Abstract
Disodium 2,4-sulphophenyl-N-tert-butylnitrone (NXY-059) is a novel free radical-trapping compound that is neuroprotective in both rodent and primate models of acute ischaemic stroke. Neuroprotection in vitro by NXY-059 has not been reported previously, and we have now investigated whether such an effect can be detected using a simple cell culture model of neurotoxicity. Neuron-like cells of the neuroblastoma-derived clonal cell line N1E-115 were exposed to the free radical-generating agent sodium nitroprusside (SNP), which produced a concentration-dependent reduction in mitochondrial complex II activity 24 h later (EC50 approximately 100 micromolar). Cell death induced by SNP (100 micromolar), assessed either by an increased proportion of apoptotic nuclear morphology or by mitochondrial complex II activity, was inhibited by a cocktail of known antioxidants (ascorbate, reduced glutathione, and dithiothreitol, all at 100 micromolar) but not by NXY-059 at a concentration known to be neuroprotective in vivo (300 micromolar). Disodium 2,4-sulphophenyl-N-tert-butylnitrone was also without effect on H2O2-mediated cytotoxicity. These results support recent data suggesting that in vivo NXY-059 probably acts at the neurovascular unit rather than at an intracellular site as a neuroprotective agent.
Introduction
Nitrone compounds were developed originally as tools for studying free radical chemistry and are able to trap free radicals to form more stable adducts (Janzen and Blackburn 1968). As there is a widespread support for the notion that free radicals play a significant role in the development of brain injury after cerebral ischaemia and reperfusion, nitrones were examined subsequently in animal models of cerebral ischaemia and there is substantial evidence that several compounds are effective neuroprotectants in animal models of acute ischaemic stroke (see Green et al, 2003). α-Phenyl-N-tert-butylnitrone (PBN) has been extensively studied in both permanent and transient focal ischaemia models in rats, and its close congener 2-sulphophenyl-N-tert-butylnitrone (S-PBN) is also an established neuroprotective agent (Green et al, 2003). Disodium 2,4-sulphophenyl-N-tert-butylnitrone (NXY-059) is another derivative of PBN that has showed an impressive neuroprotective profile in both rodent and primate models of acute ischaemic stroke (Sydserff et al, 2002; Marshall et al, 2003; Green and Ashwood, 2005).
Surprisingly, evidence for nitrones having cytoproprotective activity in vitro is sparse. α-Phenyl-N-tert-butylnitrone has been shown to have an antiapoptotic effect against damage induced by reactive oxygen species in bovine aortic endothelial cells (Kotamraju et al, 2000) and prevents protein oxidation in oligodendrocytes exposed to oxidative stress (Ernst et al, 2004). Although PBN, 2-sulphophenyl-N-tert-butylnitrone, and NXY-059 have a similar potency in trapping free radicals in aqueous solution, as assessed by electron resonance (Maples et al, 2001; Williams et al, unpublished observations), no direct test of cytoprotection by NXY-059 in vitro has been reported.
We have, therefore, now investigated whether NXY-059 is cytoprotective in an in vitro preparation using a standard neuron-like cell model of free radical-induced damage (Yamada et al, 1996; Terwel et al, 2000; Ishii et al, 2001). Cell injury was induced by the use of the free radical generator, sodium nitroprusside (SNP).
Materials and methods
Reagents
Disodium 2,4-sulphophenyl-N-tert-butylnitrone was obtained from AstraZeneca R&D Södertälje (Södertälje, Sweden). All other reagents were from Sigma-Aldrich (Poole, UK), unless otherwise stated. Stock solutions of NXY-059 and a cocktail of ascorbate, reduced glutathione and dithiothreitol were made up in phosphate-buffered saline (PBS) on the day of use. The NXY-059 solution was kept in a bottle wrapped in aluminium foil to minimise exposure to light. Addition of NXY-059 produced no change in pH of culture media. A stock solution of SNP was made up in PBS just before use.
