Abstract
The costs of alcohol dependence to the community are substantially reduced with treatment [1]. A number of treatment approaches with different treatment philosophies have been successfully applied to this chronic relapsing condition [2]. One of these, cognitive–behavioural therapy (CBT) has been effectively used in the treatment of alcohol dependence [2–4]. Cognitive–behavioural therapy is a problem-focused psychotherapeutic approach based on learning theory. It involves identification of the reinforcement processes and dysfunctional information processing, which shape and maintain maladaptive behaviour. Treatment is directed primarily at modifying distorted or maladaptive cognitions and altering environmental contingencies to diminish behavioural dysfunction. In alcohol dependence, a central tenet of CBT is that alcohol abstinence engenders a sense of personal control and selfefficacy that strengthens as abstinence continues. The identification and response to ‘high-risk’ alcohol relapse situations with the development of effective coping behaviour is intended to decrease the likelihood of return to alcohol use and has been termed, relapseprevention [5, 6]. Although Marlatt's taxonomy of relapseprecipitants [5] has not been supported by longitudinal study, prospective clinical research has identified components associated with relapse. These include positive/negative emotional states, social pressure, interpersonal conflict and craving [7].
The search for pharmacological treatments for alcohol dependence, until recently, has generally been disappointing. Evidence, after over 50 year's use, that the aversive agent disulfiram increases the proportion of patients who achieve alcohol abstinence is lacking [8]. Patients with alcohol dependence and comorbid major depression may benefit from selective serotonin reuptake inhibitors (SSRIs) [9]. This does not extend to those without depression [10, 11]. Similarly, patients with comorbid anxiety have reduced their alcohol use withbuspirone [12]. The role of the anticonvulsant medication, carbamazepine although promising, remains undefined [13]. Calcium acetylhomotaurinate (Acamprosate), a GABA analogue, has achieved widespread use in Europe. It has been demonstrated both an effective and safe adjunct to treatment in alcohol dependence [14, 15].
Clinical studies with naltrexone, a long-acting opiate antagonist, have shown both a reduction in the number of drinking days and an increase in rates of continuous alcohol abstinence [16, 17]. In a follow-up study [18], efficacy of treatment 6-months postintervention [17] showed that although naltrexone subjects (as opposed to placebo) were less likely to have relapsed, this effect did diminish over the follow-up period. A more recent study has reported the benefit of naltrexone in conjunction with weekly CBT [19]. Opiate dysregulation in animal and human studies has been implicated both in the preference for alcohol intake and craving. Alcohol use is reinforced by enhanced opiate receptor activity [20]. In Australia, on 1 February 2000, naltrexone was listed on the Pharmaceutical Benefits Scheme (PBS). The current study reports the comparative efficacy of adding naltrexone to an established CBT rehabilitation programme. While the efficacy of naltrexone in clinical trials is sound there is little research to date examining the impact of the medication in more typical outpatient settings. The current study aimed to examine the impact of adding naltrexone to an outpatient, 12-week; ‘contract’-based programme with the primary aim of alcohol abstinence. The naltrexone-treated patients are compared with historical matched controls.
Method
Naltrexone was unregistered in Australia at the time of introduction. Authorization to prescribe naltrexone was granted undersubsection 19(5) of the Therapeutic Goods Act 1989.
Subjects
Naltrexone (naltrexone + CBT) subjects
Entry criteria for the naltrexone (naltrexone + CBT) group included patients aged over the age of 18 years with alcohol dependencewith 3–30 days of alcohol abstinence. Exclusion criteria included current dependence on substances other than alcohol (except nicotine), previous naltrexone or current disulfiram, opioid or psychotropic medication use (other than benzodiazepines) and bMAST (Brief Michigan Alcoholism Screening Test) scores of < 6 (range = 0–29; scores ≥6 associated with a high likelihood of alcohol dependence). Exclusions included current suicidal ideation, psychosis, medical instability or elevated (2 × normal) bilirubin or hepatic transaminases.
