Abstract
Objective To determine the role of naratriptan in preventing migraine headache when administered during prodrome. Procedures Baseline phase: patients recorded prodrome symptoms and time of onset, time when patient knew that headache was inevitable, time of onset and severity of headache. Treatment phase: patients given naratriptan 2.5 mg to take at the time they knew headache was inevitable. Patients recorded prodrome symptoms and time of onset, time they knew headache was inevitable, time naratriptan administered, time of onset and severity of any headache. Patients treated three prodromes separated by at least 48 h. Findings Twenty patients completed both phases. During baseline phase, 59 prodromes were reported and all were followed by headache. Severity of headache: 5% mild, 51% moderate, 44% severe. During treatment phase, 63 prodromes were reported. Of these, 38/63 (60%) were not followed by headache. Among headaches that occurred, the majority occurred within 2 h of naratriptan administration, suggesting that naratriptan is more effective in preventing headache if taken early in prodrome. Severity of 25 headaches: 44% mild, 24% moderate, 32% severe. Conclusions Naratriptan 2.5 mg appears to prevent migraine headache when given early in prodrome. If headache occurs, severity appears to be reduced. □Prodrome, premonitory, aura, naratriptan, migraine
Migraine is a complex neurological event. An acute attack of migraine can be divided into a premonitory phase, a headache phase and a post-headache phase. The premonitory phase may consist of vague neurological changes, the prodrome, and/or focal neurological changes, the aura. At times the distinctions between aura and prodrome are unclear, thus the International Headache Society (IHS) uses the term ‘premonitory’ symptoms to include vague neurological symptoms and focal neurological symptoms. Many patients experi-encing premonitory symptoms report that they are harbingers of headache and impending disability. Premonitory changes may precede headache by up to 48 h (1). Early premonitory symptoms (prodrome) may include, but are not limited to, changes in mood, energy level, appetite, food craving, nausea, sense of hearing or sense of smell, swelling or fluid retention, excessive yawning, muscle pain or tenderness, irritability, confusion, extreme sleepiness, impaired speech or impaired memory (2). Auras are also premonitory symptoms but are typically more obvious and represent focal neurological change. According to the IHS, aura is defined as a visual or sensory change developing over a period of 5–20 min, lasting less than an hour and resolving within 60 min before or soon after the onset of migraine headache (1). Aura may involve visual symptoms such as flashing lights, zig-zag lines or visual distortion or sensory changes as the sensation of pins and needles around the lips or on the hand.
The reported prevalence of premonitory symptoms (prodrome and/or aura) varies considerably because of differences in definitions and reporting methods. It is generally accepted that auras occur with about 10–15% of migraine attacks (3). It is reported that the incidence of prodrome among migraineurs varies from 50% to 60% (4). Only a few studies have assessed prodrome symptoms and migraine in detail. In particular, there are few data available on the correlation between the prodrome phase and the headache phase. However, if prodromes predict the headache phase of migraine, prodromes may represent an early treatment opportunity to prevent the disability and pain of the headache phase of migraine.
Successful preventive therapy has been reported with domperidone (5), dihydroergotamine (6, 7) and zolmitriptan (8). However, distinctions between premonitory, prodrome and aura are not always clearly defined. Domperidone 30 mg prevented migraine in 66% of patients compared with 5% of patients receiving placebo if administered hours before the predicted onset of headache (5). In a study of 902 patients, 143 patients were treated with dihydroergotamine nasal spray during prodrome and/or aura. Eighty-six per cent reported either no pain or pain relief (6). In a separate study of 91 patients, 36% had no headache following administration of dihydroergotamine during prodrome or aura compared with 26% following administration of placebo (7). In a placebo-controlled study, zolmitriptan 20 mg administered during aura prevented migraine in three of 16 patients who completed the study (8).
The present study was conducted to explore the role of naratriptan in prevention of headache in migraine patients where prodrome has been documented and is reproducible.
