Abstract
Gender differences in neuropsychiatric disease are recognized but not well understood. Investigating the survival of primary rat hippocampal neurons in culture, we found significant and inverted gender differences on normoxia versus hypoxia. Male cells were more resistant under normoxia but more vulnerable under hypoxia than female cells. Male vulnerability pattern was acquired in cells from neonatally testosterone-primed females. Estrogens, acting via membrane receptors, had a higher neuroprotective power in male neurons, explained at least in part by the pronounced increase in estrogen receptor beta/alpha ratio during hypoxia in male cells only.
Introduction
For many neurologic and psychiatric diseases, gender differences in susceptibility, course, and response to treatment are known, although not well understood (e.g., Di Carlo et al, 2003; Häfner, 2003). For instance, in both animal models of hypoxia/ischemia and human stroke, male individuals show a predominance, bigger lesion size and higher mortality (Hurn and Macrae, 2000; Sadoshima et al, 1988). Estrogens, because of potent neurotrophic and neuroprotective qualities, have been associated with higher resistance of females to ischemia (Behl and Moosmann, 2002; Dubal et al, 1998; Garcia-Segura et al, 2001; Lee and McEwen, 2001; Simpkins et al, 1997). However, in addition to circulating sex hormones influencing the brain, there is sexually dimorphic gene expression in early brain development (Dewing et al, 2003), and dimorphic brain structures exist before the secretion of gonadal hormones starts (Arnold et al, 2004). At the cellular level, gender differences in morphology and function have been reported for cultured dopaminergic neurons (Reisert et al, 1989).
Using primary rat neurons derived from the hippocampus, a brain area known to be highly susceptible to hypoxia/ischemia, we report here two important basic findings: (1) neurons of male versus female rats show different survival in vitro under normoxia and hypoxia. (2) The neuroprotective potency of estrogens, mediated via estrogen receptors (ER) in the cell membrane, is higher in male neurons, possibly related to their higher ERbeta/ERalpha ratio on hypoxia.
Materials and methods
Animals
Experiments were approved by the local Animal Care and Use Committee. The gender of newborn Wistar–Imamichi rats was determined by anogenital distance and confirmed post hoc by polymerase chain reaction (PCR) genotyping for sex-determining region Y using the primers 5′-CCC GCG GAG AGA GGC ACA AGT-3′ (Hirasawa et al, 1995) and 5′-CTC TAC TCC AGT CTT GTC CG-3′ (312 bp, GenBank Acc. No. _X89730). Intermethod agreement exceeded 96%. For in vivo testosterone priming, newborn female rats received testosterone propionate (1.25 mg subcutaneously) as described (Garcia-Segura et al, 1988). Hippocampal neurons were prepared 24 h later.
Cell culture
Primary hippocampal neuronal cultures from male or female pups were prepared as described previously (Brewer, 1995) with slight modifications. Briefly, hippocampi were removed and digested with papain (Sigma, Taufkirchen, Germany), triturated, and subjected to density gradient centrifugation. Neuron-enriched fractions were resuspended in serum- and estrogen-free growth medium [NeurobasalA/B27 (Invitrogen, Karlsruhe, Germany) with 5 ng/mL basic fibroblast growth factor (bFGF, Invitrogen), 0.5 mmol/L L-glutamine (Sigma), 50 U/mL penicillin and 50 μg/mL streptomycin (Roche, Mannheim, Germany)]. For viability assessment, neurons were plated on 12 mm coverslips coated with poly-D-lysine (Sigma) at a density of 50 cells/mm2; for RNA isolation on 35 mm wells (∼70,000 cells per well). Cultures were grown in a humidified incubator at 37°C, 5% CO2, without further medium change. After 5 days, the time of experiments, 90% to 95% of cells showed positive staining for neuronal markers (β-tubulin-III, MAP-2). Contamination with glial fibrillary acidic protein (GFAP)-positive astrocytes was consistently less than 10%. Endothelial cells (factorVIII), microglia (OX-42), or oligodendrocytes (CNPase) were not detected.
Hypoxia
For experiments, medium was replaced with freshly prepared growth medium with/without 17β-estradiol (Sigma), testosterone propionate (Sigma), ICI182780 (Tocris, Bristol, UK), or BSA-estradiol (Sigma). In ICI182780 experiments, cultures were pretreated for 30 mins before hypoxia. Hypoxic conditions were induced by purging an incubator with a 95% N2/5% CO2 mixture. Cultures were exposed to hypoxia or normoxia (control) for 15 h.
