Abstract
Central nervous system (CNS) complications of human immunodeficiency virus (HIV) infection remain a serious health risk in HIV/acquired immunodeficiency syndrome despite significant advances in highly active antiretroviral therapy (HAART). Specific drugs used for HAART are substrates for the efflux transport systems, such as the multidrug resistance-associated proteins (MRPs), which are present on brain microvascular endothelial cells (BMEC) and astrocytes, that is, the main cell types that form the blood-brain barrier (BBB). Thus, drugs employed in HAART are actively removed from the CNS and do not efficiently inhibit HIV replication in the brain. To study the potential mechanisms of this process, the aim of the present research was to address the hypothesis that HIV Tat protein can contribute to upregulation of MRP expression at the BBB level. Tat is a protein produced and released by HIV-infected cells, which may play an important role in brain vascular pathology in the course of HIV infection. Among the family of MRPs, exposure to Tat specifically induced MRP1 messenger ribonucleic acid and protein expression both in BMEC and astrocytes. These alterations were accompanied by enhanced MRP1-mediated efflux functions. Furthermore, activation of the mitogen-activated protein kinase signaling cascade was identified as the mechanism involved in Tat-mediated overexpression of MRP1. These results indicate that Tat exposure can lead to alterations of the BBB functions and decrease HAART efficacy in the CNS through overexpression of drug efflux transporters.
Keywords
Introduction
The environment of the central nervous system (CNS) is maintained by the blood-brain barrier (BBB), which exists at the level of brain microvascular endothelial cells (BMEC) and astrocytes. The BBB is relatively impermeable to ions, many amino acids, small peptides, and proteins. In addition, the transcellular passage through the BBB is also limited due to the absence of fenestrations and reduced levels of pinocytic activity in BMEC. While these structural features are important for maintaining the integrity of the BBB, the influence of cellular efflux transport systems that transport selected molecules back into the bloodstream is of critical significance (Iadecola, 2004; Neuwelt, 2004; Begley, 2004; Abbott, 2005).
Among a variety of transport systems in the BBB, the family of the multidrug resistance proteins (MRPs) has recently been identified as the ATP-dependent transporters. Upregulation of these transport systems prevents the drugs from achieving therapeutic concentrations within the CNS. Indeed, MRPs can export a variety of organic anions from the cells, including drugs used for highly active antiretroviral therapy (HAART) (Thomas, 2004). Of particular interest appears to be MRP1, which is involved in the reverse transport of protease inhibitors (Dallas et al, 2004), as well as MRP4 and 5, whose substrates include nucleoside analogues (Chen et al, 2001; Jedlitschky et al, 2000).
The introduction of HAART in the mid-1990s has resulted in a 50% decline of the acquired immunodeficiency syndrome (AIDS) death rate, has decreased maternal—infant transmission rates, reduced rates of opportunistic infections, and has led to 40% to 50% decrease in the incidence of human immunodeficiency virus (HIV)-associated dementia (Sacktor et al, 2001b). In addition, several studies have conclusively shown that HAART can suppress cerebral spinal fluid (CSF) levels of HIV ribonucleic acid (RNA) (Foudraine et al, 1998) and this decline appeared to correlate with the successful reversal of neurological deficit (Ellis et al, 2000; Marra, 1999). However, despite the progress associated with HAART, HIV-associated neurological disease continues to be the major cause of morbidity and mortality in HIV patients, suggesting that HAART does not provide complete protection against neurological damage in HIV/AIDS (Dougherty et al, 2002; Kolson, 2002; Sacktor et al, 2001a). We hypothesize that the development of HAART tolerance in the CNS due to overexpression of the efflux mechanisms can be one of the major mechanisms responsible for these effects.
A compelling line of evidence indicates that the HIV-1 transactivator protein, Tat, can play an important role in the development of the AIDS-associated dementia complex. In fact, Tat was detected in the brains from AIDS patients with progressive multifocal leukoencephalopathy (Valle et al, 2000). Tat-positive mononuclear cells have also been localized to the perivascular regions in the brains of patients with HIV encephalitis (Hofman et al, 1994; Bonwetsch et al, 1999) and in rhesus macaques with Simian/human immunodeficiency virus (SHIV) encephalitis (Hudson et al, 2000). Injections of Tat in rats and mice caused perivascular cuffing with monocytic infiltration, indicating breaches in the BBB (Jones et al, 1998) and disruption of tight-junction proteins (Andras et al, 2005; Pu et al, 2005). Tat can be secreted by HIV-infected cells and taken up by the neighboring uninfected cells (Ensoli et al, 1993; Westendorp et al., 1995; Helland et al, 1991; Marcuzzi et al, 1992). These studies were performed using HIV-infected T-cell lines such as H9 and Jurkat cells, the epithelial cell line COS-1 (Ensoli et al, 1993; Marcuzzi et al, 1992), as well as peripheral blood mononuclear cells isolated from HIV-infected patients (Thomas et al, 1994). Evidence indicates that Tat is released from cells via a leaderless pathway in the absence of permeability changes (Chang et al, 1997). In addition, levels of free Tat have been detected in the serum of HIV-infected patients (Xiao et al, 2000). Regarding CNS localization of Tat, it was shown in HIV-associated encephalitis that Tat-positive immunoreactivity was present primarily in microglia and astrocytes (Bonwetsch et al, 1999). Tat is believed to be one of the potent viral neurotoxins in the pathogenesis of neuronal dysfunction in HIV-infected patients (Nath and Geiger, 1998). In addition, Tat can induce profound vascular changes by its influence on brain endothelial cells (Toborek et al, 2003; Price et al, 2005; Avraham et al, 2004; Wu et al, 2000). For example, exposure to Tat can induce inflammatory responses in BMEC and alter the expression of tight-junction proteins through the vascular endothelial growth factor receptor type 2 (VEGFR-2) and mitogen-activated protein kinase (MAPK) signaling pathways (Andras et al, 2005; Pu et al, 2005).
Owing to the importance of the efflux mechanisms at the BBB level in anti-HIV treatment, the aim of the present study was to evaluate the effects of Tat protein on expression of the MRP family in BMEC and astrocytes. We indicated that Tat can specifically induce MRP1 and that the MAPK signaling is involved in these effects.
Materials and methods
Endothelial Cell Isolation
Primary cultures of BMEC were prepared from male C57BL/6 mice (7 to 8 weeks old; weight 25 to 28 g) obtained from the National Cancer Institute (Bethesda, MD, USA) and cultured according to Deli et al. (1997) as described previously (Hayashi et al, 2005). Briefly, meninges were carefully removed from the forebrain and gray matter was minced and then digested for 2 h at 37°C with 1 mg/mL collagenase type 2 (Worthington Biochemical Corporation, Lakewood, NJ, USA) in Dulbecco's modified Eagle's medium (DMEM; Sigma, St Louis, MO, USA) containing 50 μg/mL gentamycin. The cell pellet was separated by centrifugation (1000g for 20mins) in 20% bovine serum albumin (BSA; Sigma) in DMEM. The microvessels obtained in the pellet were further digested with 1 mg/mL collagenase-dispase (Roche, Mannheim, Germany) in DMEM for 45 mins at 37°C. The microvessel endothelial cell clusters were separated on a 33% continuous Percoll (Amersham, Piscataway, NJ, USA) gradient, collected, and washed twice in DMEM before plating on 35-mm cell culture dishes coated with type IV collagen and fibronectin. Cultures were maintained in DMEM supplemented with 20% plasma-derived bovine serum (Animal Technologies, Inc., Tyler, TX, USA), 1 ng/mL basic fibroblast growth factor (Roche), 5 μg/mL heparin, and 25 μg/mL gentamycin, and incubated at 37°C in a humidified atmosphere of 5% CO2. Brain microvascular endothelial cells can dedifferentiate rapidly and lose their specific characteristics. Therefore, only primary cultures of BMEC were employed in the present study. Cultures were determined to be endothelia by uniform cobblestone morphology and determination of cellular uptake of fluorescent labeled acetylated low-density lipoprotein (DiI-Ac-LDL; Biomedical Technologies Inc., Stoughton, MA, USA).