Methods of study
Thirty-three patients from four private and general community practices were recruited to participate in an open-label, pilot study to determine if naratriptan 2.5 mg administered during prodrome could prevent headache. All patients were required to be aware of prodrome symptoms that preceded migraine by 4–24 h. Patients were also required to be able to differentiate between prodrome symptoms associated with migraine and similar symptoms not associated with migraine (i.e. normal fatigue not associated with headache). Subjects were male or female ≥ 18 years of age, met IHS diagnostic criteria for migraine with/without aura (IHS 1.2, 1.1) with at least a 6-month history of migraine with 2–6 attacks per month. Any migraine prophylactic medication was stabilized for at least 1 month prior to entry into the study. Patients were free of cardiovascular and cerebrovascular disease, hypertension, basilar or hemiplegic migraine or significant renal, hepatic, gastrointestinal, endocrine or neurological disease. Patients with more than 10 days per month of tension headache or women pregnant or breast feeding were excluded.
At screening, patients gave informed consent in accordance with the Guidelines for Good Clinical Practice. Patients were queried about symptoms considered by the patient to be prodrome, frequency of their occurrence and the time between when the patient felt the headache was inevitable and the onset of headache pain.
During the baseline phase, each patient maintained a daily diary until three prodromes followed by migraine headache pain were documented. Patients recorded prodrome symptoms, including time of onset of the first prodrome symptom, time when patient felt the headache was inevitable, time of onset and severity of headache pain. During the baseline phase, migraines were treated with the patients' usual therapy which was recorded in the diary.
During the treatment phase, patients were provided with six tablets of naratriptan 2.5 mg to treat three prodromes and three possible headaches. Patients were instructed to take naratriptan 2.5 mg at the time when they felt the headache was inevitable. Patients were instructed to administer a second dose of naratriptan 2.5 mg if headache pain followed the initial dose by 4–24 h. Headache occurring less than 4 h after initial administration of naratriptan was treated with a nontriptan, nonergot therapy determined by the investigator. Patients were required to be headache-free for 48 h before recording the onset of a new prodrome. After completing the treatment phase, patients were allowed to treat three additional prodromes with naratriptan.
Patient responses were analysed to summarize inter- and intrapatient similarities. Considered in the analysis were time of onset of prodrome symptoms, type of prodrome symptoms experienced, time naratriptan was administered, and the onset and severity of headache pain (if any) during the subsequent 24-h period.
Results
Thirty-three patients were enrolled and 20 patients completed both baseline and treatment phases. Thirteen patients did not complete the study: eight failed to treat a prodrome and five patients were either withdrawn due to a change in medical condition, failed to return before the deadline, withdrew consent or were lost to follow-up. The 20 patients had a mean age of 45 years and were comprised of 19 females and one male.
During the baseline phase, 20 patients experienced 59 prodromes. The most frequently reported prodrome symptoms were ‘change in energy’ (reported by 15 patients a total of 29 times = 49%), ‘change in mood’ (reported by 12 patients a total of 23 times = 39%), ‘irritability’ (reported by 13 patients 22 times = 37%), ‘food craving’ (reported by seven patients 16 times = 27%), ‘muscle pain/tenderness’ (reported by seven patients 16 times = 27%), ‘sleepiness’ (reported by 10 patients 16 times = 27%) and ‘change in nausea’ reported by 11 patients and ‘change in appetite’ reported by six patients (each reported 15 times = 25%).
During the treatment phase, 20 patients experienced 63 prodromes. The most frequently reported symptoms were ‘irritability’ (reported by 12 patients a total of 30 times = 48%), ‘change in nausea’ (reported by 13 patients a total of 27 times = 43%), ‘muscle pain/tenderness’ (reported by nine patients a total of 24 times = 38%), ‘change in energy level’ (reported by 11 patients a total of 19 times = 30%), ‘change in mood’ (reported by six patients a total of 15 times = 24%), ‘sleepiness’ (reported by 10 patients a total of 14 times = 22%), ‘change in appetite’ (reported by eight patients a total of 13 times = 21%) and ‘yawning’ (reported by six patients a total of 13 times = 21%). Thus, the most frequently reported symptoms were experienced by the majority of patients during both baseline and treatment phases.
