Abstract
Cocaine-induced hypoperfusion, a risk factor for ischemic stroke, has not been fully characterized during experimental drug-taking among individuals with cocaine use disorder. We sought to examine cocaine's dose-dependent, time-related effects on cerebral blood flow. In a double-blind, randomized human laboratory study with a counterbalanced order of drug administration, 31 male and female subjects with cocaine use disorder were divided into two groups receiving either (a) low-dose cocaine (0.325 mg/kg intravenously) or placebo (N = 15) or (b) high-dose cocaine (0.650 mg/kg intravenously) or placebo (N = 16). The different dose conditions were administered on test days separated by a rest period of ≥48 h. Cerebral blood flow was assessed quantitatively using H2O15 positron emission tomography. Experimentally administered low- and high-dose cocaine conditions versus their corresponding placebo conditions were associated with global and regional hypoperfusion. The trend for high- versus low-dose cocaine to be associated with greater hypoperfusion achieved statistical significance only for the dopamine-rich sublobar and midbrain regions. Cocaine's hypoperfusion effects were maximal at 8 mins after infusion (i.e., at about the expected peak of intravenous cocaine levels) and had mostly dissipated by 32 mins after infusion. Although hypoperfusion occurred throughout the brain, the left hemispheric dopamine-rich sublobar region was the most severely affected. Cocaine-induced cerebral hypoperfusion is associated with the time course of its pharmacological effects, and dopamine-rich areas, particularly in the left hemisphere, may be most vulnerable. Increasingly larger doses of cocaine may be associated with greater risk for ischemic stroke.
Introduction
An important cause of morbidity and mortality among cocaine addicts is ischemic stroke (Kaku and Lowenstein, 1990; Klonoff et al, 1989; Krendel et al, 1990; Petitti et al, 1998). The site-specific effects of cocaine-induced changes in cerebral perfusion, either measured directly or implied from studies of neuronal metabolism or activation, among cocaine addicts during drug-taking are relatively less well understood (Lamki et al, 1993; London et al, 1990a, b; Tumeh et al, 1990; Woods et al, 1991), even though the extent of the abnormalities can be severe (Holman et al, 1992; Jensen et al, 1990; Woods et al, 1991). Further, there is variation in the pattern of cerebral blood flow (CBF) abnormalities depending on whether the cocaine addict has recently stopped using cocaine or has been abstinent for a prolonged time period (Fowler et al, 1993; Holman et al, 1991; Strickland et al, 1991; Volkow et al, 1996; Weber et al, 1993). Generally, however, these studies have reported that experimentally administered cocaine in cocaine addicts is associated with global and regional reductions in CBF (Kaufman et al, 1998; Mena et al, 1990), and the regional hypoperfusion appears to be more severe in dopamine-rich brain regions (Johnson et al, 1998; Pearlson et al, 1993). While there is evidence that the amount of cocaine used in the week before imaging and cocaine-associated cue response are most correlated with activation in left hemispheric dopamine-rich areas such as the orbito-frontal cortex (Bolla et al, 2003; Bonson et al, 2002; Garavan et al, 2000; cf. Kilts et al, 2001), it is not known whether CBF changes during and immediately after drug-taking are associated with more predominant left hemispheric hypoperfusion. This effect may be because of the fact that previous studies have typically had relatively small sample sizes and, therefore, insufficient statistical power to detect such differences. Few studies have, however, used positron emission tomography (PET) or enhanced quantitative techniques now available with single photon emission computerized tomography (Johnson et al, 1998) to quantify accurately cocaine's site-specific effects on CBF. Further, relatively little is known about the dose-dependent and time-course-related effects of experimentally administered cocaine on CBF.
Using PET in a study with ample statistical power, we examined whether the effects of experimentally administered intravenous cocaine on global and cerebral perfusion were dose dependent, and whether these changes in perfusion were altered over the course of cocaine's pharmacological effects.
Methods
Subjects
We enrolled 31 male and female subjects who met DSM-IV criteria for cocaine abuse or cocaine dependence. Their mean age was 34.2 years (range, 18 to 46 years). Twenty-four subjects were men. Fifteen subjects were African American; eight were Hispanic; seven were Caucasian; and one was of multiethnic heritage. Twenty-seven subjects were right-handed and four were left-handed. Subjects were current cocaine users who were not seeking treatment. Seven of the cocaine-using subjects reported current predominantly intravenous administration; the remainder reported predominantly intranasal administration. Current use of >500 mg/day of caffeinated drinks, >1 pack/day of cigarettes, or sympathomimetics other than cocaine was exclusionary. Most subjects smoked marijuana on a regular basis. No subjects had current or past histories of diabetes or hypertension. Subjects were recruited by advertisements in newspapers, radio, and television for paid research volunteers who had experience using drugs.
Confirmation that subjects were in good physical health was ascertained by physical examination and hematological (i.e., complete blood count) and biochemical screening tests (i.e., glucose, electrolytes, liver and thyroid function tests, and urine drug screen). The research protocol was approved by the Institutional Review Board of The University of Texas Health Science Center at San Antonio. After receiving a complete description of the study, all participants gave their written informed consent to participate.
Experimental Design
Subjects were divided into two groups that received either (a) low-dose cocaine (0.325 mg/kg intravenously) or placebo cocaine (N = 15) or (b) high-dose cocaine (0.650 mg/kg intravenously) or placebo cocaine (N = 16). The required intravenous dose of cocaine was dissolved in 3 mL of 0.9% w/v saline solution and infused over 60 secs; placebo cocaine (3 mL of 0.9% w/v saline solution) also was infused over 60 secs. These two groups did not differ in demography or in pattern of alcohol or drug use (Table 1). The different dose conditions were administered on separate test days, each separated by a rest period of at least 48 h. All subjects were admitted to the University Hospital Clinical Psychopharmacology Laboratory and were maintained abstinent from cocaine, alcohol, and other drugs for the duration of the study, including at least 48 h before the start of testing. The order of drug administration was randomized, double-blind, and counterbalanced. All H2O15 PET sessions included a baseline scan and two post-cocaine or -placebo cocaine infusion scans at 8 and 32 mins.