Cell Viability
The percentage of viable neurons was determined using the Trypan blue dye exclusion method. For each condition, five distinct, nonoverlapping high-power fields on each of 2 to 3 different coverslips (i.e., 200 to 350 cells) per condition were counted.
Quantitative RT-PCR
Polymerase chain reactions were carried out on a LightCycler real-time PCR system (Roche). The reaction mixture consisted of 1 μL cDNA solution, 1 U Taq DNA polymerase (Invitrogen), 2 μL 10 × PCR buffer (Invitrogen), 0.2 mmol/L each or dATP, dCTP, dGTP, and dTTP (Roche), 2.5 mmol/L MgCl2, 500 mg/L bovine serum albumin (BSA) (New England BioLabs, Frankfurt, Germany), 50 mL/L dimethylsulfoxide (Sigma) and 1 × SYBR Green I (Molecular Probes, Eugene, OR, USA). Amplification primers were used at 0.5 μmol/L concentration. Polymerase chain reaction grade water was added to 20 μL. After initial denaturation (95°C, 30 secs), 45 cycles of (95°C, 0 sec; 55°C, 5 secs, and 72°C, 10 secs) with maximum ramp rate were performed. Primers were 5′-GACATGTTGCTGGCTACGTC-3′ and 5′-GGCCATCAAGTGGATCAAAG-3′ (ERalpha; 198 bp; GenBank Acc. No. NM_012689); 5′-AAAGTAGCCGGAAGCTGACAC-3′ and 5′-TACACCGGGACCACATTTTTG-3′ (ERbeta; 202 bp; GenBank Acc. No. NM_012754); 5′-CTG ACA CCA TGT CCG TCA CT-3′ and 5′-CAT GAC CAA GTC CAC GAC AG-3′ (rat aromatase cytochrome P450, 200 bp, GenBank Acc. No. M33986), and 5′-AAGCCCGTGCTGATGATGAA-3′ and 5′-CTTGCTGAACTTGCCGTTGG-3′ (EF-2; 372 bp; GenBank Acc. No._Y07504).
Statistical Comparisons
All values are means±s.e.m. Unless otherwise indicated, results are based on n=5 to 9 independent experiments from different preparations. Statistical analysis was performed by one-way analysis of variance (ANOVA) followed by LSD test (P<0.05 considered significant).
Results
In Vitro Gender Differences in Neuronal Survival on Hypoxia
After 5 days in culture, male and female neurons showed no morphological differences. Whereas a higher proportion of dye-positive neurons was observed in female as compared with male cultures under normoxic conditions, the opposite was true for hypoxia (Figure 1). Hypoxia increased cell death to 182% in male versus 129% in female cultures as compared with normoxia. Addition of 17β-estradiol at the start of hypoxia, in concentrations of 1, 10, 100, or 1000 nmol/L, reduced death rate in both female and, more potently, male neurons, with 10 nmol/L exerting the strongest neuroprotective effect (Figure 1, including inset). This concentration was used in further experiments. Testosterone (10 nmol/L) was not protective (not shown).
