Abstract
The interest in dual action antidepressants arose from the observation that the Selective Serotonin Reuptake Inhibitors (SSRIs) might be less efficacious than tricyclic antidepressants, especially in the treatment of more severely depressed patients. Clomipramine possesses 5-hydroxy tryptamine (5-HT) and noradrenaline (NA) reuptake dual action activity. The Danish University Antidepressant Group conducted two separate studies comparing clomipramine and the SSRIs citalopram and paroxetine. Therapeutic response with clomipramine was twice as high as with citalopram 60% vs 30% [1]. Similar results were obtained when clomipramine was compared with paroxetine [2].
The superior efficacy of tricyclic antidepressants in the treatment of more severely depressed patients was also demonstrated in two separate meta-analysis conducted by Anderson [3, 4]. The 2nd meta-analysis [4] confirmed that tricyclics with dual action on 5-HT and NA reuptake (e.g. clomipramine and amitriptymine) as compared with the tricyclics with a predominant effect on noradrenaline reuptake (e.g. desipramine and maprotiline), action had a greater efficacy than the SSRIs (p = 0.017).
There are several synaptic interactions between the two neuronal pathways, in the brain nuclei where they have their cell bodies, the locus coeruleus for the NA neurones, and the raphe nucleus for 5-HT neurones, as well as in the regions of their terminal fields, such as the cortex and hippocampus [5]. For example, in the raphé nucleus, NA released from noradrenergic terminals activates excitatory alpha-1 receptors on serotonergic neurones. Thus, activation of the NA system will facilitate serotonergic transmission, with synergy between the two-neurotransmitter systems (Fig. 1).
Interactions between noradrenaline (NA) containing and serotonin (5-HT) containing neurones in the central nervous system. (+) and (–) indicate, respectively, an excitatory or an inhibitory effect on the target neurone. (Reproduced with permission of author and the publisher, Mike Briley and David Oldfield, ADIS International, ISSN:1473–8066).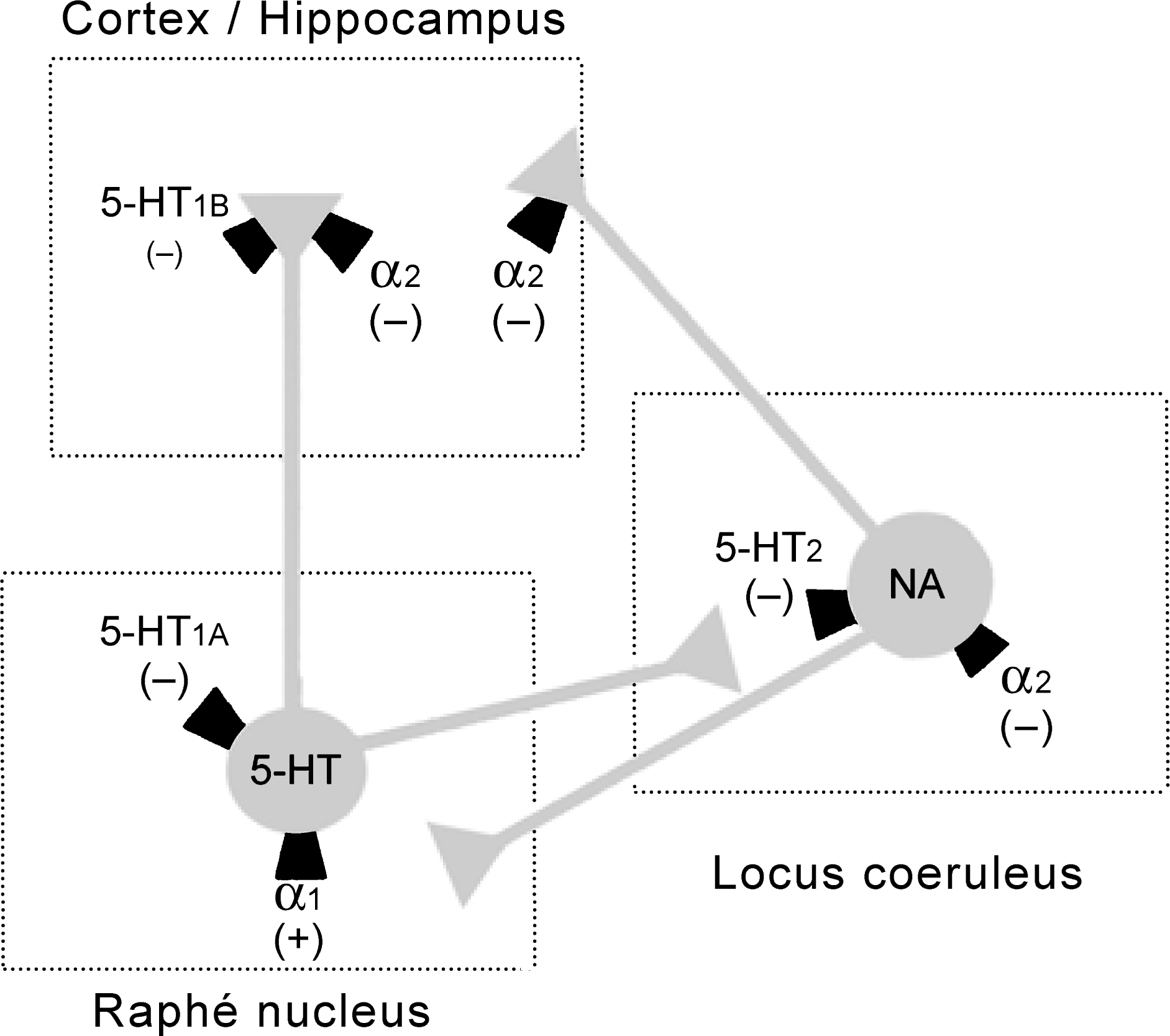
There are three examples of modern generation dual action antidepressant. Two of these, mirtazapine and venlafaxine, are available in Australia while the third, milnacipran, is not at present. The clinical evidence of the efficacy of these antidepressants is presented below along with other important issues in the long-term treatment of depression.
