Abstract
Thyroid dysfunction is a well-known contributor to psychiatric morbidity. To investigate the mechanism(s) by which thyroid hormone availability affects cerebral activity, a group of thyroidectomized individuals were studied at two points in time: when markedly hypothyroid in preparation for a thyroid cancer metastatic survey and when clinically and/or biochemically euthyroid. The analysis consisted of single photon emission computed tomography (SPECT) using a lipophilic radiopharmaceutical, technetium-99m (Tc-99m) ethyl cysteinate dimer (ECD), and measurement of mood, anxiety, and psychomotor function, at both points in time. Both increases and decreases in regional cerebral radiotracer activity were found in the hypothyroid condition relative to the euthyroid condition, and the neuropsychological assessment demonstrated significantly greater depression, anxiety, and psychomotor slowing during the hypothyroid state. Increased radiotracer activity was seen in frontal and temporal regions, posterior cingulate gyrus, thalamus, and putamen. Decreased activity was seen in the occipital cortex, and the pre- and postcentral gyri. This distribution pattern is partially consistent with findings in persons with depression and anxiety unrelated to thyroid disease, supporting the link between the symptoms observed in our subjects and their marked hypothyroidism. Finally, these results support the need to consider the effect of the thyroid state on cellular mechanisms of uptake and retention of cerebral blood flow radiopharmaceuticals when studying ‘noneuthyroid’ individuals.
Introduction
A connection between thyroid disease, particularly hypothyroidism, and neuropsychiatric disturbance has been recognized for more than a century (Hennessey and Jackson, 1996). Somatic signs and symptoms of clinical hypothyroidism are commonly associated with depressed mood and other features of clinical depression, cognitive dysfunction, and anxiety (Whybrow et al, 1969; Jain, 1972; Denicoff et al, 1990; Boswell et al, 2002). Despite significant advances in the understanding of both thyroid physiology and the human brain since publication of ‘Report on Myxedema’ in 1888 (Hennessey and Jackson, 1996), the physiologic effects of thyroid disorders on the human brain are not well understood.
Numerous investigations regarding the physiologic link between thyroid function and brain function were completed before the advent of modern nuclear imaging devices and radiotracer technologies (Madison et al, 1951; O'Brien and Harris, 1968; Scheinberg, 1950; Sensenbach et al, 1954). Although the findings were originally interpreted as resulting exclusively from changes in vascular resistance as opposed to a primary change in brain metabolism, demonstrable decreases in cerebral blood flow during hypothyroidism and the opposite change during hyperthyroidism were demonstrated as early as the 1950s (Madison et al, 1951; Scheinberg et al, 1950; Sokoloff et al, 1953; Sensenbach et al, 1954). It was not until decades later, however, that the use of nuclear magnetic resonance (NMR) spectroscopy, contemporary nuclear medicine technology (e.g., positron emission tomography (PET)), and improved statistical methodologies allowed for a reinterpretation of some of the findings from work performed much earlier. Strong support soon grew for a correlation between thyroid function and cerebral neuronal activity (Constant et al, 2001; Marangell et al, 1997; Smith and Ain, 1995), clearly not explainable by changes in vascular resistance alone (Constant et al, 2001).
Using PET to explore cerebral blood flow (with oxygen-15 water) and metabolism (with fluorine-18 fluorodeoxyglucose) in severe, transient hypothyroidism, Constant's group (Constant et al, 2001) demonstrated global decreases in both functional parameters but no significant regional changes. Using single photon emission computed tomogram (SPECT), the other standard nuclear medicine technique to investigate the brain, two groups have published studies exploring the distribution of a lipophilic, single photon emitting cerebral radiotracer—a so-called regional cerebral blood flow (rCBF) agent—in patients with hypothyroidism. Both groups, Krausz et al (2004) who examined mildly hypothyroid patients and Nagamachi et al (2004) who studied postthyroidectomy patients with marked, transient hypothyroidism, reported rCBF decrements associated with the hypothyroid state. Common to both of these groups was their use of the SPECT radiotracer, technetium-99m (Tc-99m) hexamethylpropyleneamine oxime (HMPAO). Although this agent and the other widely used lipophilic cerebral SPECT radiotracer, Tc-99m ethyl cysteinate dimer (ECD), provide an image pattern reasonably reflective of rCBF, unlike the standard PET cerebral blood flow agent O-15 water, the regional distribution of these tracers is also dependent on somewhat complex, and different, cerebral cellular mechanisms of incorporation (Verbruggen, 1998; Jacquier-Sarlin et al, 1996; Ishizu et al, 1996).