Although many symptoms did not occur during consecutive prodromes and order of onset was not always consistent, nine of 20 patients did experience at least one prodrome symptom that occurred during all of their reported prodrome phases. Prodrome symptoms did predict migraine headache for almost all subjects and patients were able to distinguish between prodrome symptoms resulting in migraine and similar interictal symptoms.
During the baseline phase, 100% of the prodromes were followed by headache. During the treatment phase, 38 of 63 (60%) treated prodromes were not followed by headache within the subsequent 24-h period. In nine of 13 (69%) patients with one or more prevented headaches, the effect was consistent across all treated prodromes (Fig. 1). Three patients prevented one headache, three patients prevented two headaches, four patients prevented three headaches, one patient prevented five headaches and two patients prevented six headaches.
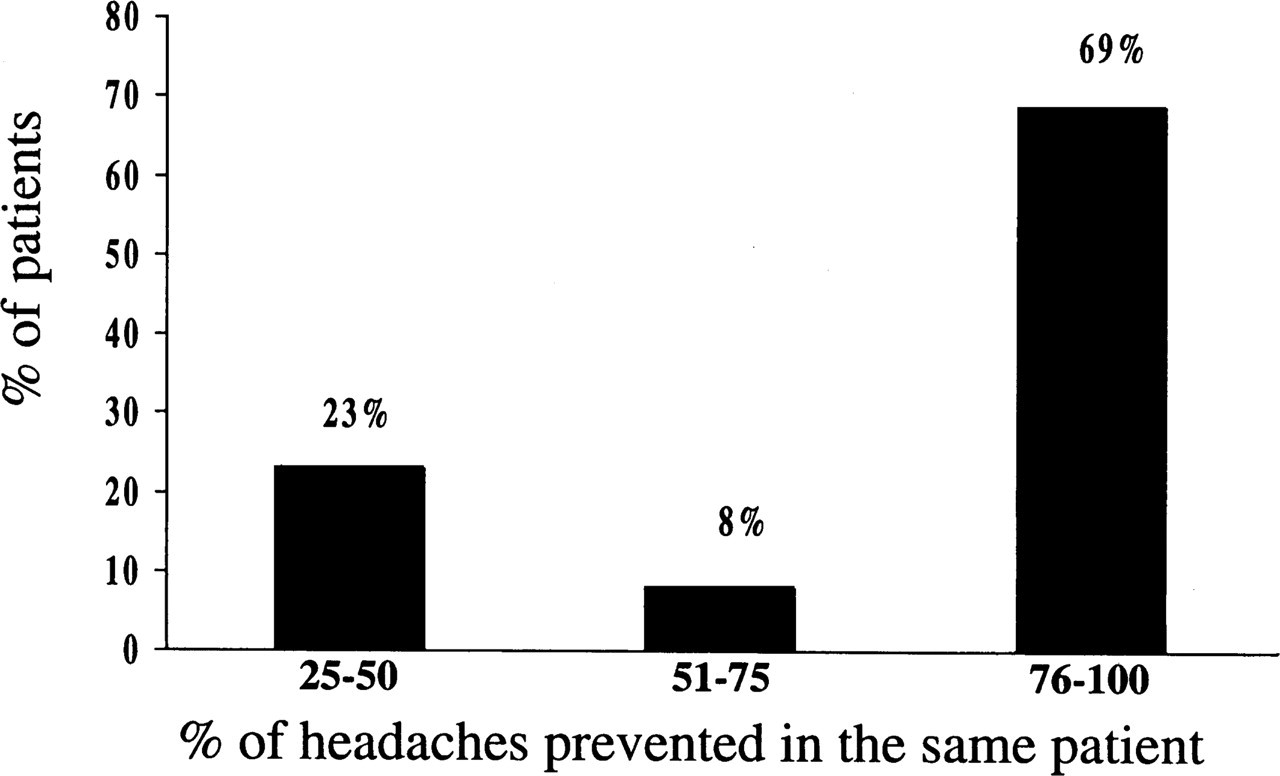
Consistency of effect of naratriptan among those patients who did not experience headache following treatment with naratriptan during prodrome.