Mirtazapine enhances noradrenergic transmission by blocking presynaptic α −2 autoreceptor and increases serotonin release by blocking α −2 heteroceptors; in addition, it antagonizes both 5-HT2 and 5HT3 receptors, thus allowing agonistic effects predominantly at 5-HT1a receptors [6–8]. Venlafaxine enhances presynaptic levels of serotonin and noradrenaline by inhibiting their reuptake. At doses up to about 200 mg per day it has serotonergic activity while at higher doses it enhances noradrenergic activity as well [9–11]. Milnacipran is a cyclopropane derivative, which acts by inhibiting noradrenaline and serotonin reuptake at presynaptic sites. No postsynaptic activity has been demonstrated [5].
Mirtazapine
Thase et al. [12] studied 156 patients in the continuation phase in a double-blind placebo controlled trial on intention to treat basis with 76 patients treated with mirtazapine and 80 with placebo. Eleven point eight per cent patients dropped out with mirtazapine due to adverse events. Nineteen point sever per cent patients relapsed while receiving mirtazapine compared to 43.8% on placebo over a period of 40 weeks.
Montgomery et al. [13] pooled data from extensions of four randomized, double-blind, three-way studies; mirtazapine versus amitriptyline versus placebo. After the initial 6-week treatment period, the responders to treatment entered the double-blind extension phase up to 2 years. At endpoint, significantly more mirtazapinetreated patients than amitriptyline- or placebo-treated patients were classified as having no depression (17-item Hamilton Depression Rating Scale < 7). A survival analysis of time-to-first-relapse showed that mirtazapine was superior to amitriptyline and placebo over a 1-year period. The relapse rate with mirtazapine (8.6%) was significantly lower than with amitriptyline (21.5%; p < 0.05) or placebo (35.5%; p < 0.0001).
The efficacy of mirtazapine in continuation treatment of depression has been compared to amitriptyline and placebo [14]. Mirtazapine was given to 43 patients, amitriptyline was given to 48 and placebo to 27 patients. The patients who received treatment with mirtazapine scored less than 7 on the Hamilton Depression Rating Scale (HDRS) (17-item) throughout the 29-week continuation period. At the endpoint of the study, significantly more patients treated with mirtazapine were classified as having no depression compared with placebo (mirtazapine 74.4%; p = 0.02, amitriptyline 62.5%; p = 0.035 and placebo 38.5%). Although a higher percentage of patients classified as having no depression was present in the mirtazapine than in the amitriptyline group, the difference did not reach statistical significance (p = 0.226).
Venlafaxine
Shrivastava et al. [15] conducted a multicentre, doubleblind, parallel-group comparison of venlafaxine and imipramine. A consistent trend in favour of venlafaxine for the therapeutic response was observed, with statistically significant differences (p < 0.05) noted at 2, 6 and 12 months. Fewer patients treated with venlafaxine had unsatisfactory treatment response and lesser adverse events in comparison to imipramine.
