Abstract
Hibernating Arctic ground squirrel (hAGS), Spermophilus parryii, survive profound decreases in cerebral perfusion during torpor and return to normal blood flow during intermittent rewarming periods without neurologic damage. Hibernating AGS tolerate traumatic brain injury in vivo, and acute hippocampal slices from hibernating animals tolerate oxygen and glucose deprivation. It remains unclear, however, if neuroprotection results from intrinsic tissue properties or from differences in response to acute trauma associated with slice preparation. The goal of this work was therefore to determine whether an intrinsic tissue tolerance persists in chronic culture of AGS hippocampal slices at 37 °C. A second goal was to address N-methyl-D-aspartate (NMDA) receptor involvement and channel arrest as potential mechanisms of intrinsic tissue tolerance. Baseline neuronal survival and tolerance to oxygen and nutrient deprivation (OND), an in vitro model of ischemia–reperfusion, were assessed in the CA1 region of hippocampal slices from juvenile, hAGS and interbout euthermic AGS (ibeAGS). Early in culture (insult onset at 3 h), slices from both hAGS and ibeAGS tolerate OND (4h deprivation followed by 20 h recovery) and 500 μmol/L NMDA plus 20 mmol/L KCl. Later in culture (insult onset at 24 h), tolerance persists in slices from hAGS but not in slices from ibeAGS. Ouabain (Na+K+ATPase inhibitor) administered 24 h in culture enhances survival of slices from hAGS (assessed 24h later). Thus, tolerance to OND in slices from hAGS is due to intrinsic tissue properties likely involving NMDA receptors and ion channel arrest.
Introduction
Stroke is the third leading cause of death in the US and the leading cause of adult disability worldwide, yet the only clinically approved therapeutic for stroke, recombinant tissue plasminogen activator (rt-PA), is indicated for use in less than 4% of stroke victims (Kleindorfer et al, 2004; Arora et al, 2005). Although experimental findings of neuroprotective therapies have been promising, the majority have failed in clinical trials. A novel approach, such as investigation of endogenous mechanisms in hibernating mammals, is well warranted for further development of stroke therapeutics.
Arctic ground squirrel (AGS) is a hibernating species that tolerates extreme fluctuations in blood flow, heart and respiratory rate (RR), and cerebral perfusion along with profound alterations in metabolism, immune response and core body temperature (T b) (Drew et al, 2001) with no evidence of neuronal pathology (Ma et al, 2005). Arctic ground squirrel rewarm to an interbout euthermic state intermittently throughout the hibernation season for approximately 24 h periods between 1- and 3-week torpor bouts.
We have previously shown that hibernating AGS (hAGS) are tolerant to traumatic brain injury (Zhou et al, 2001). This tolerance could be due to intrinsic tissue properties, circulating factors or hypothermia. Tolerance to oxygen and glucose deprivation (OGD) has been shown in acute hippocampal slices isolated from hibernating Thirteen-lined ground squirrel (TLS), S. tridecemlineatus (Frerichs and Hallenbeck, 1998). However, in this acute preparation, it was unclear if enhanced tolerance to OGD in slices from hibernating TLS was influenced by tolerance to acute trauma associated with slice preparation. Thus, the objective of the present study was to investigate whether an intrinsic tissue tolerance persists in a chronic culture of hippocampal slices of AGS at 37°C. An additional objective was to begin to address what mechanisms might be involved in this tolerance. Downregulation of N-methyl-D-aspartate (NMDA) receptor activity is a means of protection from excitotoxicity that occurs during ischemia. ‘Channel arrest’, proposed by Hochachka (1986), is a mechanism used by anoxia tolerant species to maintain ion homeostasis with minimal Na+K+ATPase activity. Therefore, we used NMDA-induced excitotoxicity and Na+K+ATPase inhibition to determine whether regulation of NMDA receptor (NMDAR) activity and channel arrest promote intrinsic tissue tolerance in AGS.
Here, we assess neuronal survival in the CA1 region of AGS hippocampal slices in response to oxygen and nutrient deprivation (OND), an in vitro model of ischemia–reperfusion. For the first time, we show that hippocampal slices from hAGS possess an intrinsic tissue tolerance to OND that persists in culture, and we provide evidence for roles of NMDAR modification and ion channel arrest in this tolerance.
Materials and methods
Animals Groups and Determination of Hibernation Status
The Institutional Animal Care and Use Committee of the University of Alaska Fairbanks (UAF) approved all procedures. Female Wistar rats were obtained from Simonsen Laboratories (Gilroy, CA, USA), quarantined for at least 7 days, and kept at an ambient temperature (Ta) of approximately 21°C until euthanasia. Arctic ground squirrel were trapped on the northern slope of the Brooks Range, Alaska, approximately 20 miles south of the Toolik Field Station of UAF (68°38′ N, 149°38′ W; elevation 809 m) in July 2003 and 2004 under permit from Alaska Department of Fish and Game. On arrival at UAF, AGS were screened for salmonella and quarantined for at least 14 days. All AGS were housed individually in cages at Ta of approximately 18°C, fed approximately 40 g of Mazuri Rodent Chow per day, and kept on natural lighting for 64 latitude where the light:dark cycle changes over days from 20:4 h to 16:8 h.
In early fall, AGS were fed 10 to 15 sunflower seeds each day for 2 weeks before being moved to environmental chambers, where they were housed at Ta of approximately 2°C and fed rodent chow ad libitum. Interbout euthermic AGS were compared with hAGS since both groups were seasonally matched, cold adapted, experienced prolonged torpor bouts of regular frequency and comparable duration, and were of similar age and weight. To avoid preconditioning effects of anesthesia, abdominal transmitters were not implanted. Instead, the ‘shavings added’ method was used to monitor the state of AGS. Shavings are placed on the back of the AGS and checked every 24 h. An AGS is considered ‘torpid’ (hibernating) if the shavings remain on its back or considered to have been through an ‘arousal’ if the shavings are disturbed or missing. This ‘shavings added’ or ‘sawdust method’ is a reliable indicator of hibernation (Lyman, 1948; Pengelley and Fisher, 1961; Willis et al, 1971). Additionally, RR and responsiveness of each AGS were recorded daily for further validation of the hibernation state. An AGS is defined as responsive if it reacts to a touch stimulus, given only if animal is motionless in the environmental chamber. Total number of days torpid, number of bouts, and average bout length were monitored. Here, we define ‘hibernating’ animals as those in the state of prolonged torpor.