Demographics and psychiatric diagnoses of subjects with cocaine use disorder
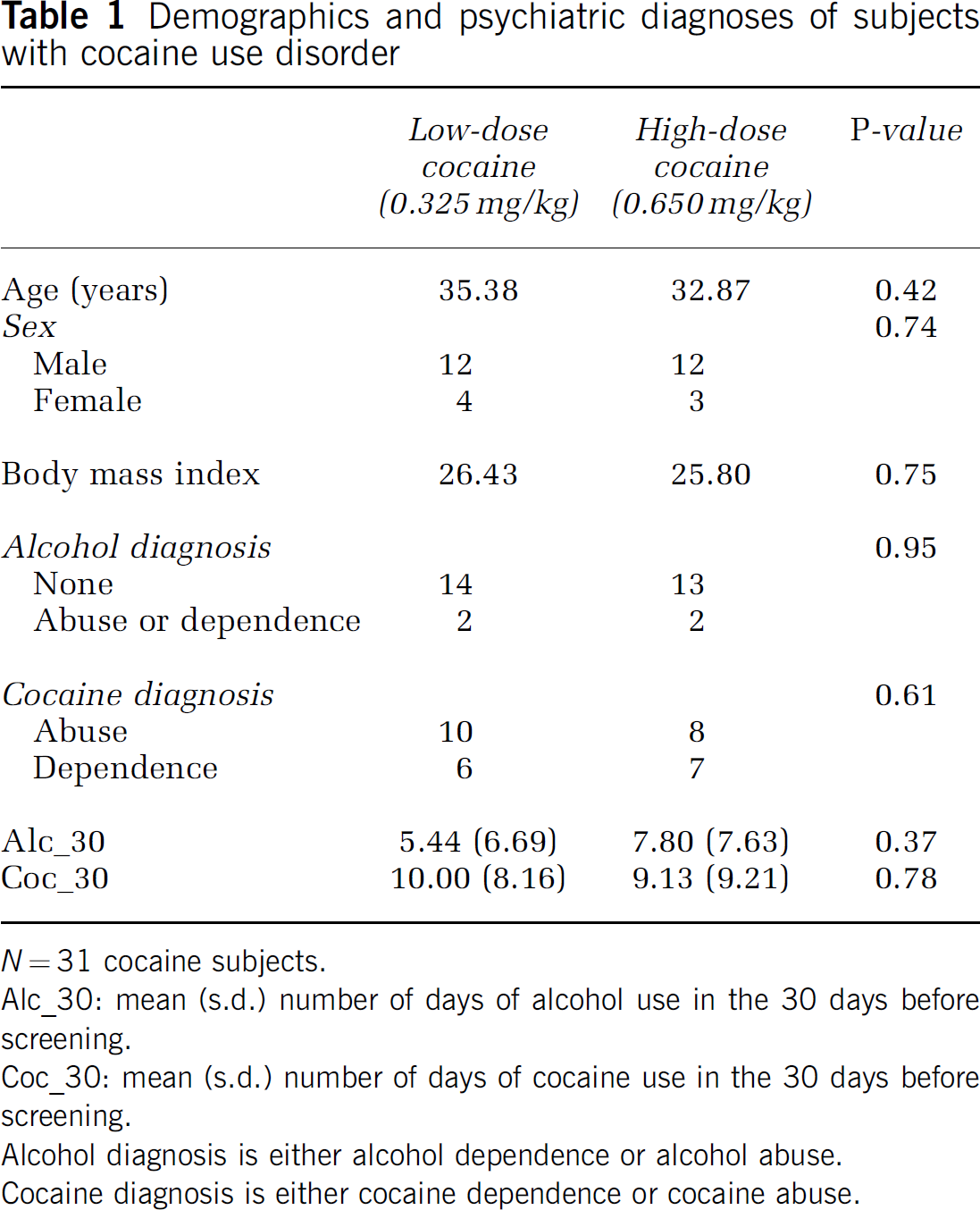
N = 31 cocaine subjects.
Alc_30: mean (s.d.) number of days of alcohol use in the 30 days before screening.
Coc_30: mean (s.d.) number of days of cocaine use in the 30 days before screening.
Alcohol diagnosis is either alcohol dependence or alcohol abuse.
Cocaine diagnosis is either cocaine dependence or cocaine abuse.
Neuroimaging Techniques
Anatomic magnetic resonance imaging: All subjects received a baseline brain magnetic resonance (MR) imaging scan to exclude those with gross neurologic abnormalities and for the purposes of coregistration with the PET images. All MR examinations of the brain were performed on a 1.9 T magnet (Elscint; Haifa, Israel). Horizontal, three-dimensional, T1-weighted (35/6: TR/TE) images were obtained to rule out gross structural lesions in gray matter, using 1.5-mm-thick slices with a 1-mm gap and a 256 × 256 matrix. Pixel size was 1 × 1 mm.
Cerebral blood flow imaging protocol: All PET images were acquired on a General Electric Scanditronix 4096 camera. Before the beginning of each scanning session, a thermoplastic facial mask immobilized the subject's head. Scanning sessions included a transmission scan using a 68Ge/68Ga pin source, to correct for attenuation for each of the three (i.e., baseline, and 8 and 32 mins post-cocaine or -placebo cocaine infusion) emission scans. Each scanning session lasted approximately 45 mins. Each PET image had the following parameters: 15 parallel slices; pixel spacing = 2 mm; center-to-center inter-slice distance = 6.5 mm full-width at half-maximum; z-axis field of view = 10 cm. Reconstruction with a Hanning filter yielded a resolution of 8 mm. For each of the perfusion scans for the subjects with cocaine use disorder, we administered an intravenous bolus of 50 to 58 mCi H2O15 tracer, with the dose determined by body weight. For all emission scans, the tracer was suspended in 5 to 10 mL of sterile, non-radioactive saline. The arrival of the tracer bolus in the brain triggered the PET scan. Images were acquired spanning 90 secs after the early vascular phase of the radiotracer.