Astrocyte Isolation
Primary cultures of astrocytes were prepared from neonatal mice as described earlier (Latzkovits et al, 1993). Briefly, meninges were removed and cortical pieces were mechanically dissociated in culture medium (DMEM supplemented with 10% fetal bovine serum and antibiotics) by passage through a 20-gauge needle. Dissociated cells were seeded into cell culture flasks without specific coating and cultured to confluence for approximately 10 days. Nonastrocyte cells were mechanically removed by vigorous shaking of confluent cultures for 2 h at 37°C, followed by a change of cell culture medium. Cultures were immunostained for glial fibrillary acidic protein (GFAP, specific intracellular marker of astrocytes) and used at passage 1.
Antibodies
Monoclonal MRPr1 (anti-MRP1) antibody was purchased from Alexis Biochemicals (San Diego, CA, USA). Anti-estrogen receptor kinase (ERK)1/2 antibody, anti-phospho-ERK1/2 (p-ERK1/2) antibody, anti-p38 antibody, anti-phospho-p38 (p-p38) antibody, and anti-c-Jun N-terminal kinase (JNK) antibody were purchased from Santa Cruz Biotechnology, Inc. Anti-phospho-JNK (p-JNK) antibody was obtained from Cell Signaling Technology, Inc. (Beverly, MA, USA).
Tat exposure
Human immunodeficiency virus Tat protein is encoded by a gene that consists of two exons. The first exon encodes neurotoxic and membrane-interactive domains. Therefore, the present study was performed using recombinant Tat protein derived from the first exon, which encodes for the first 72 amino acids (Tat1 to 72). Tat1 to 72 was synthesized and purified as described previously (Ma and Nath, 1997). Since Tat binds strongly to serum proteins, all experiments were performed in serum-free media. Brain microvascular endothelial cells remained healthy and viable under these experimental conditions.
The plasma levels of Tat in HIV-1-infected individuals were reported to be in the nanomolar range. Westendorp et al (1995) reported plasma Tat levels to be ∼ 1 to 3 ng/mL and the CSF concentrations to be ∼ 16 ng/mL. Recently, Xiao et al (2000) estimated Tat serum levels in HIV-1-infected people between 2 and 40 ng/mL. Although the exact Tat concentrations in brain tissue are not known, one could expect these levels to be markedly higher than CSF. Specifically, local Tat concentrations around HIV-infected perivascular cells (i.e., in the proximity of endothelial cells) could reach high values. We have found that 100 ng/mL of Tat can induce cytokine production in astrocytes and monocytes (Nath et al, 1999), upregulation of cell adhesion molecules (Lee et al, 2004) as well as activation of redox-responsive pathways in brain endothelial cells (Toborek et al, 2003). Therefore, similar concentrations of Tat were used in the present study. These concentrations of Tat also are consistent with literature data (Bonavia et al, 2001; Oshima et al, 2000; Prendergast et al, 2002; Speth et al, 2002).
To determine the specificity of Tat-induced effects, Tat solutions were immunoabsorbed using Tat antisera conjugated to protein-A beads (Magnuson et al, 1995). After centrifugation to remove the precipitated proteins, cells were exposed to this antibody–absorption mixture (AA Tat).
Characterization of the Brain Microvascular Endothelial Cells and Astrocytes
Brain microvascular endothelial cells cultured on chambered Permanox plastic slides (Nalge Nunc International, Naperville, IL, USA) were washed twice with phosphate-buffered saline (PBS), and incubated for 3 h at 37°C with Dil-Ac-LDL (10 μg/mL), then washed three times with PBS and fixed in 4% paraformaldehyde in PBS. The cells were again washed several times with PBS, mounted on glass slides and examined with an epifluorescence Nikon Eclipse E600 microscope. The images were captured using a Spot CCD camera system.
Astrocytes cultured on chambered Permanox plastic slides (Nalge Nunc International) were fixed with ethanol for 30 mins at 4°C. Nonspecific reactions were blocked by normal horse serum and then cells were incubated overnight with primary antibody (anti-GFAP antibody, Calbiochem, San Diego, CA, USA) at 4°C. The excess of primary antibody was removed, slides were incubated with fluorescein isothiocyanate (FITC)-conjugated secondary antibody (Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA) for 1 h at room temperature. After washing with PBS, slides were mounted in aqueous mounting media and coverslipped. Specimens were evaluated under an epifluorescence Nikon Eclipse E600 microscope and images were captured using a Spot CCD camera system.
Real-time Reverse Transcriptase-polymerase Chain Reaction (RT-PCR)
Real-time RT-PCR was employed to determine MRP gene expression both in BMEC and astrocytes. The analyses were performed as described earlier (Lee et al, 2004). Briefly, total RNA was extracted from the culture cells using the RNeasy kit (Qiagen, Valencia, CA, USA) and 1 μg RNA was reverse transcribed using the Reverse Transcription system (Promega, Madison, WI, USA) in a total volume of 20 μL. The following conditions were employed for reverse transcription: 25°C for 10 mins, 48°C for 30 mins, and 95°C for 5 mins. Specific MRP primers and probes were designed using Primer Express 2.0 (Applied Biosystems, Branchburg, NJ, USA) and custom synthesized by Integrated DNA Technologies (Coralville, IA, USA). The sequences of primers and probes are given in Table 1.
Sequences of MRP primers and probes used in real-time RT-PCR analyses

Real-time PCR was conducted on an ABI Prism 7000 system (Applied Biosystems), using 2 μL of reverse transcription product, TaqMan Universal PCR Master Mix (Applied Biosystems), primers, and probe in a total volume of 25 μL. The following thermocycling conditions were employed: 95°C for 10 mins, followed by 95°C for 15 secs, and 60°C for 60 secs. Concentration of each sample was calculated from the threshold cycle (Ct) and normalized according to the β-actin (a housekeeping gene) messenger RNA (mRNA) levels. A standard curve for each gene studied was generated by preparing serial dilutions of the cDNA sample obtained from the control groups.
