Abstract
This double-blind, placebo-controlled, parallel-group, multicentre, multinational, phase-III trial was designed to assess the efficacy and safety of a single subcutaneous injection of placebo, 2 doses of alniditan (1.4 mg and 1.8 mg) and 6 mg of sumatriptan in subjects with acute migraine. A total of 114 investigators from 13 different countries screened 2021 subjects. In total 924 patients were treated with placebo (157), alniditan 1.4 mg (309), alniditan 1.8 mg (141) and sumatriptan 6 mg (317). The lower number of subjects in the alniditan 1.8 mg group is due to the termination of this trial arm after the incidence of a serious adverse event and a subsequent protocol amendment. The number of subjects who were pain free at 2 h (primary endpoint) was: 22 (14.1%) with placebo, 174 (56.3%) with alniditan 1.4 mg, 87 (61.7%) with alnditan 1.8 mg and 209 (65.9%) with sumatriptan 6 mg. Alniditan 1.4 mg was significantly better (P < 0.001) than placebo and sumatriptan was significantly better (P = 0.015) than alniditan 1.4 mg. The number of responders (reduction of headache severity from moderate or severe headache before treatment to mild or absent at 2 h), was 59 (37.8%) on placebo, 250 (80.9%) on alniditan 1.4 mg, 120 (85.1%) on alniditan 1.8 mg, and 276 (87.1%) on sumatriptan. Response was significantly higher (P < 0.001) with alniditan 1.4 mg than with placebo, and significantly lower (P = 0.036) with alniditan 1.4 mg than with sumatriptan. Recurrence rates were: 22 (37.3%) with placebo, 87 (34.8%) with alniditan 1.4 mg, 35 (29.2%) with alniditan 1.8 mg and 108 (39.1%) with sumatriptan. Adverse events occurred in 577/924 (62.4%) subjects, i.e. in 62/157 (39.5%) with placebo, 214/309 (69.3%) with alniditan 1.4 mg, 91/141 (64.5%) with alniditan 1.8 mg and 210/317 (66.2%) with sumatriptan 6 mg. Sumatriptan was significantly better than alniditan 1.4 mg for pain free at 2 h. The difference, however, was small and clinically not important. For alniditan, a dose-dependent adverse event relationship was seen. The safety profile of alniditan 1.4 mg was similar to that of sumatriptan.
Introduction
Alniditan is a specific and powerful nonindole 5-HT1B/1D receptor agonist (1). In animal studies, alniditan causes a selective vasoconstriction in the canine carotid vascular bed (2), and alniditan selectively constricts porcine carotid arteriovenous anastomoses via 5-HT1B/1D receptors (3). In addition, alniditan attenuates neurogenic inflammation in the rat dura mater, most likely by acting on prejunctional receptors (4, 5). Alniditan is a more potent vasoconstrictor than sumatriptan in the canine carotid vascular bed (2), and is more potent than sumatriptan in attenuating neurogenic inflammation (4). Subcutaneous alniditan is quickly absorbed with peak plasma concentrations generally attained within 15 min, and the terminal elimination half-life is in the order of 8–13 h (6) (data on file JRF).
Sumatriptan, the first 5-HT1B/1D receptor agonist, undoubtedly is a significant advance in migraine therapy (7–11), and subcutaneous sumatriptan (12–14) is the most effective form of administration of sumatriptan with a therapeutic gain (percentage response for active drug minus response for placebo) of 51% after 1 h (15) for headache response, a decrease from severe or moderate headache to none or mild (16). Despite its great utilities in migraine treatment, subcutaneous sumatriptan has certain limitations. Patients want to be pain free after treatment of migraine attacks (17) and only approximately one half of patients, 52% (95% confidence intervals: 49% to 55%) (12–14), became pain-free by 2 h after treatment with subcutaneous sumatriptan. In addition, recurrence of headache occurs in up to 40% of patients who initially respond to subcutaneous sumatriptan (18, 19). In a placebo-controlled, dose-finding, phase-II study with subcutaneous alniditan in doses of 0.8, 1.0, 1.2, and 1.4 mg (6) all doses were superior to placebo, and there was evidence of a dose-dependent increase in patients who became pain-free. Therefore a comparative trial with sumatriptan as the comparator was designed.
