Abstract
It has been suggested that a vascular-like cell has multipotent regenerative and mesenchymal lineage relationships. The identity of this stem/progenitor cell has remained elusive. We report here that adult central nervous system (CNS) capillaries contain a distinct population of microvascular cells, the pericyte that are nestin/NG2 positive and in response to basic fibroblast growth factor (bFGF) differentiate into cells of neural lineage. In their microvascular location, pericytes express nestin and NG2 proteoglycan. In serum containing media primary (0 to 7 day old) CNS pericytes are nestin positive, NG2 positive, alpha smooth muscle actin (αSMA) positive, and do not bind the endothelial cell specific griffonia symplicifolia agglutinin (GSA). In serum containing media, pericytes do not undergo neurogenesis but are induced to express αSMA. In bFGF containing media without serum, CNS pericytes form small clusters and multicellular spheres. Differentiated spheres expressed neuronal and glial cell markers. After disruption and serial dilution, differentiated spheres were capable of self-renewal. When differentiated spheres were disrupted and cultured in the presence of serum, multiple adherent cell populations were identified by dual and triple immunocytochemistry. Cells expressing markers characteristic of pericytes, neurons, and glial cells were generated. Many of the cells exhibited dual expression of differentiation markers. With prolonged culture fully differentiated cells of neural lineage were present. Results indicate that adult CNS microvascular pericytes have neural stem cell capability.
Introduction
The existence of multipotent stem cells within the adult brain has been the topic of considerable research emphasis and the subject of some debate (McKay, 1997; Temple, 2001). The identity of this quiescent totipotent stem cell is still unknown. However, it is clear that stem cells tend to reside in specialized tissue (stem cell niche) and that the local microenvironment regulates their differentiation and self-renewal (Sprading et al, 2001; Doetsch, 2003). Stem cell niches appear to be relatively compartmentalized areas composed of a basement membrane, extracellular matrix, and cells thought to provide important signaling molecules that enhance and support neurogenesis (Lim and Alvarez-Buylla, 1999; Song et al, 2002). In the brain two major germinal regions have been characterized (McKay, 1997; Temple, 2001). These are the subventricular zone (SVZ) and the subgranular zone (SGZ) (Sprading et al, 2001). Both the SVZ niche and the SGZ niche are in close association with the microvasculature, as well as perivascular cells, a specialized basal lamina and an extracellular matrix bed (Doetsch, 2003; Palmer et al, 2000). These elements are common components of the microvasculature and underscore their importance to the SVZ and SGZ germinal centers. The vascular niche also provides ample opportunity for extensive cell-cell communication (Palmer et al, 2000). The role of the microvasculature in central nervous system (CNS) stem cell differentiation is, however, still unclear.
The CNS microvascular endothelium is composed of two cellular constituents the endothelial cell (EC) and the pericyte. Both cells are in close association with astrocytes, the basal lamina as well as numerous matrix proteins (Ballabh et al, 2004). In fact, the vascular pericyte synthesizes most elements of the basal lamina including a number of proteoglycans (Dienfenderfer and Brighton, 2000; Ozerdem et al, 2002). Pericyte synthesis and release of laminal proteins is thought to be a critical step in the differentiation of the blood–brain barrier (Korn et al, 2002).
Pericytes or cells with probable pericyte-like morphology have been reported to differentiate to osteogenic or osteoblast/like cells in vitro (Canfield et al, 1996; Reilly et al., 1998). In serum-containing culture medium, CNS pericytes (3- to 7-day-old) take on a macrophage/dendridic cell phenotype (Balabanov et al, 1996, 1999) and then ultimately continue differentiating to form nodules that produce mineralized bone (Canfield et al, 1996, Dore-Duffy, unpublished observations). These data support the concept that CNS pericytes may be mesenchymal stem cells. Still additional evidence suggests that pericytes may differentiate to fibroblasts (Doherty and Canfield, 1999; Gerhardt and Betsholtz, 2003), ECs (Chaudhry et al, 1978), adipocytes (Cinti et al, 1984), chondrocytes (Reilly et al, 1998), and macrophages/dendritic cells (Balabanov et al, 1996, 1999).
It has been reported that the growth factors basic fibroblast growth factor (bFGF) and epidermal growth factor (EGF) stimulate pericyte proliferation and angiogenesis (Nico et al, 2004). Both EGF and bFGF-responsive vascular stem cells have been reported in the rat microvasculature (Palmer et al, 2000; Jin et al, 2002; Louissant et al, 2002). However, direct demonstration of a precursor cell displaying stem cell characteristics has not been forthcoming.
We propose that the CNS microvasculature pericyte, has a broad developmental potential. In particular, we have investigated the neural potential of cells subcultured from isolated rat CNS capillaries. We show here that CNS microvessels contain NG2 and nestin-positive pericytes. In serum free medium pericytes are responsive to bFGF, and form clusters of adherent cells and floating spheres composed of cells of multiple neural cell lineage. These cells undergo self-renewal and increase in number after subculturing. By clonal analysis, we show that multipotent pericytes differentiate along multiple lineages and may provide trophic support and maintenance in the adult brain.
Materials and methods
Antibodies and Chemicals
Affinity-purified goat anti-mouse and anti-rabbit IgG F(ab)2 fragments conjugated to Red 613 or fluorescein isothiocyanate (FITC) were purchased from CAPPEL (Durham, NC, USA). Rabbit anti-human Von Willebrand factor (vWF) antibody (IgG) (1:500) was purchased from Dakopatts (Glostrup, Denmark) in either FITC-conjugated or unconjugated form, and mouse anti-human vWF (IgG2) (1:1,000) was purchased from Boehringer Mannheim (Indianapolis, IN, USA). Rabbit polyclonal anti-glut-1 antibody (IgG) was purchased from Calbiochem (La Jolla, CA, USA). Mouse monoclonal anti-rat nestin (IgG) was purchased from Chemicon International (Temecula, CA, USA). Rabbit antibody, directed against rodent GFAP (1:200), and rabbit antibody, directed toward neurofilament 200 (NFL-200) (1:200), were purchased from Sigma (St Louis, MO, USA). Mouse antibody directed against 5-bromodeoxyuridine (BRDU) was purchased from BD Biosciences (Franklin Lakes, NJ, USA). Cy-conjugated anti-mouse IgG was purchased from Sigma (St Louis, MO, USA). Mouse anti-NG2 chondroitin sulfate proteoglycan monoclonal antibody (IgG1) and mouse anti rat oligodendrocyte (O4) antigen (1:50) was purchased from Chemicon International (Temecula, CA, USA).
Isolation of Microvessels
Rat CNS capillaries were prepared as described previously (Dore-Duffy et al, 1994; Dore-Duffy, 2003). Briefly, brain tissue was removed within minutes of decapitation using sterile technique. Tissue was homogenized in 10 volumes of Dulbecco's modified Eagle's medium (DMEM) using a glass homogenizer and teflon plunger shaved to leave 0.25 μm between the plunger and the glass surface. After 20 up-and-down strokes at 420 r.p.m., the homogenate was centrifuged and the pellet resuspended in 15% dextran in DMEM. The suspension was centrifuged at 5,000g for 10 mins (Sorval DuPont, Wilmington, DE, USA), and the pellet resuspended and filtered through 118 μm nylon mesh. The filtrate was passed through an 80 μm nylon mesh. Microvessels (MV) were collected on the 80 μm mesh, washed vigorously from the mesh, and resuspended in DMEM. Microvessel preparations were 80% to 100% capillaries. MV were > 95% viable by trypan blue exclusion.
Purity of MV
Purity of MV was determined by visual examination and by measurement of γ-glutamyltranspeptidase activity, diagnostic kit 545 (Sigma, St Louis, MO, USA) showing enrichment over starting material. In addition, exclusion of large vessels and the capillary nature of the preparation was confirmed by analysis on the Meridian ACAS 470 laser cytometer with computer-generated size determinations (Dore-Duffy et al, 1994). Staining of microvessel capillary preparations indicated that there were no neurons or glial cell contaminants (Dore-Duffy et al, 1994; Dore-Duffy, 2003).