After AGS had been through at least three regular hibernation bouts lasting a minimum of 4 days each and three periods of interbout euthermy lasting approximately 24 h each, AGS were considered for use in slice experiments. Arctic ground squirrel that had been in their current hibernation bout for at least 2 consecutive days were confirmed as hibernating and then checked again 12 to 16 h later for evidence of persistent torpor or arousal. Natural arousal (interbout euthermy) was confirmed at the time of euthanasia using the following criteria: RR greater than 80 breaths per minute, animal quickly responsive, and rectal temperature greater than 35.5°C. The state of hAGS at time of euthanasia was confirmed using the following criteria: RR less than 5 breaths per minute, animal not responsive to touch, and rectal temperature less than 5°C. Hippocampal slice cultures were prepared from interbout euthermic (ibeAGS, n = 8) and hibernating (hAGS, n = 8) AGS. Arctic ground squirrel were female juveniles (5- to 9-month-old) that were not sexually mature. Hippocampal slice cultures were prepared from female, 28- to 34-day-old rats (n = 8) as a positive control when slices from AGS failed to respond to insult.
Slice Preparation
Before slice preparation, 1 ml of Neurobasal™ Adult media (NB; GIBCO, Grand Island, NY, USA) supplemented with antioxidant free B-27 serum substitute (GIBCO, Grand Island, NY, USA), plus 0.025 mmol/L glutamate, 0.5 mmol/L glutamine and 1% streptomycin–penicillin (Sigma, St Louis, MO, USA) was placed in each well of a six-well plate. A 30-mm diameter Millicell-CM (Millipore, Bedford, MA, USA) organotypic insert was placed in each well, and plates were pre-equilibrated for 1 h at 37°C with 5% CO2 and 99% humidity in a US Autoflow CO2 Water-Jacketed Incubator (NuAire, Plymouth, MN, USA).
Interbout euthermic AGS and rats were lightly anesthesitized with 5% halothane mixed with oxygen at a constant flow rate of 1.5 L/min, maintained on 1% to 3% halothane while rectal body temperature and weight were recorded, and then decapitated. Hibernating Arctic ground squirrel were not anesthesitized before decapitation as it is difficult to anesthetize hibernating animals to the same degree as euthermic animals due to profound differences in RR, pharmacokinetics, and response to anesthetics. Sterile technique was used throughout slice preparation. Whole brain was isolated, and hippocampi were dissected out on a chilled plate containing ice cold Hibernate™ Adult media (HIB; Brain Bits, Springfield, IL, USA) and then embedded in 3% agar. Hippocampi were kept in ice cold HIB, while slices (300 μm) were cut using a vibraslicer (World Precision Instruments, Inc., Sarasota, FL, USA). One slice was placed on each insert, and slices were cultured at 37°C with 5% CO2 and 99% humidity in the incubator.
Baseline Survival and Histology of Slices
At 3, 24 and 48 h in culture, two triplicate sets of slices were fixed in 4% buffered paraformaldehyde (PF) for 4 h at room temperature (RT) and stored in 70% EtOH at 4°C. Immunocytochemistry (ICC) for NeuN, a neuron-specific nuclear protein (Mullen et al, 1992) was performed in one of these triplicate sets to assess neuronal density and integrity in the cultured slices. Remaining slices were processed using a tissue processor (Shandon Citadel 2000, Thermo Electron Corp., Pittsburgh, PA, USA) on a modified biopsy schedule (15 mins per station), embedded in paraffin, cut at a thickness of 5 μm, and stained with hematoxylin and eosin (H&E).
Analysis and Quantification of Neuron Survival
To identify neurons, free floating ICC for NeuN was performed in slices using a protocol adapted from Bengzon et al (1997). Slices were kept at RT in solution with gentle rotation during incubations and washes. Slices were incubated for 1 h in 10% normal horse serum (NHS; Vector Laboratories, Burlingame, CA, USA) and 0.40% Triton × in PBS followed by overnight incubation in anti-NeuN monoclonal antibody (Chemicon, Temecula, CA, USA) at a 1:100 dilution in 5% NHS and 0.10% Triton × in PBS. Slices were then washed with PBS three times for 10mins each and incubated for 1 h in biotinylated horse anti-mouse IgG at a 1:100 dilution (Vector Laboratories, Burlingame, CA, USA). Slices were washed with PBS three times for 10mins each, incubated for 1h in avidin D-Texas Red at a 1:100 dilution (Vector Laboratories, Burlingame, CA, USA), kept in the dark, rinsed with PBS three times for 10mins each, and coverslipped on slides with Vectashield mounting media (Vector Laboratories, Burlingame, CA, USA). Using a LSM 510 Laser Scanning Confocal microscope (Zeiss, Thornwood, NY, USA), three images across CA1 were taken in the middlemost layer of neurons in the slice using a × 40 objective. Neuron survival was quantified by counting NeuN positive cells in nine oval regions of interest placed along CA1 (i.e., 3 ovals per × 40 image).
Analysis and Quantification of Cell Death
Propidium iodide (PI; Molecular Probes, Eugene, OR, USA) staining was used to quantify percent cell death in the CA1 region of the slices. Since PI can only penetrate cells with a permeable membrane, it is used as an indicator of dead or damaged cells. Triplicate slices were assessed with PI at time points of 3, 24 and 48 h in culture or following insults. Slices were kept on inserts throughout entire PI staining procedure. Slices were incubated in 1.5 μmol/L PI in NB at 37°C for 1 h before imaging using a Zeiss Axioplan 2 Imaging microscope equipped with Axiovision 3.0.6.1 software (Zeiss USA, Thornwood, NY, USA). A × 2.5 objective was used to allow visualization of the entire slice in one field. Zeiss's Atto Arc 2 HBO 100W mercury lamp at 5% output was used with a Texas Red filter to obtain PI fluorescent images while a halogen bulb was used to obtain bright field images corresponding to each fluorescent image. After imaging, slices were immediately fixed in 4% PF for 4 h at RT (to permeabilize all cell membranes) and then restained with 1.5 μmol/L PI in PBS for 1 h at RT. Texas Red fluorescent images indicating maximum PI fluorescence (100% cell death) and bright field images were then acquired.
Percent cell death in the CA1 region of each slice was quantified as follows: %CELL DEATH = (FA-FC)/(FW-FY) × 100 where FA is the PI fluorescence of the test condition, FW is the maximum PI fluorescence, and FC and FY are the background PI fluorescence of the insert for FA and FW, respectively (Strasser and Fischer, 1995; Laake et al, 1999). Image J 1.30V software (NIH, Bethesda, MD, USA) was used to manually outline regions of interest on the bright field image, overlay the region of interest on the PI fluorescence image, and then calculate the average PI fluorescence intensity within each region of interest.
