Abstract
The purpose of this study was to investigate the effect of enriched environment on motor function after experimental stroke in mice, and to determine whether time in enriched environment affects functional recovery. Earlier investigations have shown that rats placed in an enriched environment after focal ischemia, remarkably improve motor function, but similar observations in mice have not been reported. In this study, we show that placing mice in an enriched environment for 3 h daily for 2 weeks, after transient (50 mins) occlusion of the middle cerebral artery, enhanced neurologic outcome. Continuous postischemic housing in the enriched environment likewise improved motor function, but mortality increased. Two weeks exposure to enriched environment followed by housing the mice in standard cages for 2 weeks, resulted in a loss of the improved motor function. In contrast, 4 weeks exposure to enriched environment led to an improved motor function and to a better maintenance of neurologic recovery. The expression levels of the immediate-early gene nerve growth factor-induced gene A at 2 to 3 weeks of recovery decreased in animals housed in enriched environment, implying this transcription factor in the recovery process. We conclude that housing mice in an enriched environment after experimental stroke improves functional outcome. Also, the presented experimental procedure is useful for further studies of the genomics of functional recovery after experimental stroke.
Introduction
Stroke leads to tissue degeneration and dysfunction of neurotransmission resulting in sensory, motor, and cognitive deficits. Most stroke patients improve neurologic function spontaneously over the first months after stroke, albeit to a variable degree (Kotila et al, 1984). Functional recovery of motor skills after stroke can be enhanced by forced (Liepert et al, 2000, 1998) or robot-aided training (Krebs et al, 1998). Still, simply the social and physical environment of stroke patients has been suggested to influence the recovery process (SUTC, 1997).
In the experimental setting, training (Biernaskie and Corbett, 2001), as well as social and physical environment, can positively influence the recovery of function after brain damage (Johansson, 2004; Will et al, 2004; Will and Kelche, 1992). Housing several rats in an enriched environment after an experimental stroke, allowing both social interaction and physical activity, improves functional outcome compared with rats housed in standard cages (Johansson, 1996; Ohlsson and Johansson, 1995). This effect of enriched environment is not because of a decrease in infarct volume because the size of the brain infarct is the same in animals housed in standard cages and in those in enriched environment (Grabowski et al, 1995; Ohlsson and Johansson, 1995). Rather, the enriched environment causes structural changes of neurons such as increased spine density, dendritic branching, and cell genesis (Biernaskie and Corbett, 2001; Johansson and Belichenko, 2002; Komitova et al, 2002). Also, the expression of several genes associated with synaptic plasticity is changed when rodents are housed in an enriched environment after brain injury (Keyvani et al, 2004).
Mice can be genetically modified, and are therefore well suited for studies of the effect of particular genes on recovery process after stroke. So far, the effect of different housing conditions on motor function and gene expression after focal ischemia has not been studied in mice. Hence, we investigated whether exposure to an enriched environment after a transient occlusion of the middle cerebral artery (MCA) in the mouse enhanced functional outcome in a sensori-motor test. Also, we investigated whether the effect of enriched environment on functional recovery was preserved after discontinuation of the exposure.
When intact rats are housed in an enriched environment, the expression of the neurotrophic factors NGF, neurotrophin-3 and BDNF (Ickes et al, 2000), and the inducible transcription factors nerve growth factor-induced gene A (NGFI-A) and Arc (Olsson et al, 1994; Pinaud et al, 2001, 2002; Wallace et al, 1995) are upregulated. These results suggest that plasticity-related genes are activated by an enriched environment and contribute to the enhanced performance seen in rats after exposure to enriched environment. In contrast to the findings in intact brains, NGFI-A mRNA levels markedly decrease in the rat brain when rats are housed in an enriched environment the first 3 weeks after middle cerebral artery occlusion (MCAO) (Dahlqvist et al, 1999) but not at 1 month (Dahlqvist et al, 2003, 1999). Hence, we also investigated the changes in NGFI-A mRNA expression in the brain of mice housed in an enriched environment.
Materials and methods
Focal Ischemia
Male C57BL/6 mice, 8 to 10 weeks old and weighing between 20 and 35g, obtained from Taconic M&B, (Ry, Denmark) and B&K, (Stockholm, Sweden) were housed under diurnal light conditions with free access to food and water before surgery. The ethical committee at Lund University approved the experiments.
A modified model of MCAO (Hara et al, 1996) was used. Mice were anesthetized with 2.5% halothane in N2O:O2 (70:30) in a chamber and anesthesia was maintained by allowing the animals to breathe 1.5% halothane through an inhalation mask. Body temperature was regulated at 36°C to 38°C with the help of a heating pad and a lamp. The right hemisphere skull was exposed and a flexible optical filament (Probe 318-I, Perimed, Stockholm, Sweden) was fixed to the skull 4 mm lateral from midline and 2 mm posterior from bregma with instant glue (LOCTITE 415 glue and LOCTITE TAK PAK accelerator 7452, Gothenburg, Sweden) and attached to a laser Doppler (Periflux System 5,000, Perimed, Stockholm, Sweden) for regional cerebral blood flow (rCBF) measurements. Using a microscope, a silicon-coated filament (6-0 nylon filament, Xantropren L, and activator universal, Heraeus Kulzer, Hanau, Germany) was introduced into the internal carotid artery through an incision in the external carotid artery (ECA). The filament was advanced to block the origin of the MCA, where the placement was confirmed by a reduction in laser Doppler flow. Halothane concentration was lowered to 0.8% during the 50mins period of occlusion.
After the occlusion period, recirculation was accomplished by withdrawing the filament. After the removal, the origin of the ECA was permanently ligated. Thereafter, the laser-Doppler probe was removed and wounds in the neck and on the head were sutured. Only mice with an immediate reduction in laser Doppler flow during occlusion and with a recirculation 5 mins after filament removal were included in the study.
After recirculation, mice were kept in an incubator for 2 h in 35°C and injected with 0.5 mL of 5% glucose in Krebs subcutaneously. The temperature in the incubator was set to 30°C overnight. The following day, animals received an additional injection of glucose and were transferred to room temperature. The body temperature was measured at 1, 2, 24, and 48 h after recirculation.