Primary Pericyte Cultures
Capillaries were prepared as detailed above and suspended in DMEM (Sigma, St Louis, MO, USA) supplemented with 50 mg/mL pen-strep, 2.5 mg/mL nystatin and 0.1% collagenase type II (Worthington corp., Freehold, NJ, USA) and incubated at 37°C overnight in a shaker bath. After incubation, disrupted MV fragments and single cells were vigorously pipetted to disrupt any remaining fragments then centrifuged. Cells were pelleted and resuspended in DMEM plus 2% fetal calf serum (FCS) and 1% antibiotics and negatively purified by fluorescence-activated cell sorting (FACS) (Balabanov et al, 1996; Dore-Duffy, 2003). To confirm purity, cells (105/mL/plate) were allowed to adhere for 4 h on coverslips then stained for EC-specific and pericyte markers. Cells were plated on uncoated plastic and then nonadhered cells vigorously washed off. Nonadhered cells were again washed off after 24 h. Pericytes began to proliferate by 24 to 48 h and became confluent by days 7 to 8 depending on the plating density. Purity of pericyte cultures was assessed after dual staining of cultured FACS sorted cells for griffonia symplicifolia agglutinin (GSA)-FITC binding or factor VIII and for alpha smooth muscle actin (αSMA) after 3 days in culture (Herman and D'Amore, 1985). FACS sorted GSA – cultures were 100% pericytes (GSA-αSMA +) (Dore-Duffy, 2003). GSA + cell populations contained roughly 0% to 3% αSMA + cells. No GFAP +, NF1 + or O4 + cells were observed.
Primary EC Cultures
Microvessels were digested with 0.1% collagenase for 4 to 6 h at 37°C. MV were vigorously pipetted to produce tiny fragments. Approximately 104 MV fragments per plate were allowed to adhere overnight on collagen-coated Petri dishes. Nonadhered fragments and loosely adhered cells were vigorously shaken and washed off after 24 h. Using this technique, cultures were routinely 90% to 96% EC. Total removal of pericytes required flow cytometric removal of freshly isolated CD11b-positive cells (Balabanov et al, 1996). Even with flow cytometry it was almost impossible to completely eliminate pericytes as GSA + cell populations usually have small percentages of αSMA + cells after 3 days in culture.
Culture of bFGF-Responsive Cells
The viability of cells was determined by trypan blue exclusion. Viable cells (200/cm2) were plated in 35 mm Petri dishes in DMEMF-12 with N2 supplement plus 20 ng/mL human recombinant bFGF (medium described below) (Gibco, Grand Island, NY, USA). Fresh medium was carefully added every 2 to 3 days after centrifugation of the plates at 800g for 10 mins. The number of free-floating spheres was counted after 20 to 25 days in culture. Some spheres were cultured on poly-L-ornithine (Poly-O)-coated glass coverslips for immunocytochemistry. Other spheres were used in experiments to establish self-renewal and in subcloning experiments detailed below. Pericyte spheres were transferred to conical test tubes in bFGF-supplemented medium and diluted to yield one sphere/tube. Spheres were mechanically disassociated by vigorous up and down pipetting. One cell was added per microwell using a limiting dilution technique. Cells were incubated in bFGF containing medium. Approximately 10% of cells survived and produced new spheres. Spheres were transferred to Poly-O-coated slides as detailed below. A second subcloning step was used to make a third group of spheres.
Immunocytochemistry
Pericyte spheres were grown on Poly-O-coated coverslips for 5 days in bFGF containing medium then transferred to medium with no growth factors. Coverslips were fixed for 30 mins at room temperature in 4% paraformaldelyde then washed 3 × in phosphate-buffered saline (PBS). Coverslips were treated 10 mins with 0.01% Triton X-100 in the presence of blocking antibody. After washing 3 × in PBS, the coverslips were MV and primary cultured EC and pericytes were allowed to dry on alcohol washed glass coverslips or poly-O and fixed with 3% paraformaldehyde. Coverslips were washed in PBS with 1% bovine serum albumin, permeabilized with Triton X-100 and then stained with antibody directed against indicated markers, or with control antibodies. All primary antibodies were used at antibody excess and secondary antibodies at predetermined saturation density (1:100 dilution for nestin, GFAP and 1:200 dilution for antibodies directed against CD11b, NF1, and vWF). Coverslips were incubated 30 mins at room temperature, washed 3 × then incubated with fluorochrome-conjugated secondary antibody (affinity-purified anti-IgG F(ab)′2 fragments 1:100 dilution) as appropriate. Second antibody alone and/or isotype control antibody was used as a control. In dual label experiments, species similar immunoglobulins were not used. Immunofluorescence was examined with a fluorescence microscope Orthoplan-2 (Zeiss, Germany) using × 25 and × 60 oil objectives. Percentage of positive MV or cells was calculated after counting a minimum of 300 cells or MV. Coverslips were photographed with a 35 mm CONTAX 165 MT camera (Kyocera Corp., Japan)
Fluorescence-Activated Cell Analysis/Sorting (Dore-Duffy, 2003)
After enzyme treatment, single cell suspensions were incubate with EC binding FITC-conjugated GSA 1:100 for 60 mins in the cold. GSA-FITC-positive cell populations were removed. For analysis, suspensions were dual labeled with anti-rat nestin antibody and GSA-FITC. Cy-conjugated anti mouse IgG was used as second antibody. Cell suspensions were fixed then analyzed by a Becton-Dickerson fluorescence activated cell sorter. Isotype similar nonspecific antibody was used as controls and values for percentage subtracted.
Polymerase Chain Reaction Techniques
Reverse transcriptase polymerase chain reaction (RT-PCR) was performed using total RNA from rat brain (Mullis and Faloona, 1987). Synthesis of cDNA was performed as previously described (Mullis and Faloona, 1987). Two microliters of cDNA per assay point was amplified using forward and reverse gene-specific primers listed in Table 1. PCR products were isolated on 1.5% agarose gels. Quantitative (real-time) PCR (Kwok and Higuchi, 1989; Faloona et al, 2005) was performed on selected samples as indicated in the text. Experiments were performed in the Wayne State University DNA facility using Applied Biosystems Inc. Prism 7700 sequence Detection System (Foster City, CA, USA). Parameters include 10 mins, 95°C, 40 cycles of 30 secs at 95°C, 2 mins at 60°C using 2 mL cDNA, 400 nmol/L target gene primers and 12.5 mL of SYBR GREEN 1 dye. An internal reference dye (Rox) was used to normalize for non-PCR related fluctuations in fluorescence that may occur from well to well.
Primers used for RT-PCR and real-time PCR

Results
Purity of Microvessel and Primary Pericyte Cultures
Central nervous system capillary preparations were isolated and enzymatically disrupted as detailed in Methods. If capillaries are carefully isolated they can be prepared with no cellular contamination. However, if isolation techniques are not performed carefully or performed incorrectly then cellular contaminants may include neurons. Poorly controlled preparations may also include astrocytes as well as glial cells. While the laboratory routinely isolates pure capillary preparations for pericyte subculture, care was taken to document purity to prove the validity of subsequent data. Capillary digests were examined for purity using immunocytochemistry and then analyzed by FACS analysis (Figure 1). Capillary cell preparations did not contain O4-positive cells, GFAP-positive cells, or neurofilament antigen-positive cells. Capillaries were positive for GSA lectin-binding cells (ECs) and GSA lectin—negative/NG2-positive cells (pericytes) (Balabanov et al, 1996; Dore-Duffy, 2003; Balabanov and Dore-Duffy, 1998). Muscle-specific actin (αSMA), although an in vitro marker for pericytes, is not expressed on 100% of pericytes in vivo. Thus, αSMA was not used to identify pericytes in stained capillaries. Using the above FACS criteria as well as additional tests performed for capillary purity (Methods), we concluded that our capillary preparations were pure and devoid of glial and neuronal cell contamination.