Oxygen and Nutrient Deprivation
Triplicate slices from each animal were subjected to OND either 3 or 24 h in culture. For OND, media was removed from the well and slice was submerged in PBS (1 ml below the insert and 1 ml on top of the slice) that had been bubbled with N2 gas for at least 10 mins. The six-well plate was quickly wrapped with parafilm, then Saran wrap, put in a Ziploc bag, and placed in the incubator similar to the procedure described by Frantseva et al (1999). After 4h of OND, PBS was removed and replaced with fresh NB for a 20 h recovery period after which slices were assessed with PI. For control (CON) conditions, media was changed at both time points at which OND and fresh media were administered to other slices. Oxygen partial pressure (PO2) was measured at 37°C with a Clarke type oxygen electrode (5300 Biological Oxygen Monitor, Yellow Springs Instrument Co., Inc., Yellow Springs, OH, USA) in normoxic media and in PBS immediately after bubbling with N2 and after 1.5 and 4h in the 37°C incubator.
Excitotoxicity Via N-methyl-D-aspartate
Triplicate slices were treated with 500 μmol/L NMDA (Sigma, USA) plus 20 mmol/L potassium chloride (NMDA/KCl) or vehicle (VEH) for 24 h commencing at 3 or 24 h in culture and assessed with PI 24 h later. Neurobasal™ Adult media was removed from each well and replaced with an equal volume of NB containing NMDA/KCl or VEH.
Na+K+ATPase Inhibition Via Ouabain
Triplicate slices were treated with 100 μmol/L ouabain (OUA, Sigma, St Louis, MO, USA) for 30mins commencing 24h in culture and assessed 24h later with PI. After OUA treatment, slices were washed, and inserts placed in fresh NB. Media of CON slices was changed at the same time as was the media for slices given OUA.
Statistical Analysis
Data were analyzed with two-way ANOVA, and significant main effects or interactions were followed by Student–Newman–Keuls post hoc analysis using Sigma-Stat 3.0 (Systat Software, Inc., Point Richmond, CA, USA). Level of significance was P < 0.05. Data are expressed as mean ± s.e.m.
Results
We evaluated group characteristics of animals from which hippocampal slices were prepared (Table 1). There were no statistically significant differences between body weight, total days torpid, number of bouts, or bout length of hAGS and ibeAGS. As expected, Tb and RR were decreased in hAGS compared with ibeAGS.
Characteristics of animal groups
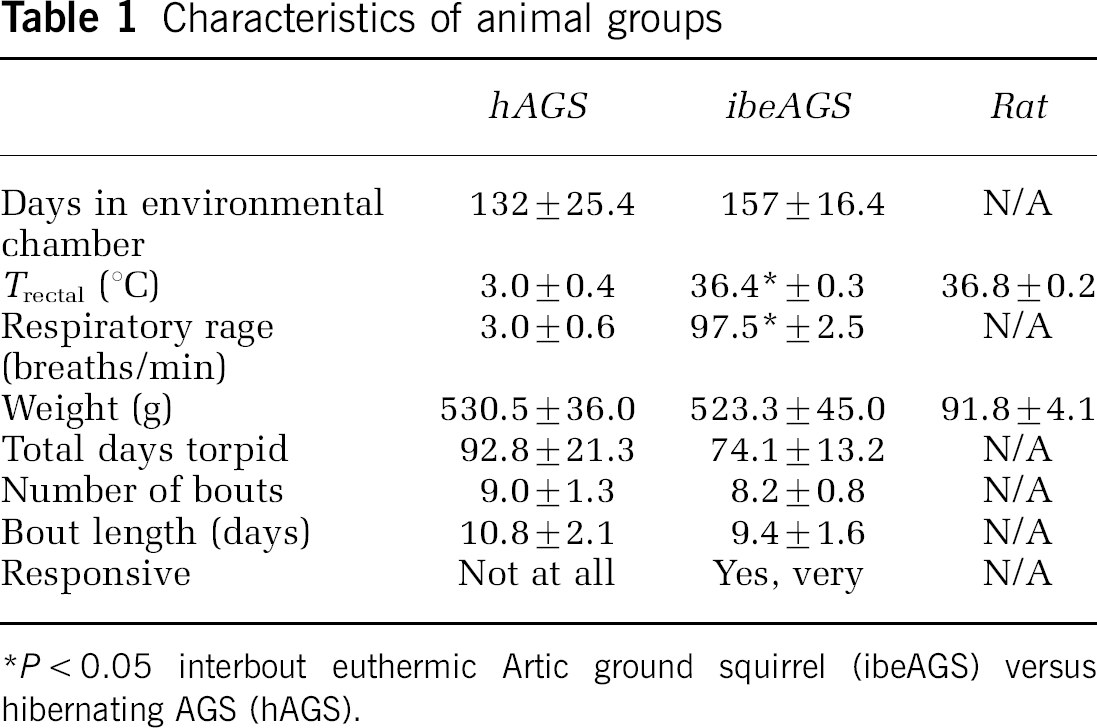
P < 0.05 interbout euthermic Artic ground squirrel (ibeAGS) versu hibernating AGS (hAGS).
As expected from mature hippocampus, baseline cell death was approximately 50% in this chronic culture. However, intensity of PI fluorescence (Figure 1A) or number of NeuN positive cells (Figures 1B and 2) did not change significantly in slices from hAGS or ibeAGS over 48 h in culture. While NeuN and PI were not meant to be directly comparable, results from both measurements suggest that cell viability remains constant from 3 to 48 h of culture. Additionally, there was no significant difference in baseline CA1 PI intensity or neuronal density of slices from hAGS compared with slices from ibeAGS over 48 h in culture. Also as expected from the cell death data, H&E staining of AGS slices cultured for up to 48 h showed some ghost or pyknotic neurons but normal morphology in remaining neurons (data not shown).