After 48h, animals were returned to standard (20 × 37 × 14 cm3) cages with maximum four mice in each cage. The enriched group was either housed continuously in a multilevel enriched cage (88 × 65 × 140 cm3) (EE24h) or transferred from standard cages to an enriched cage for 3 h daily (EE3h). The enriched cage contained plastic tubes, ropes, chains, ladders, platforms, running-wheel, and toys, which were changed every second day (Figure 1). Animals in standard cages were handled once a day.

The enriched environment, consisting of a large (88 × 65 × 140cm3) multilevel cage containing plastic tubes, ladders, chains, ramps, and platforms that were changed every second day. The enriched environment consisted also of social stimulation and housed 10 to 15 mice at the same time.
Behavioral Test
Rotating pole test: Gross motor function was evaluated by the ability of the mouse to traverse a rotating pole as described for rats previously (Ohlsson and Johansson, 1995) with the following modifications. The pole had a diameter of 15 mm and a length of 1,500 mm. It was elevated 750 mm above the ground and rotating at 10r.p.m. to the right or left, respectively. The animals were trained to transverse the pole to reach a platform on the opposite side, with no pole rotation and with a rotation of 3r.p.m. At a rotation speed of 10r.p.m., the following scoring system was used. A score of 0 to 6 was given for each rotation direction; 0—the animal is unable to transverse the pole and falls off almost immediately; 1—the animal has severe difficulties to cross the pole and when crossing the paws embrace the pole. The animal is unable to reach the platform; 2—the animal embraces the pole with its paws while crossing the pole, and manages to transverse the pole; 3—the animal crosses the pole with the body close to the pole, jumping with the hind legs or slipping with its feet; 4—the animal transverses the pole with approximately 10 foot slips; 5—the animal crosses the pole with around 3 to 7 foot slips and a normal posture and; 6—the animal transverses the pole perfectly, with 0 to 2 foot slips. All test sessions were video recorded (Sony digital handycam) for evaluation by a masked investigator.
Urine Collection and Corticosterone Analysis
Every fourth to fifth day, urine samples were collected for corticosterone analysis from uninjured mice. The mice were placed in a plastic cage (20 × 26 cm2) until the animals urinated, but not longer than 60mins. The urine was collected with a syringe and stored in plastic tubes at −20°C until analysis (Van de Weerd et al, 1997). All urine samples were collected between 09.00 and 10.00h, and before EE3 h mice were transferred to the enriched environment. Corticosterone levels were measured using an ELISA-kit (EA 66, Oxford biomedical research, Oxford, USA) according to the manufacturer's instructions.
Mortality
Animals that were showing signs of severe discomfort or illness, with no prospects of recovering were killed and included in the mortality group together with animals that spontaneously died.
Infarction Assessment with Immunohistochemistry
Fourteen to sixty days after MCAO, mice were anesthetized with 4.5% halothane and perfusion fixated with saline followed by 4% cold paraformaldehyde (PA). The brains were removed and allowed to fixate in PA for 24 h followed by saturation in 25% sucrose. Free-floating coronal sections of 40μm were cut with a distance of 560μm with the help of a microtome (Leach SM, 2000R Houston, USA); alternatively five sections were cut at bregma 1.18, 0.38, −0.82, −1.58, and −2.54 mm. The sections were stained by immunohistochemistry for a neuronal-specific antigen (Neu N) for infarction evaluation. The sections were rinsed in buffer, and 3% H2O2 and 10% methanol were applied to quench endogenous peroxidase activity. After preincubation with normal horse serum, sections were incubated with monoclonal anti-Neu N antibody (1:100, Chemicon, Temecula, USA) overnight at 4°C. The following day, the sections were rinsed and incubated with secondary biotinylated horse anti-mouse antibody (1:200, Vector Laboratories Inc., Burlingame, USA) and thereafter reacted with avidin–biotin–peroxidase complex (ABC kit, Vector Laboratories Inc., Burlingame, USA). Diaminobenzidine (DAB) staining was developed in a nickel-intensified peroxidase reaction and infarction was evaluated with the help of a stereo-logical microscope and software (Olympus Bx51, Cast2) at 10 × magnifications by a masked investigator. The border between the infarct and viable brain tissue was outlined so that the infarct area contained no NeuN-stained neurons. The area of the injured brain was attained by subtracting the normal brain tissue on the damaged side by the total brain area on the contralateral side. Infarct volume for each section was calculated as infarct area × slice thickness (distance between slices in μm).
In Situ Hybridization
After a breif exposure to halothane, mice were decapitated and the brain was quickly removed and frozen at −80°C. The EE3 h mice were killed immediately after the rotating pole testing session, while the EE24 h mice were decapitated 1 week after the testing session. Coronal sections were taken at the level of the striatum and hippocampus in a cryostat and the 14μm thick sections were thaw mounted onto Super Frost Plus slides (Menzel-Gläser, Germany) and stored at −20°C. Nerve growth factorinduced gene A probe was obtained from invitrogen (Stockholm, Sweden) and has previously been described (Pinaud et al, 2002): 5‘-CCGTTGCTCAGCAGCATCATCTC CTCCAGTTTGGGGTAGTTGTCC-3’. Slides were fixed in PA and dehydrated in graded ethanol before hybridization. They were then hybridized with NGFI-A probe, DTT, and hybridization buffer overnight at 42°C in humidified sealed boxes. After hybridization, the slides were washed in 1 × SSC at 55°C. They were finally washed in water and then dehydrated in ethanol and then air-dried before exposure to film (KODAK, BioMax MR film).
Quantification of In Situ Hybridization
The level of NGFI-A mRNA was assessed by measuring the optical density in the parietal cortex from the rhinal to the sagital fissure in the hemisphere contralateral to the infarct, using an image analysis system (ImageJ 1.32j, NIH, USA). The quantification of radioactivity was made by plotting a standard curve calculated from the carbon-14 autoradiographic micro-scales (Amersham Life science, Buckinghamshire, England).