From July 1998, 53 consecutive patients fulfilling the DSM-IV criteria for the diagnosis of alcohol dependence were prescribed naltrexone. This was an adjunct to an established outpatient ‘contract’- based abstinence CBT rehabilitation programme implemented over the previous decade. Some patients had recently been detoxified, usually as an outpatient under the care of their general practitioner. All had achieved alcohol abstinence for at least 3 days. Exclusions from the analysis included one woman initially elected to participate but who later withdrew before taking naltrexone. Two other women withdrew, one after 1 weeks' treatment because of dysphoria and the other transferred to the private sector soon after naltrexone was introduced.
Control (CBT) subjects
A sample of 734 historical controls offered the same ‘contract’- based abstinence programme and delivered by the same group of therapists was screened on identical exclusion criteria as the naltrexone group. This resulted in a sample of 301 subjects. From these subjects, each naltrexone patient was individually matched (blind to any personal identifiers and outcome measures) to a control subject on the basis of: (i) gender (male/female); (ii) previous drug or alcohol rehabilitation treatments undertaken (yes/no); (iii) if alcohol detoxification was undertaken immediately prior to commencing treatment (yes/no); (iv) usual alcohol consumption (average g/day) categories (male < 80 g; female < 60 g; male ≥ 80 g; female ≥ 60 g); (v) age categoriesof 5-year increments (e.g. 20–24, 25–29)
Forty of the 50 control subjects were matched on the above criteria. Where there was more than one possible match within the control sample, a random selection option was employed by the statistical program used (SPSS v9, SPSS, Chicago, IL, USA). The 10 remaining naltrexone subjects were matched on criteria one to four. From this selection, the control with the closest age to the naltrexone subject was matched.
The mean age of the naltrexone subjects was 43 ± 11 and for the controls 42 ± 10, demonstrating that the subjects' age did not differ significantly between groups (t = 4.10, df = 1,98, p = 0.682). Patients in both programmes also reported similar for usual alcohol consumption (t = −1.339, df = 1,98, p = 0.184), maximum alcohol consumption (t = 0.429, df = 1,92, p = 0.669), age of onset of problem drinking (t = 0.819, df = 1,98, p = 0.415), years of use (t = −0.018, df = 1, 98, p = 0.986) and bMAST scores (t = 0.223, df = 1, 98, p = 0.824). These data are reported in Table 1.
Drinking and treatment parameters by treatment group
Design
A non-randomized historical cohort comparison between current patients (receiving Natrexone + CBT) and former patients (receiving CBT alone) was conducted. This design is typically utilized in the early development of a treatment, or in examining the use of a treatment of established efficacy in a new setting [21, 22]. While claims of treatment efficacy based entirely upon historical controls are questionable [23] given that the efficacy of naltrexone is well established, a historical control design was considered appropriate. The current study examines the impact of naltrexone outside of a research setting. It is acknowledged that this design is likely to underestimate modest treatment effects and that there is less experimental control than with a randomized controlled trial [24].
Pharmacological and cognitive behavioural therapy treatment
Following informed consent all patients in the naltrexone group were prescribed naltrexone 50 mg orally daily. Prescriptions were collected before each scheduled therapy session. The abstinencebased treatment programme contains defined cognitive and behavioural components conducted by Masters-level clinical psychologists. Comprehensive supervision and training in line with the treatment manual is provided to each therapist by the senior psychologist (RMcDY) who has been involved in the CBT programme since inception. It includes weekly therapy sessions lasting 1 h for thefirst 4 weeks. Patients are seen fortnightly for the subsequent 4 weeks, completing eight sessions over the 3 months of the programme. Three further appointments at 3-monthly intervals were offered tocomplete 12 months of review. All subjects received this programme.
Alcohol abstinence was estimated by a combination of factors including patient self-report, clinic visit alcohol breathalysation and programme compliance. In this we chose a high threshold of 12 weeks complete abstinence, rather than a return to previously harmful (pretreatment) drinking, to define successful programme completion.
Data analysis
Data analysis was performed using SPSS version 9.0. Log rank analyses were performed to compare programme attrition and relapse across treatment groups over the duration of the programme. Chisquared analyses were employed to compare the final outcome measures (i.e. in week 12) of attendance and abstinence between the two treatment groups.