Methods
This was a randomized, double-blind, placebo-controlled, multinational, multicentre, phase III trial using a parallel-group design, which evaluated 2 dose levels of alniditan (1.4 mg and 1.8 mg) and 1 dose of sumatriptan (6 mg) vs. placebo. Subjects received one single treatment. Subjects' eligibility for the trial was ascertained in a pretrial screening visit (visit 1). Those meeting the entrance criteria at the screening were asked to report to the investigator twice: once, as soon as possible after the start of their next migraine attack for the treatment visit (including a 2-h observation period – visit 2) and a second time for the follow-up visit (visit 3) after the treatment. Informed consent had to be obtained prior to any trial specific procedure. Subjects who presented at the clinic (visit 2) with a moderate to severe migraine headache were (if it had been ascertained that the treatment criteria were satisfied) randomly allocated to one of the four treatment groups (a subcutaneous injection of 1.4 or 1.8 mg of alniditan, 6 mg of sumatriptan or placebo). The trial was approved by the ethics committees of all participating centres. All patients gave informed consent.
Subjects were discharged from the clinic at the earliest 2 h after the trial drug administration (the observation period). During this period the headache severity and presence or absence of associated symptoms was scored at specific time points. In addition, cardiovascular safety parameters were recorded. Starting at 2 h after the trial drug administration, subjects were allowed to take rescue medication, if needed. However, sumatriptan or ergot derivatives were not allowed until 24 h after dosing. From the time of discharge from the clinic until 48 h after trial drug administration, the subjects had to keep a headache card on which they had to specify the severity of their headache, associated symptoms, recurrence of migraine headache, any untoward effect experienced and the use of concomitant medication. They completed the headache card and returned it to the investigator at the follow-up visit. Subjects were contacted by the investigator or his/her designee between 24 and 48 h after treatment, to ensure that they had completed the headache card. The follow-up visit (visit 3), at which the headache card was to be discussed with the subject, was scheduled no later than 2 weeks after the treatment. In August 1996, following three reports of chest pain with ischaemic ECG-changes after 1.8 mg alniditan (two in another trial), it was decided to withdraw the 1.8 mg dose from this trial. Inclusion criteria were as follows: migraine with or without aura, as defined by the International Headache Society diagnostic criteria (20), at least a 6-month history of 1–6 migraine attacks per month, between 18 and 65 years of age, in good health and physical condition as determined by medical history, physical examination, vital signs, ECG and laboratory evaluation, written informed consent. Before dose administration the following inclusion criteria were applied: headache identified as migraine with at least one of the following symptoms: nausea, vomiting, photophobia, phonophobia, severity of the headache was moderate or severe, time lapse between the onset of the migraine attack and the sc injection of the trial medication had to be ≤ 8 h. Exclusion criteria were: Inability to differentiate between migraine headaches and other types of headaches if other headache types coexisted, a pattern of headache-free intervals of less than 24 h, history or presence of a significant cardiovascular disorder, history or presence of a neurological disorder other than migraine (including subarachnoidal bleeding, stroke or brain tumour), or a clinically significant psychiatric disorder, other serious diseases, including hepatic, renal, gastrointestinal, pulmonary, metabolic or endocrine disorders. Subjects on long-term prophylactic migraine therapy with methysergide, tricyclic antidepressants, MAO inhibitors were excluded (prophylactic therapy with flunarizine, pizotifen or beta-blockers started before the trial was not a reason for disqualification). Other exclusion criteria were: regular use (> 10 days per month) of medication for the treatment of the acute migraine attack (ergotamine or ergot derivatives, sumatriptan, aspirin, NSAIDs), history or suspicion of drug or alcohol abuse, pregnancy or breast-feeding in women, absence of adequate contraception (e.g. oral contraception or IUD) in female subjects with child-bearing potential, subjects with a known hypersensitivity to sulphonamides, subjects who have used any treatment for the present attack. In migraine attacks with aura, trial medication could only be given, when the aura symptoms had resolved.
Statistics
The primary objective of this trial was to compare the proportion of subjects who were pain free at 2 h after treatment with alniditan 1.4 mg and 1.8 mg sc to that with sumatriptan 6 mg sc and placebo. Since the effect sizes were expected to be different between alniditan vs. sumatriptan and alniditan vs. placebo, the sample size calculation was based on two separate procedures. Both procedures used Bonferroni adjusted alpha (0.0125) assuming four primary comparisons (alniditan 1.4 mg vs. sumatriptan 6 mg, alniditan 1.8 mg vs. sumatriptan 6 mg, alniditan 1.4 mg vs. placebo, and alniditan 1.8 mg vs. placebo). The sample size calculation for the three active treatment groups was based on the expected treatment difference between the best alniditan group and the sumatriptan group. The sample size calculation for the placebo group was based on the expected treatment difference between the best alniditan group and the placebo group.