Statistics
For presentation of the rotating pole test results, the mean value for each group was calculated. Differences on the rotating pole scores between groups were analyzed with the Kruskal–Wallis and the Mann–Whitney U-test. Infarction volumes, rCBF, body temperatures, mRNA expression, and corticosterone levels are expressed as mean ± s.d. and were analyzed with ANOVA, Scheffé's post hoc test or Student's t-test. Differences in mortality rate were analyzed with the χ2 test. A P-value of >0.05 was considered statistically significant.
Results
Effect of Enriched Environment on Functional Recovery on the Rotating Pole Test and Infarct Volume
Mice subjected to focal ischemia by transient occlusion for 50mins of the right MCA and housed in either enriched environment for 3 h a day, or in standard cages were tested on the rotating pole 14 days after surgery. Mice in the enriched environment performed significantly better in crossing the pole when the pole was rotating to the right (5.1) as well as to the left (4.6) when compared with standard housed mice (2.1 to the right and 1.8 to the left) (Figures 2A and 2B).
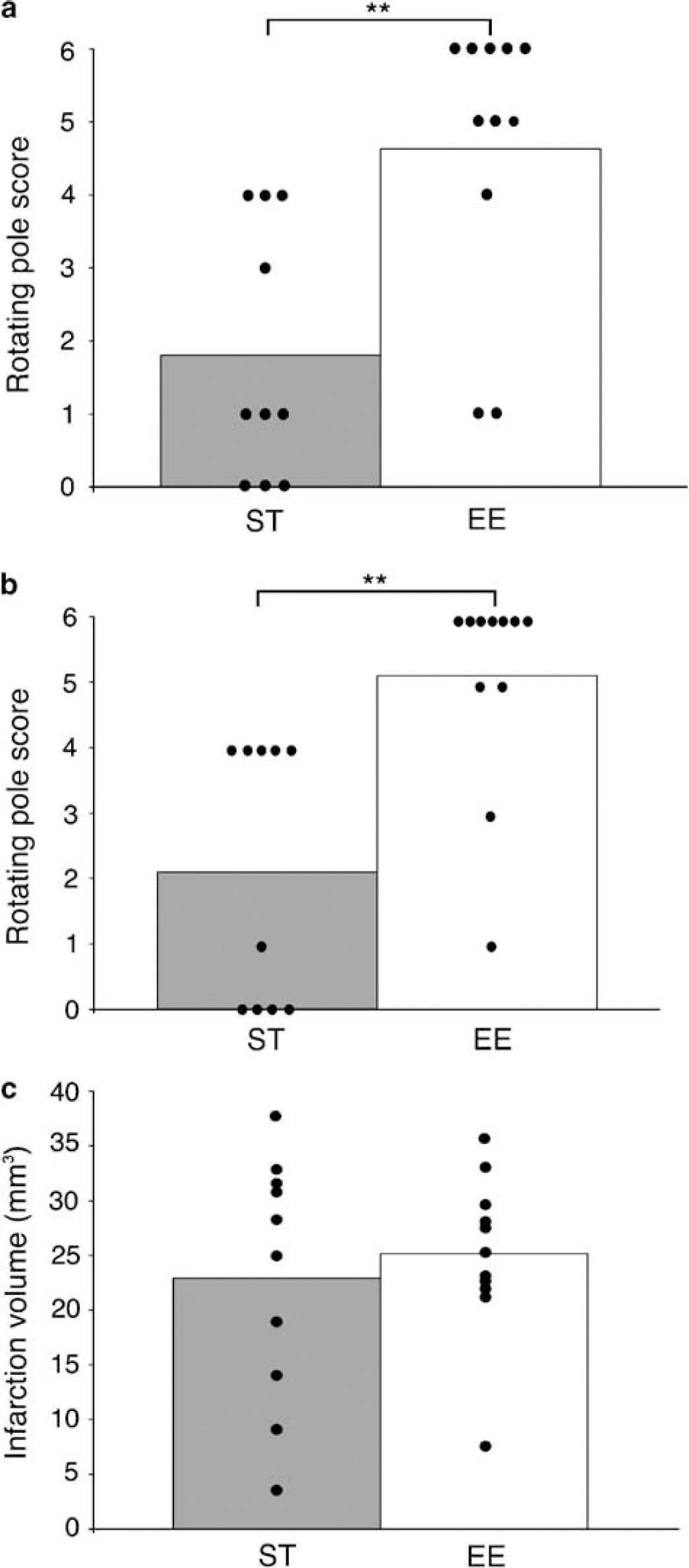
Effect of enriched environment on the rotating pole test and infarction volume. Fifty minutes of right middle cerebral artery occlusion (MCAO) was induced in mice, which were then housed in standard cages (n = 10) or in an enriched environment (n = 11) 3 h a day for 2 weeks. The mice were then tested on the rotating pole turning 10 r.p.m. to the left (
No significant difference in infarct volume was found between enriched and standard housed animals (Figure 2C). Body temperature 1 h after occlusion did not differ between groups. At 2 h of recirculation, there was a small but significant difference in body temperature between groups (Table 1). The mortality rates for mice in enriched environment and standard cages were 21% and 23%, respectively.