Image registration and spatial normalization: The MR and PET images were spatially normalized to match the position, orientation, and dimensions of the 1988 Talairach Atlas brain (Talairach and Tournoux, 1988). Each PET image within a study was first assessed for interscan, intrasubject movement, and corrections were made using automated image registration software (Woods et al, 1993). A mean PET image was formed for each subject from his/her coregistered PET images. The three-dimensional anatomic MR image for each subject was spatially normalized using a nine-parameter affine transform with a landmark-based method (Lancaster et al, 1995). The mean PET image for each subject was then spatially transformed to match this spatially normalized MR image using a convex hull surface-based method (Lancaster et al, 1999). This resulted in a single transform that was applied to each subject's PET images, thereby completing spatial normalization processing.
Imaging Analysis
Primary dependent measures: The primary dependent measures of global CBF and regional CBF (rCBF) were extracted from our previous work on the cocaine-induced changes in rCBF (Johnson et al, 1998). These areas included analyses of the whole brain and both hemispheres. Also analyzed were lobar regions including the limbic lobe (basal ganglia, caudate), sublobar region, occipital lobe, temporal lobe, parietal lobe, and frontal lobe, as well as the brainstem and midbrain.
Imaging data processing: To assess global and regional changes in CBF that might be encountered in pharmacological studies without the use of arterial sampling, voxel-wise tissue activity of [15O] water was value-normalized to a fixed whole-brain value (Raichle et al, 1983). Positron emission tomography images were first obtained at a threshold of 30% of the maximum voxel count to define the brain region of the scan volume. Areas outside this volume were set to zero. Each voxel in the PET image was divided by the injected activity to correct for minor fluctuations. This procedure produced voxel units of counts/mCi. The corrected voxel-wise tissue activity was assumed to be proportional to rCBF.
Standard regions of interest (ROI) were defined by the 1988 Talairach Atlas in the Talairach Daemon (Lancaster et al, 1997); see also Figures 1 and 2 (Lancaster et al, 2000). The mean count for each ROI within a PET image was assumed to be proportional to the mean rCBF.
A typical set of three-dimensional data used to generate the volume occupancy Talairach labels around the z = +1 level. Openings in lobe through cell levels were provided to emphasize the three-dimensional nature of data at each level. (From Lancaster et al (2000), copyright © 2000. Reprinted by permission of Wiley-Liss, Inc., a subsidiary of John Wiley & Sons, Inc.)
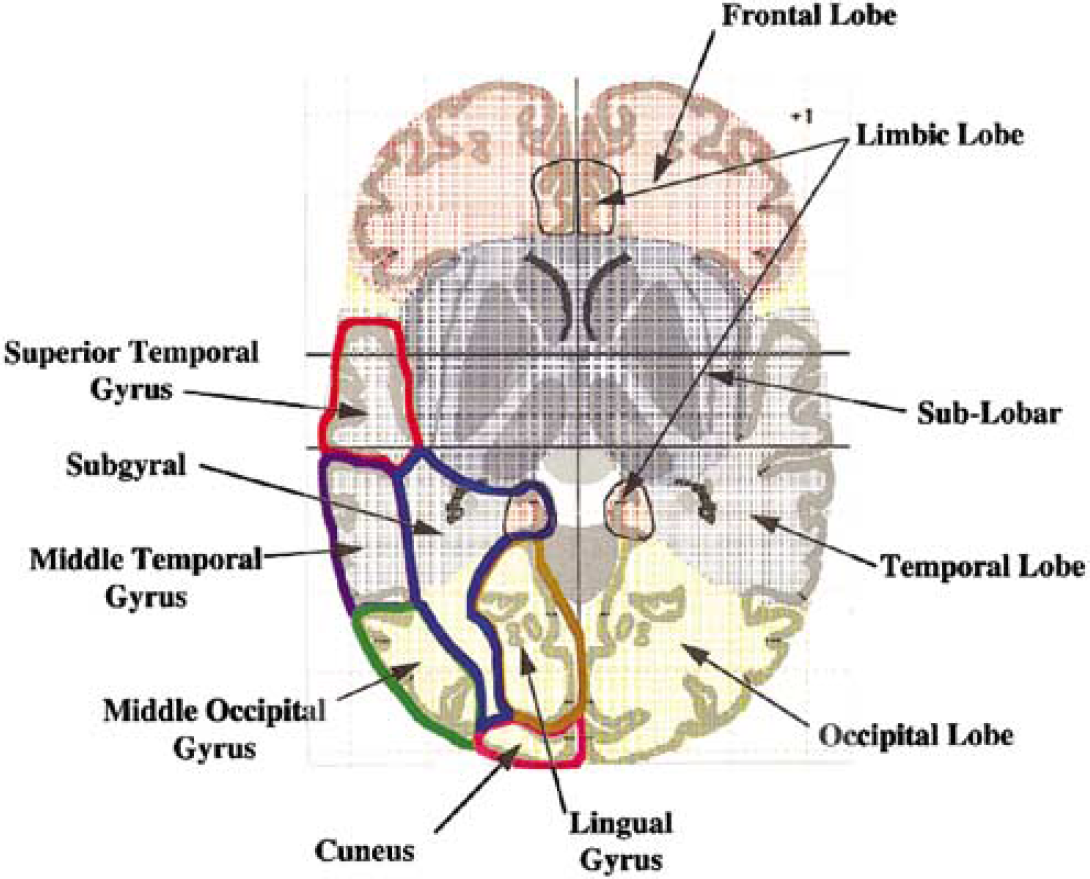
An example of volume occupancy Talairach labels for a Talairach Atlas section image at the z = +1 level. Lobar levels are demarcated by patterned colors. Bold color outlines are used to demarcate gyral levels on the bottom left of the image. (From Lancaster et al (2000), copyright © 2000. Adapted by permission of Wiley-Liss, Inc., a subsidiary of John Wiley & Sons, Inc.)