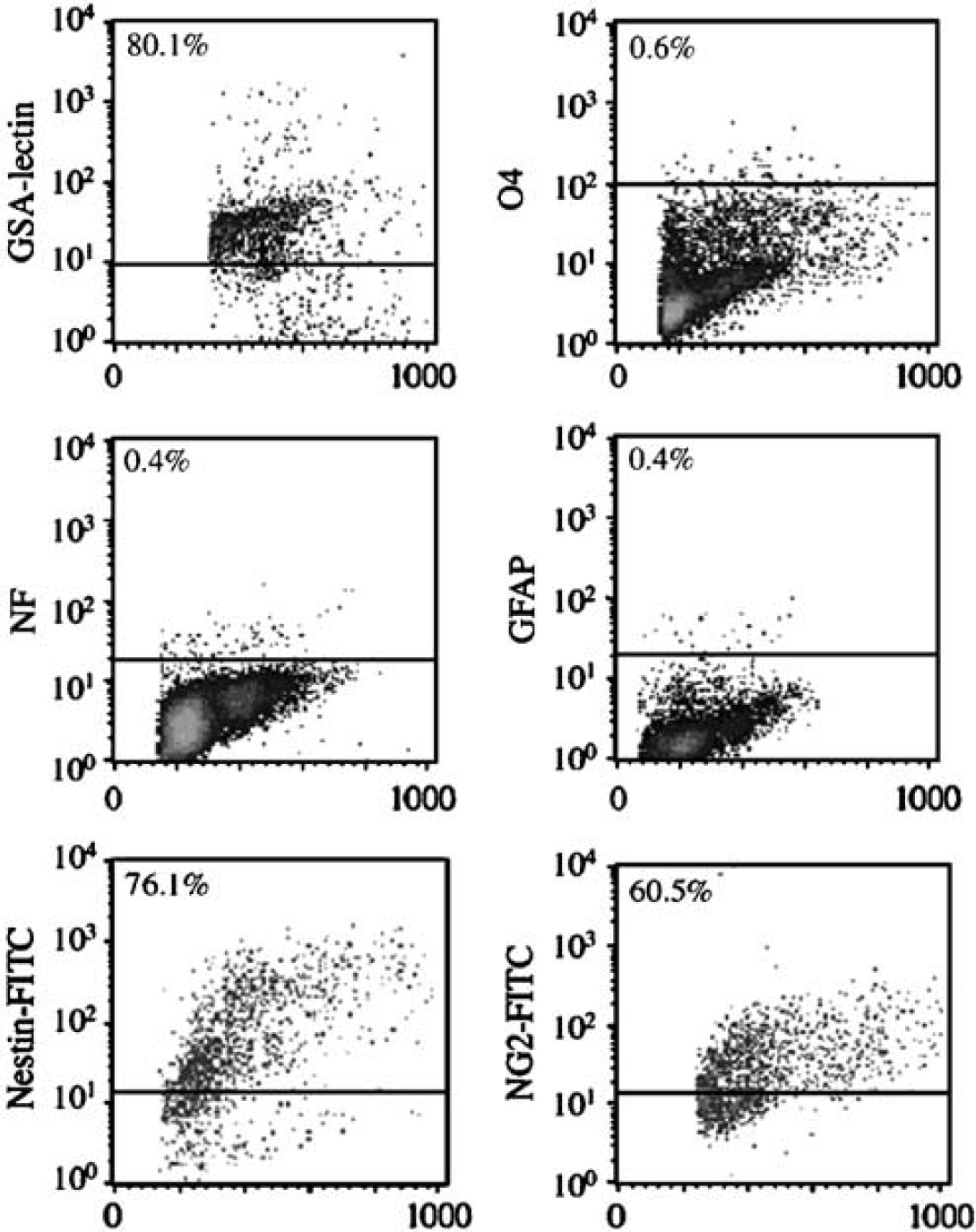
Purity of freshly isolated CNS capillary suspensions. Freshly isolated CNS capillary suspensions were enzymatically disrupted as detailed in Methods (Dore-Duffy, 2003). After disruption, single-cell suspensions were stained using immunocytochemical techniques and the antigenic properties of cell populations analyzed by flow cytometry. CNS capillary digests contained cells that bound FITC-GSA lectin (ECs) and stained positively for the expression of vWF (data not shown in this figure). Preparations also contained nestin-positive cells and NG2-positive cells. Capillary preparations did not contain O4 + cell populations, cells that stained positive for expression of neurofilament(NF), or cells that expressed GFAP Cultured FACS sorted GSA– cells expressed αSMA (data not shown in this figure).
Capillaries and Primary Pericytes Express Nestin/NG2
Pure capillary preparations were prepared as detailed above and subcultured to produce primary pericytes as detailed in Methods. Capillaries were dual stained for expression of EC-specific markers (vWF; GSA lectin binding activity) and nestin. Capillaries were also dual stained for expression of NG2/nestin. Primary pericytes (7-day-old) were dual stained for expression of αSMA/nestin, and NG2/nestin. 4,6-diamidino-2-phenylindole (DAPI) was used to stain nuclei. Results are shown in Figure 2. Capillaries expressed Von Willebrand factor (vWF) and nestin (Figure 2A). Von Willebrand factor staining is distributed throughout the vessel, whereas nestin staining appeared to concentrate near round versus elongated nuclei. This pattern of nestin staining within the vessel is consistent with localization of antigen in pericytes (Balabanov et al, 1996). There was no evidence of colocalization of nestin with vWF Capillary preparations also stained positively for NG2/nestin (Figure 2B) and vWF/NG2 (Figure 2C). NG2 staining appeared to be localized to pericytes confirming previously published data (Ozerdem et al, 2002). To confirm that microvascular capillaries contain a nestin-positive cell population, we performed a second set of experiments. Freshly isolated capillaries were enzymatically disrupted and single cells resulting from these digests were dual stained with FITC-conjugated GSA lectin and antibody directed against nestin. Dual stained cell suspensions were examined using FACS analysis (Figure 2D). Nestin expression was localized in GSA– cell populations (Figure 2D) consistent with localization within pericytes (Dore-Duffy, 2003). When GSA–/nestin + cells were plated, 100% became αSMA + by 4 to 7 days in culture, confirming previous studies by our laboratory (Balabanov et al, 1996). These cells expressed immunologically reactive αSMA/nestin and αSMA/NG2 as shown in dual stains (Figures 2E and 2F). Capillaries and primary pericyte cultures also expressed nestin and NG2 transcripts by RT-PCR (Figures 3A and 3B). Taken together, results indicated that CNS capillary pericytes express nestin and NG2 in situ and in primary culture. Pericyte expression of nestin transcripts decreased with time in culture when assayed by quantitative real-time PCR (Figure 3A, right panel). However, NG2 mRNA, transcripts were expressed up to 3 weeks (Figure 3B, right panel).