Immunoblotting
Western blot assay was performed using a standard technique as described previously (Hayashi et al, 2001, 2005). Briefly, homogenates of cultured BMEC were prepared in lysis buffer (20 mmol/L Tris-HCl, pH 7.4, 150mmol/L NaCl, 0.5% Triton X-100, 0.1 μg/mL phenyl-methylsulfonyl fluoride, 0.5% Nonidet 40, 1 mM ethylene diamine tetraacetate (EDTA), 2.5 μg/mL leupeptin, and 1 μg/mL aprotinin). After centrifugation at 15,000g for 15 mins, supernatants were collected and protein concentrations were determined using the Bradford protein assay. Samples (50 μg/lane) were separated on sodium dodecyl sulfate (SDS)-polyacrylamide gel, blotted onto nitrocellulose membranes (Bio-Rad Laboratories, Hercules, CA, USA), and incubated with the primary antibody. After washing with Tris-buffered saline three times, the blots were incubated with secondary antibody, conjugated with horseradish peroxidase (Santa Cruz Biotechnology, Inc.) for 1 h at 20°C. For visualization, immunoblots were analyzed using an ECL Western blot detection kit (Amersham Biosciences, Piscataway, NJ, USA). To quantify the relative levels of protein expression, the intensity of the specific bands was estimated by Scion image analysis software (Scion Corporation, Frederick, MD, USA).
Drug Efflux Assay
The efflux function of MRP1 in BMEC and astrocytes was measured by glutathione bimane (GS-B) assay as described previously (Homma et al, 1999). The cells were seeded onto 24-well culture plates and, after reaching confluence, the cultures were treated with Tat for 12 h. The media was then removed and cells were incubated with monochlorobimane (MCB; 20 μmol/L, Molecular Probes, Eugene, OR, USA) in 0.5 mL of Krebs–Ringer glucose buffer for 60 mins at 37°C. Selected cultures were incubated with the specific MRP1 inhibitor, MK571 (Calbiochem, 50 μmol/L). The incubation was terminated by removing the MCB solution and washing the cells three times with ice-cold PBS. Cells were solubilized with 0.5% Triton X-100. Cellular levels of GS-B were determined by fluorescent measurement (excitation at 386 nm and emission at 476 nm).
Statistical Analysis
All data are expressed as mean 7 standard error of the mean (s.e.m.). Statistical comparisons were made by analysis of variance (ANOVA) with Fisher's protected least significant difference using SigmaStat 2.03 (SPSS, Chicago, IL, USA). Statistical significance was accepted at P < 0.05.
Results
As BMEC and astrocytes are major components of the BBB, cultures of brain endothelial cells and astrocytes have been developed as convenient in vitro model systems for investigating a variety of BBB functions. Figure 1 depicts the characteristics of cultures employed in the present study. Phase-contrast microscopy shows a typical morphology of endothelial cells, such as a fusiform shape and cobblestone appearance (Figure 1A). In addition, they were shown to take up specific fluorescent probe DiI-Ac-LDL (Figure 1B). The tight-junction proteins, such as occludin, claudin-5, and zona occludens-1 (ZO-1), which are the special features of BMEC, are also well developed in these cultures and concentrated primarily at cell–cell borders (Hayashi et al, 2005). Furthermore, transendothelial resistance of the BMEC monolayers used in the present study was more than 100 Ω/cm2, indicating the formation of a functional barrier. Astrocyte cultures were characterized by larger cell size, multipolar shape, and positive immunoreactivity for GFAP (Figures 1C and 1D).
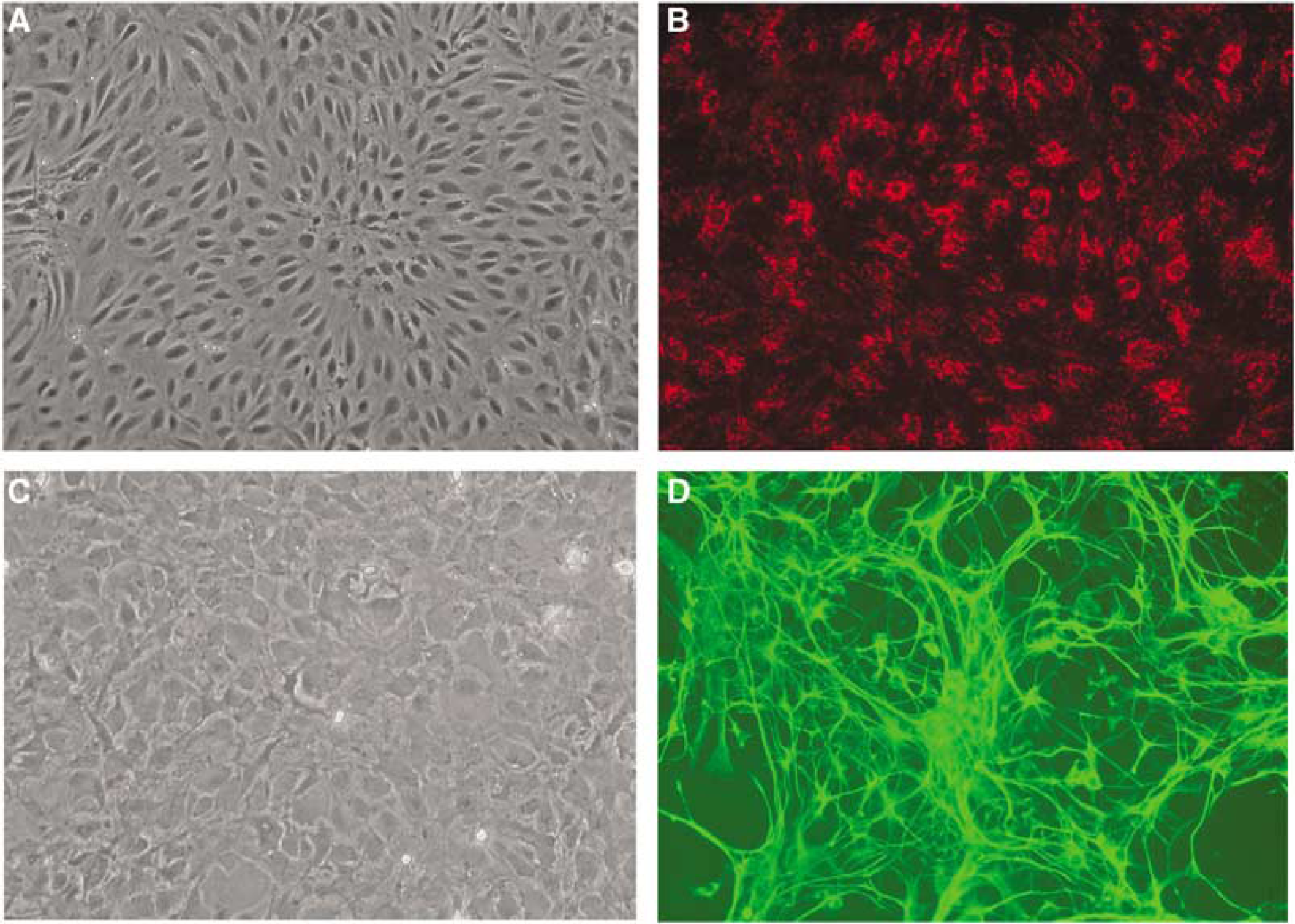
Morphological characterization of primary cultures of BMEC and astrocytes employed in the present study. (
Expression Profile of Genes Encoding for Multidrug Resistance-Associated Proteins in Brain Endothelial Cells and Astrocytes
The expression profile of MRPs in cell types that create the BBB is unknown. Therefore, we determined mRNA levels of MRP1 to 7 in normal cultures of brain endothelial cells and astrocytes, and the results were normalized as the percentage of β-actin expression to compare relative mRNA levels. All seven main members of the MRP family investigated in the present study were expressed at the mRNA levels both in BMEC and astrocytes (Figure 2). However, MRP1 appears to be the dominant MRP isoform both in brain endothelial cells and astrocytes. Other members of the MRP family with high mRNA levels are MRP3 in astrocytes and MRP4 in brain endothelial cells. In contrast, the levels of mRNA expression for MRP2, 5, 6, or 7 are low or only minimal in cells of the BBB.