As previously mentioned, patients with hypothyroidism typically exhibit symptoms and signs of depression and anxiety (Whybrow et al, 1969; Jain, 1972; Denicoff et al, 1990; Boswell et al, 2002). These are often alleviated or reversed, however, once the individual receives adequate thyroid hormone replacement therapy (Whybrow, 1996). To determine the underlying pattern of brain function produced in association with thyroid hormone removal in relation to the thyroid hormone replacement condition, we designed this study to compare the Tc-99m ECD brain accumulation pattern in patients after thyroidectomy (hypothyroid) with their brain accumulation patterns after hormone replacement therapy (euthyroid). Based on functional brain imaging studies of persons with depression (Hornig et al, 1997) and anxiety (Lucey et al, 1997), we expect that the hypothyroid condition will produce differential regional patterns of activity relative to the euthyroid condition. We also expect that the Tc-99m ECD tracer's unique mechanism of cellular accumulation, not heretofore examined in hypothyroid patients, may help to further our understanding of the effects of hypothyroidism on cerebral physiology.
Materials and methods
Subjects
This study includes data from nine subjects who had undergone a thyroidectomy for treatment of thyroid carcinoma and were presenting to the Nuclear Medicine Service for a metastatic thyroid cancer survey using radioactive iodine. The subjects included four women and five men with a mean age (± s.d.) of 33.3 ± 8.7 years. Each subject was free of current or past significant medical problems with the exception of thyroid cancer. This included no history of neurologic or psychiatric illness, and no subject was taking psychotropic medication. Magnetic resonance imaging (MRI) of each subject's brain was negative for structural abnormalities. Likewise, no subject evidenced brain metatases on MRI or radioactive iodine scintigraphy. All of the subjects were right handed and, educationally, each had at least a high school diploma. The length of time that each subject had been without thyroid hormone before his or her ‘hypothyroid brain’ SPECT evaluation was consistent across the group.
The study was approved by the Institutional Review Board of the National Naval Medical Center, the procedures followed were in accordance with the ethical standards of the Institutional Review Board, and all of the subjects provided written informed consent.
Study Design
The study design consisted of subject evaluations performed at two points in time: when each subject was markedly hypothyroid and after each subject had received appropriate thyroid hormone replacement. At the time of initial evaluation, all subjects had been without thyroxine for at least 7 weeks and without triiodothyronine for at least 3 weeks to achieve their markedly hypothyroid condition. At the follow-up evaluation, each of these subjects had been receiving thyroxine for at least seven weeks. The initial and follow-up assessments for the subjects consisted of a battery of thyroid function tests; evaluations of mood, anxiety level, and psychomotor function; and a SPECT of the brain.
Thyroid Function Measurements
The following laboratory tests were performed as part of both the hypothyroid and thyroid hormonereplaced evaluations: thyroid-stimulating hormone (TSH), measured by microparticle enzyme immunoassay (Abbot Laboratories) with a lower limit of detectability of 0.08 μIU/mL, an interassay coefficient of variation of 0.7% to 3.6%, an intraassay coefficient of variation of 3.1% to 4.4%, and a normal range of 0.49 to 4.67 μIU/mL; free thyroxine (free T4), measured by microparticle enzyme immunoassay (Abbot Laboratories) with a lower limit of detectability of 0.4 ng/dL, an interassay coefficient of variation of 0.8% to 4.6%, an intraassay coefficient of variation of 2.6% to 4.5%, and a normal range of 0.71 to 1.85 ng/dL; and free triiodothyronine (free T3) (Quest Diagnostic, Inc.) with a lower limit of detectability of 160 pg/dL, an interassay coefficient of variation of 2.35%, an intraassay coefficient of variation of 2.87%, and a normal range of 230 to 420 pg/dL.
Neuropsychological Testing
Mood and anxiety assessments included the Beck Depression Inventory (BDI) and the state portion of the Spielberger State-Trait Anxiety Inventory (STAI), respectively. The Beck Depression Inventory is a 21 item, self-report questionnaire used to screen for depression (Beck et al, 1961) with a well-established history of use in clinical and research settings (Blacker, 2005). The STAI is a 40-item self-report inventory that has been used in more than 2000 studies (Spielberger, 1983). The subjects completed the ‘state anxiety’ portion of the test, a 20-item scale for measuring state anxiety (i.e., evaluating how the respondent currently feels). The measures of interest in this study were the total scores on the mood and anxiety scales, with the scores being directly related to the degree of depression and anxiety, respectively.