Data Analytic Strategies
Analyses were conducted to determine cocaine-induced and time-related changes in CBF at global, hemispheric, and lobar levels of analysis. Cocaine and placebo effects were measured as the CBF (e.g., counts/mm3). Cerebral blood flow data were normalized by the square root transformation. Within the statistical model, we adjusted for age, gender, and body mass index-weight (kg)/height2 (m2).
Before examining for cocaine dose and time effects, we planned to examine whether there were differences in CBF based on laterality at the hemispheric and lobar levels of analysis. We measured the magnitude of changes in CBF by calculating the ratio of CBF at 8 or 32 mins after cocaine infusion divided by the 8- or 32-min placebo condition, after adjusting for baseline CBF.
The general statistical method was the use of a repeated-measures analysis of variance followed by paired t-test contrasts, adjusted by Bonferroni corrections for the numbers of contrasts being made. Contrasts were of three types. First, we examined for significant differences between cocaine dose and placebo within time points (e.g., at 8 and 32 mins postinfusion). Second, we analyzed the differences in CBF across time within cocaine dose level for postinfusion PET studies. Third, we contrasted the differences in high- versus low-dose cocaine at 8 and 32 mins after infusion.
Results
Global, Hemispheric, and Lobar Effects of Cocaine on Cerebral Blood Flow
To understand the changes in counts/mCi after cocaine infusion, we present the raw data means and standard deviations of PET results for 8 mins before infusion, 8 mins after infusion, and 32 mins after infusion (Table 2). We also wanted to understand whether the results were similar to those found in infarct zones and peri-infarct zones. Therefore, we compared our results for global CBF in our cocaine-using subjects with those in 20 age- and sex-matched controls. We assumed an rCBF of 50 ml/min per 100 g for controls, with the mean count for controls of 2349.66 (s.d. = 725.35). Using this scaling factor, we estimated the global CBF at 8 mins before infusion and at 8 and 32 mins after infusion (Table 3). The corrected rCBF values appear similar in magnitude to rCBF values found in borderzone and territorial infarcts (De Reuck et al, 2004; Powers, 1991; Sette et al, 1989).
Counts/mCi by hemisphere and lobe
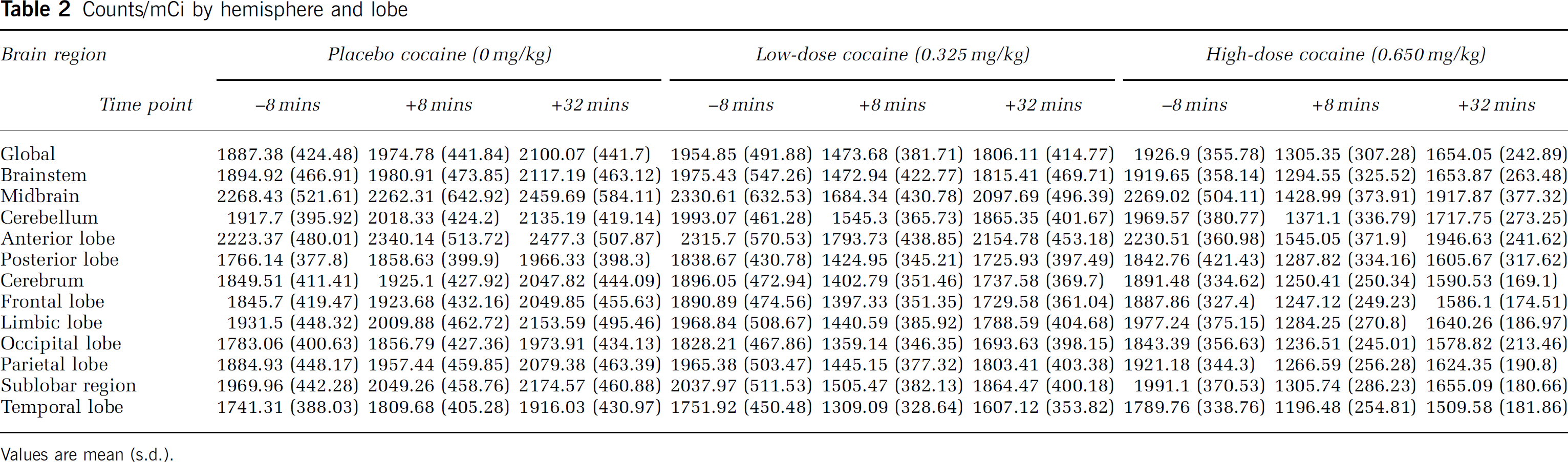
Values are mean (s.d.).
Scaled global CBF for cocaine subjects

Values are mean (s.d.).
CBF, cerebral blood flow, PET, positron emission tomography.
Experimentally administered intravenous cocaine was associated with significant dose-dependent reductions in global CBF and rCBF-the higher cocaine dose being associated with the greatest level of hypoperfusion (Tables 4 and 5; Figures 3 and 4).