If it is assumed that the complete response rate would be 72% for the best alniditan group and 55% for the sumatriptan 6 mg group, the required sample size to show superiority, with alpha=0.05/4 = 0.0125 and 80% power, was 189 subjects for each active treatment group. Similarly, assuming the complete response rate would be 72% for the best alniditan group and 40% for the placebo group, the required sample size, with alpha=0.05/4 = 0.0125 and 80% power, would be 58 subjects for the placebo group. To balance the randomization, a total of 665 subjects (i.e. 190 in the active treatments and 95 in placebo) were required in this trial. All statistical tests were interpreted at the 5% significance level (2-tailed), unless specified otherwise. All randomized subjects were included in the analysis of safety, demographic, and baseline characteristics data. Analysis of efficacy and quality of life data were based on the intent-to-treat population (ITT) defined as all randomized subjects who took at least one dose of double-blind trial medication and who provided any follow-up data for one or more key efficacy variables (headache intensity, associated symptoms, and functional ability); in turn, ITT subjects are allocated to treatments actually received. The comparability between the treatment groups was evaluated with respect to the demographic and baseline variables. For continuous variables (e.g. age, weight, etc.), a two-way analysis of variance (
The primary efficacy parameter was the number of subjects with who were pain free at 2 h after sc injection. Pain free was defined as a reduction in headache intensity from score 3 or 2 prior to therapy to score 0 (no pain) at 2 h after treatment. The incidence rate in each of the alniditan groups was compared to the incidence rate in the sumatriptan group and the placebo group by means of a Cochran-Mantel-Haenszel test for general association controlling for country. In addition, the Breslow-Day test for homogeneity of odds ratios across countries was executed. Subsequently the Fisher's exact probability test was performed. All other parameters were regarded as secondary parameters: with the number of subjects required in this trial a significant difference between alniditan and sumatriptan was also expected with respect to the number of subjects with recurrence. Recurrence was defined as an increase in headache severity from 0 or 1 at 2 h to 2 or 3 within 2–24 h after treatment. Additional secondary outcome parameters were: proportion of subjects pain free at 2 h and without deteriorating within 24 h after drug administration, proportion of responders at each efficacy evaluation time point after drug administration (response is defined as a reduction in headache severity from score 3 or 2 prior to therapy to score 0, no pain or score 1, mild pain after treatment), effect on associated symptoms (nausea, vomiting, phono- or photophobia), effect on functional ability, time to onset of effect of trial medication (time lapse till a decrease in headache severity score by one), proportion of subjects using rescue medication, duration of response (difference in time between the onset of the effect and recurrence of symptoms to moderate or severe pain), quality of life assessment (15 items across 5 clusters: work functioning, social functioning, energy/vitality, migraine symptoms, feeling/concerns), reuse of trial medication. For the nominal parameters (e.g. event rates), a Cochran-Mantel-Haenszel test for general association controlling for country was applied. The Breslow-Day test for homogeneity of odds ratios across countries was also executed. Additionally, a Fisher's exact probability test was performed for the comparison between alniditan and sumatriptan and between alniditan and placebo. For ordinal categorical variables, treatment groups were compared using the Van Elteren test, controlling for country.
Results
The trial was run from March 1996 to January 1997. A total of 114 investigators from 13 different countries screened 2021 subjects. Of these, 924 received treatment: 157 were treated with placebo, 309 with alniditan 1.4 mg, 141 with alniditan 1.8 mg and 317 with sumatriptan 6 mg. Three subjects dropped out before trial completion. One subject on alniditan 1.4 mg and one on sumatriptan 6 mg were lost to follow-up, whilst one on placebo was noncompliant. Apart from early withdrawals, described above, protocol deviations were noted in 16 subjects. All protocol deviations were related to the selection criteria. More in particular, rescue medication was taken by 10/16 subjects within 2 h after drug administration (4 placebo, 3 alniditan 1.4 mg, 1 alniditan 1.8 mg, 2 sumatriptan 6 mg).
The majority (86.4%) of the 924 treated subjects were female, and the ages in the four groups ranged from 18 to 64 years (median: 41 years). The usual migraine attack was initiated by an aura in 35.6% of all subjects, pain unilaterally located in 86%, of pulsating quality in 85%, aggravated by routine physical activity in 92% and associated by symptoms such as photo- and phono-phobia, nausea and vomiting in 100%. Migraine had started at the median age of 18 years and the median duration of a migraine attack was 48 h. The subjects had on average 3 attacks a month. Demographic data and baseline disease characteristics were not significantly different in the 4 groups, except for unilateral location of headache (P = 0.024), the incidence being the highest in the alniditan 1.4 mg group (90.6%) and the lowest in the sumatriptan 6 mg group (82.3%). The current migraine attack was initiated by an aura in 14.2% of the subjects. Headache severity was moderate in 57.3% of the patients treated with placebo, 55.3% with 1.4 mg alniditan, 56.7% with 1.8 mg alniditan and 53% with sumatriptan. The median time elapsed between the onset of the current attack and the sc injection was 4 h. Concomitant treatments were taken by over 60% of each treatment group. Conjugated oestrogens were the most frequently reported concomitant medications.