A psychomotor assessment was performed using the Grooved Pegboard Test. Subjects are required to insert small metal pegs into a pegboard containing a 5 × 5 set of slotted holes angled in different directions. Scores are determined for each hand, dominant (the right hand for each of our subjects) and nondominant, and are based on time to complete the task. This is a sensitive measure of general psychomotor slowing, and although the test can aid in identifying lateralized impairment (Matthews and Klove, 1995), performance may be bilaterally impaired in the setting of unilateral brain insult owing to the test's high demands for cognitive integration (Swanda and Haaland, 2005).
Paired samples t-tests were used to compare the scores for each of these measures at the two points in time.
Single Photon Emission Computed Tomography of the Brain
The first scan was performed during hypothyroidism and the other after at least 7 weeks of thyroid hormone replacement (with a time separation range of 7 to 11 weeks and a mean (± s.d.) of 58.4 (± 8.2) days apart. In preparation for the scan, each of the participants was placed in a dimly lighted room in a supine position on a comfortable cot with his or her eyes open. An intravenous 18 or 20 gauge catheter was inserted into an antecubital vein and normal saline was infused at a ‘KVO’ rate. At 30mins after the intravenous catheter insertion, approximately 1110 MBq (30 mCi ± 10%) of Tc-99m ECD was administered in 1.5 ml of normal saline for 15 to 20 secs through the intravenous catheter. At 45 mins after the administration of the radiopharmaceutical, SPECT was performed of the subject's brain. A triple-headed rotating SPECT camera (PICKER PRISM 3000) with low-energy/high-resolution collimators and a system spatial resolution (CFOV at 10 cm) of 8.7-mm full-width at half-maximum (FWHM) was used for all acquisitions. A total of 40 projections were acquired per camera head (3° per stop in ‘step and shoot’ mode) in a 128 × 128 matrix. These data were reconstructed with attenuation and motion correction, and using a 360° filtered back projection algorithm. Postreconstruction 3D filtering was performed using a Wiener filter.
The SPECT scans for all subjects were realigned and spatially normalized into standard stereotactic space and smoothed to a FWHM of 10, 10, and 10 mm in the x, y, and z planes. To distinguish brain from nonbrain areas (i.e., ventricular and subarachnoid spaces), the raw images were masked to include only those voxels with intensity values that fell within 80% of the maximum intensity of the image. Pixel values were then scaled using the ratio adjustment method. The image data were analyzed using Statistical Parametric Mapping (SPM2; Welcome Department of Cognitive Neurology, London, England), where voxel by voxel t-test comparisons were performed to determine significant changes in signal intensity between the two conditions (P < 0.01) (Friston et al, 1995). To control for multiple comparisons, cluster analyses were performed on each contrast to determine the significance of activated voxels based on the magnitude of activation (Z = 2.36) and spatial extent (P < 0.05) (Poline et al, 1997).
Results
Thyroid Function Tests
During the hypothyroid condition, the TSH ranged from 55.3 to 297.0 μIU/mL. The mean TSH, free T4, and free T3 (± s.d.) during hypothyroidism were 157.1 (± 93.8) μIU/mL, <0.40 ng/dL, and <160 pg/dL, respectively. During thyroid hormone replacement, the TSH ranged from <0.04 (considered ‘0’ for purposes of analysis) to 4.19 μIU/mL, the free T4 from 0.76 to 2.01 ng/dL, and the free T3 from 260 to 422 pg/dL. The mean TSH, free T4, and free T3 were 1.2 (± 1.6) μIU/mL, 1.5 (± 0.41)ng/dL, and 335.2 (± 61.3) pg/dL, respectively: subject ‘3’ had a free T4 value slightly above normal (i.e., 2.01 ng/dL), and subject ‘5’ had a free T3 value slightly above normal (i.e., 422 pg/dL). These results are shown in Table 1.
Thyroid function test values showing mean (standard deviations) of free T4, TSH, and free T3 serum concentrations during hypothyroidism and thyroid hormonereplaced conditions

All measured values were below threshold of assay detectability.