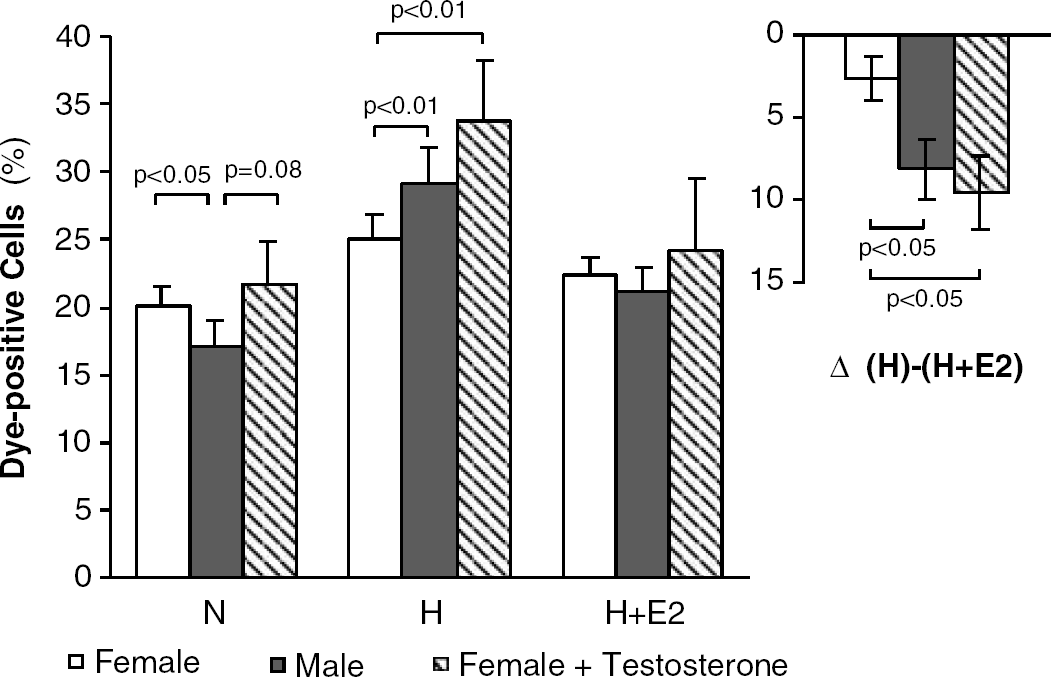
In vitro gender differences in neuronal survival upon hypoxia. Dead cells (absolute percentage of Trypan blue-positive cells) in primary hippocampal neuron cultures from female (open bars; n=23), male (closed bars; n=23), and testosterone-primed female rats (striped bars; n=4) under normoxia (N), hypoxia (H), or hypoxia plus 17β-estradiol (H+E2) (10 nmol/L). Inset denotes the protective effect of 17β-estradiol. Means±s.e.m.
In vivo testosterone priming of newborn female rats did not change neuronal survival under normoxia but led to increased cell death (‘male pattern’) on hypoxia (Figure 1). Culturing female neurons in the continuous presence of testosterone (10 nmol/L or 10 μM; ‘in vitro priming’) did not induce a male vulnerability pattern.
To examine whether the protective effect of 17β-estradiol involves intracellular ERs, 17β-estradiol bound to BSA (BSA-estradiol; 10 nmol/L) and the ER antagonist ICI182780 (1 μmol/L) were used. Bovine serum albumine-estradiol, unable to pass the cell membrane, showed an efficacy in reducing hypoxia-induced cell death comparable with 17β-estradiol (78%±6% versus 76%±3% of hypoxia; n=8). ICI182780 did not alter the effect of 17β-estradiol or of BSA-estradiol on neuronal survival (not shown).
Expression of mRNA for Estrogen Receptor alpha, Estrogen Receptor beta and Aromatase
Under normoxic conditions, ERalpha was expressed at a higher level than ERbeta in both female and male neuronal cultures (1878±616 and 1298±172 copies/106 EF-2 for ERalpha, but 452±135 and 353±25 copies/106 EF-2 for ERbeta, respectively, with no difference between genders: P=0.4 and P=0.5). Whereas no significant change was noted in ERalpha expression, a significant increase in ERbeta mRNA was observed on hypoxia and hypoxia plus 17β-estradiol in male cultures only, leading to a significantly increased ratio of ERbeta/ERalpha (Figure 2). Aromatase mRNA expression tended to be higher (overall P=0.08) in female than male neurons (3086±848 versus 1960±306 copies/106 EF-2 for normoxia; n=4) and did not change with experimental conditions.