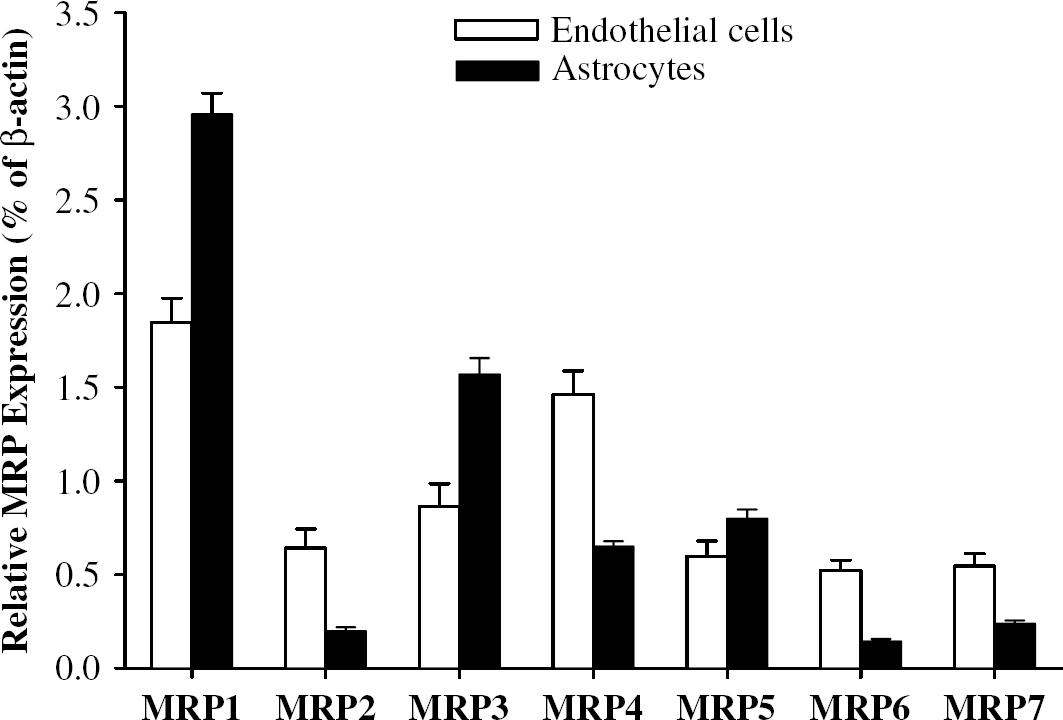
Expression profile of specific MRP genes in BMEC and astrocytes. Total RNA was extracted from confluent BMEC or astrocyte cultures and subjected to real-time RT-PCR analysis. The levels of mRNA for specific MRPs were normalized according to β-actin mRNA expression as described in the Materials and methods. Values are mean ± s.e.m. (n = 4).
Tat Specifically Upregulates Multidrug Resistance-Associated Protein 1 Messenger Ribonucleic Acid Expression in Brain Microvascular Endothelial Cells and Astrocytes
To examine the effects of Tat on MRP expression at the mRNA levels, BMEC and astrocytes were exposed to increasing concentrations of this protein for 6 h and MRP mRNA levels were evaluated using real-time RT-PCR. As reflected in Figure 3A, treatment of BMEC with Tat at the concentration of 200 ng/mL specifically induced MRP1 mRNA expression. In contrast, mRNA levels of MRP2 to 7 in BMEC were not affected by Tat exposure.
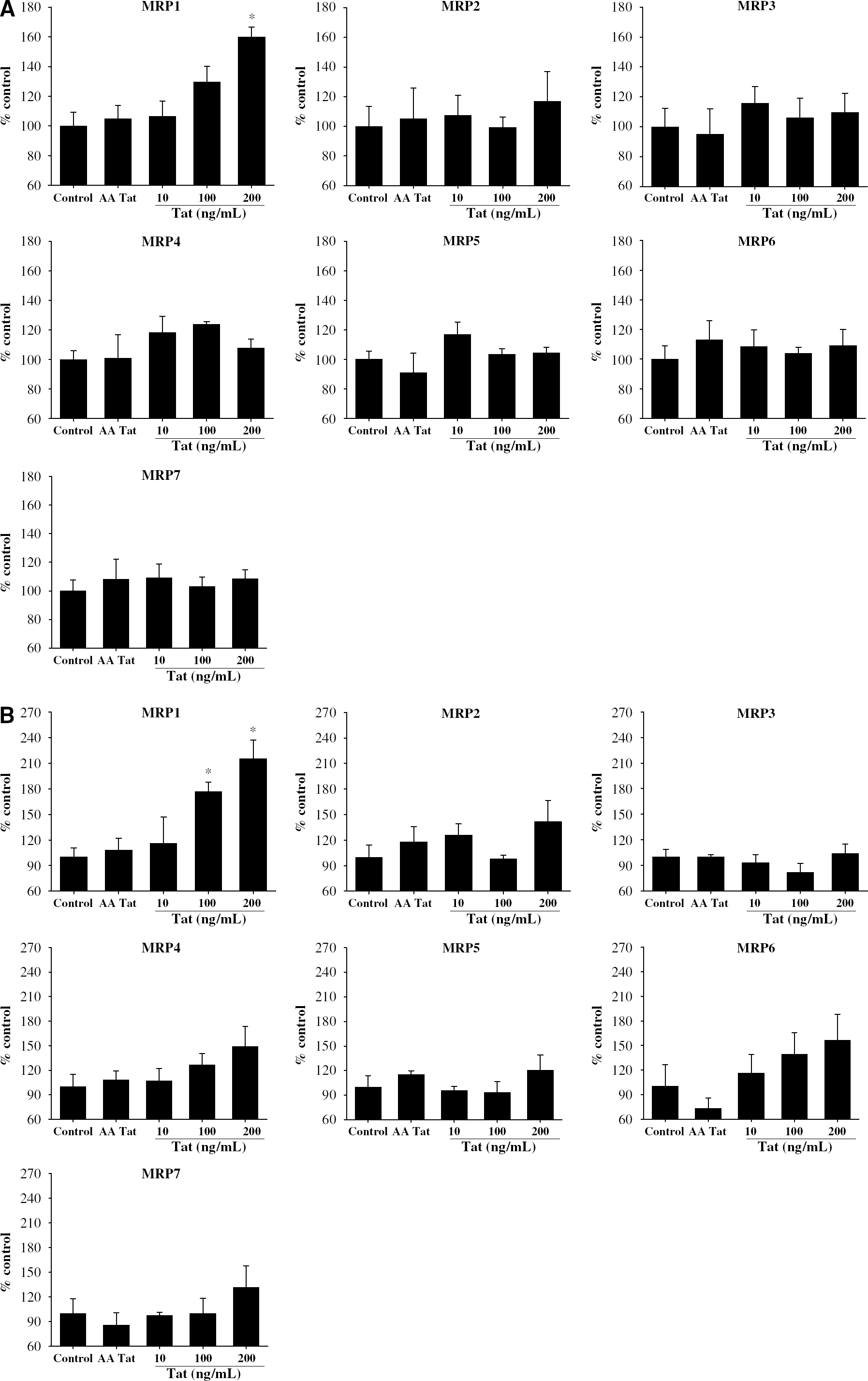
Effects of Tat on MRP mRNA expression in BMEC (
In cultured astrocytes, treatment with Tat at the dose of 100 ng/mL also markedly increased MRP1 mRNA expression. This effect was even higher (approximately a twofold increase) in astrocytes exposed to 200 ng/mL of Tat. Treatment with Tat did not alter mRNA levels of MRP2 to 7.