This results from the fact that four of the subjects had TSH values below the limit of detectability and were thus given the value of ‘0’.
Neuropsychological Data
The subjects were significantly more depressed (P < 0.005), anxious (P < 0.05), and psychomotorically slowed (P < 0.05) during hypothyroidism than when thyroid hormone replaced. These results are shown in Table 2.
Psychological assessment values showing mean (standard deviations) scores on the Beck Depression Inventory, State Anxiety Inventory, and Pegboard Test for the dominant (the right hand for each of the subjects) and nondominant hands during hypothyroidism and thyroid hormonereplaced conditions
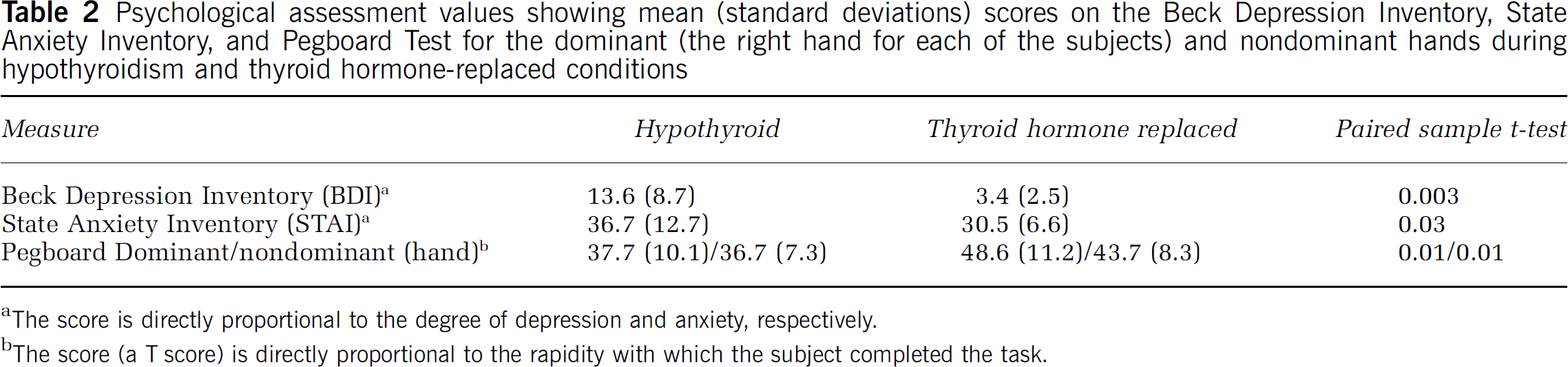
The score is directly proportional to the degree of depression and anxiety, respectively.
The score (a T score) is directly proportional to the rapidity with which the subject completed the task.
Single Photon Emission Computed Tomography Data
Significant differences in radiotracer accumulation were observed during the hypothyroid condition relative to the baseline thyroid hormone replaced condition. Increased activity was seen in the medial frontal gyrus, middle frontal gyrus (Brodmann Area 6 or BA 6), posterior cingulate gyrus (BA 31), and thalamus of the left hemisphere; the medial frontal gyrus (BA 6), insula, superior temporal gyrus (BA 38), and putamen of the right hemisphere.
Decreased accumulation was also seen during hypothyroidism. Regions exhibiting decreased activity included the bilateral middle occipital (BA 19) regions, and the precentral (BA 4) and postcentral (BA 43) gyri of the left hemisphere. These results are shown in Table 2 and Figure 1.