Cell Culture
Murine neuroblastoma cells of the N1E-115 cell line were grown in 1:1 (v/v) Dulbecco's modified Eagle medium: Ham F-12 medium, supplemented with 10% (v/v) heatinactivated foetal bovine serum (Gibco-Invitrogen, Paisley, UK) and 1% v/v glutamax II (Gibco-Invitrogen), without antibiotics, maintained in a humidified 5% CO2 atmosphere at 37°C, and divided every 7 to 10 days. Cells were plated into 96-well plates and used for experiments 6 to 9 days later. The following were added to cell culture medium 30 mins before injury stimuli: NXY-059 (final concentration 300 μmol/L in most experiments); or a cocktail of known antioxidant agents (ascorbate, reduced glutathione, and dithiothreitol, all at 100 μmol/L final concentration); or an equivalent volume of PBS (10 μL). Sodium nitroprusside (final concentration 100 μmol/L in most experiments) or an equivalent volume of PBS (10 μL) was added to appropriate wells 30 mins later.
Cell Survival Assays
After 24 h, cell injury was assayed either by the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay of mitochondrial activity (Terwel et al, 2000) or by nuclear morphology counting. 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide is a yellow water-soluble monotetrazolium salt that is reduced by active mitochondrial complex II to form an insoluble purple formazan pigment, whose abundance can be assayed spectrophotometrically. Decreased MTT formazan density indicates decreased mitochondrial activity and, as such, is a global indicator of reduced cell survival. Hoechst 33342 chromatin stain was used to identify apoptotic cells on the basis of nuclear morphology. Cells with a bright, round nucleus or with three or more nuclear fragments were considered to be