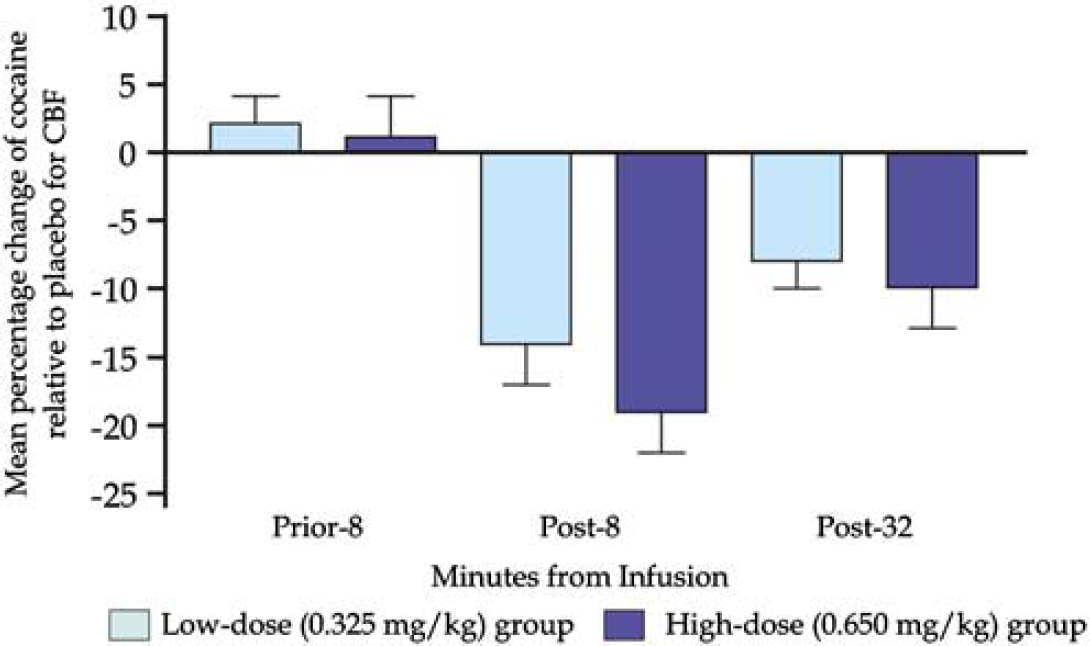
Mean percentage change in global cerebral blood flow (CBF) assessments among those receiving low-dose cocaine versus placebo or high-dose cocaine versus placebo.
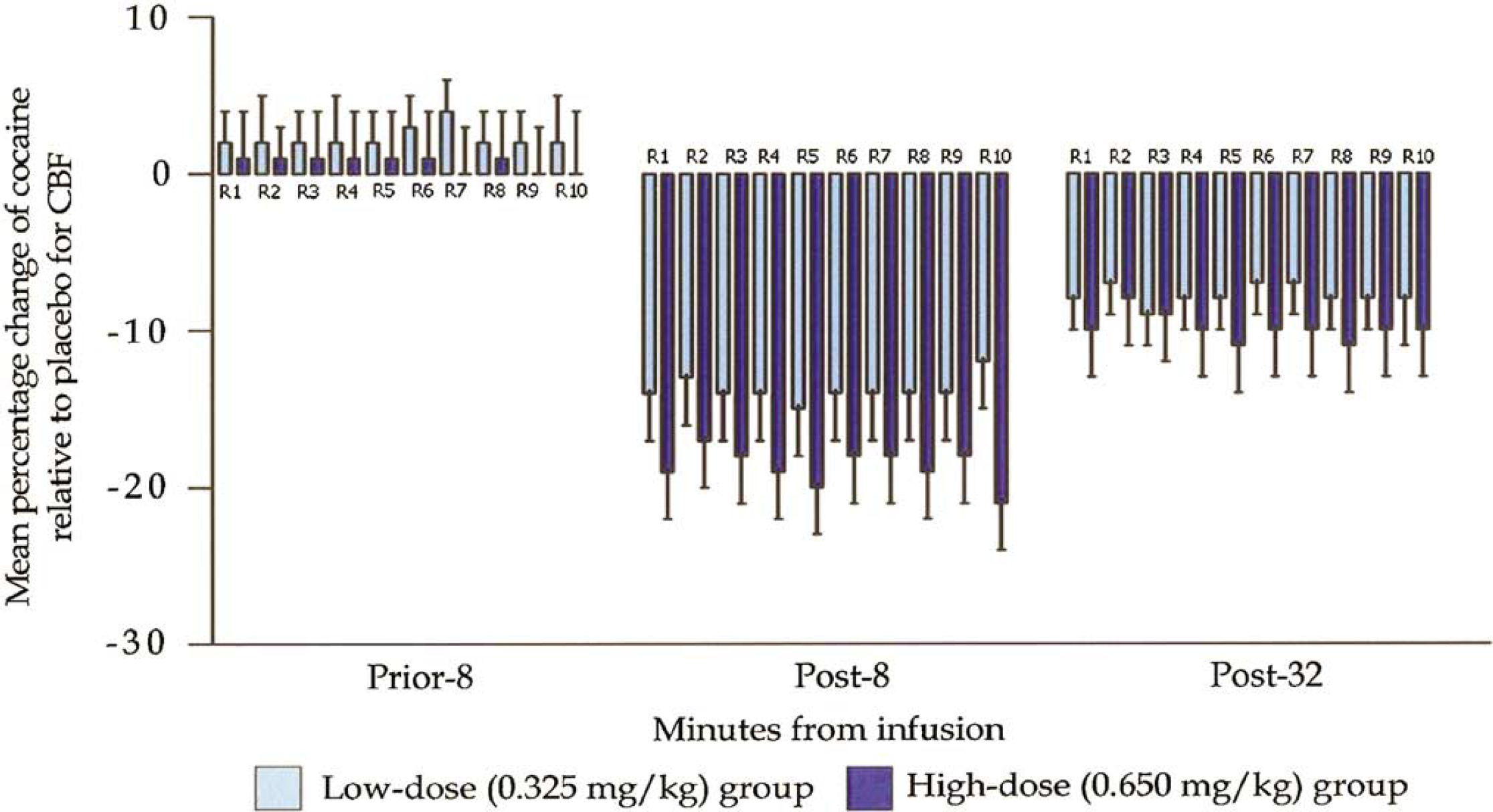
Mean percentage change in regional cerebral blood flow (CBF) assessments among those receiving low-dose cocaine versus placebo or high-dose cocaine versus placebo. R1 = cerebrum; R2 = cerebellum; R3 = brainstem; R4 = frontal lobe; R5 = limbic lobe; R6 = parietal lobe; R7 = occipital lobe; R8 = sublobar region; R9 = temporal lobe; R10 = midbrain.