Immunoabsorbed Tat (AA) was used in all experiments as negative control. As indicated in Figures 3A and 3B, AA Tat did not affect the expression of MRP mRNA levels in BMEC and astrocytes. Therefore, this negative control treatment was discontinued in the remaining experiments.
Tat Stimulates Multidrug Resistance-Associated Protein 1 Protein Expression
As MRP1 mRNA levels both in BMEC and astrocytes were specifically and significantly induced by Tat exposure, our studies on protein expression of the MRP family have focused on MRP1. The dose- and time-dependent effects of Tat on MRP1 protein expression in BMEC and astrocytes as measured by Western blot are reflected in Figures 4A and 4B, respectively. In the dose-dependent experiments, cells were treated with Tat for 12 h. In BMEC, enhanced levels of MRP1 were observed when cells were exposed to Tat at the concentration of 200 ng/mL. Although the levels of Tat as low as 10 ng/mL showed a tendency to increase MRP1 protein levels, these effects were not statistically significant. In the time-dependent experiments, treatment of BMEC with 200 ng/mL of Tat for 6 and 12 h markedly increased MRP1 protein levels (Figure 4A).
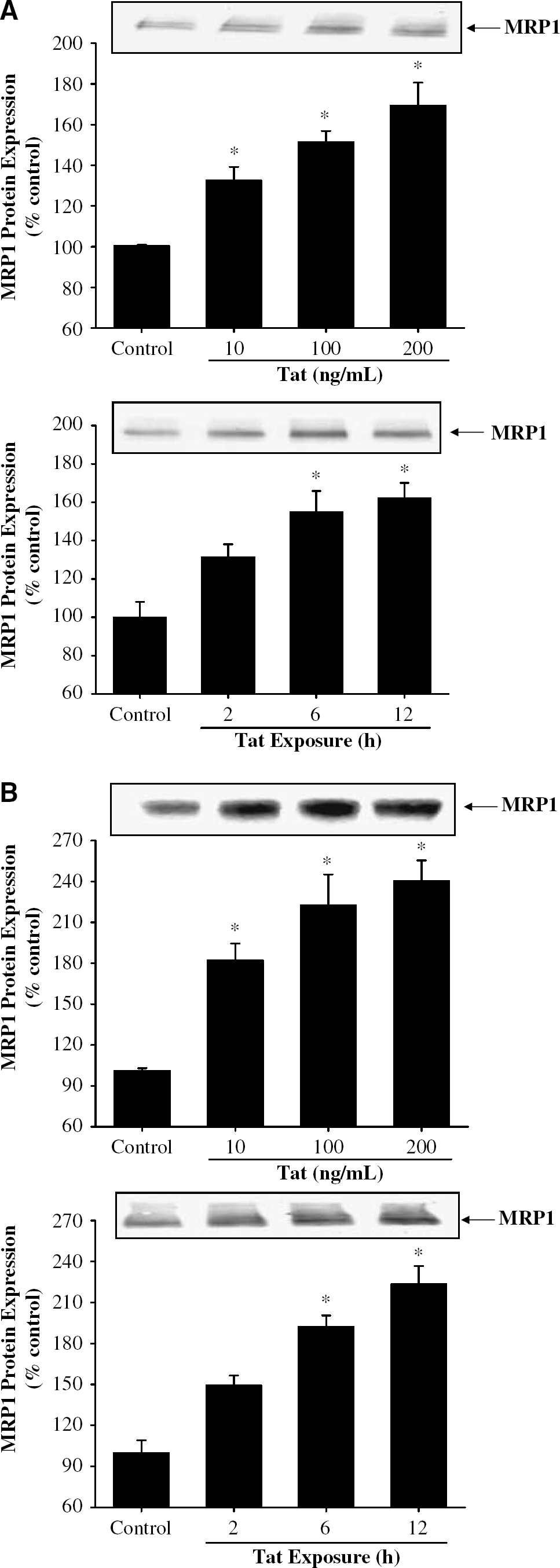
Dose- and time-dependent effects of Tat on MRP1 protein expression in BMEC (
As compared with BMEC, astrocytes appeared to be more susceptible to upregulation of MRP1 protein levels in response to Tat treatment (Figure 4B). Specifically, increased expression of MRP1 was observed in astrocytes treated with Tat at the concentration as low as 10 ng/mL. The maximal increase in MRP1 protein levels was approximately 2.5 times over control values in astrocytes exposed to 200 ng/mL of Tat. Similar as in BMEC, statistically significant upregulation of MRP1 protein in astrocytes was observed as the result of treatment with 200 ng/mL of Tat for 6 or 12 h (Figure 4B).
Tat Upregulates Multidrug Resistance-Associated Protein 1 Efflux Functions in Brain Microvascular Endothelial Cells and Astrocytes
Transporter activity of MRP1 was studied using the GS-B efflux assay. The assay is based on the principle that MCB is conjugated intracellularly with glutathione to produce GS-B. Then, GS-B is transported out of the cells in the MRP1-dependent reaction. As illustrated in Figures 5A and 5B, treatment with Tat at the dose of 200 ng/mL significantly decreased cellular accumulation of GS-B by approximately 30% in BMEC and 40% in astrocytes, indicating an accelerated removal of this dye. To confirm the specificity of this functional assay, BMEC and astrocytes were treated with MK571, a specific inhibitor of MRP1. As expected, MK571 blocked removal of GS-B, which was reflected by markedly increased intracellular GS-B accumulation.