undergoing apoptosis-like cell death (‘Hoechst-positive‘). An observer, blind to treatment condition, counted the cells observed in four or more randomly chosen, non-overlapping fields (× 200 magnification) to a total of at least 200 cells in each well of a 96-well plate. The fraction of apoptotic cells was expressed as number of Hoechst-positive cells per hundred total cells.
Statistical Analysis
Bar charts show mean + 1 s.e.m. of three or more independent experiments. Statistical analyses were carried out using Microsoft Excel and GraphPad Prism software. Significant differences (P <0.05) were determined by Student's t-test.
Results
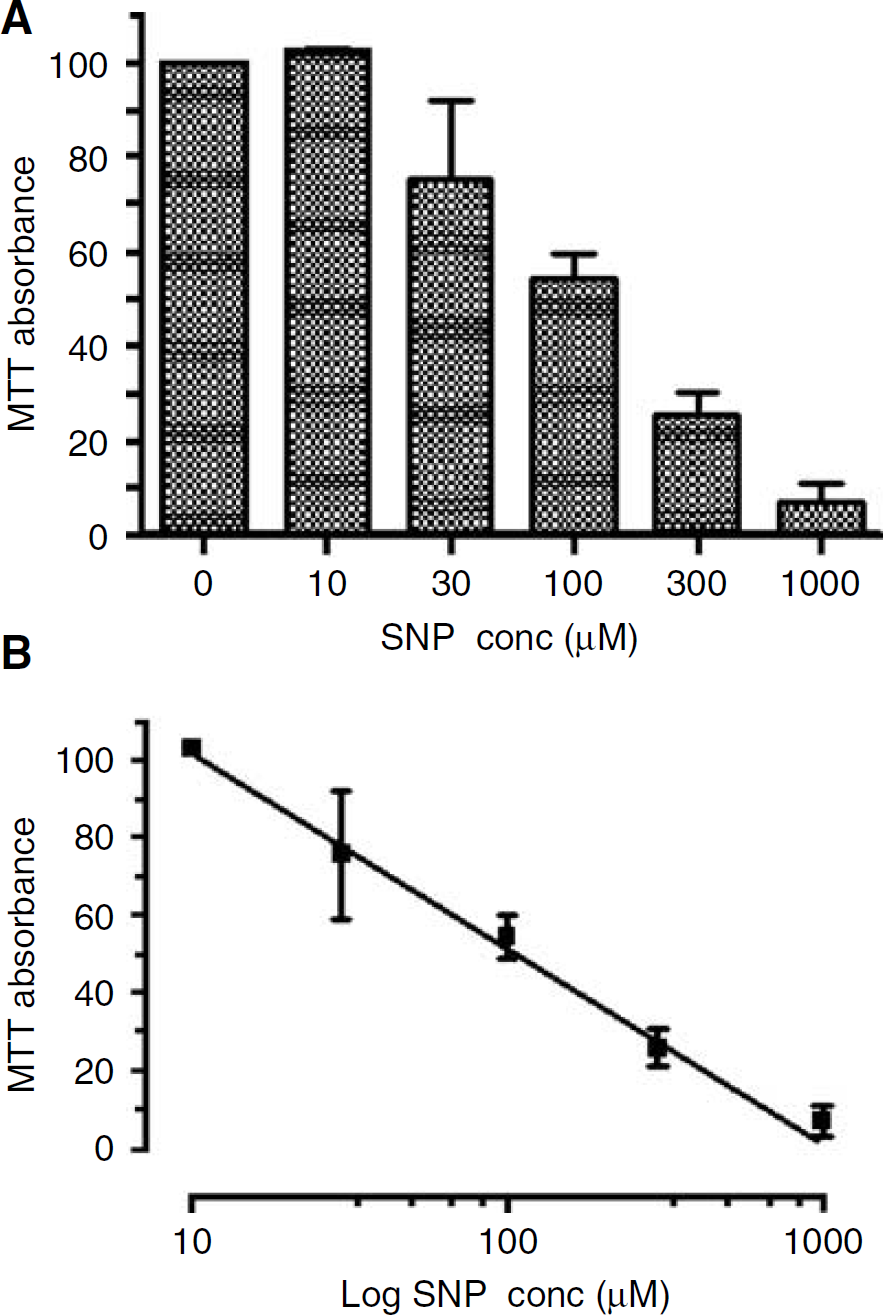
Exposure to SNP (10 to 1,000 μmol/L) for 24 h produced cell injury in N1E-115 neuroblastoma cells, as determined by the MTT assay (Figure 1A). This effect was concentration-dependent with near-complete cell death at 1 mmol/L and a half-maximal effect at a concentration of approximately 100 μmol/L (Figure 1B).
When the intermediate concentration of SNP (100 μmol/L) was used, the antioxidant cocktail produced significant protection, using either MTT conversion (Figure 2A) or nuclear morphology (Figure 2A) as the end point (P <0.05). In contrast, pretreatment with NXY-059 (300 μmol/L) was without effect on SNP-induced cell injury using either end point (Figure 2). Furthermore NXY-059 (1.0 or 3.0 mmol/L) was without effect on the cytotoxicity of SNP as measured by MTT conversion (data not shown), or by nuclear morphology when added at a concentration of 1.0 mmol/L (16.8 ± 1.39% apoptotic nuclei, n = 3). There was no significant effect of the antioxidant cocktail or NXY-059 on cell survival in the absence of SNP, as determined by either method (Figure 2).