Cocaine effect; comparison of mean square root of blood flow (counts/mm3) (adjusted by baseline, age, sex, and body mass index) between placebo and low-dose cocaine (0.325 mg/kg); N = 16
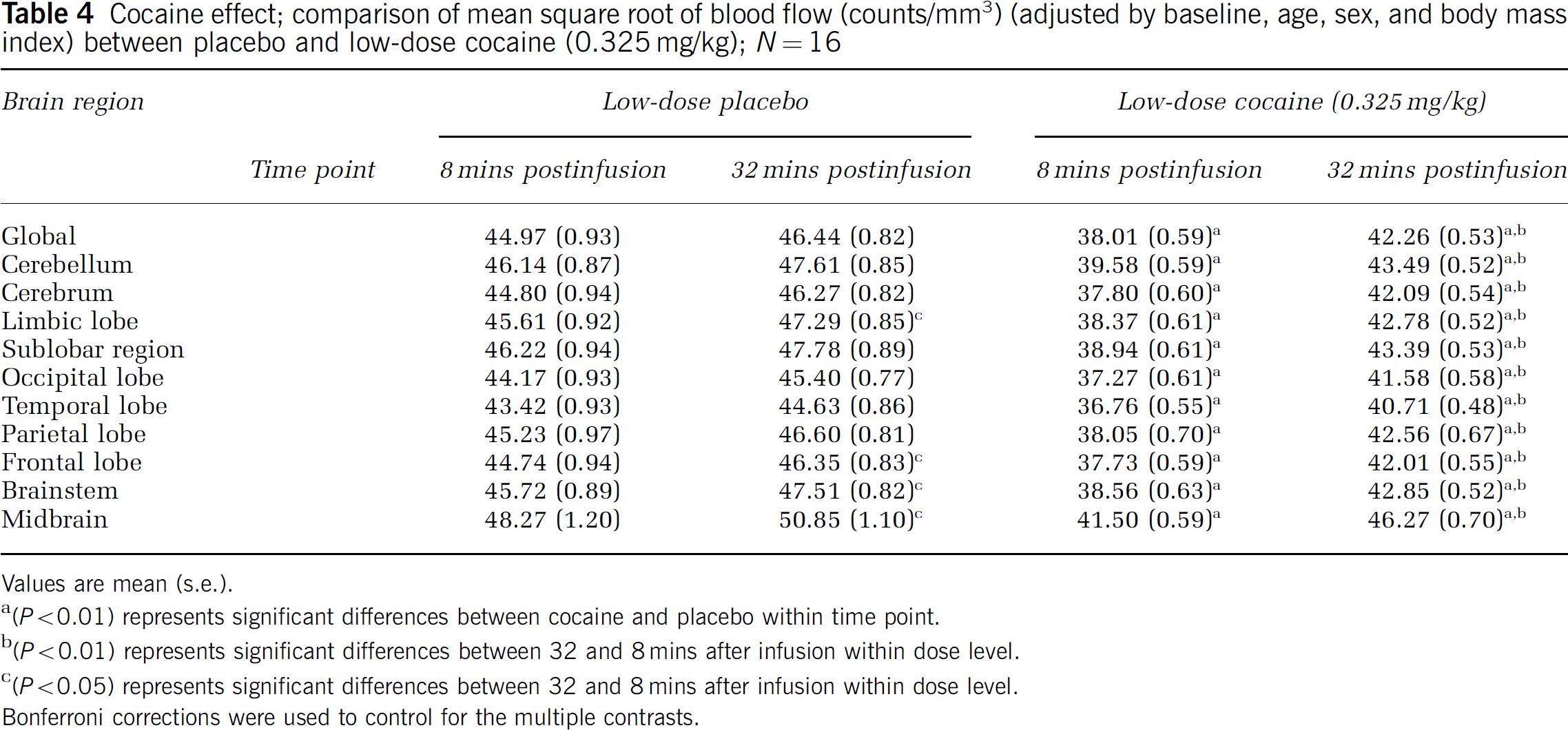
Values are mean (s.e.).
(P<0.01) represents significant differences between cocaine and placebo within time point.
(P<0.01) represents significant differences between 32 and 8 mins after infusion within dose level.
(P<0.05) represents significant differences between 32 and 8 mins after infusion within dose level.
Bonferroni corrections were used to control for the multiple contrasts.