Results
Attendance over programme duration by treatment group
Attendance across each of the eight treatment sessions was recorded for each patient. If the patient withdrew, the week of withdrawal was noted. Log rank analysis investigating the attrition across the programme by treatment group identified significantly higher attrition in the control group. (Log rank = 31.22, df = 1,98, p < 0.001)
Abstinence over programme duration by treatment group
Patient's self-reports and blood alcohol level screening at each session were employed to determine abstinence for control samples. If a relapse occurred, the week of the relapse was noted. The patients retained the choice to continue the programme or withdraw. If a patient withdrew from the treatment without advising clinical staff, this was recorded as a relapse. The log rank analysis showed that patients in the control group relapsed sooner and more frequently across the programme duration than did patients in the naltrexone group (Log rank = 45. 90, df = 1,98, p < 0.001).
A Kaplan–Meier survival curve (Fig. 1) shows comparative cumulative proportion of subjects by treatment group withdrawing from the programme and those relapsing during the 12-week abstinence-based programme.
The comparative cumulative proportion of subjects withdrawing and those relapsing duringthe 12-week abstinence-based programme by treatment group. ▴, naltrexone attendance; •, control attendance; △, naltrexone abstinence; ○, control abstinence.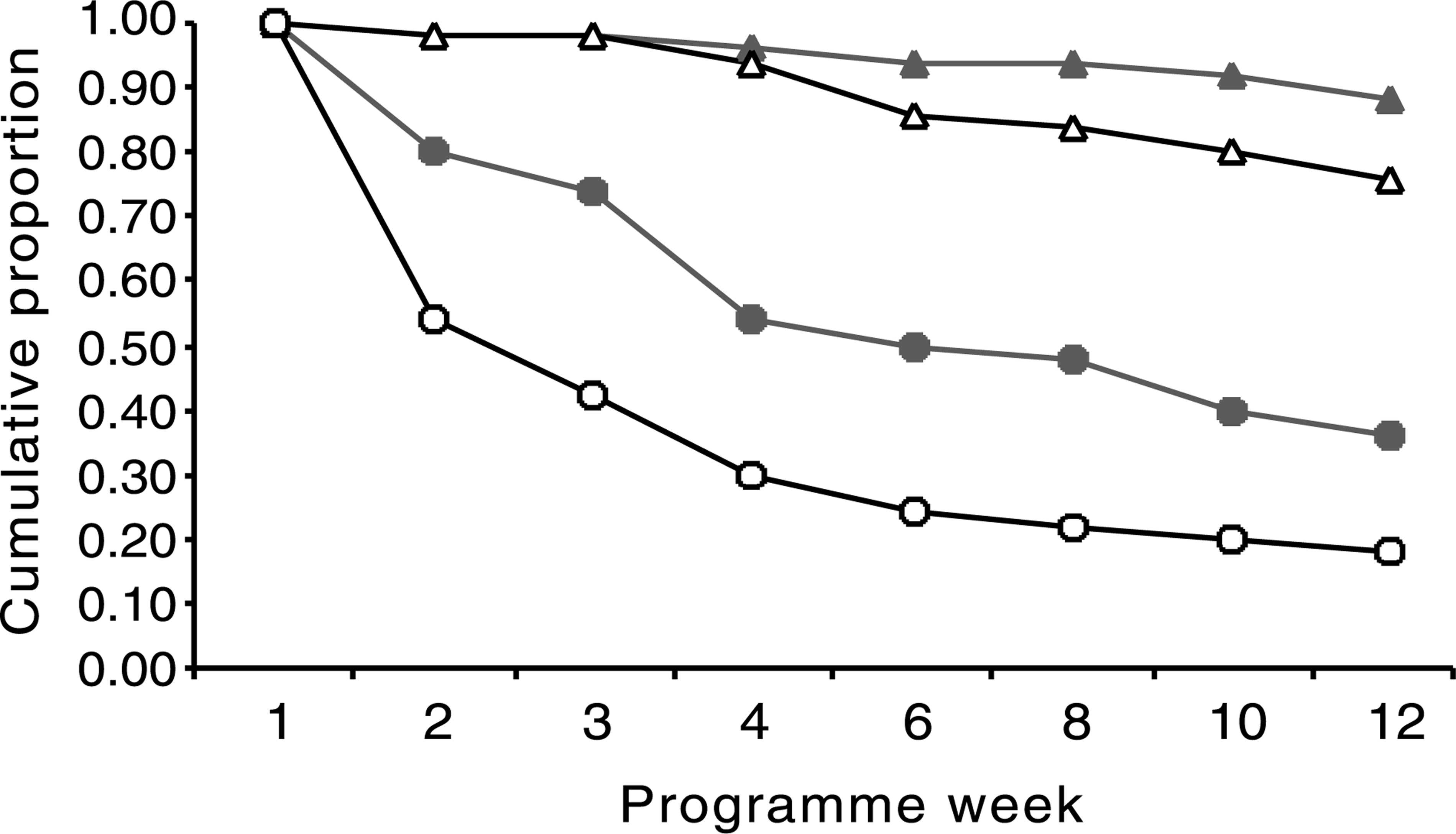
Programme completion by treatment group
The proportion of patients completing the 12-week programme by treatment group as defined by their attendance at each scheduled follow-up session was determined by χ2 analysis. This ratio was 44/50 (88%) in the naltrexone + CBT group and 18/50 (36%) in the CBT group. The difference in proportions between the two groups was significant (χ2 = 28.69, df = 1,98, p < 0.001).