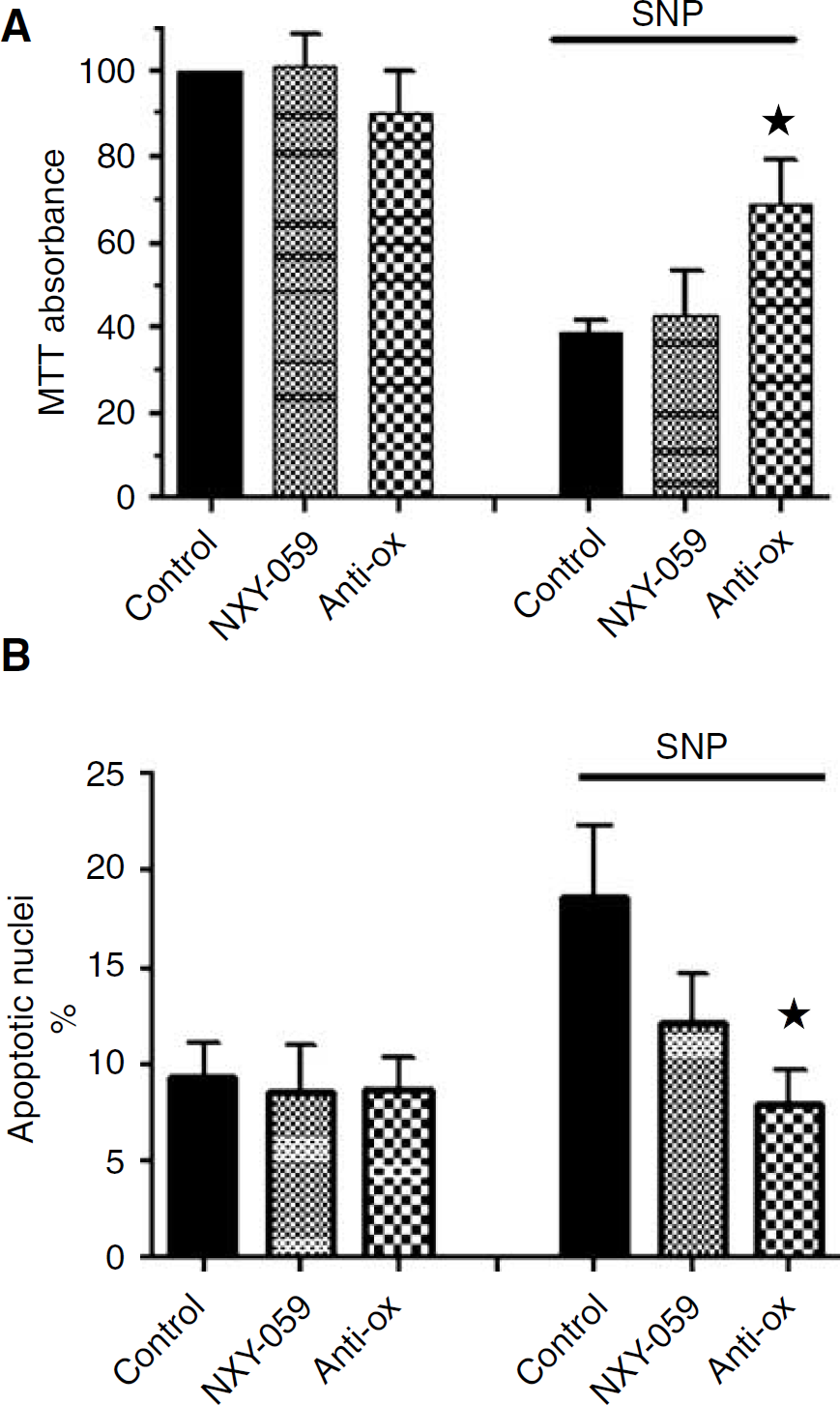
Effect of NXY-059 on SNP-induced cell death in N1E-115 neuroblastoma cells, assayed by (
Disodium 2,4-sulphophenyl-N-tert-butylnitrone was also without effect on H2O2-mediated cytotoxicity: cell survival assessed by MTT assay after 24 h treatment with 90 μmol/L H2O2 was 47 ± 5.2% in the absence and 42 ± 7.5% (mean, s.e.m., n = 3; P > 0.05) in the presence of NXY-059 (300 μmol/L).
To test for free radical-neutralising activity in a cell-free system, we measured formation of 3-nitrotyrosine on treatment of
Discussion
Sodium nitroprusside can produce a range of oxidant and free radical species in aqueous solution, including NO, superoxide, H2O2, peroxynitrite, and hydroxyl radical (Dawson et al, 1991; Yamada et al, 1996; Terwel et al, 2000). Other workers have observed SNP-induced delayed cell death in neuronal and neuroblastoma cells under similar conditions to those used here, presumably due to a combination of extracellular and internal free radical species (Dawson et al, 1991; Yamada et al, 1996; Terwel et al, 2000). In general, higher concentrations were used in those earlier studies and dose-response data were not generated, whereas we have been able to show a half-maximal concentration as low as 100 μmol/L.
