medication other than naratriptan was taken 4–24 h after an initial 2.5 mg naratriptan dose in a mean 19% of attacks (median 15%).
Overall rating of study medication
Patients rated naratriptan tablets as good or excellent in a mean 64% of 14 671 attacks treated with 2.5 mg ± 2.5 mg.
Discussion
The results of this study demonstrate that naratriptan tablets 2.5 mg taken for acute migraine attacks over a 1‐year period are well‐tolerated and effective. The vast majority (84%) of attacks treated during the 1‐year study were not associated with adverse events. The percentage of attacks with adverse events did not increase as a function of number of naratriptan 2.5 mg doses used to treat a given attack (1 vs. 2) or time in the study (first 6 months vs. second 6 months).
The most common adverse event in this study was nausea, which was reported in 3% of attacks. The occurrence of this adverse event, which is a symptom of migraine in many patients, highlights the fact that adverse events were reported regardless of their suspected causality in this study. Migraine symptoms not attributed to administration of the study drug could be reported as adverse events.
The use of naratriptan tablets 2.5 mg for the acute treatment of migraine attacks for 1 years was associated with an extremely low incidence (< 1%) of adverse events typically associated with ‘triptan’ compounds, including chest symptoms (e.g. tightness, pressure) and sensations such as pressure or tingling in the limbs. A similarly low incidence of ‘triptan’‐associated adverse events has been observed in placebo‐controlled trials with naratriptan tablets 2.5 mg (1–3).
Headache relief 2 and 4 h post‐dose was reported in 49% and 70%, respectively, of moderate or severe migraine attacks treated over the 1‐year study period. The efficacy of naratriptan did not wane as a function of time in the study (first 6 months vs. second 6 months) or frequency of use of naratriptan (for more than vs. fewer than 36 attacks). The efficacy data from this open‐label study are consistent with those from placebo‐controlled, short‐term studies, in which approximately 65% of naratriptan‐treated patients compared with approximately 30% of placebo‐treated patients reported relief of headache and associated symptoms 4 h post‐dose (1–3). Thus, there is no evidence of tachyphylaxis with naratriptan.
The results of this study extend previous findings by demonstrating that naratriptan 2.5 mg is also effective in the treatment of mild attacks: headache relief 4 h post‐dose was reported in a median 86% of mild attacks. This outcome suggests that patients may also benefit from administration of naratriptan early in an attack, rather than waiting for the migraine to become moderate or severe.
The mean number of naratriptan 2.5 mg tablets taken per attack in this study was 1.2 (median 1.0). A second dose of naratriptan was used to treat headache recurrence in a mean 16% (median 8%) of attacks. These results suggest that a single dose of naratriptan provides potent, sustained headache relief. The data corroborate results of sumatriptan‐ and placebo‐controlled trials (1, 3–6) in which naratriptan 2.5 mg was associated with a low incidence of headache recurrence (17% to 28% across phase II and III placebo‐controlled trials compared with the ∼35% recurrence rate with other migraine therapies (7–10) with long duration of headache relief. The pattern of results suggests that low recurrence, in addition to high tolerability, distinguishes naratriptan among the 5HT1 agonists and other acute migraine medications.
The open‐label design and the long duration of this study were employed to more closely approximate the pattern of actual use of naratriptan in clinical practice than a controlled clinical trial could. The results of this study provide evidence that the high degree of tolerability and consistent efficacy with naratriptan observed in controlled clinical trials will extend to clinical practice.