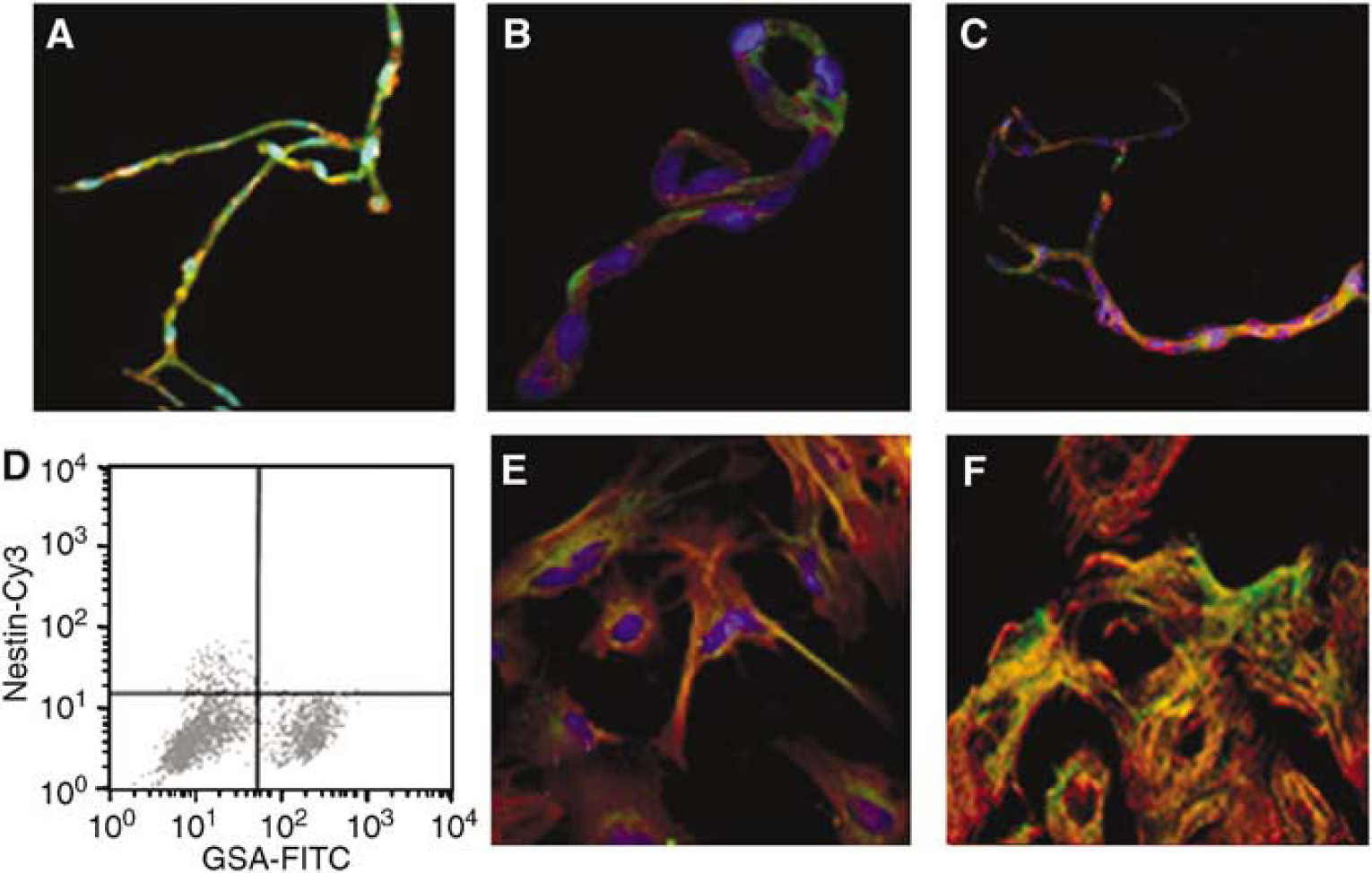
Characterization of nestin/NG2-positive cells in CNS capillaries. Freshly isolated CNS capillaries were dual stained for expression of vWF(green)/nestin(red) (
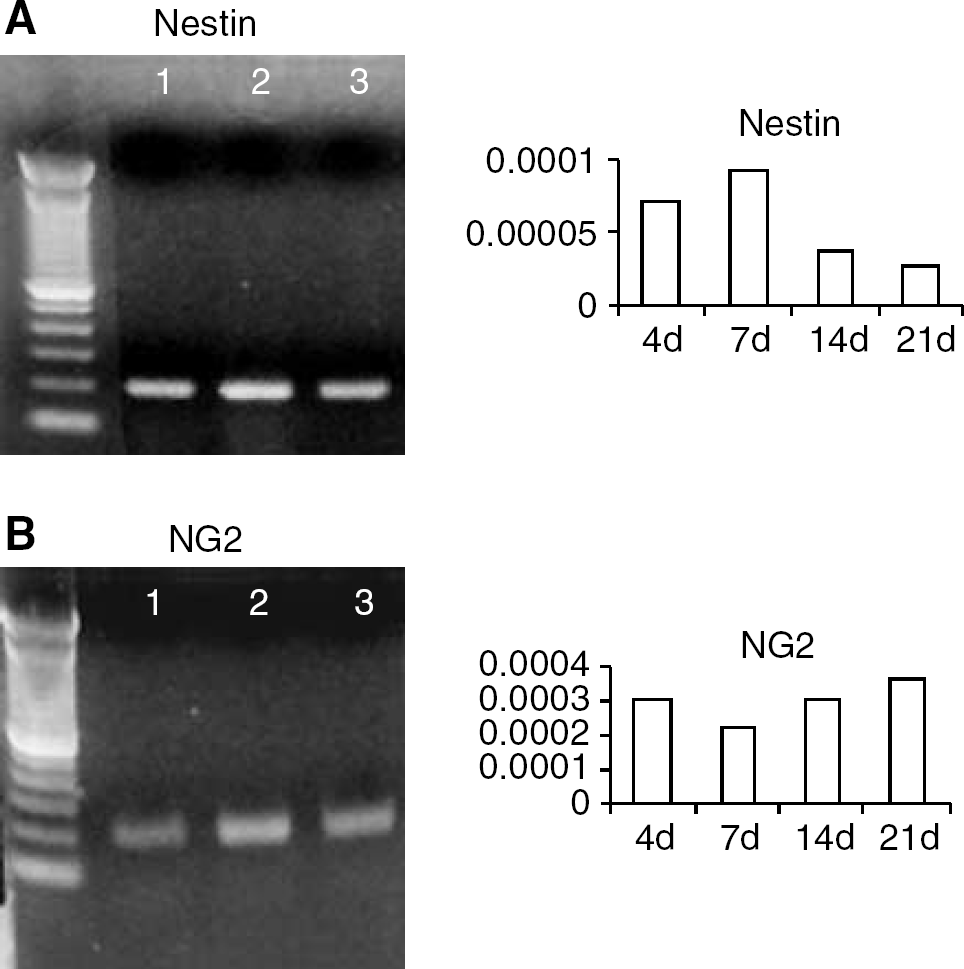
Expression of nestin and NG2 transcripts in cultured CNS primary pericytes. Freshly isolated CNS capillaries were enzymatically digested and pericytes subcultured as detailed in Methods. Primary pericytes were cultured in DMEM plus FCS and in N2bFGF medium. Total RNA was isolated from capillaries pericytes and from pericyte derived spheres. Reverse transcriptase polymerase chain reaction analysis indicated that capillaries (lane 1), pericytes (lane 2), and spheres (lane 3) expressed mRNA transcripts for nestin (
Primary Central Nervous System Microvascular Pericytes Proliferate in Response to Basic Fibroblast Growth Factor
Primary pericytes were plated at a low density (200 cells/cm2) in normal culture medium, and in DMEM F12 (no serum) with N2 supplement with and without bFGF (20 ng/mL). In normal culture medium (DMEM + 20% FCS), pericytes display typical morphology. On plastic, pericytes appear large with multiple short processes (Balabanov et al, 1996, 1999; Balabanov and Dore-Duffy, 1998). However, in N2 medium supplemented with bFGF, pericytes displayed altered culture morphology (Figure 4). Few cells adhered to the culture dish and approximately 1/4 of the cells died. Trypan blue-positive cells appeared by 2 days. Adhered as well as nonadhered cells formed small spherical clusters that, with continued growth, lifted from the culture dish. The majority of growing spheres remained in suspension growing in size as shown in Figures 4A–4C. Pericyte spheres were nestin + (Figure 4D) and labeled with BRDU (Figure 4E). Pericyte-derived spheres expressed nestin and NG2 transcripts (Figures 3A and 3B, lane 3).