A meta-analysis of four comparative clinical trials in which patients received venlafaxine or comparator placebo for up to 12 months after completing short-term treatment (6 weeks) was performed [16]. The primary objective of this analysis was to compare the rate of relapse/recurrence of depression in patients treated with venlafaxine (n = 185) to that experienced by patients treated with placebo (n = 119). At the end of 12 months of long-term treatment, the cumulative relapse rates were 20% in the venlafaxine group and 34% for the placebo group. There was a significant difference between the relapse rate for venlafaxine and that for placebo (p = 0.026), however, there were no significant differences between the rate for venlafaxine and the active comparator medications.
Several other reports describe the efficacy of venlafaxine to prevent recurrence of depression. A 12-month, double-blind, randomized, placebo-controlled study assessed the effectiveness of venlafaxine 100–200 mg/day, to prevent recurrence in patients with recurrent major depression. Patients who had responded to an 8-week, acute trial of venlafaxine were continued on open-label therapy for a total of 6 months, and then randomized to double-blind treatment with either venlafaxine or placebo for up to 12 months. Discontinuation due to lack of efficacy occurred in 48% of the placebo group and 21% of the venlafaxine group. Interestingly, the incidence of common adverse events was similar in the placebo and venlafaxine group during the double-blind phase [17].
Milnacipran
A total of 214 recovered patients were randomized to receive either milnacipran (50 mg b.i.d.) or a placebo for a 1-year recurrence prevention phase. Milnacipran was effective in reducing recurrences (Odds Radio (OR) = 1.5; p < 0.05), with a more marked effect in recovered patients with few residual symptoms (OR = 3.0). Responders who continued treatment with milnacipran had a considerable improvement in their quality-of-life (QoL). The patients with residual symptoms still experienced some deterioration in their QoL (recreation, emotional, social, alertness and home assistance scores). Even recovered patients having zero score on the HDRS-21-item had some QoL deterioration. Overall, there was a significant improvement in the milnacipran group in comparison to the placebo on the mobility, communication and the psychosocial measures [18].
Side-effects of dual action antidepressants
As with tricyclic antidepressants and the SSRIs, the dual action antidepressants come with their own sideeffect profile. Important aspects are discussed here.
Previous studies [3, 4, 19–22] have examined the relative side-effects of various groups of antidepressants and the bearing these may have on treatment adherence and outcome. They show that successive classes of antidepressants have increased the proportion of patients benefiting from treatment. The newer antidepressants are devoid of the cardiovascular and autonomic side-effects of tricyclic antidepressants, with the exception of venlafaxine, which can produce hypertension doses above 300 mg [23]. Further, venlafaxine also facilitates sympathetic neuro-transmission and may cause dysuria which also occurs in approximately 3% of patients treated with milnacipran [24].
Nausea is a frequently reported side-effect of venlafaxine [25, 26] and occurs to a esser degree with milnacipran [27]. The relative propensity of these two dual action antidepressants to cause nausea probably reflects their relative inhibitory potency on serotonin uptake.
Sedation is one of the most common side-effects of tricyclic antidepressants and is responsible for the 50% of all tolerability-related treatment discontinuations (15% of treatment courses) for tricyclic antidepressants attributable to ‘lethargy’ [24]. Tricyclic antidepressants may interact with up to three monoamine receptors known to influence arousal, the α 1 adrenoreceptor, the muscarinic acetylcholine receptor and the H1 histamine receptor, cumulating their sedative potential. These sedative properties are also shared by mirtazapine, which has potent H1 histamine receptor antagonist properties [28]. A lack of α 1-adrenergic antagonistic activity and the effect on H1 histamine receptor is related to the reduced incidence of somnolence and fatigue observed in milnacipran treated patients. The incidence of somnolence and fatigue is less than those treated with placebo [24]. Venlafaxine does not interact with a number of neurotransmitter sites and therefore lacks the anticholinergic side-effects. Sedation is not a regular sideeffect. A single dose of alcohol or diazepam given with Venlafaxine did not significantly effect central nervous system activity including psychomotor performance, information processing and short-term recall [29].