Body temperature, rCBF, and mortality in mice housed in standard cages (ST) and in an enriched environment (EE)
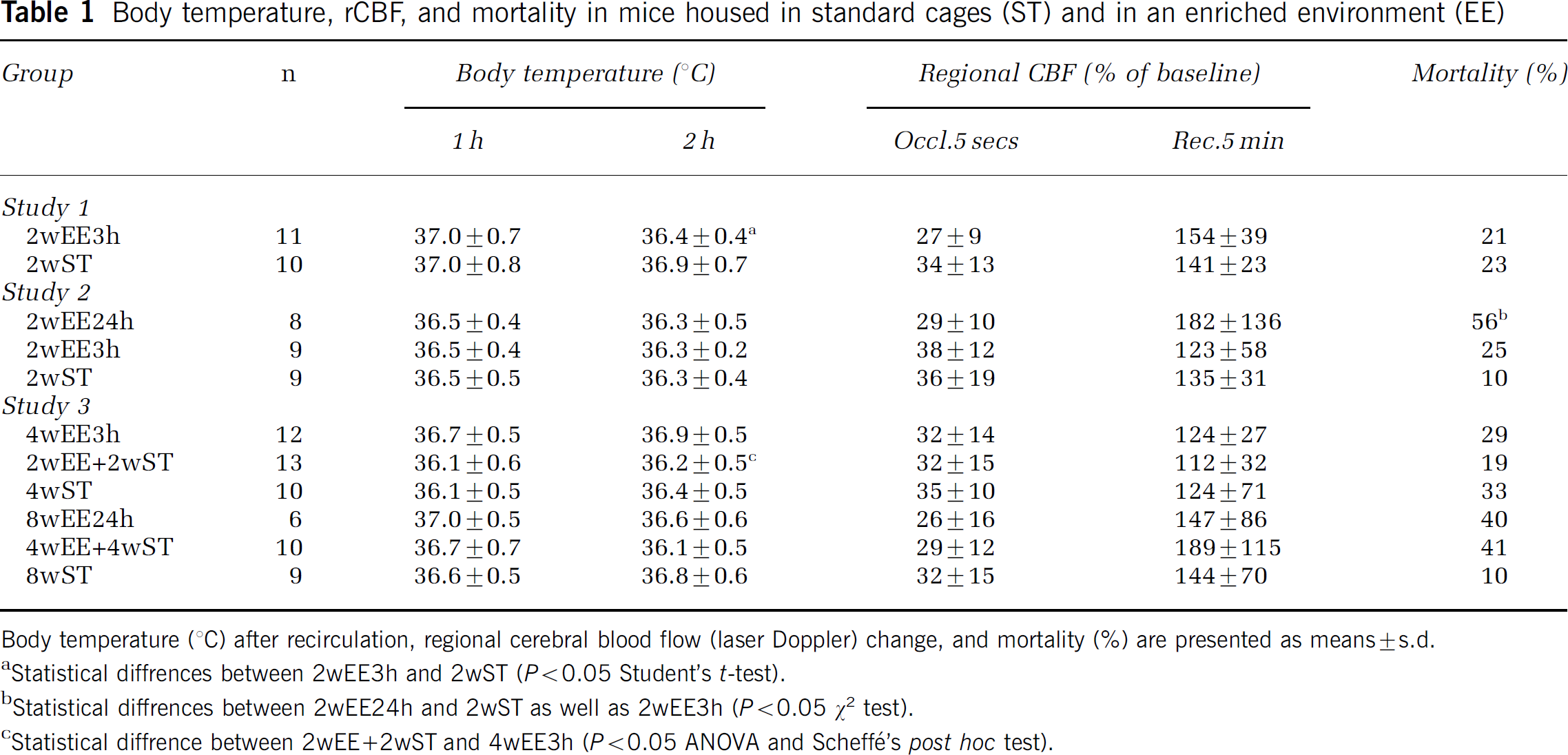
Body temperature (°C) after recirculation, regional cerebral blood flow (laser Doppler) change, and mortality (%) are presented as means±s.d.
Statistical diffrences between 2wEE3 h and 2wST (P < 0.05 Student's t-test).
Statistical diffrences between 2wEE24 h and 2wST as well as 2wEE3 h (P < 0.05 χ2 test).
Statistical diffrence between 2wEE+2wST and 4wEE3 h (P < 0.05 ANOVA and Scheffé's post hoc test).
Effect of Exposure to Enriched Environment for 3 and 24 h a Day on Functional Recovery in the Rotating Pole Test
Mice subjected to focal ischemia by 50mins of MCAO and housed in either enriched environment for 3 h a day, 24 h a day or in standard cages were tested on the rotating pole 14 days after surgery. Mice exposed to enriched environment for 3 h a day in enriched environment performed significantly better in crossing the pole when rotating to the right (5.0) as well as to the left (4.9) when compared with standard housed mice (2.1 to the right and 2.2 to the left). Mice housed 24 h a day in enriched environment were also significantly better than mice in a standard environment on the rotating pole test (4.6 to the right and 4.3 to the left) (Figure 3). However, mice housed in an enriched environment during the entire day for 2 weeks had a significantly higher mortality (56%) than mice housed only 3 h a day in enriched environment (25% mortality) and standard housed mice (10% mortality). There was no mortality among intact control animals housed in an enriched environment for 3 and 24h, or in standard environment.