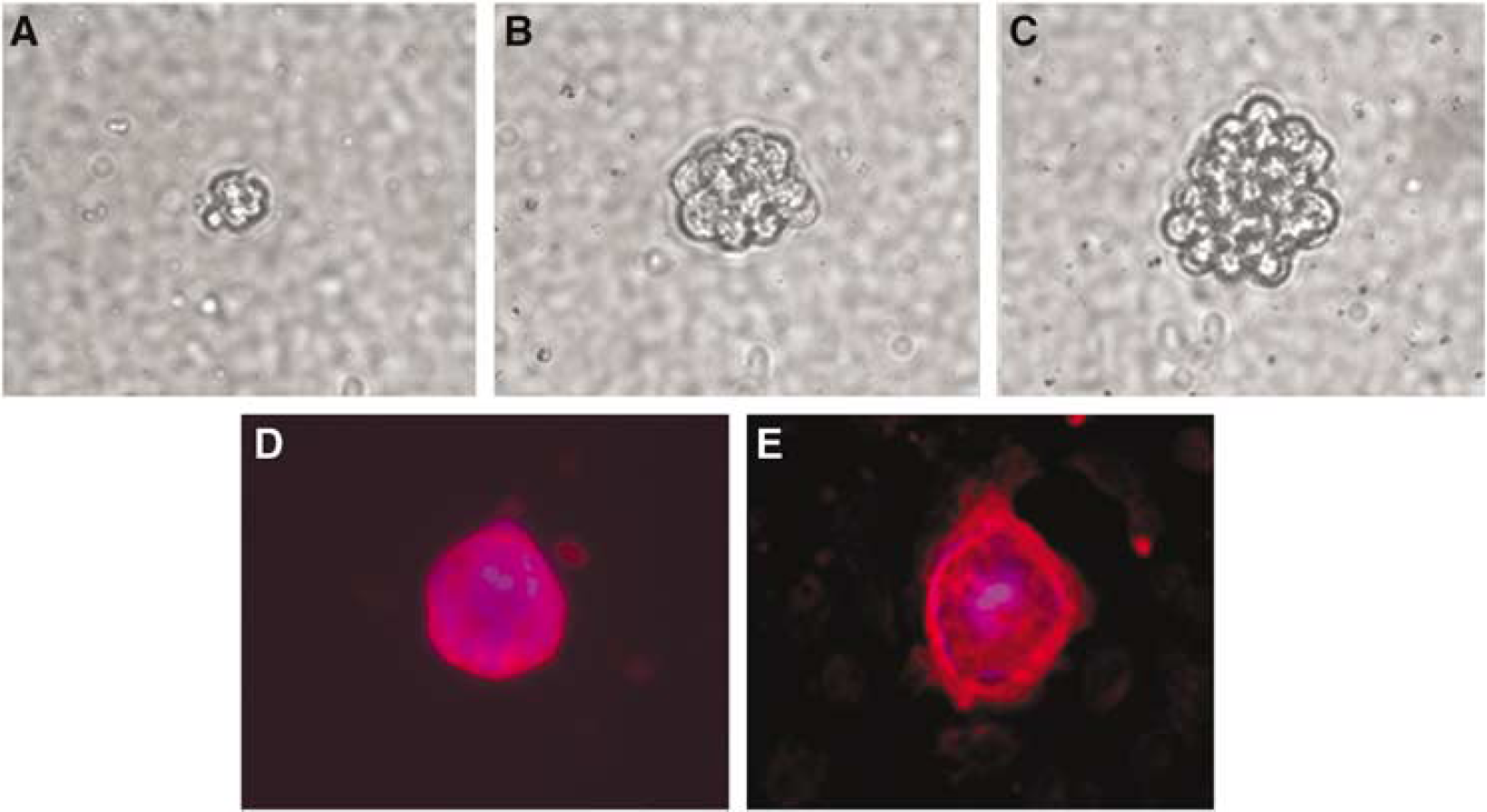
Primary CNS pericytes are responsive to bFGF Primary CNS pericytes were cultured in medium with N2 supplement and bFGF for variable periods of time as detailed in Methods. The majority of cells remained in suspension although a few adhered to the culture dish. Both adhered and floating cells replicated forming spheres that increased in size (
Basic Fibroblast Growth Factor-Responsive Central Nervous System Pericytes are Multipotent and Differentiate
We investigated whether bFGF-responsive CNS pericyte-derived spheres could differentiate into the major cell populations present in the CNS. Individual 20 to 25 day old spheres were transferred to glass coverslips coated with poly-O. Coverslips were prepared for immunocytochemistry and stained for astroglial, oligodendroglial, neuronal markers and the chondroitin proteoglycan NG2. Results indicate that each primary BRDU + sphere differentiated into multiple cell types (Figure 5, top panels). Astroglial cells were identified by expression of GFAP, oligodendrocyte precursors by O4-positive staining, and neuronal differentiation by expression of neurofilament (Figure 5, top panels). All spheres were nestin positive. All spheres also expressed NG2 (data not shown). Differentiation of pericyte spheres into cells of neurolineage was confirmed by FACS analysis (Figure 5, bottom panels) and by analysis of specific transcripts by quantitative real-time RT-PCR (gels not shown).
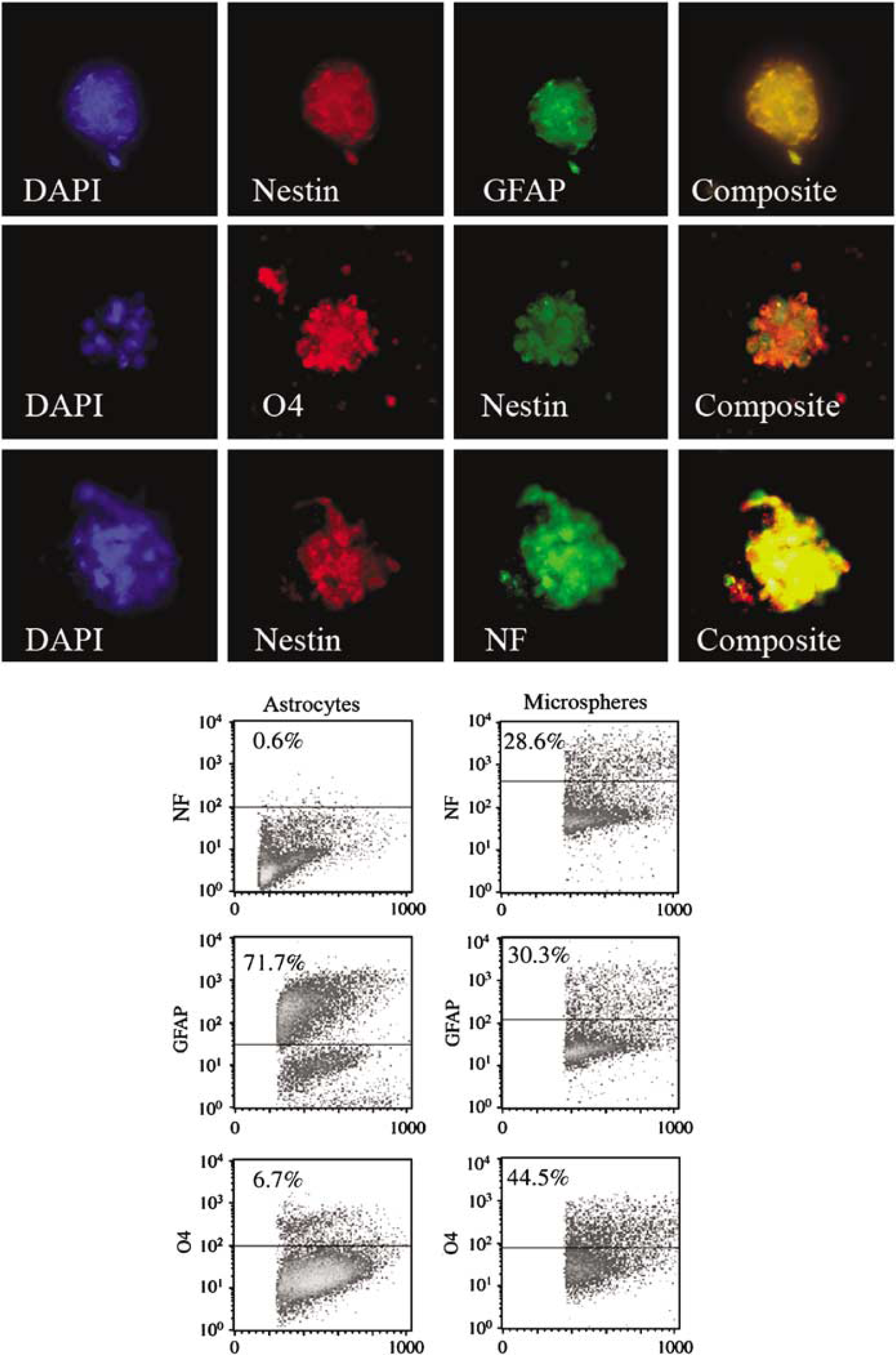
Characterization of differentiated pericyte-derived spheres. Two- to three-week-old pericyte-derived spheres were characterized by immunocytochemistry (top panels) and by flow cytometry (bottom panels). Pericyte-derived spheres contained cells that stained positively for dual expression of nestin/GFAP (top row, top panels); nestin/04 (middle row, top panels) and nestin/NF (bottom row, top panels). Pericyte-derived spheres were disrupted, stained and analyzed by flow cytometry (bottom section, right panel). Primary rat astrocyte preparations were similarly analyzed as controls(bottom section, left panel). FACS analysis indicated that spheres showed evidence of differentiation to the neural lineage. Pericyte derived spheres contained cells that expressed GFAP, NF and O4 +. Astrocyte control cell suspensions expressed GFAP, a small percentage of 04 positive cells and no cells expressing NF.
Self-Renewal of Multipotent Basic Fibroblast Growth Factor-Responsive Central Nervous System Pericytes
To show that self-renewal, bFGF-responsive pericyte-derived spheres were serially subcloned as detailed in Methods. Individual cells from the primary isolation produced spheres (Table 2). When these cells were disrupted, serially diluted, and plated at low density in the presence of bFGF, new secondary spheres and a few small adherent clusters were formed. A second generation as well as a third generation of spheres could be produced. Secondary and tertiary spheres were nestin positive and displayed evidence of differentiation (Table 2). The percentage of differentiated spheres increased somewhat with serial subcloning.