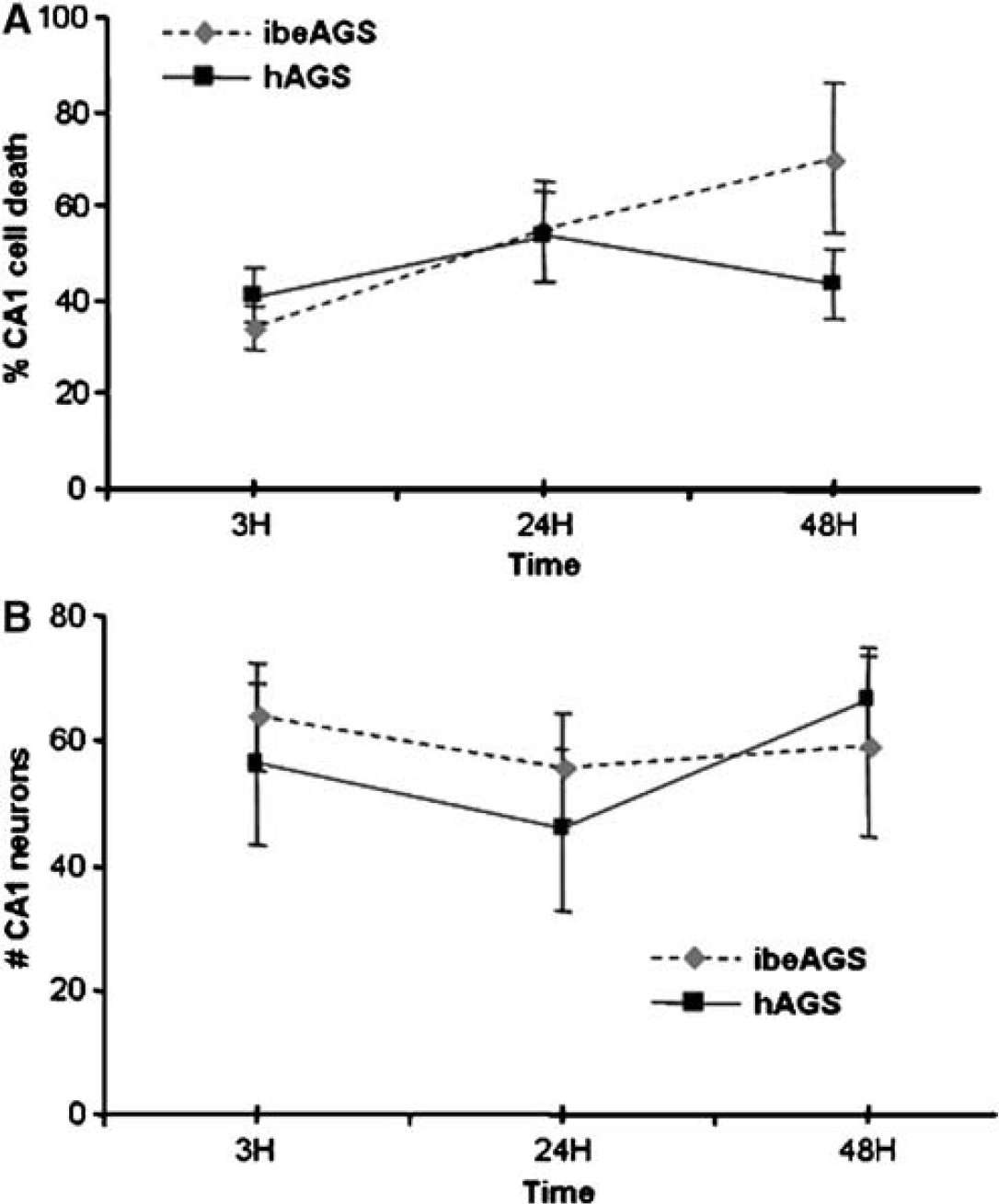
Stability of chronic 37°C culture of Arctic ground squirrel (AGS) hippocampal slices. No significant change in baseline survival of CA1 neurons in either slices from hibernating AGS (hAGS) or slices from interbout euthermic AGS (ibeAGS) over 3 to 48 h in culture is shown by (
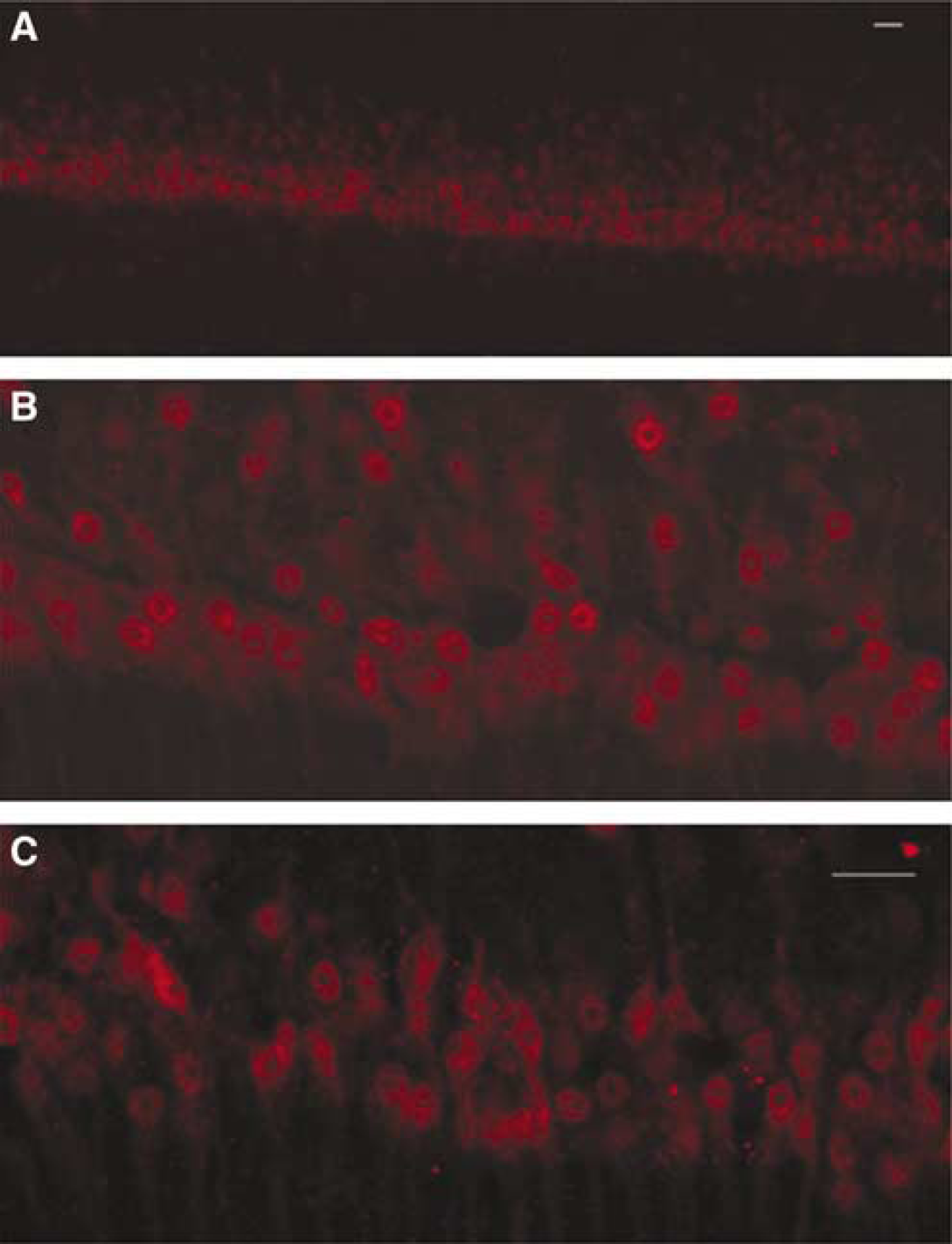
Identification of neurons in cultured AGS slices via immunocytochemistry for NeuN. Representative images of CA1 are shown; all images used for quantification were acquired in the center of the slice using confocal microscopy. Three images taken along CA1 using a × 40 objective were used for counting. (
Oxygen partial pressure was measured in normoxic media and in deoxygenated PBS to determine the degree of hypoxia to which the slices were exposed. Oxygen partial pressure was found to be 154.5 ± 7.0 mm Hg in normoxic media, 12.7 ± 2.3 mm Hg in PBS immediately after bubbling with N2, 126.2 ± 4.3 mm Hg in PBS after 1.5 h in the incubator, and 131.7 ± 1.2 mm Hg in PBS after 4 h in the incubator. Oxygen partial pressure in deoxygenated PBS was significantly less than in normoxic media at all time points tested (P < 0.05 compared with normoxia control, t-tests, n =3). Therefore, slices were exposed to severe hypoxia at the onset of OND at a level similar to that reported previously (Bonde et al, 2002, 2003a, b; Frantseva et al, 1999), and slices were exposed to less severe hypoxia over the duration of treatment.