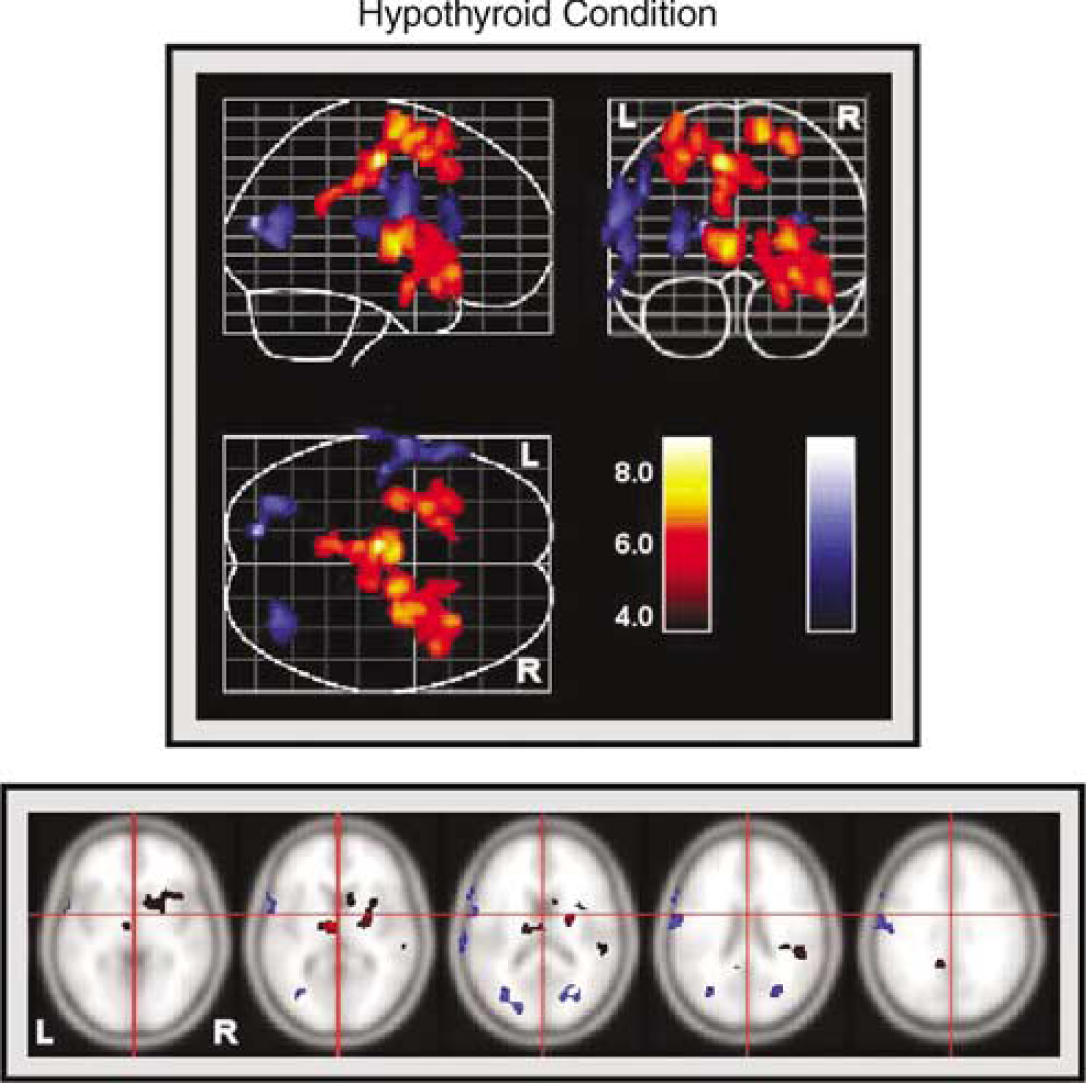
Brain activity patterns during hypothyroidism relative to the thyroid hormonereplaced condition. Lateral, horizontal, and coronal views are shown along with sample slices through the brain. Right and left hemispheres are indicated. Increases in tracer accumulation are indicated in red; and decreases in accumulation are shown in blue. Color bars represent the t-value of regional activity changes.
Discussion
We found multiple regions of both increased and decreased radiotracer retention in the brains of transiently, but markedly, hypothyroid patients. Neuropsychological symptoms related to hypothyroidism—depression, anxiety, and psychomotor slowing were also seen.
Among the most robust changes seen in our cohort was increased tracer activity in the bilateral medial frontal gyri during hypothyroidism. This area (BA 6), part of the supplemental motor area, has shown increased metabolic activity in mood-disordered patients with depression and anxiety (Osuch et al, 2000). In fact, the left medial frontal area of the cerebral cortex has been repeatedly implicated in the pathophysiology of depression (Kumari et al, 2003; Larisch et al, 1997; Bench et al, 1992). Brodmann Area 6 is also considered critical for the initiation of movement and emotional expression (Boswell et al, 2002; Delong, 1996). Flattening or dulling of affect and psychomotor retardation has been described in many hypothyroid patients (Constant et al, 2001), and was observed in our subjects when presenting for their metastatic surveys. Taken together, these findings seem to directly corroborate involvement of this region of the brain (Table 3).