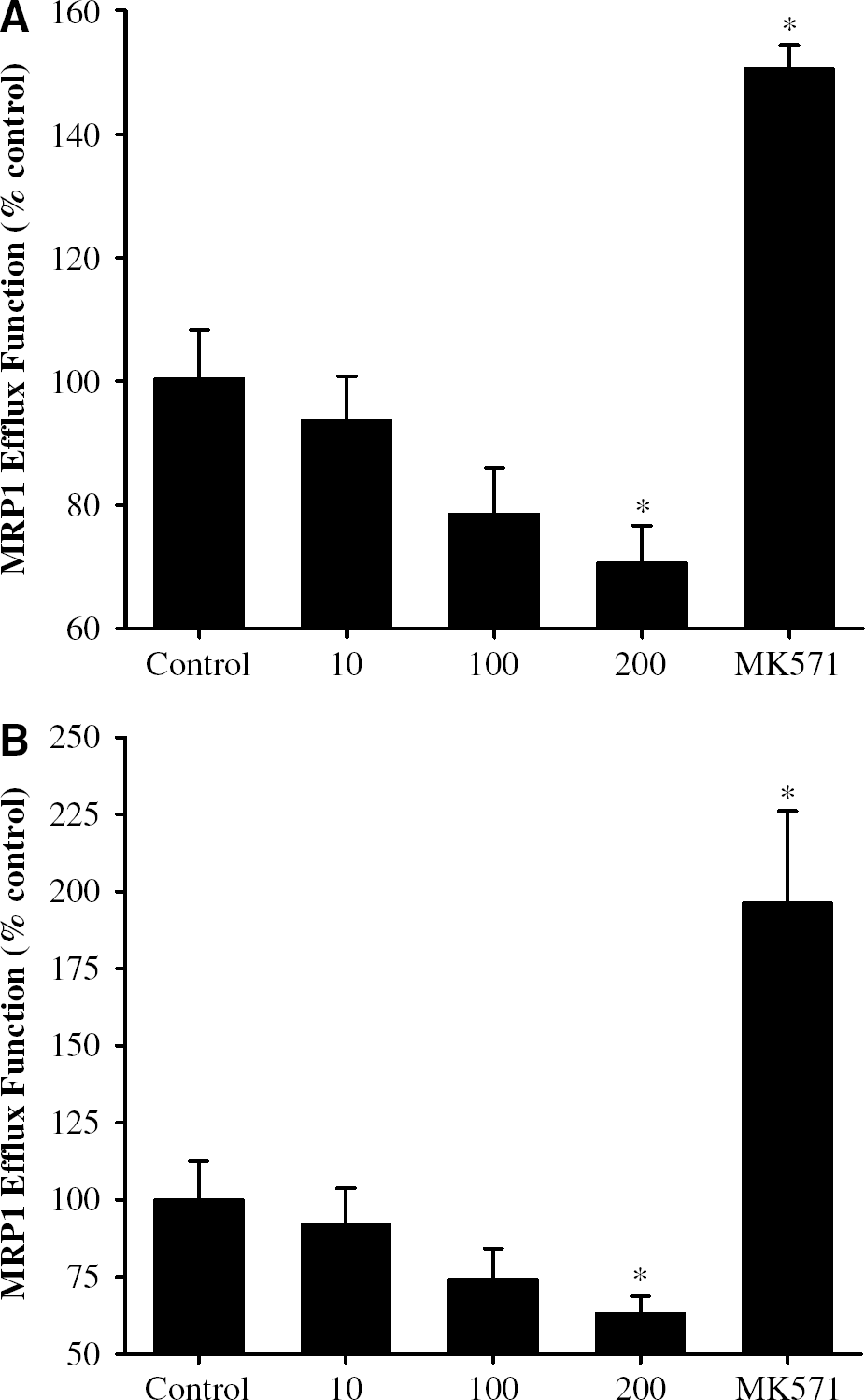
Effects of Tat on MRP1-mediated efflux functions in BMEC (
Tat-Induced Multidrug Resistance-Associated Protein 1 Expression is Mediated by the MAP-Kinase-Dependent Mechanism
To test the hypothesis that the MAPK signaling cascade can be involved in Tat-induced MRP1 expression, we first examined activation of the MAP kinases in BMEC and astrocytes exposed to Tat. Activation of MAPK was determined by Western blot with anti-p-ERK1/2, anti-p-p38 MAPK, or anti-p-JNK antibodies in a series of time-dependent experiments (Figure 6). Treatment with Tat for 30 or 60 mins induced phosphorylation of ERK1/2 and JNK in BMEC (Figure 6A) and astrocytes (Figure 6B). In contrast, phosphorylation of p38 MAP kinase was not affected by Tat exposure in these cell types.
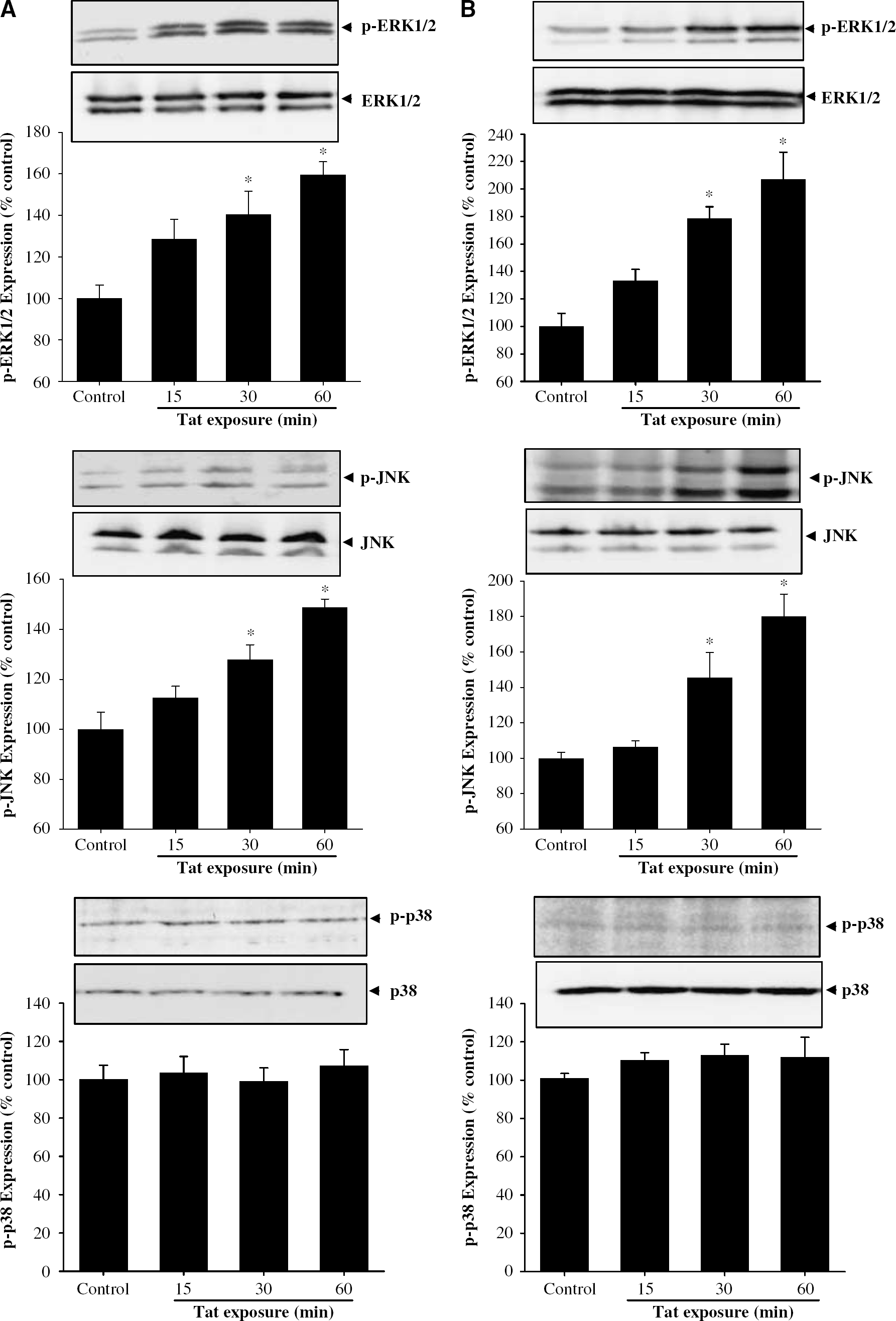
Effects of Tat on activation of the MAP kinase signaling pathways in BMEC (
To investigate whether Tat-mediated MAP kinase activation can participate in upregulation of MRP1, mRNA levels of MRP1 were measured in BMEC and astrocytes treated with Tat in the presence of mitogen-activated protein (MAP) kinase kinase (MEK) inhibitor PD98059 or JNK inhibitor SP600125. Cells were pretreated with these inhibitors for 30 mins and then treated with 200 ng/mL Tat for an additional 6 h. As illustrated in Figures 7A and 7B, PD98059 inhibited Tat-induced upregulation of MRP1 mRNA levels in BMEC and astrocytes. In contrast, SP600125 attenuated Tat-induced MRP1 mRNA expression in astrocytes but not in BMEC, suggesting a distinct signaling for MRP1 induction that depends on cell type.