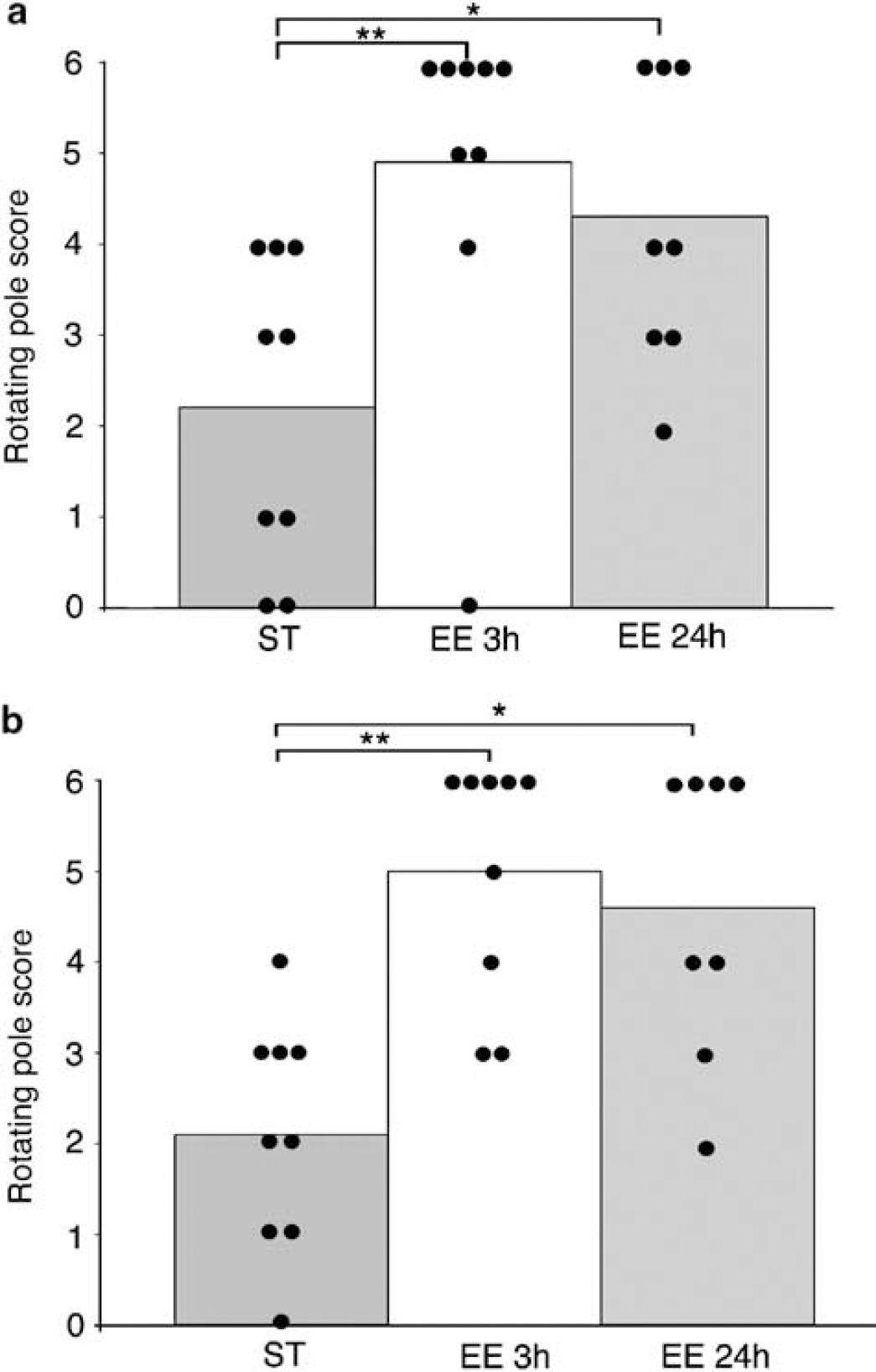
Performance on the rotating pole test after different exposures to enriched environment. Mice were subjected to 50 mins middle cerebral artery occlusion (MCAO) and then exposed to an enriched environment (n = 8) for 24 h a day, 3 h a day (n = 9), or housed in standard cages (n = 9) for 2 weeks in the rotating pole test when rotating to the left (
Urine corticosterone levels from control mice with different housing conditions: Urine corticosterone levels from control mice housed in standard cages (n = 5), enriched environment 3 h a day (n = 5), or enriched environment 24 h a day (n = 5) were measured every fourth to fifth day during 2 weeks. There was a tendency to lower corticosterone levels in mice exposed to enriched environment (3h) compared with standard conditions, but the difference was not statistically significant (Table 2). Standard housed mice and animals exposed to enriched environment for 3 h a day required longer time periods before urinating, while enriched environment 24 h mice urinated almost immediately.
Urine corticosterone levels at different time points in uninjured mice with different housing conditions
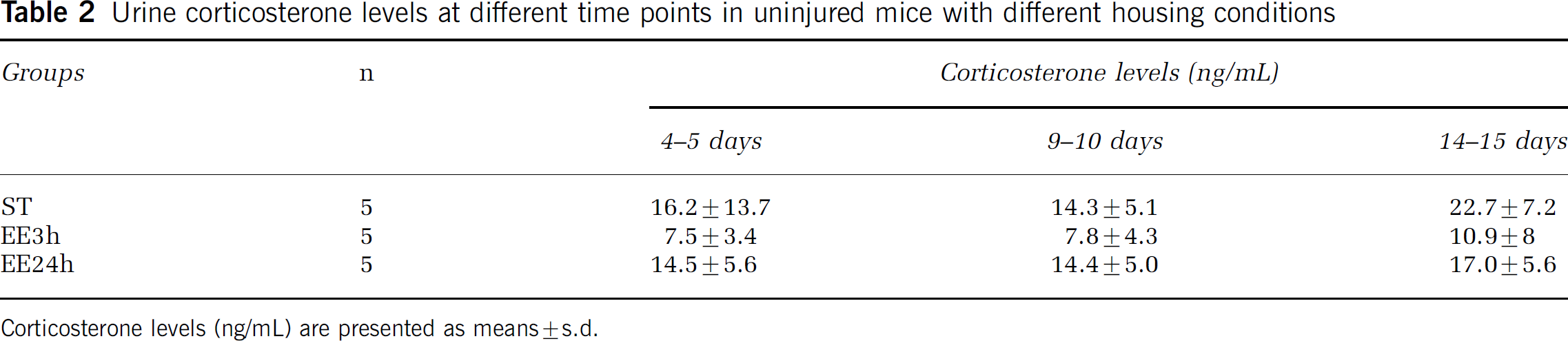
Corticosterone levels (ng/mL) are presented as means±s.d.
Exposure of Enriched Environment Followed by Standard Environment
After MCAO, mice were exposed to enriched environment for 3 h a day for 2weeks and tested on the rotating pole. Thereafter, they were transferred to a standard cage for another 2 weeks. After 2 weeks of exposure to EE (EE2w), mice had a rotating pole score of 5.2 to the left, which significantly decreased to 3.5 after 2 weeks in standard cages (EE2w + ST2w). One group was continuously housed in standard environment and tested at 2 weeks (ST2w) and 4 weeks (ST4w) and obtained a score of 2.7 and 3.0, respectively. One group was housed in an enriched environment for 4 weeks and obtained a score of 5.2 and 4.8, when tested at 2 (EE2w) and 4 weeks (EE4w), respectively (Figure 4). Infarct volume did not differ between groups (data not shown), but there was a small difference in body temperature after recirculation (Table 1). Mortality was similar between groups (Table 1).