Self-renewal properties of pericyte bFGF-responsive cells
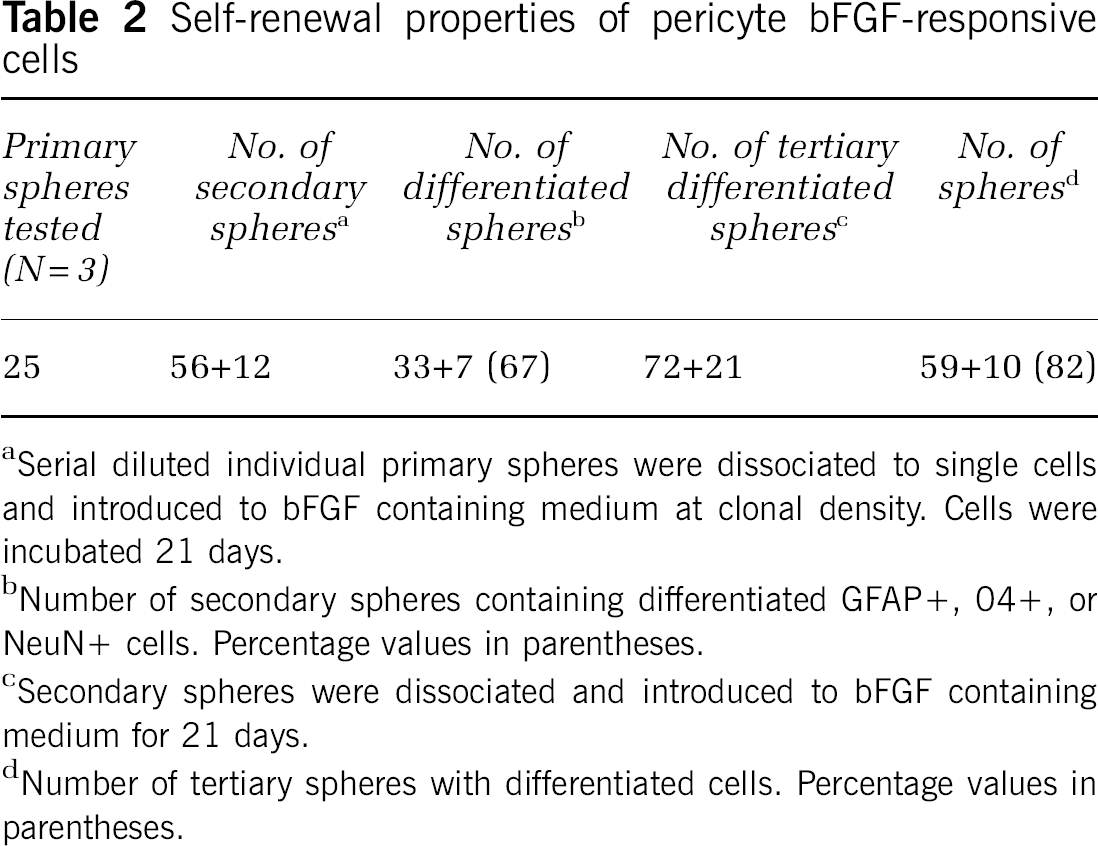
Serial diluted individual primary spheres were dissociated to single cells and introduced to bFGF containing medium at clonal density. Cells were incubated 21 days.
Number of secondary spheres containing differentiated GFAP +, 04+, or NeuN+ cells. Percentage values in parentheses.
Secondary spheres were dissociated and introduced to bFGF containing medium for 21 days.
Number of tertiary spheres with differentiated cells. Percentage values in parentheses.
Differentiated Spheres Returned to Serum-Containing Medium
Primary and secondary spheres were disrupted and returned to DMEM supplemented with serum and allowed to adhere to coverslips for 4 to 7 days (Figure 6). Coverslips were prepared for immunocytochemistry to document the presence of differentiated cells of neural lineage. Double and triple labeling techniques revealed that each sphere contained cells that, after plating in serum-containing medium, continued to exhibit neuronal, astroglial, and oligodendroglial markers (Figures 6A–6H). Each sphere contained cells that were αSMA/NG2 positive and nestin/αSMA positive and displayed a morphological appearance consistent with pericytes (Figures 6A and 6B). Many αSMA + cells coexpressed GFAP, O4, or NF (Figures 6C–6E). We also observed a number of nestin-positive cells that exhibited dual expression of neuronal and glial markers, as well as cells that expressed NF and morphologically resembled neurons (Figure 2B). Dual expression of O4/GFAP, NF/O4, and GFAP/NF were observed (Figures 6F–6H). The percentage of each cell population was determined at 5 days of culture and is shown in Table 3. The majority of cells exhibited pericyte morphology (30% NG2). Dual expression of αSMA and GFAP was seen in approximately 9% of cells. Coexpression of αSMA and O4 was observed in 6% of cells. Differentiated O4-positive cells represented approximately 6% of the population. αSMA/NF dual labeled cells compose approximately 11% and total NF + cells equal 12%. Cells dual labeled for GFAP and αSMA represent 9%. GFAP + cells with morphology characteristic of astrocytes represented only 3% or less. A proportion of cells were also αSMA +, nestin +, NF + (3%). GFAP + /O4+ cells (5%) were observed, as were NF/O4 + cells (7%) and GFAP/NF + cells (8%). It is unclear whether the percentages of differentiated cells changes with time in culture or with defined culture medium.
Cultures from differentiated spheres a
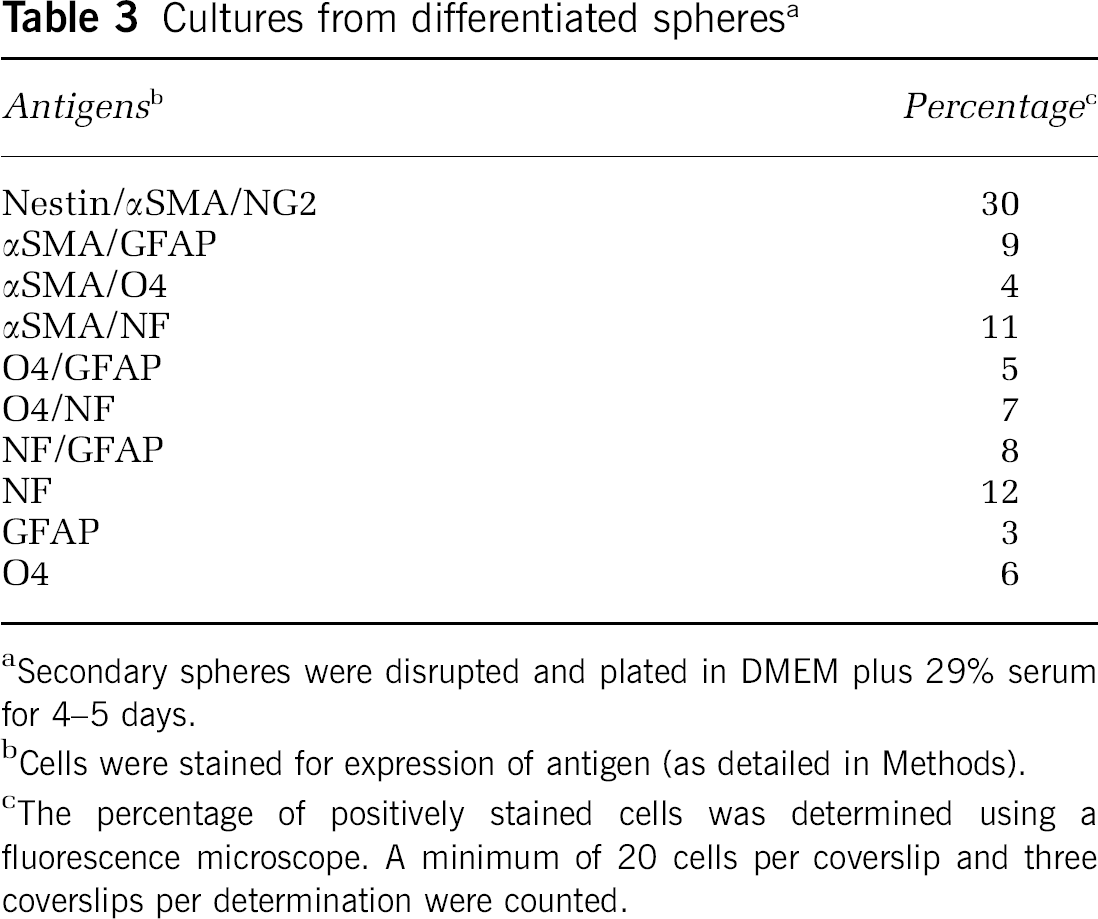
Secondary spheres were disrupted and plated in DMEM plus 29% serum for 4–5 days.