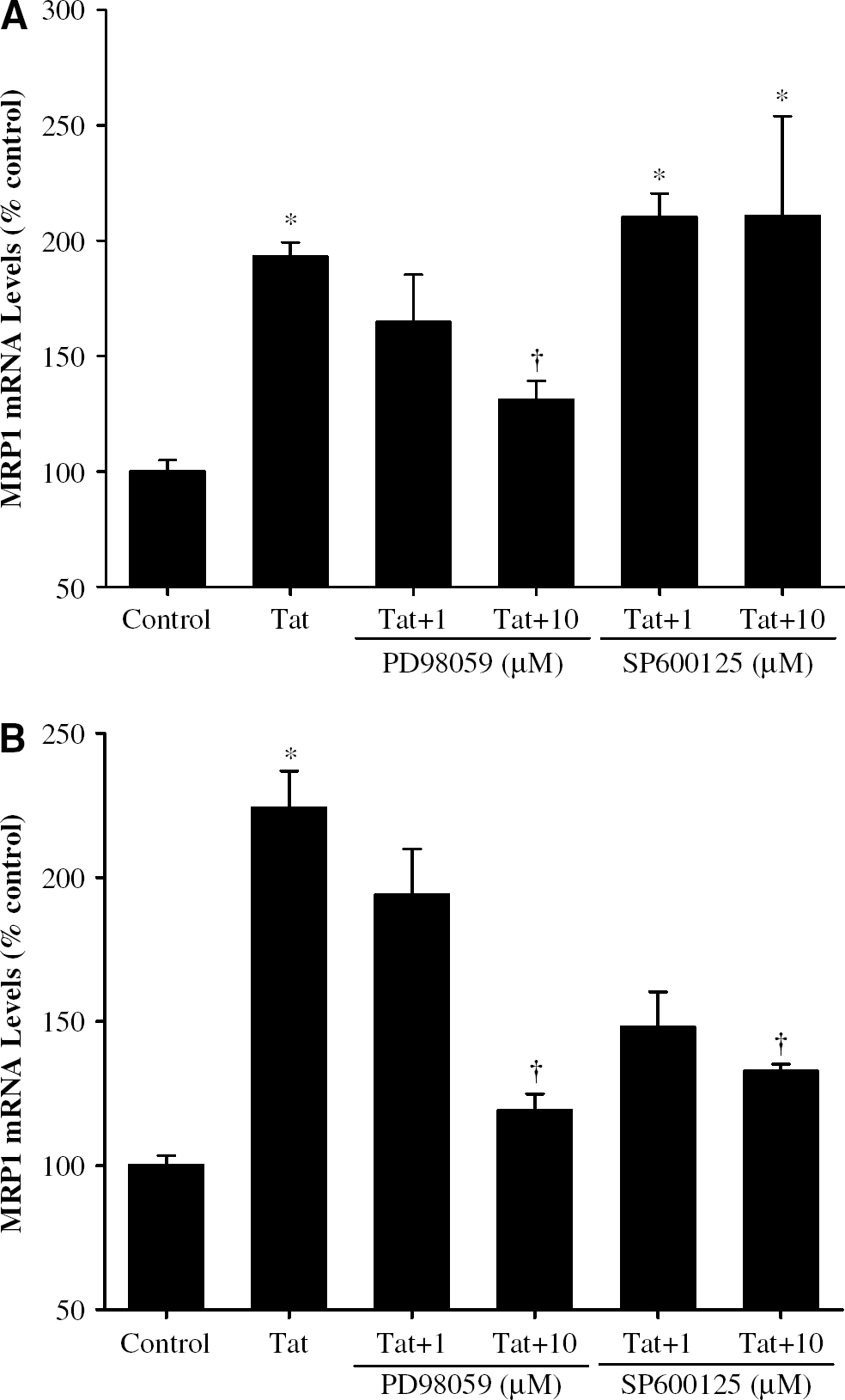
Effect of MAP kinase inhibition on Tat-induced upregulation of MRP1 mRNA levels in BMEC (
Discussion
The entry of drugs into the brain is tightly regulated by the BBB, a barrier which exists between the blood 1059 and brain interstitial fluid compartments. The BBB is formed by the microvascular endothelium surrounded by pericytes and end-feet astrocytes, and is characterized by the presence of tight junctions that seal adjacent endothelial cells together (Abbott, 2002). Other factors that regulate the penetration of drugs into the CNS are related to (a) lipid solubility (lipophylic drugs being readily diffusable across the BBB), (b) binding to plasma proteins that restrict drug entry into the CNS, and (c) activity of efflux drug transporters, such as P-glycoprotein and MRPs, which remove drugs from the CNS (Wynn et al, 2002). Normally, the presence of efflux transporters in the BBB prevents drugs and toxins from accumulating in the brain. However, their efflux activity restricts the entry of specific drugs, including those used for HAART, into the CNS and contributes to the phenomenon that the brain is intrinsically resistant to treatment with several cytotoxic and antiviral drugs (Haimeur et al, 2004), including those used for HAART.
Current guidelines for HAART recommend the combination of at least three agents from at least two different therapeutic classes. This therapy usually includes either a protease inhibitor (e.g., indinavir) or a nonnucleoside reverse transcriptase inhibitor (NNRTI, e.g., nevirapine) in combination with two nucleoside reverse-transcriptase inhibitors (NRTIs, e.g., zidovudine and abacavir) (Carr, 2003). This drug combination is highly effective in reduction of the viral load in plasma and many tissues of infected patients (Dougherty et al, 2002). However, due to the pharmacokinetic properties of drugs and the activity of the efflux mechanisms of the BBB, the efficacy of HAART is limited in the CNS as compared with other compartments. For example, protease inhibitors are lipophilic drugs; however, they usually have very high binding properties to plasma proteins (∼98%), which restrict their entry into the CNS (Wynn et al, 2002). Protease inhibitors are also very good substrates for the efflux transporters, such as P-glycoprotein and the MRPs (Jorajuria et al, 2004a). Another class of anti-HIV drugs are the NRTIs. These drugs tend to be hydrophilic and do not readily diffuse across the BBB. In addition, they are substrates for the efflux transport systems (Wynn et al, 2002; Jorajuria et al, 2004a). Finally, NNRTIs tend to be lipophilic and weak substrates for the efflux transport systems. However, their entry into the CNS is restricted due to high binding to plasma proteins. The exception is nevirapine, which is approximately 60% protein bound (Wynn et al, 2002).