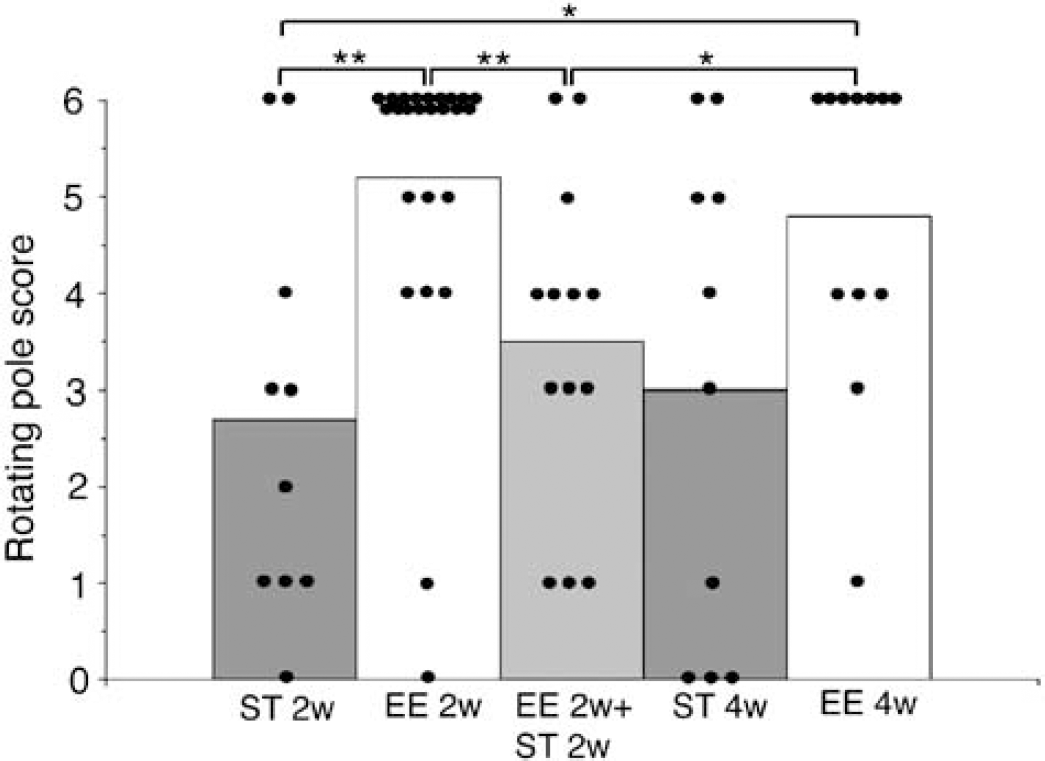
Effect of intermittent exposure to enriched environment. Rotating pole test score of mice subjected to 50mins middle cerebral artery occlusion (MCAO) and exposed to an enriched environment 3 h a day for 2 weeks, and then transferred to standard cages for another 2 weeks (n = 13, EE2w + ST2w). Control animals were either housed in standard cages (n = 10, ST2w and ST4w) during the whole 4 weeks or in enriched cages (EE2w n = 25, and EE4w n = 12) and tested at 2 and 4 weeks (**P < 0.01, *P < 0.05, Mann–Whitney U-test).
A prolonged experimental period with exposure to enriched environment for 24 h a day for 4 weeks and then transfer to standard cages for another 4 weeks resulted in a sustained effect in improvement of motor function on the rotating pole test (Figure 5). Animals in enriched environment had a score of 5.3 on the rotating pole test after 4 weeks (EE4w), which declined to a mean score of 4.0 after transfer to standard cages (EE4w + ST4w). Mice housed in standard environment for 8 weeks and tested at 4 (ST4w) and 8 weeks (ST8w) on the rotating pole obtained a score of 1.4 and 2.3, respectively. Multiple comparison among the experimental groups showed that the EE4w + ST4w group was not significantly different from the EE4w group and EE8w group (4.8). Also, the scores at EE4w and EE4w + ST4w were both significantly higher than the score from ST4w (1.4). However, at 8 weeks in standard environment the performance on the rotating pole improved spontaneously (2.3) but was still less than in the EE4w + ST4w (4.0). This difference was not statistically significant. In this study, mice that were exposed to enriched environment for 24 h a day had a higher mortality rate than standard housed animals (40% versus 10%), but the difference was not statistically significant.
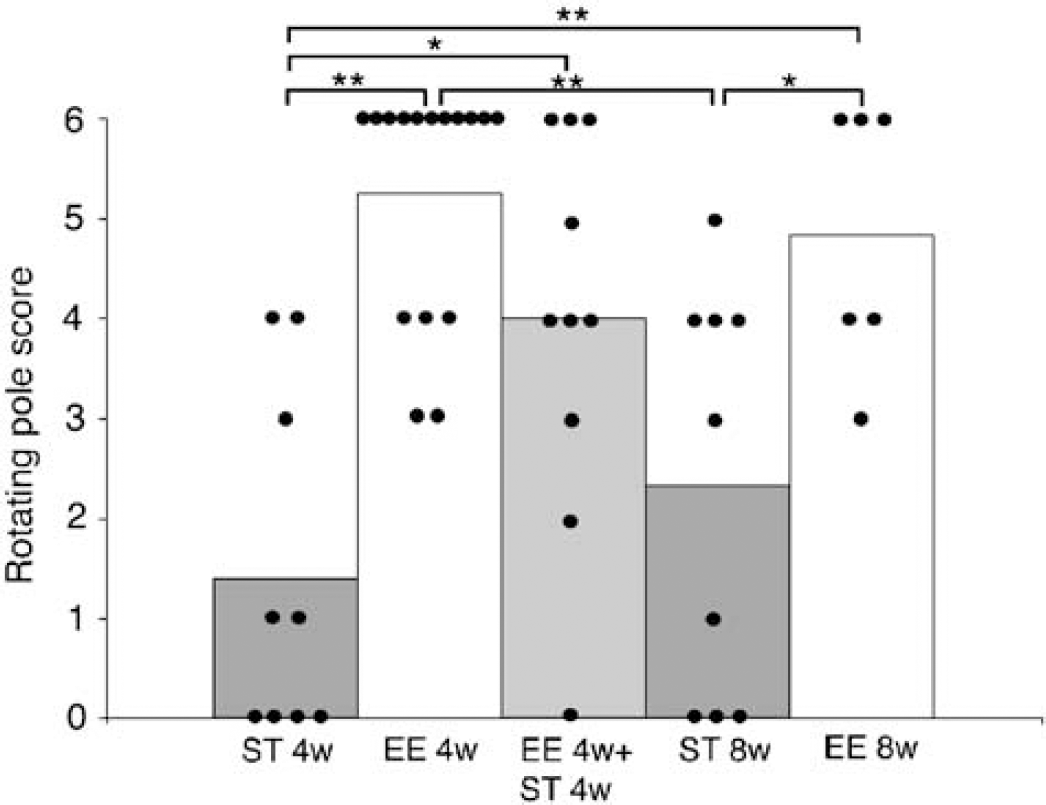
Effect of intermittent exposure to enriched environment. Rotating pole score of mice subjected to 50 mins middle cerebral artery occlusion (MCAO) and housed in standard cages for 4 weeks (ST4w) to 8 weeks (ST8w), or exposed to an enriched environment 24 h a day for 4 weeks (EE4w) to 8 weeks (EE8w), or in enriched environment for 4 weeks and then standard environment for 4 weeks (EE4w + ST4w) (**P < 0.01, *P < 0.05, Mann–Whitney U-test).