N1E-115 cells are a homogenous clonal source of neuron-like cells, derived from a mouse neuroblastoma. On depletion of serum, as in the aged cultures used here, these cells differentiate into a neuron-like morphology, exhibiting neurite outgrowth and electrical excitability (Ishii et al, 2001). They undergo apoptosis-like cell death after cellular injury stimuli, including treatment with SNP (Yamada et al, 1996). The sensitivity to SNP reported here is similar to that of primary cortical neurons (Dawson et al, 1991) and the nuclear morphology changes observed are similar to those in cultured cortical neurons after treatment with nitric oxide donors (Bonfoco et al, 1995).
Here, the cytotoxic action of SNP-induced free radical production was prevented by a known antioxidant mixture at a concentration (100 μmol/L of the three compounds) that is considerably lower than that used by Terwel et al (2000). In contrast, NXY-059 was ineffective at a concentration (300 μmol/L), which both markedly exceeds that required in plasma for a maximum neuroprotective effect in animal models of stroke in vivo (150 μmol/L; Sydserff et al, 2002) and is also greater than the targeted steady-state concentration (260 μmol/L) in the recent Phase III trials of NXY-059 in acute stroke (Lees et al, 2006). Direct nitration of amino acid tyrosine by peroxynitrite was also abolished by the antioxidant cocktail, but was unaffected by NXY-059. Disodium 2,4-sulphophenyl-N-tert-butylnitrone has previously displayed antioxidant activity in a cell-free, cuvette-based system using Fe(II)/H2O2 to generate hydroxyl radicals and to measure salicylate oxidation; in that earlier study, an extrapolated IC50 of 298 μmol/L was obtained for NXY-059 (Maples et al, 2001). We saw no effect of NXY-059 on tyrosine nitration at concentrations well above the maximal concentration of 200 μmol/L used by Maples et al (2001). A recent paper (Wang et al, 2006) did claim that low concentrations of NXY-059 were cytoprotective in a brain slice preparation, but the nitrone studied was in fact PBN (DC Lo, personal communication).
Our study is therefore the first test for a cytoprotective effect of NXY-059 on neuronal-type cells in vitro. Previous in vitro studies (Dehouck et al, 2002) observed very limited membrane permeability. Despite this, the compound affords substantial brain protection in vivo when administered at up to 4 h after occlusion in rat and primate models of stroke (Sydserff et al, 2002; Marshall et al, 2003). The lack of effect in vitro that we have observed may reflect low cell penetration, an interpretation supported by studies in organotypic hippocampal preparations where PBN was cytoprotective (Barth et al, 1996), but the much more hydrophilic 2-sulphophenyl-N-tert-butylnitrone, even at very high concentration (1,000 μmol/L), was without effect (Wilde et al, 2000).
In view of the current data, it is interesting to speculate on why NXY-059 is such an effective neuroprotectant in animal models of stroke (Green and Ashwood, 2005). Recent studies have shown that NXY-059 protects the brain endothelium after a simulated ischaemic insult in vitro (Culot et al, 2006). It therefore seems reasonable to suggest that NXY-059 acts as a neuroprotectant in animal models of stroke primarily by an action at the neurovascular unit (del Zoppo, 2006). The present study suggests that the translation from in vitro to in vivo preclinical models of stroke requires further research, just as clearly as is the case for translation from in vivo animal models to the clinical condition of acute ischaemic stroke, for while the first Phase III trial of NXY-059 in 1,700 patients (Stroke-Acute Ischemic NXY Treatment I) was positive (Lees et al, 2006), the second definitive trial in over 3200 patients (Stroke-Acute Ischemic NXY Treatment 2) was negative (see press release at www.astrazeneca.com/pressrelease/5279.aspx, October 2006).