For those headaches that did occur, the majority occurred within 2 h of naratriptan administration (Fig. 2).

Time between administration of naratriptan 2.5 mg during prodrome and the onset of headache pain in those patients who experienced headache.
The distribution of headache severity during the baseline phase was 3(5%) mild, 30(51%) moderate and 26(44%) severe. For the 25 prodromes that were followed by headache during the treatment phase, headache severity was 11(44%) mild, 6(24%) moderate and 8(32%) severe, representing a reduction from what would have been predicted from the baseline assessment (Fig. 3).

Severity of headaches during baseline phase (n = 59) compared with severity of headaches during treatment phase (n = 25) following administration of naratriptan during prodrome. □= baseline; ▪= treatment.
Discussion
The pathophysiology of migraine is not completely understood. However, evidence is accumulating that central factors are involved. Recently, the central nervous system of migraineurs has been shown to be different from normal individuals in its inherent excitability (9, 10). Furthermore, activation of pain and cardiovascular control centres in the brainstem during migraine suggests that these regions may be the ‘migraine generator’ (11). These central models are attractive because they could explain the diverse central and systemic symptoms experienced with migraine. Cumberbatch et al. suggest that changes in central pain signal processing occur as a result of vasodilation during a migraine. In these studies, electrophysiological recordings were made from single convergent (receiving nociceptive input from the dura and non-nociceptive input from the vibrissae) neurones in the caudal brainstem of anaesthetized rats. A marked facilitation of the non-nociceptive response to a jet of air blown onto the whiskers of the rat was observed when vasodilation of meningeal vessels was induced by CGRP. This phenomenon is termed ‘winding up’. Interestingly, pre-treatment with a brain penetrant 5-HT 1B/1D receptor agonist (L741 604) prevented this vasodilation-induced sensitization of pain (12). Although highly speculative, such a ‘wind up’ phenomenon and amplification of sensory processing may explain some of the prodrome and aura symptoms observed with migraine and provides a rationale for evaluating whether administration of naratriptan during prodrome can prevent migraine headache.
Sumatriptan has been shown to prevent menstrual migraine when initiated in scheduled regimens around menses (13). Dowson reported that high-dose zolmitriptan administered during aura prevented headache in three of 16 patients (8). Interestingly, in the present study it appeared that failure to prevent headache occurred when naratriptan was administered within short proximity (less than 2 h) of the onset of headache. This may explain why so few headaches were prevented when treated during aura in the Dowson study (8). A similar explanation may also explain why subcutaneous sumatriptan did not prevent headache when administered during aura (14).
This study has numerous limitations. It is an open label study without the benefits of a placebo control. The data represent a population of highly selected migraine patients since each was selected on the basis of previously identifying prodromes that preceded headache. Nonetheless, 60% of predicted headaches
Another limitation is the lack of prospective data on prodromes and the specificity of prodrome to headache in a more general population. The question of a prodromal threshold that would reliably predict headache can not be accurately assessed from this study. This remains a question for future studies. It should be noted, however, that during the baseline phase, patients reported with considerable accuracy the association of prodrome and headache and a majority of the patients did consistently ‘prevent’ the headache in at least two of the three attempts.
Conclusion
Naratriptan 2.5 mg appears to prevent the headache phase of migraine when given early in prodrome. If headache ensues, there appears to be a reduction in headache severity. Selected patients appear better able to identify prodrome symptoms and distinguish them from similar symptoms not associated with headache. Use of naratriptan during prodrome has the potential to prevent headache, thereby reducing disability and, possibly, the need for daily prophylactic therapy. However, more research is needed before clinical treatment of prodrome can be recommended.
Footnotes
Acknowledgements
We acknowledge the contributions and assistance of Timothy Kuhn PharmD, Paul Stang PhD, Carolyn Hart BSN and M.E. Beach LPN. Funding was provided by GlaxoWellcome Inc.