Programme abstinence by treatment group
The proportion of patients within the two treatment groups that remained abstinent across the 12-week programme was also determined by χ2 analysis. The abstinence ratio was 38/50 (76%) in thenaltrexone + CBT group and 9/50 (18%) in the CBT group. The difference in proportions between the two groups was significant (χ2 = 33.76, df = 1,98, p < 0.001).
Figure 2 shows the number of patients in the naltrexone or control groups that attended all sessions and those that completed abstinent.
The proportion of patients in the naltrexone or control groups that attended all sessions and those that completed the program abstinent. □, abstainers; ▪, non-abstainers.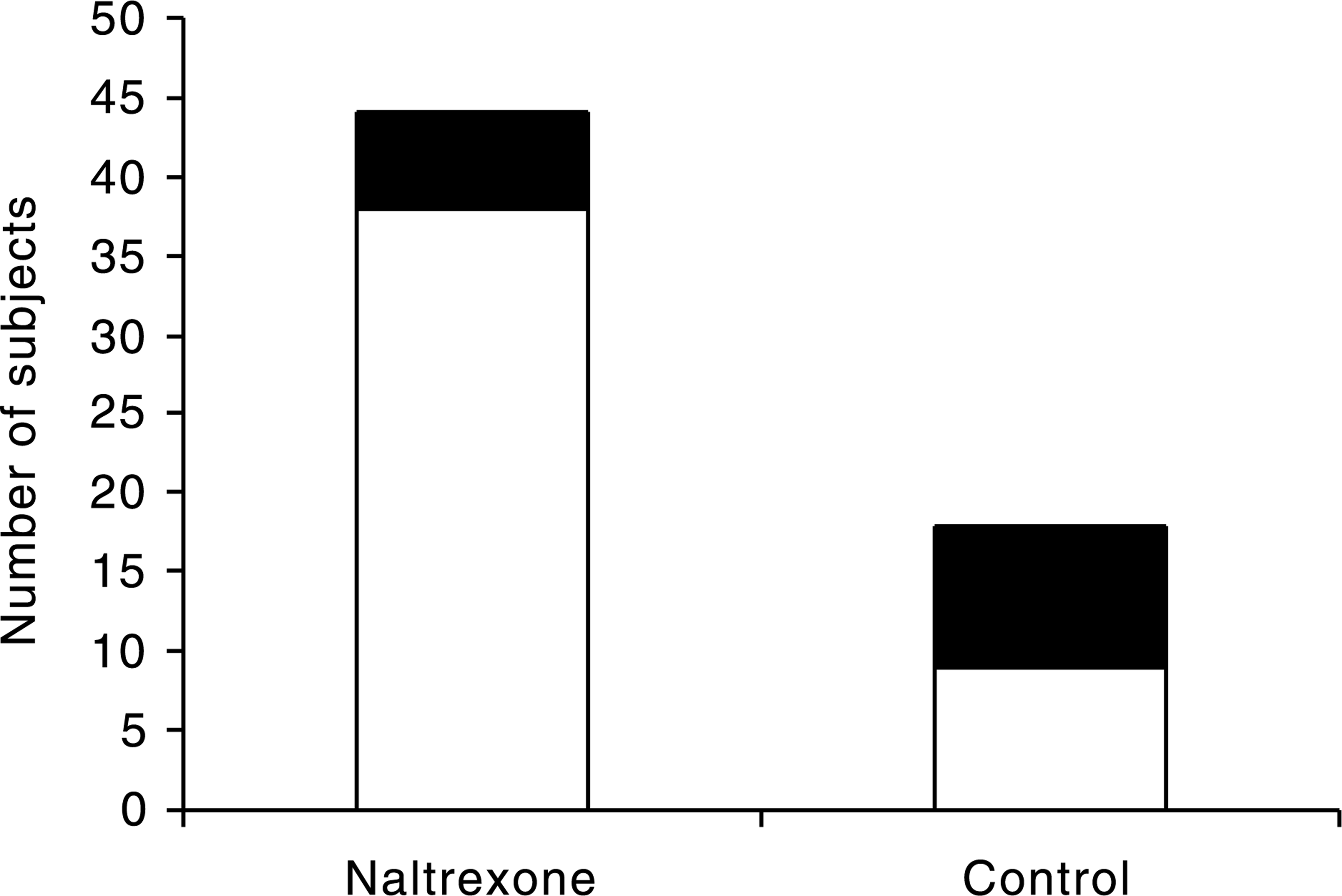
Discussion
Following the initiation of abstinence in patients with alcohol dependence, relapse is the most common outcome. After alcohol detoxification, relapse (without treatment) to any alcohol use at 12 weeks follow up has been reported at 76% [25]. Cumulative relapse rates for patients treated for alcohol dependence have been reported as 35% at 2 weeks and 58% at 3 months following treatment [26]. In the two original naltrexone, placebocontrolled clinical trials, the placebo groups both reported high relapse rates at 12 weeks (54% and 79%) [16, 17]. Where psychosocial treatments have been evaluated, short-term relapse rates remain high. In project MATCH, CBT, 12-step facilitation (TSF) and motivational enhancement therapy (MET) were randomized and by the end of the 12-week treatment phase, 59% of the CBT/TSF groups and 72% of the MET group had returned to problem drinking [27]. The relapse rate when CBT was the only therapy offered was 67% in the placebo arm compared with 53% in the naltrexone-treated group [19]. Although on current evidence there appears to be little difference in outcome by type of treatment [2], the addition of the anticraving medication, naltrexone, has the potential to improve outcomes across treatments [16, 17].