We found that OND did not produce a significant increase in CA1 neuronal death in slices from hAGS or ibeAGS early in culture (Figure 3A). However, when OND commenced 24 h in culture, a significant increase in cell death was observed in slices from ibeAGS but not in slices from hAGS (Figure 3B), indicating that the combined hypoxia exposure and nutrient deprivation was sufficient to induce cell death in this group.
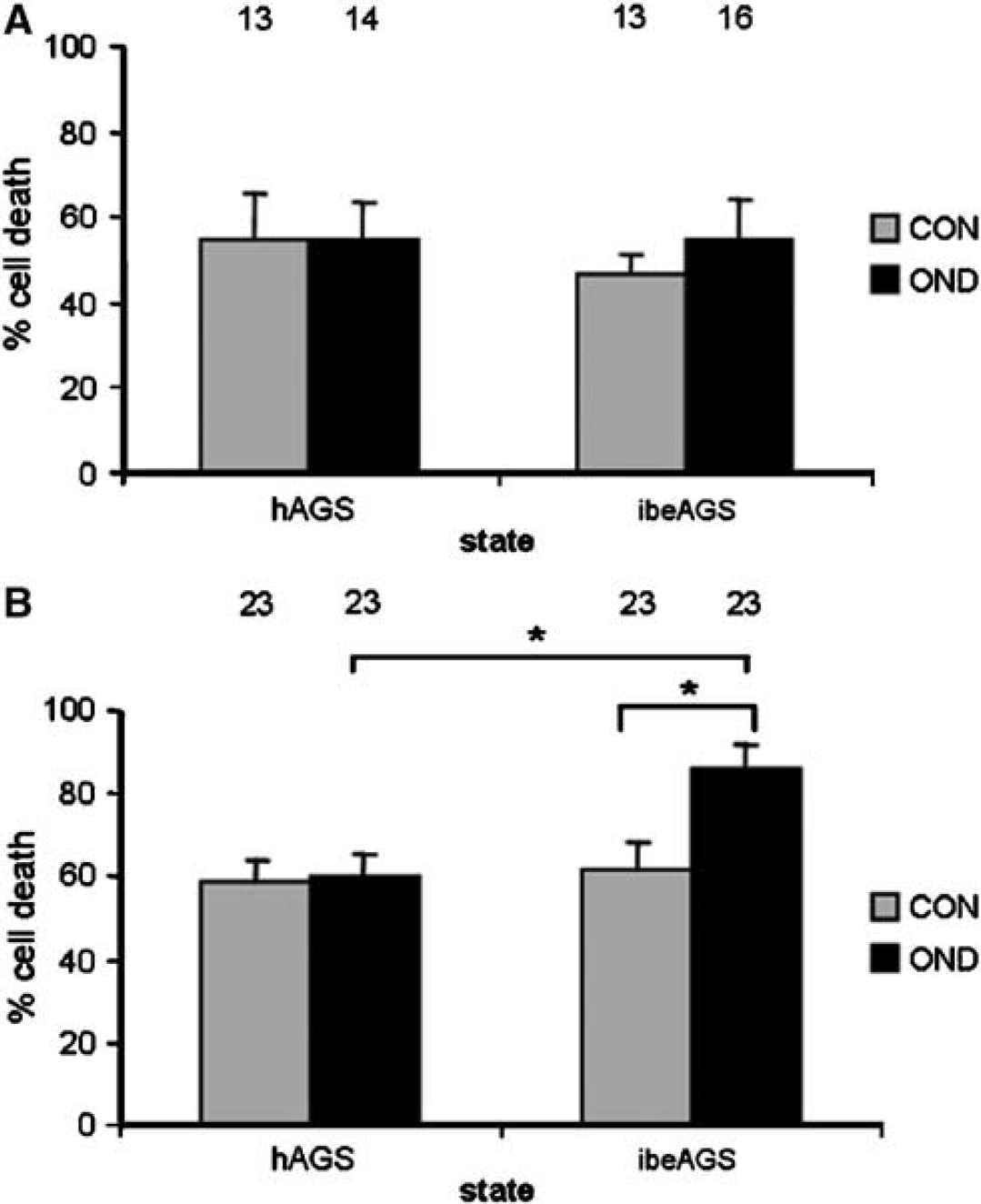
Intrinsic tissue tolerance is present in CA1 of slices from ibeAGS and hAGS early in culture and persists in slices from hAGS later in culture. Tolerance of CA1 to oxygen and nutrient deprivation (OND) is assessed with PI. (
Because excitotoxicity is a key mechanism of ischemia-induced neuronal cell death, we investigated whether AGS slices tolerate NMDA/KCl. Preliminary data indicated AGS slices tolerated high doses of NMDA without significant cell death, so 20 mmol/L KCl was added to remove Mg2+ block of NMDAR and enhance excitotoxicity. N-methyl-D-aspartate/potassium chloride administered 3 h in culture and assessed 24 h later failed to produce an increase in CA1 neuronal death in slices from hAGS or ibeAGS (Figure 4A). In contrast, rat slices, used as a positive control, showed a significant increase in cell death with NMDA/KCl. Rat slices were not used as a species with which to compare to AGS but were instead used to show that the dose of NMDA and KCl was indeed high enough to cause a significant increase in cell death in a more established experimental model. Later in culture (24h), slices from ibeAGS, but not slices from hAGS, showed a significant increase in NMDA/KCl induced cell death (Figure 4B).