At baseline, headache was moderate to severe in all but one subject, who was therefore excluded from the analysis. There were no baseline differences between the four groups. The proportion of patients who became pain free, i.e. a reduction in headache severity from moderate or severe before treatment to absence of headache at 2 h after drug administration, is given in Table 1. Alniditan 1.4 mg was significantly better than placebo, whilst sumatriptan was significantly better than alniditan 1.4 mg. Already at 15 min, headache was mild or absent in 12 (7.6%) subjects on placebo, 65 (21%) on alniditan 1.4 mg, 36 (25.5%) on alniditan 1.8 mg and 85 (26.8%) on sumatritan 6 mg. At this time point, both doses of alniditan were significantly better (P < 0.001) than placebo, whilst the difference between alniditan and sumatriptan was not significant. Differences between both doses of alniditan and placebo remained significant up to 12 h after the sc injection. Differences between alniditan 1.4 mg and sumatriptan, in favour of sumatriptan, were seen at 2 h (P = 0.008) and 4 h (P = 0.013).
Patients who were pain free at 2 h (ITT population)

The response rate at different time intervals is summarized in Table 2. Response was defined as a reduction in headache severity from moderate or severe before treatment to mild or no pain after drug administration. Alniditan 1.4 mg was significantly better than placebo from 15 min up to 12 h after the sc injection, whilst sumatriptan was significantly better than alniditan 1.4 mg at 2 h. The median time to response was 4.0 h in the placebo group and 0.5 h in the three other groups. The median time to onset of effect, defined as a lowering of the headache severity score by 1, was 1.5 h in the placebo group and 0.5 h in the three other groups. The number (%) of subjects with 24 h pain free, defined as absence of headache from 2 h up to 24 h after injection, irrespective whether the subject took any rescue medication or not, were 8 (5.1%) with placebo, 83 (26.9%) with alniditan 1.4 mg, 43 (30.5%) with alniditan 1.8 mg and 78 (24.6%) with sumatriptan. The difference between alniditan 1.4 mg and placebo was significant (P < 0.001). There was no difference between alniditan 1.8 mg and sumatriptan. The numbers (%) of responders at 2 h with recurrence within 24 h after trial drug administration were 22 (37.3%) with placebo, 87 (34.8%) with alniditan 1.4 mg, 35 (29.2%) with alniditan 1.8 mg and 108 (39.1%) with sumatriptan. No intergroup differences were seen. The median time to recurrence could not be calculated because the number of responders with recurrence within the interval set was too low. The numbers (%) of subjects using rescue medication within 24 h after trial drug administration were 123 (78.3%) with placebo, 142 (46.0%) with alniditan 1.4 mg, 65 (46.1%) with alniditan 1.8 mg and 155 (48.9%) with sumatriptan. There was a significant difference between alniditan 1.4 mg and placebo (P < 0.001), in favour of alniditan. No difference was seen between alniditan 1.9 mg and sumatriptan. Of the responders at 2 h, 36/59 (61.0%) subjects on placebo, 91/250 (36.4%) on alniditan 1.4 mg, 46/120 (38.3%) on alniditan 1.8 mg and 120/276 (43.5%) on sumatriptan took rescue medication within 24 h after the sc injection.
Response rate (ITT population)

The evolution of the associated symptoms (nausea, vomiting, photo- and phono-phobia) is given in Table 3. All subjects reported at least one symptom at the start of the trial. After one hour, the presence of associated symptoms had lowered to less than 50% in all treatment groups except in the placebo group, and had lowered further to approximately 25% (except again in the placebo group) at 2 h. There was a significant difference between alniditan 1.4 mg and placebo, but not between alniditan 1.4 mg and sumatriptan. As can be seen in Table 4, the number of subjects whose functional ability was severely impaired or who required bedrest was greatly reduced from before treatment to one hour after treatment. No significant differences were seen in the scores of the clusters of the migraine quality of life questionnaire between alniditan 1.4 mg and sumatriptan.