In this study we assessed the influence of the addition of naltrexone to an established CBT programme where alcohol abstinence was the primary therapeutic goal. All these patients had alcohol dependence, long drinking histories and were unsuitable for drinking reduction approaches. Alone, CBT (control) outcomes at 3 months had high attrition rates with approximately one-third of patients completing the programme. Completion rates are more than doubled with naltrexone use. Differences were more pronounced with measures of alcohol abstinence. The magnitude of this difference is considerable (by a factor of four) and occurs without programme change other than the addition of an anticraving medication. In this case naltrexone has contributed substantially to improving treatment effectiveness. Attention to this agent followed two randomized controlled trials, of different design, both of which reported significant benefit in outcome with naltrexone use [16, 17].
This study differs as it uses a historical control group. This is predicated on the assumption that factors relevant to successful outcome closely approximate in both groups. We used multiple indicators and provided the best match between individuals on these indices. We included reported predictors of abstention in alcohol dependence (age, age of onset, gender, past rehabilitation) [28]. However, historical controls may be misleading. There are a number of potential sources of bias favouring the naltrexone-treated group. It is possible that the control group contained more impaired patients, had shorter abstinence periods before beginning rehabilitation and may have been at higher risk of relapse. Over time, increasing staff expertise in delivering CBT may have benefited the most recently treated (naltrexone) group. As this was the only regional hospital rehabilitation unit with access to naltrexone, patients aware of this and self-referral may reflect a more motivated and capable patient group. In addition, therapist confidence, expectation and perception may have been altered by the presence of naltrexone and could have influenced treatment outcomes.
Recognizing that abstinence is just one measure of positive outcome, our results with naltrexone are generally better than currently reported [16, 17, 19]. Overall these findings confirm the trend in recent studies that naltrexone is a useful adjunct to CBT treatment for alcohol dependence and its use should be encouraged.