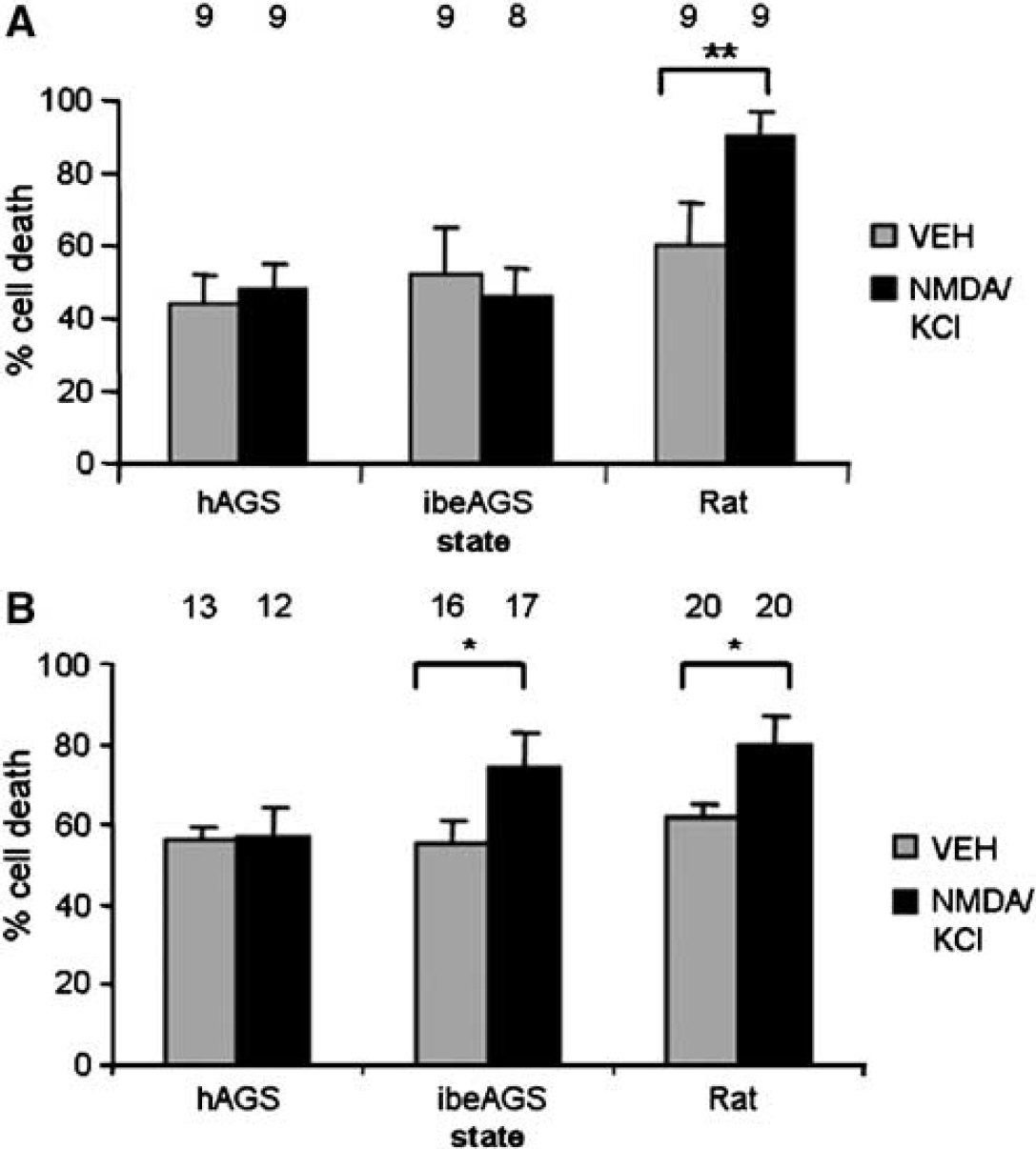
Evidence of protection in AGS at the N-methyl-D-aspartate (NMDA) receptor level. CA1 of slices from both hAGS and ibeAGS tolerate 500 μmol/L NMDA plus 20 mmol/L KCl (NMDA/KCl) early in culture; this tolerance persists in slices from hAGS later in culture. N-methyl-D-aspartate/potassium chloride is administered for 24 h and assessed with PI. (
To investigate evidence for additional channel modifications, specifically channel arrest, which could contribute to the persistent tolerance seen in slices from hAGS, we used OUA to inhibit Na+K+ATPase activity. The relatively high baseline PI intensity in cultures of mature hippocampus was taken as evidence of membrane disruption. Thus, we expected that Na+K+ATPase was active and was therefore a significant sink for ATP in these cultures. We therefore predicted that inhibiting Na+K+ATPase would conserve ATP and enhance survival if Na+K+ATPase was not necessary for ion homeostasis. As predicted, slices from hAGS showed significantly less CA1 neuronal cell death than slices from ibeAGS after OUA administration at 24 h in culture (Figure 5).
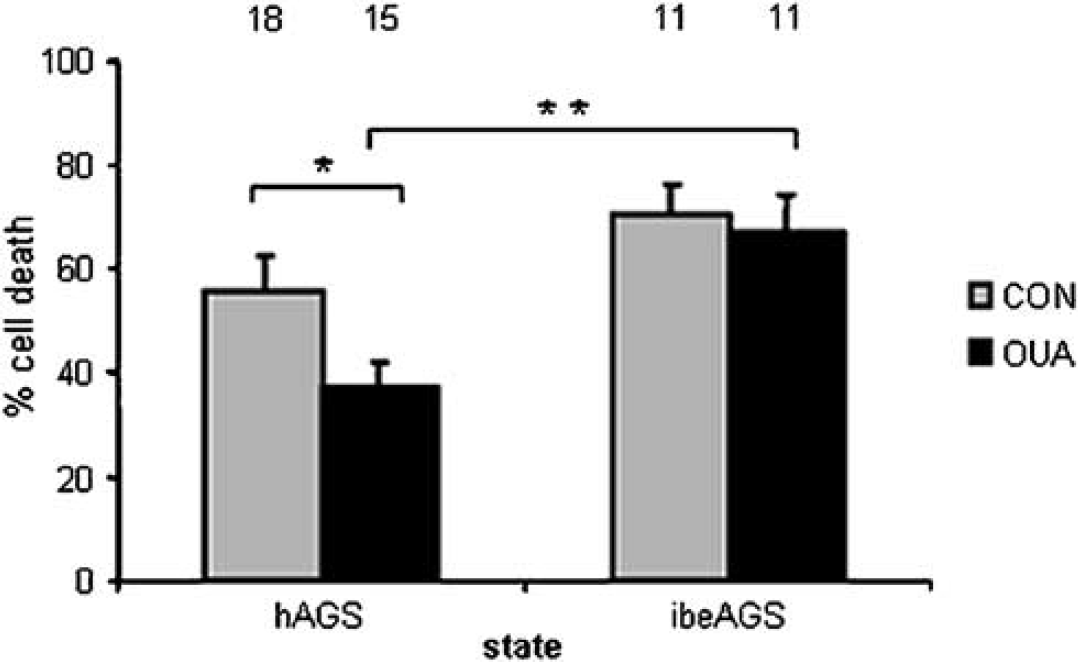
Suggested role of channel arrest as a protective mechanism in hAGS. CA1 of slices from hAGS survive significantly better than CA1 of slices from ibeAGS with Na+K+ATPase inhibition (100 μmol/L ouabain, OUA, given for 30 mins) at time 24 h in culture and assessed with PI 24 h later. In metabolically challenged, cultured AGS slices, preservation of ATP due to decreased Na+K+ pump activity would result in better survival only if decreased ion flux, due to channel arrest, was also present. Main effect of state P < 0.001, *P <0.05, and **P < 0.005 (4 hAGS and 3 ibeAGS). Numbers above bars indicate number of slices studied.