Local maxima table listing areas of significant increases and decreases of Tc-99m ECD accumulation during the hypothyroid condition relative to the baseline thyroid hormonereplaced condition
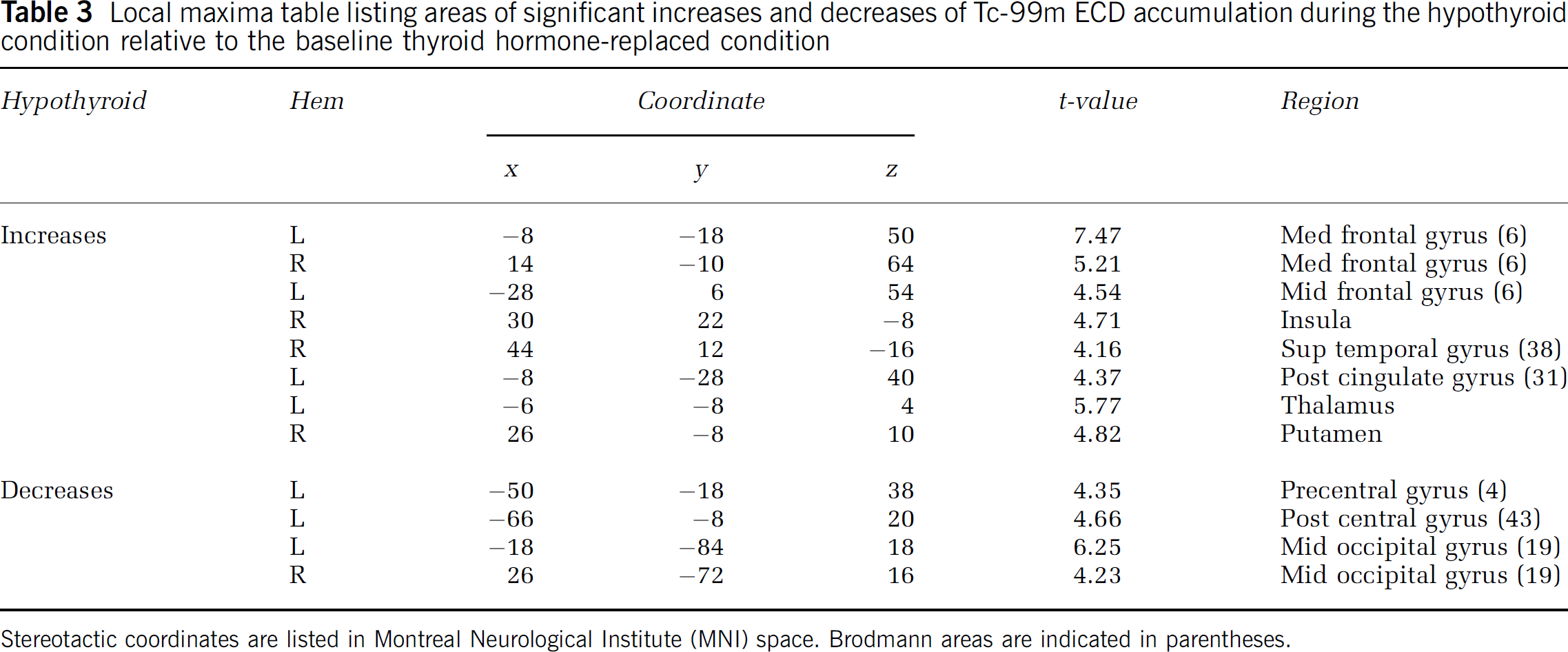
Stereotactic coordinates are listed in Montreal Neurological Institute (MNI) space. Brodmann areas are indicated in parentheses.
Another area of significant increase in radiotracer concentration during the hypothyroid condition was the anterior right insula. In a study by Mayberg et al (1999), the anterior insula demonstrated decreases in metabolic activity in association with recovery from major depression and increased blood flow during the provocation of induced sadness in healthy volunteers. This area, also known as the island of Reil, is involved primarily with autonomic function (Henley and Vladic, 1997; Matsukawa et al, 1993; Dillmann, 1989). It is interesting to note that along with the mood-related effects of hypothyroidism, depressed thyroid function is also known to significantly affect the autonomic nervous system (Foley et al, 2001).