Cells were stained for expression of antigen (as detailed in Methods).
The percentage of positively stained cells was determined using a fluorescence microscope. A minimum of 20 cells per coverslip and three coverslips per determination were counted.
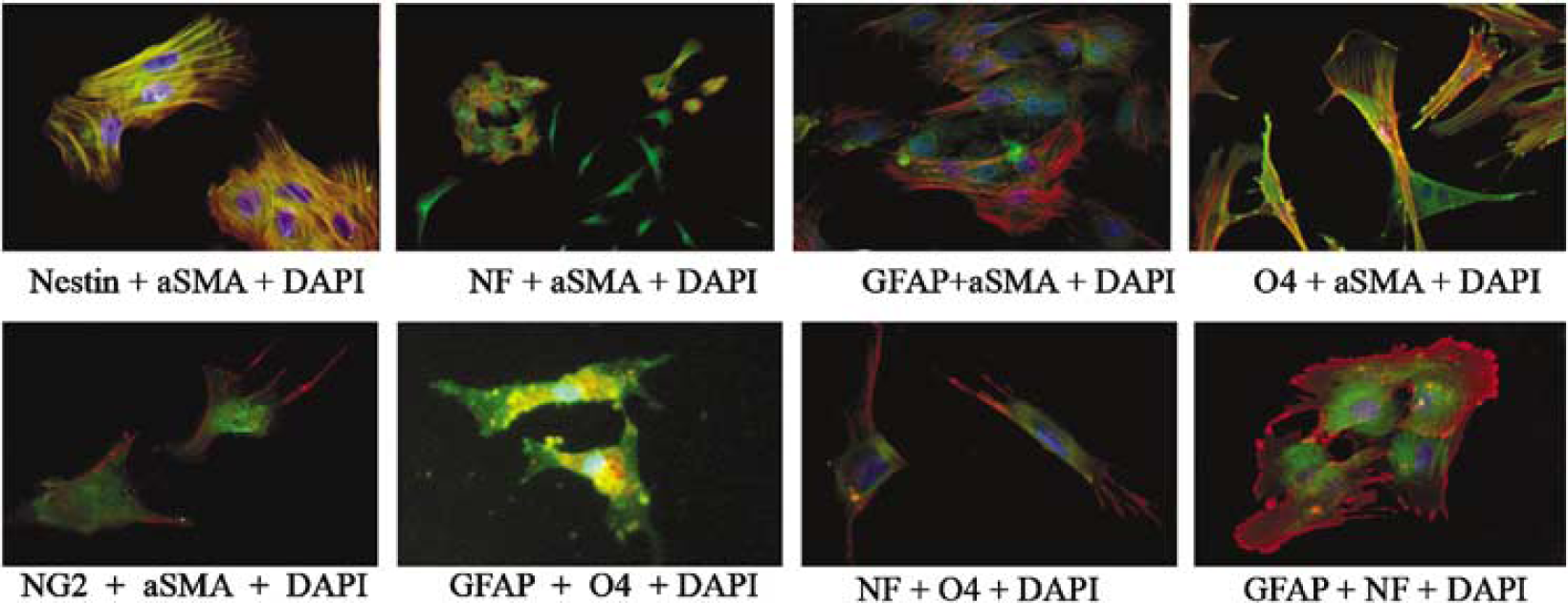
Antigenic properties of cells cultured from pericyte derived spheres. CNS primary pericyte-derived spheres (21 days old) were disrupted and cultured for 5 to 7 days in DMEM supplemented with 20% serum. Cells were dual stained for expression of indicated markers using immunocytochemistry. Nuclei were stained with DAPI. Differentiated spheres yielded cells that exhibited expression of: αSMA(red)/nestin(green); αSMA(red)/NF(green), NF(green); αSMA(red)/GFAP(green); and αSMA(red)/04(green) (top left to right). Spheres also generated αSMA(red)/NG2(green) positive cells (bottom left) that were also nestin positive (not shown). Of interest is that spheres generated cells that exhibited dual expression of GFAP(green)/04(red), 04(green)/NF(red) and GFAP(green)/NF(red) (bottom panel left to right). Spheres also generated cells that expressed only NF (seen in the top panel second from left)seen by 4 days in culture.
Discussion
While considerable evidence exists characterizing the role of the microvascular EC in the brain, very little is known of the role played by the CNS microvascular pericyte. With the development of culture techniques (Balabanov and Dore-Duffy, 1998; Gitlin and D'Amore, 1984; Shepro and Morel, 1993), investigators have begun to unravel the mysteries surrounding this intriguing cell and to decipher the nature of crosstalk mechanisms between pericytes and cells of the neurovascular niche. In its vascular location pericytes are thought to be important to maintenance of vascular homeostasis and hemostasis (Balabanov and Dore-Duffy, 1998; Shepro and Morel, 1993; Thomas, 1999). In response to infection, injury or inflammation pericytes release a wide variety of regulatory molecules and migrate from their vascular location into the perivascular space (Dore-Duffy et al, 2000). After migration in the perivascular space or in primary culture, the CNS pericyte responds to signaling molecules that induce differentiation. In this paper, we show that CNS pericytes exhibit multipotential stem cell characteristics. Pericytes replicate in the presence of bFGF and differentiate into cells displaying the antigenic properties of astrocytes, neurons, and oligodendrocytes as detailed in a flow diagram (Figure 7).
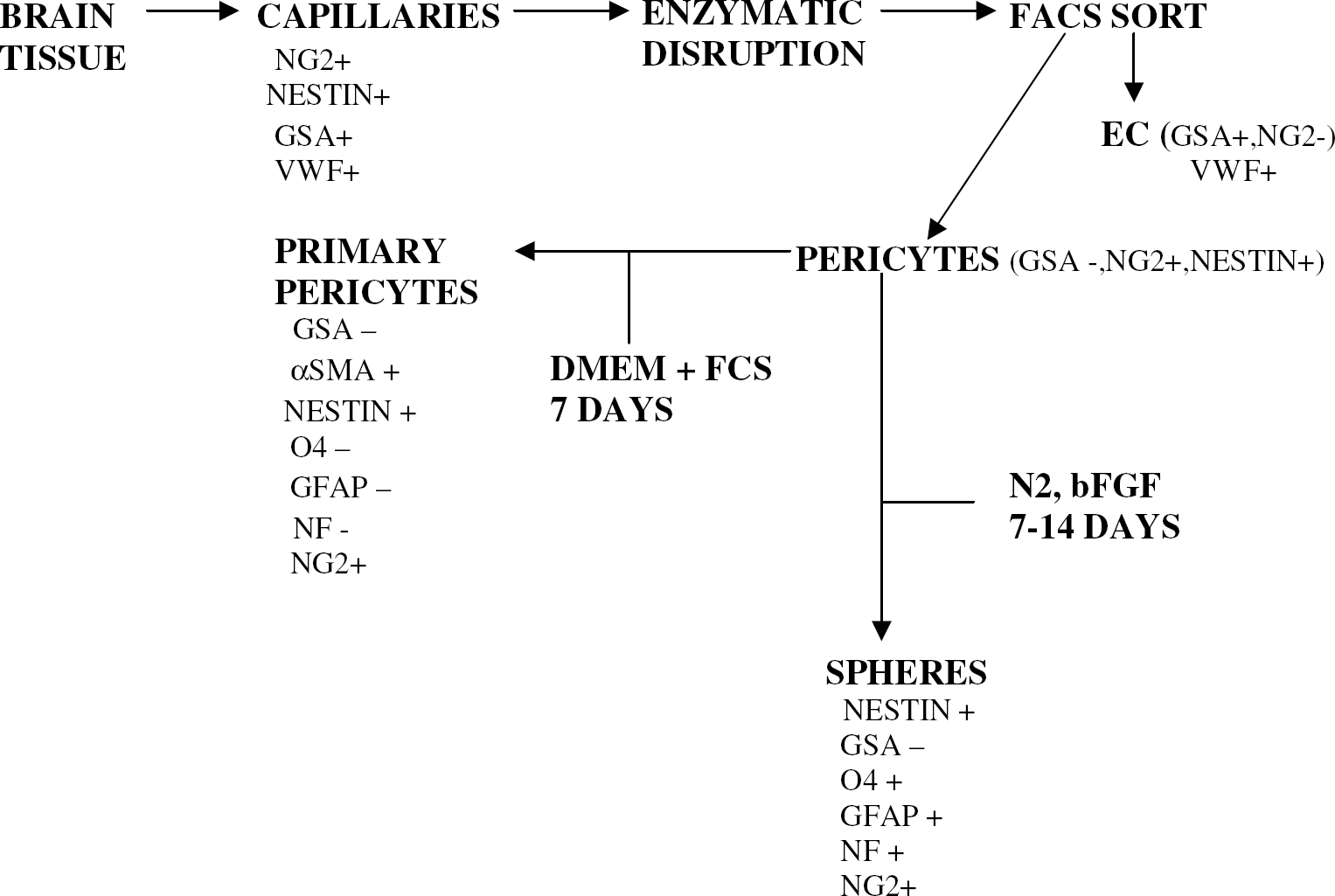
Flow diagram depicting the antigenic properties of CNS capillaries and primary pericytes. The sequence represents the isolation and culture of primary pericytes in serum containing medium and pericyte-derived spheres in N2, bFGF + medium without serum.