The MRP family creates the major efflux system involved in reverse transport of anti-HIV drugs from the CNS into the bloodstream. All MRPs are energy-dependent efflux pumps that belong to the family of ATP-binding cassette transporters; however, they differ in substrate characteristics. Conjugates of lipophilic compounds with glutathione, glucuronate, or sulfate are the preferred physiological substrates of MRP1 (Borst et al, 2000), 2 (Konig et al, 1999), and 3 (Hirohashi et al, 1999). Substrates for MRP4 and 5 include cyclic adenosine monophosphate (AMP), cyclic guanosine monophosphate (GMP), and nucleoside analogs (Chen et al, 2001; Jedlitschky et al, 2000). Multidrug resistance-associated protein 6 has been reported to transport the anionic cyclopentapeptide BQ123, but none of the prototypical MRP1 substrates (Madon et al, 2000). Functions of MRP7 are currently unknown; however, MRP7 chromosomal localization (6p12 to 21) is shared by several genes whose proteins are involved in glutathione metabolism, suggesting that MRP7 is a member of the MRP subfamily of amphipathic anion transporters (Hopper et al, 2001).
The expression profile of MRPs in the BBB has not been well characterized; therefore, part of our study focused on mRNA expression of MRP1 to 7 in BMEC and astrocytes, that is, the major cell types that form the BBB. The relative levels of MRP1 to 7 mRNA (Figure 2) illustrate that MRP1 is the dominant member of the MRP family both in astrocytes and BMEC. Consistent with a previous report (Decleves et al, 2000), MRP1 mRNA expression was detected at higher levels in astrocytes by approximately 60% as compared with BMEC. Based on constitutive expression, MRP4 and 3 also appear to be important efflux mechanisms of the BBB. Multidrug resistance associated protein 3 mRNA is primarily expressed in astrocytes and MRP4 mRNA is mainly present in BMEC.
Available evidence suggests the absence of MRP2 in the human brain (Kool et al, 1997; Hirrlinger et al, 2002). However, our analysis showed the presence of low amounts of MRP2 mRNA both in BMEC and astrocytes. This discrepancy might be attributed to the different detection techniques used. Real-time RT-PCR used in the present study is a much more sensitive technique than the RNase protection assay or Northern blot employed in the previous studies (Kool et al, 1997; Hirrlinger et al, 2002). The relatively low mRNA levels of MRP2, 6, and 7 both in BMEC and astrocytes suggest that these MRPs might not be important transporters in the BBB.
The comparison of the expression profiles of MRPs in astrocytes and BMEC illustrates the fact that these cell types closely interact in the generation of a unique brain microvascular environment. It is well recognized that the microvascular-facing membranes of the astrocyte processes are polarized and their transporters play a critical role in capillary functions. In fact, glucose transporter-2 and aqua-porin-4 are expressed dominantly in astrocytes, contributing to glucose and water transport within the capillaries. A high level of MRP1 expression in astrocytes suggests that it may markedly contribute to the overall drug removal from the CNS. In addition, a high expression of MRP1 in astrocytes may compensate for low levels of phospho (P)-glycoprotein, another major efflux system of the BBB, in this type of cell (Hayashi et al, 2005).
One of the major findings of the present study was that exposure of BMEC or astrocytes to Tat can markedly increase expression of MRP1 both at mRNA and protein levels. These effects are highly specific because other members of the MRP family evaluated in the present study were not affected by Tat treatment. Elevated levels of MRP1 mRNA and protein (Figures 3 and 4, respectively) were associated with increased efflux function of this transporter by approximately 30% to 40% (Figure 5). These observations are of high clinical significance, because MRP1 is involved in reverse transport of protease inhibitors, such as indinavir or retinavir (van der Sandt et al, 2001). They are also supported by literature data showing that MRP1 is overexpressed in human macrophages infected with HIV (Jorajuria et al, 2004b). Most interesting, the increase in MRP1 mRNA coincided with the peak of viral replication. Such results may indirectly indicate the role of Tat in MRP1 overexpression, since Tat is a transactivating protein involved in virus replication (Jorajuria et al, 2004b). To confirm the role of Tat in upregulation of efflux transporters at the levels of the BBB, we showed that this protein can lead to overexpression of P-glycoprotein in BMEC and in brain capillaries (Hayashi et al, 2005).
Tat, by inducing cellular oxidative reactions, can activate a variety of redox-mediated signaling pathways and thus affect vascular functions (Toborek et al, 2005). For example, activation of the ERK1/2 cascade can be involved in Tat-mediated alterations of tight-junction expression and entry of the inflammatory cells into the CNS. In addition, literature data suggest that oxidative stress, alterations of glutathione metabolism, and activation of the MAPK can be involved in MRP expression (Cripe et al, 2002; Guan et al, 2004; Yamane et al, 1998). Therefore, our studies also focused on the involvement of the MAPK signaling cascade in Tat-mediated overexpression of MRP1. The main components of the MAPK signaling are: (a) the ERK1/2 pathway, (b) the JNK pathway, and (c) the p38 MAPK pathway (for a recent review, see Kyosseva, 2004). Our data indicated that treatment with Tat specifically phosphorylates two of the main components of the MAPK signaling, namely ERK1/2 and JNK. However, it does not affect phosphorylation of p38 MAPK. Specificity of these responses is confirmed by the fact that such a pattern of phosphorylation of major MAPK components was observed both in BMEC and in astrocytes (Figures 6A and 6B, respectively). Another major finding of this study was that the ERK1/2 pathway is involved in Tat-mediated overexpression of MRP1 in BMEC. Indeed, pharmacological inhibition of the ERK1/2 signaling by PD98059 blocked the effects of Tat on MRP1 mRNA expression (Figure 7). The JNK inhibitor did not affect Tat-induced MRP1 expression, indicating that the JNK signaling is not involved in MRP1 expression in BMEC. Conversely, both the ERK1/2 and JNK pathways appear to participate in Tat-mediated overexpression of MRP1 in astrocytes. A marked attenuation of Tat-induced MRP1 mRNA overexpression in this cell type was observed after pretreatment with pharmacological inhibitors of both ERK1/2 and JNK (Figure 7). The differences in the regulatory mechanisms of MRP1 induction in BMEC and astrocytes illustrate that the effects of Tat on the BBB are finely tuned and depend on the specific cell types.
In summary, we have provided evidence that the HIV transcriptional regulator, Tat protein, can specifically upregulate MRP1 mRNA and protein expression in BMEC and astrocytes through the mechanisms regulated by the MAPK signaling. The changes in MRP1 expression were associated with increased transport function of this protein. These data suggest that the release of Tat into the vascular microenvironment by HIV-infected cells may contribute to the development of resistance to HAART due to upregulation of MRP1 at the BBB level. Such alterations may contribute to the development of HIV-related neurological complications, including dementia, at the later stages of the disease.
Footnotes
Acknowledgements
The authors thank Dr Avindra Nath (Johns Hopkins University, Baltimore, MD, USA) for providing Tat protein.