Left posterior cingulate radiotracer accumulation was also increased during hypothyroidism. Increased activation of this region has been consistently seen in functional imaging studies examining the effect of emotional stimuli, thereby supporting both an affective, and possibly an anxiety-related function, for this region of the brain (Maddock et al, 2003; Fischer et al, 1996).
The thalamus, particularly the left thalamus, is another area of the brain about which multiple investigators have demonstrated mood state correlations. Regional blood flow increases to the left thalamus have been observed in both induced sadness in subjects with no psychiatric history, and during the depression phase of a patient with rapid-cycling bipolar disorder. Evidence of increased cellular energy metabolism in the left thalamus in mood-disordered patients has also been demonstrated with magnetic resonance spectroscopy (Juckel et al, 2000; Boswell et al, 2002; Deicken et al, 2001). We, likewise, observed increased activity in the left thalamus of our subjects when markedly hypothyroid.
The right putamen was another area of increased activity seen during hypothyroidism in our subjects. Investigators have shown generalized increased metabolic activity and blood flow in the striatum (the caudate nucleus and the putamen) of patients with major depression and in healthy subjects during the induction of transient sadness (George et al, 1995; Boswell et al, 2002), and Mayberg et al (1997, 1999) have proposed that reductions in activity in the putamen and globus pallidus along with other ventral paralimbic areas of the brain are a necessary part of recovery from major depression. Nevertheless, a functional change specifically in the right putamen in patients with affective symptoms has not been demonstrated. Interestingly, however, the right putamen has been specifically implicated as abnormal in a post-mortem volumetric brain study of patients with mood disorders compared with normal controls (Baumann et al, 1999).
Hypothyroid-related anxiety, including both panic attacks and generalized anxiety, is widely described (Hollander et al, 1994; Boswell et al, 2002). In this context, it is noteworthy that the right superior temporal gyrus, an area demonstrating increased blood flow in patients with panic disorder during anticipatory anxiety and at rest, and an increased volume in patients with generalized anxiety disorder (Boshuisen et al, 2002; De Bellis et al, 2002) accumulated significantly more tracer during hypothyroidism than during the baseline condition. In a study using functional magnetic resonance imaging (fMRI), this same region demonstrated increased signal during negative emotional arousal in normal subjects (Kuniecki et al, 2003).
Areas of decreased activity during the hypothyroid condition were predominately in the left hemisphere, including foci in the middle occipital gyri, and the pre- and postcentral gyri of the left hemisphere representing primary motor and somatosensory gyri, respectively. None of these regions have consistently demonstrated decreased metabolic activity or decreased blood flow in persons with diagnosed neuropsychiatrie disorders or transient neuropsychiatrie symptoms.
When considering the strikingly similar phenomenology associated with hypothyroidism and major depressive disorder as well as some types of anxiety disorders (Parsey and Mann, 2003; Boswell et al, 2002), it is not surprising that we would see alterations in cerebral activity similar to those seen in functional imaging studies of patients with depressive and anxiety illnesses. Indeed, our subjects demonstrated depressive and anxiety symptoms, as well as psychomotor slowing, when markedly hypothyroid. These findings support other studies where depression, anxiety, and psychomotor slowing were found during similar hypothyroid states (Denicoff et al, 1990; Constant et al, 2001; Nagamachi et al, 2004).
Our findings are in partial contrast to the other published studies using lipophilic SPECT radiotracers to examine rCBF during hypothyroidism. These studies included mild hypothyroidism in one study, which might in itself have a different effect on the brain than severe, transient hypothyroidism (Krausz et al, 2004), and marked, transient hypothyroidism (Nagamachi et al, 2004) similar to our subjects. Like us, each of these groups of investigators showed regional decreases in radiotracer activity; and our data do, in fact, corroborate one (but no other) finding by Nagamachi et al (2004) of decreased activity in parts of the occipital lobes. However, unlike our group, neither of these groups of investigators demonstrated areas of increased tracer accumulation. A key component to the explanation for these differences may be the choice of radiotracer: Tc-99m ECD used by our group versus Tc-99m HMPAO used in the other two studies. The published regional variations in Tc-99m HMPAO distribution in hypothyroidism, and now the regional variations seen in our cohort using Tc-99m ECD, may be related to differential regional metabolism and retention of these radiotracers in the setting of different states of thyroid hormone availability. In a direct comparison of Tc-99m ECD with oxygen-15 water, Ishizu et al (1996) demonstrated a retention fraction and permeability surface area product (PS) independent of rCBF in healthy volunteers. Results from this and other groups (Jonsson et al, 1998) have demonstrated that both Tc-99m ECD and Tc-99m HMPAO fail to provide a precise map of cerebral perfusion. Discrepancies between the patterns seen with these different tracers in hypothyroid subjects is, in fact, not unexpected when one considers that Tc-99m ECD and Tc-99m HMPAO have been shown to demonstrate different patterns of accumulation in the same brains, both healthy and diseased (Hyun et al, 2001; Siennicki-Lantz et al, 1999; Komatani et al, 1998; Asenbaum et al, 1998).