Discussion
Here for the first time, we report evidence of intrinsic tissue tolerance in hippocampal slices from hAGS that persists in culture. We show that slices from hAGS and ibeAGS both tolerate OND and excitotoxicity (NMDA/KCl) early in culture, and that tolerance to OND and excitotoxicity persists in slices from hAGS, but not in slices from ibeAGS. Additionally, we show that slices from hAGS survive significantly better than slices from ibeAGS with Na+K+ATPase inhibition by OUA. Taken together, our data suggest that modulation of NMDAR and maintenance of ion homeostasis contribute to intrinsic tissue tolerance in AGS.
Using chronic slice culture, we confirmed that protection in hAGS is not the result of differences in acute trauma associated with slice preparation and in this way extend earlier work by Frerichs and Hallenbeck (1998). Additionally, the chronic preparation allowed maximum manifestation of delayed cell death that occurs 24h after ischemia or excitotoxicity (Kirino, 1982; Wallis and Panizzon, 1995). Furthermore, the use of a chronic slice culture minimized any residual effects of anesthesia that may have been present in the acute time frame after slice preparation.
The present study also revealed that hAGS possess intrinsic tissue tolerance exclusive of the protective effects of hypothermia by using an in vitro model that allowed for manipulation of temperature not possible in hibernating species in vivo. Since AGS do not hibernate until at least 4 months of age, a prolonged organotypic culture using immature neural tissue was not feasible for these studies. As expected, basal neuronal death was high in cultures prepared from mature hippocampus. Accordingly, histologic analysis of cultured slices showed some large intercellular spaces and pyknotic neurons in CA1, likely a result of slice preparation. Nonetheless, H&E and NeuN staining showed that remaining neurons had normal morphology. Furthermore, survival of CA1 neurons in slices from both hAGS and ibeAGS were stable and similar over 48 h in culture. Because similar survival in culture is seen in slices from both hAGS and ibeAGS but tolerance to OND and NMDA only persists in culture in slices from hAGS, different mechanisms are suggested to contribute to tolerance to OND and survival in culture. Oxygen deprivation after submersion in deoxygenated PBS was less severe than would be expected with a hypoxia chamber and subsided to mild hypoxia during the 4 h incubation period. Nonetheless, the degree and duration of hypoxia achieved was sufficient to induce a significant degree of CA1 neuronal cell death in slices from ibeAGS, but not hAGS, when applied after 24 h in culture.
Here, we also show evidence of intrinsic tissue tolerance in AGS hippocampus at the level of the NMDAR. This mechanism likely contributes to the observed tolerance to OND in slices from AGS. In an ischemia-sensitive species, deleterious events downstream of ischemia include loss of energy supply, deterioration of ion homeostasis, neuronal depolarization, and excitotoxicity via activation of NMDAR resulting in excess intracellular Ca2+. In AGS, tolerance to excitotoxicity is present concomitant to tolerance to OND early in culture in slices from both hAGS and ibeAGS and later in culture in slices from hAGS. Anoxia tolerant turtle neurons show protective downregulation of NMDAR activity called ‘silencing’, which can result from dephosphorylation, a process that occurs within minutes of insult (Bickler et al, 2000). Other regulation that can rapidly take place at the NMDAR level includes specialized subunit expression as shown in hypoxia-tolerant neonates (Bickler et al, 2003) and ischemia-tolerant NR2A knockout mice (Zhang et al, 1997) or dissociation of the receptor from the postsynaptic density complex (Aarts et al, 2002). Consistent with our evidence of protection at the level of the NMDAR in AGS, previous findings in hamsters and preliminary results in AGS show downregulation of NMDAR function during hibernation (Igelmund et al, 1996; Zhao HW, Ross AP, Christian SL, Buchholz JN and KL Drew, unpublished observations).
Further research is necessary to elucidate the mechanisms by which NMDAR are modified in AGS. Although clinical trials, to date, of NMDAR antagonists as stroke therapeutics have proved unsuccessful, the NMDAR antagonist memantine has recently shown great promise since it acts as an open channel blocker that reduces excitotoxicity while allowing normal NMDAR function (Lipton, 2004). In treatment of traumatic brain injury, studies have found that beneficial intervention with NMDAR antagonism is very short lived after injury, while delayed NMDAR antagonism exacerbates injury, and delayed NMDAR stimulation improves function (Biegon et al, 2004). These findings, together with our evidence of tolerance at the NMDAR level in AGS, suggest NMDAR modification in AGS may avoid excitotoxicity. This modification would generate a functional receptor that allows only moderate Ca2+ influx consistent with the model proposed by Bickler and Fahlman (2004).
Our findings of tolerance to OND and excitotoxicity in slices from ibeAGS early in culture are supported by evidence from other investigators that heterothermic species express a protected phenotype even when in the euthermic state (and at euthermic temperature of 37°C). Euthermic AGS are able to tolerate global ischemia, induced via cardiac arrest, without loss of CA1 neurons (Dave et al, 2005). Additionally, organs from TLS, whether in the euthermic or torpid state, tolerate cold ischemia–reperfusion injury (Lindell et al, 2005). Furthermore, acute hippocampal slices from euthermic TLS tended to tolerate OGD better than slices from rat at 36°C; however, this tendency was not statistically significant (Frerichs and Hallenbeck, 1998). Taken together these findings suggest that tolerance to ischemia–reperfusion is not exclusively dependent on prolonged torpor.
It is unclear why tolerance persists in hAGS, but not in ibeAGS. Comparable tolerance to OND and excitotoxicity in slices from both ibeAGS and hAGS early in culture could be due to similar or distinct mechanisms that persist longer in slices from hAGS. Further understanding of these protective mechanisms early in culture will be necessary to address this question. Effects of OUA (i.e. Na+K+ATPase inhibition) were only assessed later in culture so it is unknown if channel modifications, in addition to NMDAR modifications, occur in ibeAGS at this early time point.