Nerve Growth Factor-Induced Gene A Messenger RNA Expression
Nerve growth factor-induced gene A mRNA was expressed as two distinct layers in the cortex and in all hippocampal subregions. Sham-operated animals housed in either enriched (n = 3) or standard cages (n = 3) had high cortical expression of NGFI-A transcripts and there was no difference in the expression between the two treatment paradigms (434±70 and 406±42 nCi/g, respectively). These two sham-operated groups were therefore pooled. Ischemia resulted in a decrease in the expression of NGFI-A after prolonged periods of recovery in mice housed in enriched environment (Figure 6). Brains from ischemic mice housed in enriched environment (EE24 h n = 3, EE3 h n = 3) and with a rotating pole score of 4 or above had an expression of 265 ± 51 and 315 ± 64 nCi/g, respectively, where EE24 h had a significant lower expression than sham-operated animals. Standard housed mice (ST24 h n = 3, ST3 h n = 3) with a rotating pole score of below 3 had an expression of 384 ± 12 and 388 ± 24nCi/g. Although there was a tendency of lower expression values they were not significantly lower than sham.
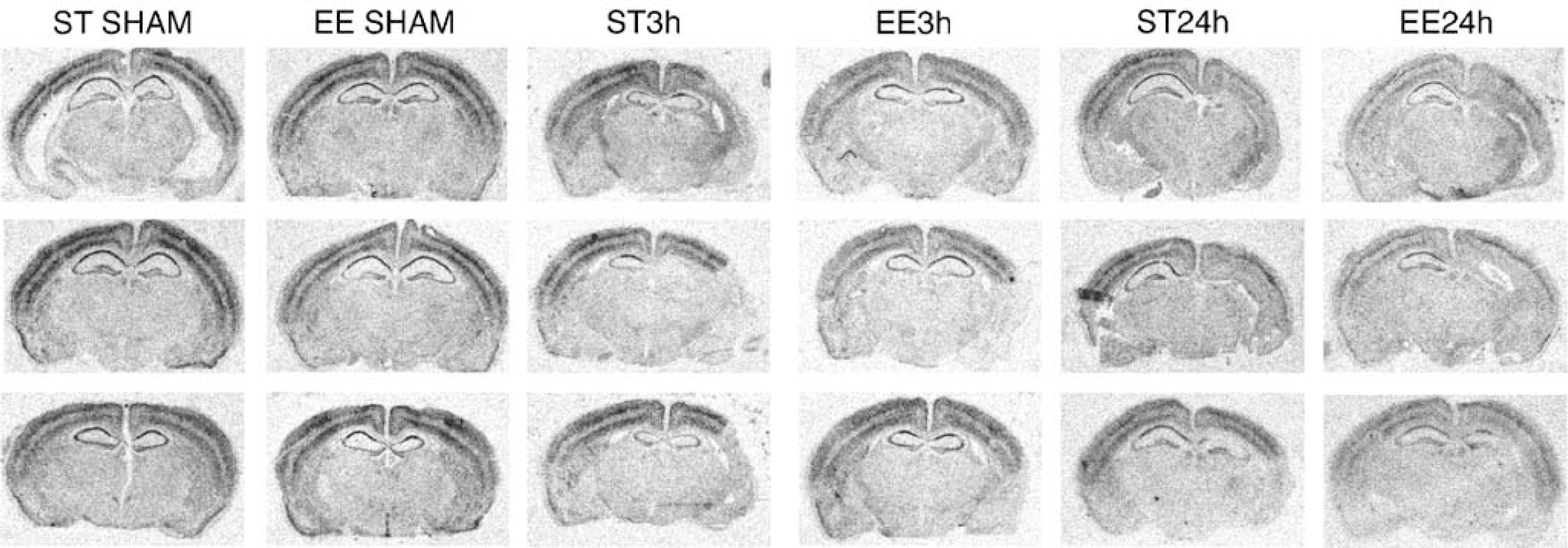
Effect of enriched environment on nerve growth factor-induced gene A (NGFI-A) mRNA expression. Autoradiograms of NGFI-A mRNA in situ hybridizations on brain sections from control mice housed in standard cages, control mice housed in enriched environment, MCA-occluded mice housed in ST environment for 14 days, MCA-occluded mice exposed to enriched environment 3 h a day for 2 weeks, MCA-occluded mice housed in ST cages for 3 weeks, and MCA-occluded mice housed in enriched environment for 3 weeks. All hybridizations were from the same autoradiogram.