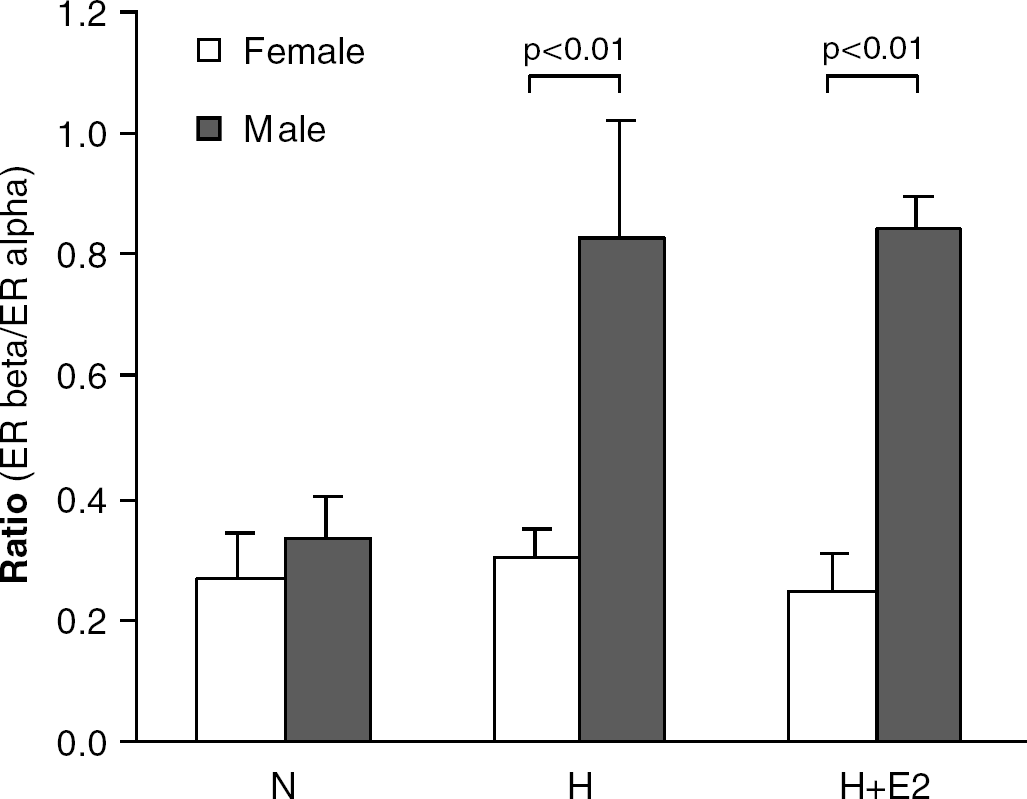
Ratio of ERbeta/ERalpha expression levels in neuronal cultures from female (open bars; n=4) and male (closed bars; n=4) rats under normoxia (N), hypoxia (H), hypoxia plus 17β-estradiol (H+E2). Means±s.e.m.
Discussion
Our data show intrinsic gender differences in the survival of cultured hippocampal neurons both under normoxia and hypoxia. More resistant under normoxia as compared with female cells, male neurons were more vulnerable to hypoxia. Similarly, a higher neurotoxic effect of dopamine was reported for male dopaminergic neurons in vitro (Lieb et al, 1995). The male pattern of neuronal vulnerability toward hypoxia in vitro could be achieved by systemic testosterone priming of female pups, proving that in the perinatal period, neurons are subject to masculinizing effects of circulating steroids. The molecular mechanisms underlying this phenomenon are still unknown. Interestingly, when the present paper was about to be submitted, an online publication found higher vulnerability of male neurons linked to their incapacity to maintain intracellular levels of reduced glutathione (Du et al, 2004).
The higher protective effect of 17β-estradiol in male cells observed here may, in part, be related to the increased ERbeta/ERalpha ratio found under hypoxia. In ERalpha as well as in ERbeta transfected fibroblasts, BSA-estradiol activated MAPK, indicating that both receptor subtypes are membrane-situated. This effect of BSA-estradiol was blocked by ICI182780 in ERalpha but not in ERbeta transfected cells (Wade et al, 2001). Similarly, ICI182780 was not able to block the neuroprotective effect of 17β-estradiol or of BSA-estradiol in our cultures. Assuming that the situation is comparable among cell types, membrane-located ERbeta would be the predominant candidate for mediating protection of rat hippocampal neurons during hypoxia in vitro.
Whereas the presence of membrane ERs in neurons has been convincingly shown previously (Xu et al, 2003), the ER subtype involved in neuroprotection, ERalpha, ERbeta, or both, appears to differ among species, brain regions, or disease models used. As an example, Dubal et al (2001) proposed the ERalpha subtype as a critical mechanistic link in mediating the protective effects of physiologic levels of estradiol in the injured mouse brain. In contrast, very recently, a selective ERbeta agonist has been identified as a powerful neuroprotectant in a mouse model of global ischemia (Carswell et al, 2004).
To conclude, the present study may help to explain, at a cellular level, gender differences in neuropsychiatric diseases.
Footnotes
Acknowledgements
The authors thank Sandra Hartung for skillful technical assistance.