The incidence of self-reported weight gain after 12 weeks of therapy with mirtazapine is 21%. It rose to 30% among patients who continued to take active medication during the continuation phase [12]. There was no difference between groups in the incidence of new-onset weight gain during the continuation phase (mirtazapine, 7.9%; placebo, 7.3%). The weight gain during the 40 weeks of mirtazapine continuation therapy was only about half the amount of the weight gain observed during the first 8–12 weeks of therapy [12]. This was, on average, 2.5 kg. A 12-week study by Silverstone and Ravindran comparing venlafaxine and fluoxetine showed no significant weight gain among patients treated with either agent [30]. In patients who continued venlafaxine beyond 5 months, mean weight increases from baseline of up to 2.5 kg were observed by month 20 of therapy [31]. However, if a patient is well advised and counselled some or most of the weight gain can be controlled. Since milnacipran is a relatively new antidepressant there is little data avaliable with regard to its effect on weight gain and it does not appear to be an issue so far.
Sexual side-effects with SSRIs and venlafaxine occur in as many as 58–73% of patients [32–35]. Decreased sexual desire and activity is frequently associated with depression itself [36], so it is important that this is not further impaired by treatment. Treatment with SSRIs interferes with the ease of orgasm and orgasmic satisfaction in men. It has been demonstrated that SSRIs also lead to anorgasmia in women, and this may in fact be more frequent than in men [37]. In addition to their deleterious effects on orgasmic ability, SSRIs also appear to depress libido in both men and women [33]. It should be mentioned that SSRIs have been proposed to be of benefit in the treatment of premature ejaculation [38].
The dual action antidepressants appear to be relatively well tolerated in this respect. The use of venlafaxine produces some degree of delayed orgasm, but the frequency is considerably lower than with SSRIs [39]. In the case of milnacipran, no systematic surveys have been carried out. However, studies in volunteers [32] have not revealed any such effects, and no reports have arisen from the clinical development programme or from pharmacovigilance data acquired over a three year period [40]. The difference between venlafaxine and milnacipran in this respect may be due to the relative selectivity of the former towards serotonin uptake and of the latter towards noradrenaline uptake.
The 5HT2 antagonism of mirtazapine seems to almost totally eliminate sexual dysfunction. In order to manage treatment emergent sexual side-effects one may consider waiting for tolerance to the side-effect to develop or alternatively switch to an antidepressant with 5HT2 antagonism activity. Drug holidays or a reduction in the dose of prescribed antidepressant are likely to be counterproductive by increasing the risk of relapse or recurrence of depression.
Withdrawal syndrome
Sudden cessation of continuing long-term therapy with any of these agents may precipitate withdrawal symptoms. Such symptoms were well known to occur with TCA as well as with the SSRIs [41]. There have been case reports of psychiatric withdrawal symptoms (panic attack and hypomania) after cessation of mirtazapine treatment [42, 43]. A few case reports of a withdrawal syndrome similar to the serotonin syndrome seen with SSRIs have appeared with venlafaxine [43–45]. Typical venlafaxine discontinuation symptoms include a subjective experience of substantial distress, agitation, anxiety, confusion, dry mouth, fatigue, parasthesiaes, vertigo, hypomania, diarrhoea, insomnia, sweating and nervousness. Clinical experience with milnacipran is limited and a withdrawal syndrome does not appear to be an issue.
Conclusions
Studies indicate that dual action antidepressants may bring further tolerability and efficacy advantages compared to the SSRIs and the tricyclics in the treatment of moderate to severe major depression. Some experts still regard tricyclics as most useful for the more severe and melancholic subtype of depression [46–48]. However, this issue is controversial, especially with the availability of the dual action antidepressants.
Adverse events are an important consideration in treatment adherence, especially in long-term therapy, as patients when feeling well are reluctant to continue with medicines that cause them problems. Most SSRIs and venlafaxine cause sexual dysfunction and do not cause significant weight gain, whereas dual action mirtazapine results in weight gain in a proportion of patients without causing significant sexual dysfunction. An effective drug free of side-effects and toxicity should facilitate that process but none is yet available.
Emerging areas of research in the treatment of depression indicate that there is genetically determined variability in serotonergic gene expression which influences complex behaviour leading to increased anxiety depression and aggression. Pharmacogenetics are bound to target novel strategies aiming toward modification of such serotonergic function [49–51].
Finally, Cassano [52] and colleagues have shown that serious changes to the individual personality take place (described as ‘postaffective personality’) and often complicate the course of recurrent mood disorders. Therefore, treatment of depression is not merely a matter of medication. Paykel and Priest [53] emphasize that antidepressants should be only part of the approach and that a programme of treatment should be negotiated with the patient, whose choice is taken into account. Combined with antidepressants, psychotherapeutic interventions may also improve outcome, as would social rehabilitation [54].