No. of subjects whose functional ability was severely impaired or who required bedrest (ITT population)
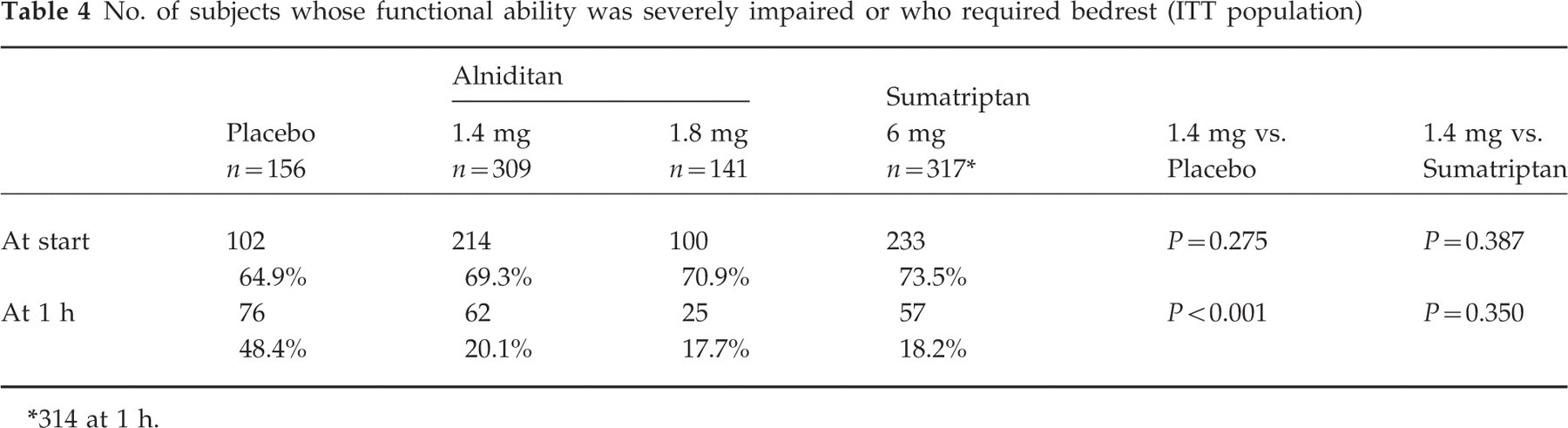
314 at 1 h.
Presence of any associated symptom (ITT population)

Safety
Adverse events occurred in 577/924 (62.4%) subjects and related to treatment groups in 62/157 (39.5%) with placebo, 214/309 (69.3%) with alniditan 1.4 mg, 91/141 (64.5%) with alniditan 1.8 mg and 210/317 (66.2%) with sumatriptan 6 mg. The most frequently reported adverse events are listed in Table 5. Chest symptoms were more common with alniditan, application site reactions more common with sumatriptan. On the whole, most AE were mild or moderate. The number of subjects of whom the most important AE were reported as severe are given in Table 6. Two serious adverse events were reported. In one subject, belonging to the placebo group, a myometrial increase and a uterine fibroid were reported. One subject, who had no prior history of coronary heart diesease in the alniditan 1.8 mg group reported chest pain. A cardiological examination revelaed a coronary artery disorder. Neither alniditan nor sumatriptan had effects on heart rate and blood pressure, or ECG. No consistent changes in blood chemistry or haematology were observed. In summary, for alniditan, a dose-dependent adverse event relationship was seen. The safety profile of alniditan 1.4 mg was similar to that of sumatriptan in this trial.
Adverse events reported as severe

Total no. of subjects reporting this AE.
Adverse events occurring in > 5% of all subjects

Discussion
For pain free at 2 h, alniditan 1.4 mg was significantly better than placebo, whereas sumatriptan was significantly better than alniditan 1.4 mg. The same was true for headache response. The difference, however, was small and clinically not important. The objectives of the trial (higher efficacy and lower recurrence rate for alniditan vs. sumatriptan) were not met. For alniditan, a dose-dependent adverse event relationship was seen. The safety profile of alniditan 1.4 mg was comparable to that of sumatriptan. In an earlier dose-finding trial of sc alniditan (6) a higher rate of headache free patients (72%) were observed with 1.4 mg alniditan than in this trial (56%). The fact that 1.4 mg alniditan was inferior to 6 mg sumatriptan and that the 1.8 mg dose of alniditan was not further developed due to a few serious adverse events resulted in termination of the study programme. One important pharmacological aspect is the fact, that alniditan has almost no activity at the 5-HT1F receptor. This indicates that the 5-HT1F receptor plays no crucial role in the treatment of acute migraine attacks.
Footnotes
Acknowledgements
The study was supported by a grant from Janssen Research Foundation, Belgium