Discussion
Here we show that daily exposure to enriched environment after MCAO significantly enhances the motor function in mice, and that enriched environment does not affect infarct volume. We also show that enhanced functional outcome from enriched environment is not sustained if mice are transferred to standard cages 2 weeks after an experimental stroke. If mice are exposed to enriched environment for 4 weeks, the improvement of motor function is maintained more efficiently, but still there is a decrease in functional outcome on the rotating pole test when animals are transferred to standard cages. Finally, we found that the levels of NGFI-A mRNA after MCAO were lower in brains from animals exposed to enriched environment for 3 weeks, than from mice in standard cages.
This is the first study to show that enriched environment has a beneficial effect on motor function recovery after transient focal ischemia in mice. In general, our results are in agreement with previous studies in rats (Ohlsson and Johansson, 1995) and humans (Ernst, 1990), which show that social interaction and/or physical activity in a stimulating environment results in improved motor function after stroke. Importantly, and also in agreement with previous studies (Grabowski et al, 1995; Ohlsson and Johansson, 1995), the exposure to an enriched environment does not affect infarct size in our experiments. A small (approx 0.6°C) but significant difference in temperature between two experimental groups 2 h after recirculation was seen. This decrease in temperature is coincidental, and most probably does not affect the functional outcome because infarct size was unaffected by the temperature difference. Also, the decrease in temperature is too small and occurs too late after MCAO to be protective. Mild hypothermia is neuroprotective only if initiated within 1.5 h after occlusion of the MCA and if brain temperature is 33°C (Krieger and Yenari, 2004).
The motor dysfunction seen after MCAO in mice is similar to that seen in rats, and likewise, the rotating pole test adapted to mice serves as an indicator of gross motor functions. Moreover, the test monitors sensori-motor function, is easy to use, provides consistent data, and extensive training before induction of ischemia is not necessary. However, handling of animals during the whole experimental time is important, because unhandled animals tend to become stressed; this affects their performance on the rotating pole test. Also, repetitive testing sessions on the rotating pole influence performance and should be avoided.
Mice housed in an enriched environment during the entire day for the whole experimental period had a higher mortality, compared with mice housed only 3 h a day in enriched environment, or with standard cage housed animals. One possible explanation for the higher mortality rate is that mice housed in large cages, with large groups of animals or animals housed in enriched environment are more aggressive, have unstable hierarchies, and are more stressed as indicated by the elevated level of plasma corticosterone (Haemisch et al, 1994; Van Loo et al, 2001). However, we did not find any difference in corticosterone levels in mice housed under different conditions. Evidently, a short (3h) daily exposure compared with continued housing in enriched environment is preferred because the mortality in injured mice is lower, while there is equal recovery of function. Also, when not exposed to an enriched environment for 3h, the mice are housed in small cages with a maximum of three cage mates, which is optimal for preventing aggressive behavior (Van Loo et al, 2001).
Animals that were kept for 2 weeks in enriched environment (EE3h) after transient MCAO, did not maintain their improved motor function, while mice housed continuously in enriched environment for 1 month maintained their functional improvement better. It thus appears that at least 4 weeks in enriched environment is necessary for a stable neuronal rewiring in the mouse brain to take place after experimental stroke. It has been shown that during recovery after brain injury, the synaptic connections of the injured rat brain are reorganized, which include axonal sprouting, synapse formation, and cortical remapping (Carmichael et al, 2001; Chen et al, 2002; Jones, 1999). Also, formation of new synapses starts at 3 to 4 weeks after a brain lesion (Jones, 1999; Jones et al, 1996; Jones and Schallert, 1992). Indeed, housing rats in an enriched environment significantly increases the number of dendritic spines in the superficial cortical layers of the somatosensory cortex (Johansson and Belichenko, 2002). The half-life of spines vary from only a few days to several months (Trachtenberg et al, 2002), and sensory deprivation modulates spine turnover, decreasing the pool of stable spines (Trachtenberg et al, 2002). Hence, functional improvement after exposure to enriched environment could be because of stabilization of the spine pool, which may be lost if the stimulation is not maintained. The data are in accord with the clinical findings that longer periods of rehabilitation of stroke patients are mandatory for a sustained beneficial effect (Ernst, 1990).
In normal, intact mice, housed in an enriched environment, several genes are regulated (Rampon et al, 2000). Nerve growth factor-induced gene A is a transcription factor that is involved in brain plasticity and that increases in the brain of rats housed in an enriched environment (Olsson et al, 1994; Pinaud et al, 2002; Wallace et al, 1995). We did not find a similar effect in the intact brain of mice housed in an enriched environment. In fact, sham-operated mice housed in standard environment or enriched environment had similar levels of NGFI-A mRNA expression.
However, the observed decrease in NGFI-A to below control levels 2 to 3 weeks after MCAO that we observed in the mouse brain after housing in enriched environment is similar to that reported in the rat (Dahlqvist et al, 1999). Still, the levels of NGFI-A in the brain of mice housed in standard cages for 2 or 3 weeks after MCAO were not different from sham-operated animals, in contrast to the findings in the rat brain where NGFI-A levels were elevated (Dahlqvist et al, 1999). However, this elevation was transient and at 4 weeks the levels of NGFI-A were similar to that seen in sham-operated rats. The results indicate that the observed differences in NGFI-A expression in the brain of mice and rats in response to the environmental changes do not seem to be because of differences in some fundamental mechanisms of NGFI-A regulation, but rather because of differences in the dynamics of the postischemic gene expression, which is strain dependent or influenced by the intensity of insult, and the impact of the environment.
In conclusion, this is the first study to show that enriched environment enhances motor function after experimental stroke in mice. The enhancement subsides gradually if animals are not maintained in the enriched environment for at least 1 month. Also, we show that housing conditions markedly affect gene expression during the recovery phase. Hence, we have established a mouse model for future studies of the influence of particular genes on the functional recovery in mice after a focal brain ischemia.
Footnotes
Acknowledgements
The authors thank Professor Barbro Johansson and Maj-Lis Smith for valuable suggestions and discussions.