Cocaine effect; comparison of mean square root of blood flow (counts/mm3) (adjusted by baseline, age, sex, and body mass index) between placebo and high-dose cocaine (0.650 mg/kg); N = 15
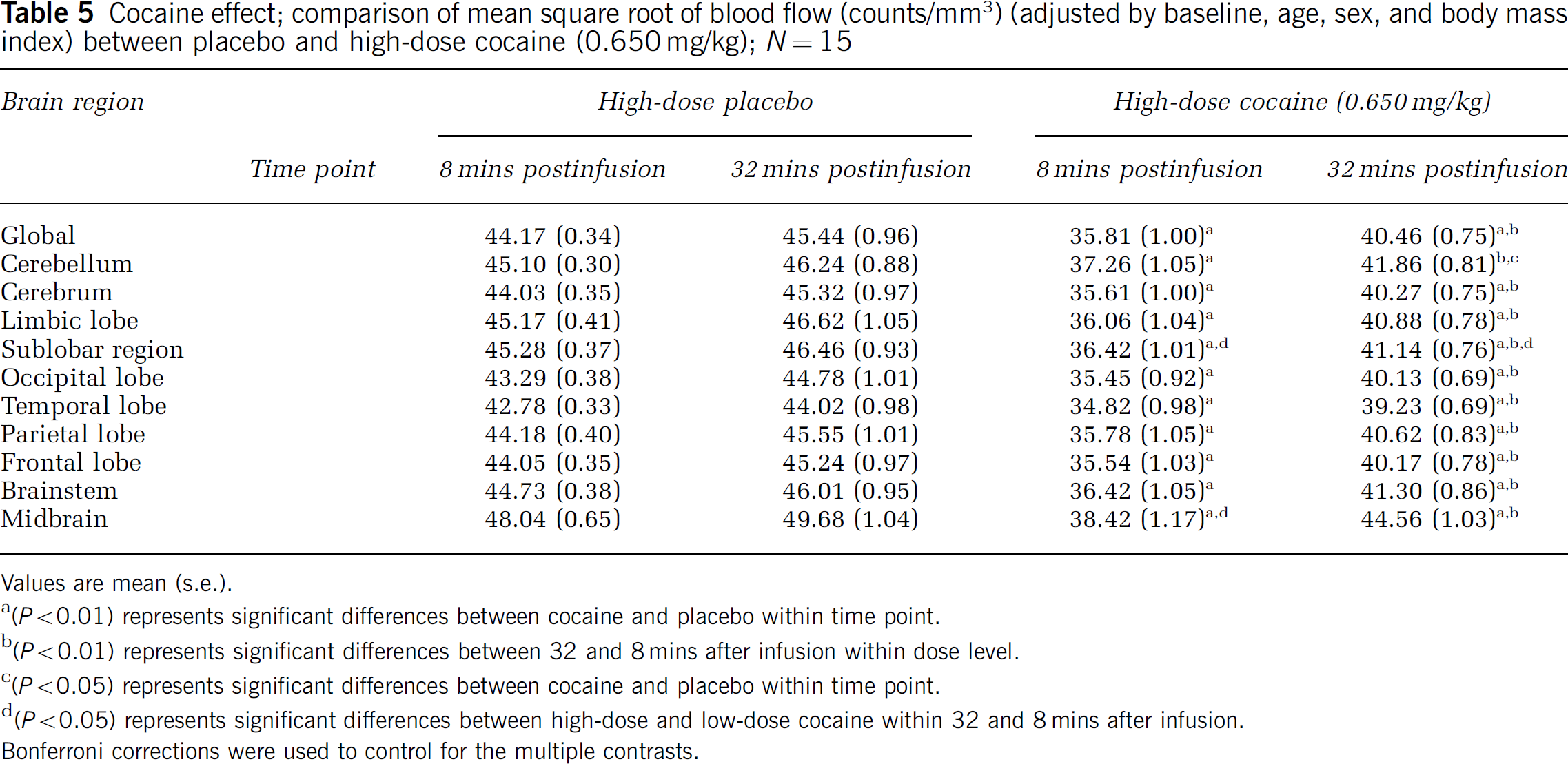
Values are mean (s.e.).
(P<0.01) represents significant differences between cocaine and placebo within time point.
(P<0.01) represents significant differences between 32 and 8 mins after infusion within dose level.
(P<0.05) represents significant differences between cocaine and placebo within time point.
(P<0.05) represents significant differences between high-dose and low-dose cocaine within 32 and 8 mins after infusion.
Bonferroni corrections were used to control for the multiple contrasts.
Cocaine-related decreases in CBF were significantly greater on the left compared with the right sublobar region (F = 5.65, df = 26, P = 0.025). Further, these effects were predominantly seen at the high but not the low dose of cocaine (mean difference (1-{32-min scan for cocaine/32-min scan for placebo}/{8-min scan for cocaine/8-min scan for placebo} in the left-to-right ratio of high-dose cocaine relative to placebo = 0.012 {s.e. 0.004}; t = 2.74, P = 0.01)). For all other regions-including the cerebellum, cerebrum limbic lobe, occipital lobe, temporal lobe, parietal lobe, frontal lobe, and midbrain-there were no significant differences in the laterality of cocaine effects. As shown in Figure 2 (Lancaster et al, 2000), the sublobar region contains the insula, caudate, thalamus, lentiform (putamen), and claustrum.
As can be seen in Tables 4 and 5 and Figure 4, significant differences were observed between high-dose cocaine (0.650 mg/kg intravenously) and placebo, and between low-dose cocaine (0.325 mg/kg intravenously) and placebo, within each time point; these cocaine-placebo differences were seen at the hemispheric (cerebellum, cerebrum), lobar (limbic, occipital, temporal, parietal, frontal, sublobar), and midbrain levels of analysis. The postinfusion time-trend analysis also showed significant differences in cocaine-induced decreases in CBF in all hemispheric, lobar, and midbrain regions examined. Significant differences between high- and low-dose cocaine were seen at both 8 and 32 mins after infusion, but only in the dopamine-rich sublobar and midbrain regions (Table 5). At 8 mins after infusion, high-dose cocaine produced significantly greater decreases in CBF, compared with low-dose cocaine, in the sublobar region and in the midbrain. At 32 mins after infusion, dose-dependent effects on CBF were seen only in the sublobar region.