It has long been suggested that pericytes may have stem/precursor cell potential (as reviewed Korn et al, 2002). We have previously shown that CNS pericytes differentiate to a macrophage/dendritic cell phenotype in primary culture (3- to 5-day-old) and express CD80 and CD86 costimulatory molecules (Balabanov et al, 1996, 1999). Imitola et al (2004) reported that neural stem cells expressed the same co-stimulatory molecules. We have also reported that CNS pericytes express CD11b (Balabanov et al, 1996). Pericytes have been described as mesenchymal stem cells because of reports that they can differentiate into osteoblasts, chondroblasts, fibroblasts, adipocytes, and smooth muscle cells (Korn et al, 2002; Canfield et al, 1996; Reilly et al, 1998; Terskikh et al, 2001). These studies were performed in mixed culture systems. In our hands CNS pericytes cultured for prolonged periods in the presence of serum exhibit multipotentiality and differentiate to a macrophage-like phenotype then, with a prolonged culture period, differentiate to an osteogenic phenotype. CNS pericytes form nodules that express bone-specific protein by 18–21 days culture (Dore-Duffy, unpublished observations). Similar results have been reported for retinal pericytes (Reilly et al, 1998).
Adult multipotential stem cells have been described in tissue from brain (Mckay, 1997; Temple, 2001; Sprading et al, 2001), bone marrow (Terskikh et al, 2001), skin (Goodyer et al, 2001), skeletal muscle (Peng and Huard, 2004), and periostium (Simon et al, 2003). While the concept of a vascular stem cell has been discussed, much of this literature has focused on new vessel formation during angiogenesis in non-CNS tissue, as well as the possible stem cell nature of the EC (Reilly et al, 1998; Lutton et al, 2002). In our hands, nestin-positive cells within microvascular capillaries are GSA–/vWF–, and, on culture in serum containing medium, form αSMA + cells. However, coculture of EC and pericytes enhanced the number of differentiating primary spheres (data not shown), confirming data by Shen et al (2004). These authors also reported that smooth muscle cells did not enhance neurogenesis. We would support this hypothesis, as most fully differentiated αSMA + pericytes are not multipotent and do not form neurospheres when removed from serum-containing medium and cultured in bFGF-containing N2 medium without serum. Pericytes at this stage of differentiation may be committed to the mesenchymal lineage.
Neurogenesis occurs in foci (niches) in close association with microvessels (Doetsch, 2003; Palmer et al, 2000). These multicellular regulatory niches promote self-renewing cell populations. The nature of CNS stem cell niches and the mechanisms governing targeted differentiation are not well understood. We propose that the cellular constituents of the microvasculature are important in regulation of differentiation. Our results suggest that the microvasculature is a source of adult stem cells and that these cells are the pericytes. That pericytes are found in most tissue may help explain the transdifferentiation of adult stem cells from one tissue type to another.
Adult stem cell proliferation and differentiation is induced in response to environmental stress (Zhu et al, 2005). Proliferating stem cells are often found in a perivascular location (Song et al, 2002). However, the exact mechanism that regulates induction and proliferation, as well as the role of the stem cell in adaptation to stress injury is not known. Pericytes migrate from their vascular location in response to stress injury (Dore-Duffy et al, 2000) and remain in a perivascular location where they proliferate during the angiogenic response (Gerhardt and Betsholtz, 2003). Pericytes regulate angiogenesis (Balabanov and Dore-Duffy, 1998; Gerhardt and Betsholtz, 2003; Otani et al, 2000; Nehls et al, 1992; Hirschi and D'Amore, 1997; Yonekura et al, 1999) during: (1) initiation (Otani et al, 2000); (2) sprout formation and migration (Nehls et al, 1992); and (3) maturation and termination (Gerhardt and Betsholtz, 2003; Hirschi and D'Amore, 1997; Yonekura et al, 1999). Pericyte signaling molecules that are involved in regulation of angiogenesis are also involved in neurogenesis (Jin et al, 2002; Louissant et al, 2002). Pericytes synthesize the proangiogenic cytokine vascular endothelial cell growth factor (VEGF) (Yonekura et al, 1999). VEGF augments periycte proliferation in an autocrine fashion (Yonekura et al, 1999) and promotes differentiation of multipotent chondrocytic stem cells (de la Fuente et al, 2004). Pericytes are also responsive to growth factors and other signaling molecules important in regulation of neurogenesis (Nico et al, 2004; Balabanov and Dore-Duffy, 1998; Yonekura et al, 1999). Thus, it is possible that the response to injury and stress at the tissue level is coregulated with neurogenesis in the adult and tied into regulation of the cell cycle.
Regulation of the cell cycle is important to cell repair and survival and is an integral component of the stress response. The arresting of G1-S transition involves a complex interaction of cullins, p53-responsive proteins, glucose response proteins, cyclins and other regulatory elements that render the cell competent to proliferate after reception of a subsequent signal (Sciandra et al, 1984; Amellelum and Peterson, 1991; Little et al, 1994; Jackson, 1996). It is likely that a similar complex sequence of regulatory factors and cell cycle regulation are important in differentiation during neurogenesis and is involved in mobilization of adult stem cell activity.
Pericytes synthesize heparin sulfate proteoglycans as well as other matrix proteins (Dienfenderfer and Brighton, 2000; Ozerdem et al, 2002) and are thought to be responsible for the laying of the basal lamina during development (Balabanov and Dore-Duffy, 1998). We report here that CNS pericytes are NG2 + confirming previous observations in other tissue (Ozerdem et al, 2001). Adult neurogenesis is thought to be due to an NG2 + progenitor cell that is intrinsically multipotent (Chekenya et al, 2002; Ffrench-Constant and Raff, 1986; Wolswijk and Noble, 1998; Belachew et al, 2003; Aguirre et al, 2004). Heparin sulfate proteoglycans tether and present many factors involved in adult neurogenesis (Hienola et al, 2004). Pericyte synthesis of extracellular matrix proteins may therefore have profound effects on the presentation of regulatory molecules within the stem cell niche and suggests that pericytes have an important role in homeostasis as well as neurogenesis.
In conclusion, we have shown that CNS microvascular pericytes are nestin/NG2 positive and are a source of adult multipotential progenitor cells, adding further credence to the supposition that pericytes are of hematopoietic origin (Hess et al, 2004). It is unclear at this stage of the studies whether these cells are comparable to multipotent NG2 + type C-like cells identified in the SVZ of early postnatal and adult animals (Belachew et al, 2003; Aguirre et al, 2004; Miller, 1999). However, the CNS pericyte may be a source of purified viable multipotent stem cells with the potential of directed neurogenesis and could be important to future therapeutic strategies.
Footnotes
Acknowledgements
The authors thank DeShawn Williams for typing this manuscript.