Since our AGS slices exhibited high baseline PI fluorescence, an indication of permeable membranes, we expected that metabolic demands of in vitro conditions would cause Na+K+ATPase in AGS slices to function at a high rate. Thus, by inhibiting Na+K+ATPase, the presence of reduced ion leakage could be investigated in AGS slices. We showed that slices from hAGS survived significantly better with Na+K+ATPase inhibition than slices from ibeAGS. Na+ K+ pump function was not necessary for survival in hAGS slices since ion homeostasis was likely preserved as a result of channel arrest. Other evidence that channel arrest occurs in hibernating species includes downregulation of voltage-gated Q-type Ca2+ channels in brain of hibernating TLS (Gentile et al, 1996). Moreover, activity of Na+K+ATPase is decreased 40% to 60% in skeletal muscle, liver and kidney of hibernating Golden-mantled ground squirrel (S. lateralis) (MacDonald and Storey, 1999), which when coupled with channel arrest, would be expected to prevent excess consumption of ATP and preserve energy balance. Further studies addressing channel arrest in hibernating species are well warranted and may yield strategies for preserving ion homeostasis and energy balance in species that do not tolerate ischemia.
Channel arrest has been shown in anoxia tolerant turtles (Edwards et al, 1989; Bickler, 1992; Perez-Pinzon et al, 1992; Pek and Lutz, 1997), where decreased membrane ion permeability conserves ATP and prevents depolarization (Chih et al, 1989). Additionally, in anoxia tolerant turtle brain, a profound conservation of energy results from decreasing Na+K+ pump activity (Doll et al, 1993) since maintenance of ion homeostasis in brain consumes 70% of active metabolism (Astrup et al, 1981). Downregulation of voltage-gated sodium channels is one mechanism of preserving ion homeostasis without Na+K+ATPase activity in anoxia tolerant turtles (Perez-Pinzon et al, 1992).
In conclusion, we report novel findings that hibernating animals possess an intrinsic tissue tolerance that is likely due to mechanisms involving modifications of ion channels. A model integrating our NMDA and OUA findings in context of tolerance to OND is shown in Figure 6. Our findings complement a recent report from Stenzel-Poore et al (2003) showing that regulation of ion channel activity as well as several other mechanisms associated with protective aspects of hibernation confer enhanced tolerance to ischemia in nonhibernating species after preconditioning. Perhaps heterothermic species, in a nonstate dependent manner, express an endogenous ‘preconditioned’-like tolerance. Other mechanisms in hibernating species that are associated with a more persistent protection may depend on the animal being in a state of prolonged torpor. Enhancement of ‘endogenous neuroprotection’ (Dirnagl et al, 2003) suggests that neuronal injury resulting from ischemia may be attenuated by induction of protective mechanisms intrinsic to but not normally expressed in nonhibernating species. Therefore, in the pursuit of stroke therapeutics, further investigation into protective mechanisms used by hibernating species and application of these mechanisms in nonhibernating species are promising.
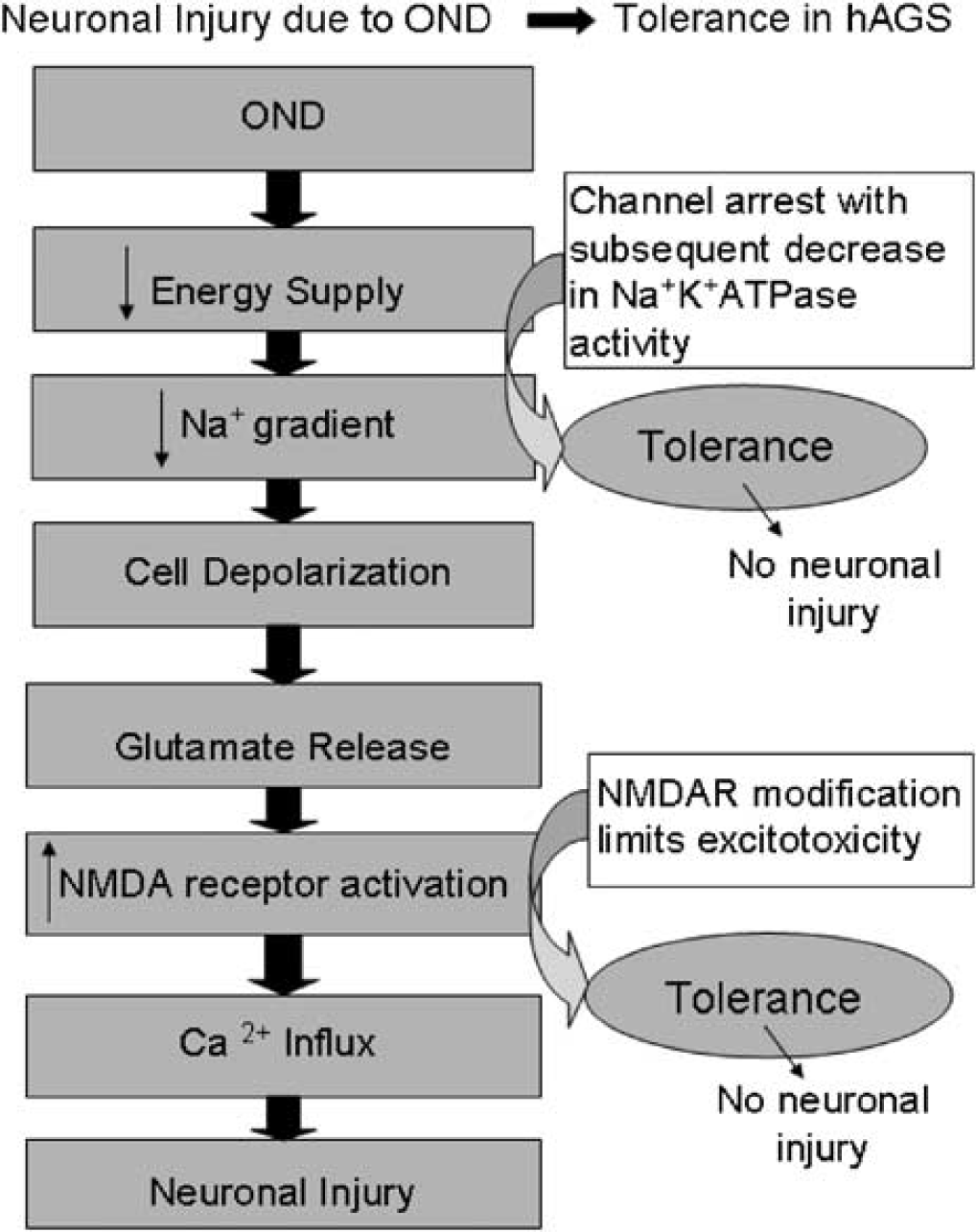
Mechanisms involving ion channel modifications are suggested to contribute to the intrinsic tissue tolerance to OND in hibernating animals. By maintaining ion homeostasis (potentially due to channel arrest) and protection from excitotoxicity (potentially due to NMDAR modifications), a tolerant animal such as a hibernating species is protected from the deleterious cascade of events resulting from OND.
Footnotes
Acknowledgements
The authors thank Ron Barry for discussion on statistical analysis, John Pender and Kara Nance for computer technical assistance, Johanna Herron for assistance with animal handling, Jennifer Whorton for assistance in histology, and Barbara Taylor for technical assistance and helpful comments on the manuscript.