Discussion
We observed that experimentally administered intravenous cocaine was associated with dose-dependent reductions in global CBF and rCBF; the higher cocaine dose was associated with a greater level of hypoperfusion. Hypoperfusion was greatest at 8 mins after cocaine infusion, which corresponds to about the expected time of the peak of intravenous cocaine levels (i.e., 4 to 8 mins) (Evans et al, 1996; Foltin and Fischman, 1991). Because peak arterial cocaine level, which is achieved at approximately 15 secs after cocaine infusion, is more likely than peak intravenous cocaine level to represent the cocaine level in the brain (Evans et al, 1996), the magnitude of hypoperfusion seen at 8 mins might have led us to underestimate the maximal reductions in CBF that occurred after cocaine dosing. Cerebral blood flow changes at 32 mins after cocaine infusion for both the high- and low-dose cocaine conditions were similar to their corresponding placebo conditions. Despite the fact that cocaine's hypoperfusion effects were widespread, the greatest percentage changes were still seen in left hemispheric dopamine-rich areas of the sublobar region (Johnson et al, 1998). Indeed, although there was a consistent trend for greater hypoperfusion at 8 versus 32 mins after cocaine infusion, these differences only achieved statistical significance for the dopamine-rich sublobar and midbrain regions. Dopamine may control local blood flow by inducing vasospasm of smooth muscles lining the cerebral vessels (Anday et al, 1993; He et al, 1994; Johnson et al, 2001; Kaufman et al, 1998; Kurth et al, 1993; Madden et al, 1995), particularly those of the middle cerebral artery (He et al, 1994). In the living brain, pericytes containing contractile elements located proximal to dopamine nerve endings, which are also responsive to vasoactive substances in the endothelium, may be mechanistically responsible for this process (Haefliger et al, 1994; Shepro and Morel, 1993). Essential to this process appears to be an initial transmembrane loss of magnesium followed by a rapid rise in intracellular calcium concentration. The severity of the perfusion deficit is, therefore, typically correlated with the relative strength of these neurochemical changes (Zhang et al, 1996). Second, cocaine-induced reductions in cerebral metabolism may lead to feedback downregulation of blood flow (Rogers and Nahorski, 1973).
While our findings are consistent with the suggestion that dopamine-mediated processes are important for the control of CBF (Krimer et al, 1998), this effect is obviously not entirely dopamine dependent, and other neuronal mechanisms and vasculitis (Krendel et al, 1990) may play a role. For instance, while intracortical vessels are preferentially innervated by dopamine, those located extra-parenchymally receive their neuronal supply from norepinephrine-containing cells in the superior cervical ganglia (Krimer et al, 1998). That is, deep cortical brain regions may be more susceptible to dopamine's vasospastic effects, whereas reductions in CBF subsequent to constriction of large cranial arteries may be more dependent on norepinephrine facilitation (Bement et al, 1989; Langner et al, 1988). Cocaine-associated increases in serotonin level also may contribute to vasospasm of large- and medium-sized cranial arteries. Hence, multi-factorial mechanisms may be associated with the development and extension of cerebral vasoconstriction.
It would be noteworthy to consider the pathophysiologic significance of cocaine-associated hypoperfusion. There is a strong temporal relationship between cocaine use and the development of ischemic stroke, with most events taking place in the distribution of the middle cerebral artery (Daras et al, 1994; Johnson et al, 1998; Levine et al, 1990)-thus, medications that antagonize cortico-mesolimbic dopamine-mediated processes may be potential therapies. Despite the parametric strength of the H2O15 PET approach to quantifying hypoperfusion, this method alone, because it was not coupled with contemporaneous measurement of cellular metabolism, and longitudinal contrasts with structural MR, would not enable us to segregate hypoperfusion caused by vasospasm, neuronal downregulation, or infarction. Nevertheless, the magnitude of the decreases in CBF was about the same as, but not identical to, that of the reported reductions in cerebral glucose metabolism, also measured by PET, in a study using a similar maximal dose of cocaine challenge (London et al, 1990b), thereby suggesting that there may be a predictable relationship between these two parameters. Thus, simultaneously measuring blood flow and metabolism-and their relationship with cellular oxygen demand (i.e., oxygen extraction ratio)-in different brain regions might be a practical and elegant method for segregating areas of ischemia from those with infarction or changes in neuronal metabolism. Such studies are currently being conducted in our laboratory.
We observed greater left- compared with right-sided hypoperfusion in the sublobar region, which encompasses part of the orbito-frontal cortex. We suggest that this laterality might be because of the following: increased right orbito-frontal cortex neuronal loss, which has been shown in recently abstinent cocaine users (Matochik et al, 2003)-thus, there is relatively greater dopamine innervation in the left orbito-frontal cortical area; greater neuronal activation and metabolism associated with enhanced cocaine-induced reinforcement and related cues, which have been proposed as being mediated via activation of the left orbito-frontal cortex (Garavan et al, 2000); preponderance of right-handed subjects and, therefore, greater left hemispheric dopamine (Mohr et al, 2003); or some combination of the three possibilities. In essence, more left-sided orbito-frontal cortex dopamine innervation in these cocaine-using individuals, irrespective of the exact mechanism, would reasonably be expected to result in greater left-sided orbito-frontal cortex hypoperfusion after the administration of cocaine.
We encountered two experimental challenges in this study. First, the experimental complexity required to administer cocaine safely under controlled conditions to human subjects, and to synchronize the manufacture of the H2O15 isotope with PET scanning, was accomplished by intensive training and practice by the research team. Second, while we included both cocaine abusers and cocaine-dependent individuals in the study, we did not consider this to be a disadvantage because their patterns of recent cocaine and alcohol use were similar, and there was no obvious baseline difference in CBF assessments between them.
An important advantage of this study over other PET studies of CBF among individuals with cocaine use disorder is that, to our knowledge, this is the largest sample to date. Based on our estimates, this was an adequately powered experiment for testing contrasts in CBF from the hemispheric to the lobar level while still maintaining protection against type 1 statistical errors. Advantages of this study over most previous imaging studies of CBF include the use of intravenous cocaine dosed by body weight, which enabled clear temporal evaluation of cocaine's vascular effects, and testing two (i.e., low and high) pharmacologically active doses, which provided determination of the magnitude of CBF change as a function of dose.
In summary, the experimental administration of intravenous cocaine is associated with dose-dependent global and regional hypoperfusion. Cocaine's direct acute pharmacological effects, therefore, appear to be an important risk factor for ischemic stroke, and this potential may rise with increasing doses of cocaine. Nevertheless, the consequence and timing of an ischemic stroke among cocaine addicts may not only depend on actual cocaine-taking but also be related to the individuals' prior vascular status and proneness to thrombus formation (Johnson et al, 2001).
Footnotes
Acknowledgements
We thank the staff of the South Texas Addiction Research and Technology Center for their skilled technical assistance and dedication to this project. We are also grateful to Mr Robert Cormier for his assistance with the preparation of this manuscript.