Ethyl cysteinate dimer, because of its high lipophilicity, passes through the blood–brain barrier via the endothelial cell membrane (Weiner and Thakur, 2003). On entrance into the cellular space, metabolism to one or more polar Tc-99m-complexes occurs via enzymatic deesterification, with this process being directly affected by the location of the esterase activity (i.e., membrane bound versus free in the cytosol) (Verbruggen, 1998; Jacquier-Sarlin et al, 1996). Cell culture studies have shown that the amount of cell membrane-bound esterase activity is inversely proportional to Tc-99m ECD uptake (Walovitch et al, 1994). Thyroid axis hormonal changes have been demonstrated in animal studies to modulate esterase production and activity in both a positive and a negative manner, depending in part on the particular type of esterase and the biochemical milieu (Goswami and Rosenberg, 1985; Banerjee and Ray, 2002; Takahashi et al, 2001; Sarkar and Ray, 2001). Given the aberrations in acid-base balance and electrolyte metabolism known to occur during hypothyroidism (Moses and Scheinman, 1996), the probable resultant pH changes, and the pH-related ionization and trapping of ligands with the same N2S2 structure as ECD (Verbruggen, 1998), it is plausible that a pH change in the biochemical brain milieu, with secondary effects on esterase production and function, may contribute to local changes in Tc-99m ECD retention during hypothyroidism.
Whatever the complete and precise mechanism, it would seem to follow that the areas of the brain with disproportionately high (and low) cerebral accumulation of Tc-99m ECD are regions where the processes responsible for uptake and retention of the ECD are most significantly affected by hypothyroidism, relative to the baseline thyroid hormone replaced state. The areas of increased activity therefore suggest that, contrary to the inference from previous SPECT investigations, mechanisms resulting in ‘hyperaccumulation’, or a type of ‘hypermetabolism’, are regionally operative.
In summary, significant regional changes in the brain occur in patients with marked, short-term hypothyroidism compared with the thyroid hormonereplaced condition (i.e., typically clinical and/or biochemical euthyroidism). We hypothesize that these findings are the result of regional differences in uptake and retention of radiotracer brought about primarily by state-dependent physiological effects of differential availability of thyroid hormone during these two contrasting conditions, and not imaging the subjects at two different points in time (Van Laere et al, 2001). Our study also shows regional cerebral increases in radiotracer accumulation, suggesting regional hypermetabolism, in addition to the hypometabolism seen in other studies of the hypothyroid state. Our subjects were more depressed and anxious when hypothyroid, and our findings are actually more in keeping with functional brain imaging results in depression and anxiety: primary psychiatric conditions that are often mimicked by hypothyroidism. In fact, many of the particular regions demonstrating significant increases in tracer accumulation in our cohort during hypothyroidism have demonstrated increased ‘activity’ in cohorts of patients with depression and anxiety. Likewise, psychomotor slowing, seen in our subjects, in other cohorts of hypothyroid patients, and in patients with major depression, was reflected in a pattern of regional tracer accumulation with a strong functional neuroanatomy correlate. A more complete understanding of the effects of hypothyroidism on the brain and its relationship to nonthyroid-related psychiatric illness awaits prospective series involving larger numbers of patients with thyroid-related illness, with and without psychiatric symptomatology, sampled at more points in time. This improved understanding of the effects of hypothyroidism on the brain may likely come about, in part, through a better understanding of the cellular-level effects of thyroid hormone on the accumulation and retention of the radiotracers used in functional neuroimaging investigations.
Footnotes
Acknowledgements
The opinions expressed in this work are those of the authors, and do not reflect the official policy or the position of the Department of the Navy, Department of Defense, or the US government.
