Abstract
The effects of sodium nitroprusside (SNP), a potent hypotensive agent, on cerebral blood flow (CBF) have been extensively studied in clinical and experimental situations but the results remain controversial. Whereas its properties would predict a dilatation of cerebral blood vessels, most studies report either no change or a decrease in CBF. The aim of this study was to investigate the effects of SNP on CBF, cerebral blood volume (CBV), and cerebral oxygen metabolism (CMRO2), by means of positron emission tomography in the anaesthetized baboon. Measurements were performed during normotension (mean arterial pressure (MABP): 97±16 mm Hg) and repeated following SNP-induced hypotension (MABP: 44±9 mm Hg). Sodium nitroprusside led to an increase in CBF and CBV (+30% and +37%, respectively, P<0.05), whereas no change in CMRO2 was noted. Linear regression analysis of CBF values as a function of MABP confirmed that CBF increases when MABP is reduced by SNP. The comparison between these cerebrovascular changes and those found during trimetaphan-induced hypotension in our previously published studies further argues for a direct dilatatory effect of SNP on cerebral blood vessels.
Keywords
Introduction
Sodium nitroprusside (SNP) is a powerful vasodilatatory agent, which produces a dose-dependent dilatation of arteries and veins by releasing nitric oxide (NO) into the blood stream (Feelisch and Kelm, 1991). Its action on systemic haemodynamics is essentially that of immediate hypotension. Studies in vitro (Auer, 1978; Harkin et al, 1997) and in vivo (cats, Auer (1978), baboons, Fitch et al, (1988), rodents, Zhang and Iadecola (1993)) have shown a direct dilatatory effect of SNP on cerebral blood vessels. Based on the above, the cerebrovascular effect of SNP is the consequence of two interdependent but opposing processes: systemic hypotension and cerebral vasodilatation. On the one hand, systemic hypotension should produce either no change (because of the phenomenon of autoregulation) or a slight decrease in cerebral blood flow (CBF). However, direct cerebral vasodilatation should induce an increase of CBF. The literature, about the overall cerebrovascular effects of SNP is rather controversial: studies report no change, an increase, or a decrease in CBF after an intravenous administration of SNP in humans (Griffiths et al, 1974; Larsen et al, 1982; Pinaud et al, 1989; Bünemann et al, 1991) and in laboratory animals (McDowall et al, 1974; Fitch et al, 1976, 1988; Akerman et al, 1976; Candia et al, 1978; Grubb and Raichle, 1982; Sivarajan et al, 1985; Hartman et al, 1989; Hamaguchi et al, 1992; Tsutsui et al, 1995; Salom et al, 2000; Joshi et al, 2002a) (for a review see Joshi et al, 2002b). In clinical practice, SNP is used for acute hypertension especially associated with encephalopathy, controlled hypotension during anaesthesia and heart failure. Recently, the vasodilatatory properties of SNP have been used to treat vasospasm after subarachnoid haemorrhage (Thomas et al, 1999; Vajkoczy et al, 2000; Raabe et al, 2002). Despite its clinical usefulness, SNP remains a drug with ambiguous cerebral haemodynamic effects.
The aim of this study was to provide a detailed and extensive assessment of cerebrovascular changes under SNP-induced hypotension in the non-human primate. The effects of SNP on CBF and cerebral blood volume (CBV) were measured, for the first time, through the use of positron emission tomography (PET). This technique allows coupled measurements not only of cerebral haemodynamics but also of cerebral oxygen consumption (CMRO2) and is well suited for such measurements in non-human primates (Touzani et al, 1995; Schumann et al, 1996, 1998). The present study was undertaken in the etomidate-anaesthetized baboon and a partial sympathetic blockade was used to limit the sympathetic reaction to SNP-induced hypotension. In addition, to elucidate whether the SNP-mediated changes in cerebral haemodynamics are related to either the cerebral or the systemic effects of the agent, we compared the cerebrovascular changes elicited by SNP-induced hypotension with those induced by trimetaphan (TRIM)-induced hypotension (published in a previous work of our own group, Schumann et al, 1998). Trimetaphan is a ganglioplegic drug devoid of major effects on the cerebral vasculature and can therefore be used as a reference to describe the cerebrovascular changes elicited by a ‘pure’ hypotension (Strandgaard et al, 1973; Schumann et al, 1998).
Methods
Anaesthesia and Physiological Monitoring
Studies were performed on 10 adolescent male baboons (Papio anubis) weighing 14 to 22 kg. The experiments conformed to the Guiding Principles in the Care and Use of Animals as approved by the University of Caen. The baboons were first sedated with methohexitone (20 mg/kg, intramuscularly, Brietal, Lilly, Suresnes, France). After placement of polyethylene catheters into the saphenous veins, the baboons were anaesthetized as follows: induction was achieved with a nonbarbiturate hypnotic (etomidate, 3 mg/kg, intravenously bolus, Hypnomidate, Janssen-Cilag, Issy-Les-Moulineaux, France) and a slow perfusion of clonidine (0.08 mg over 10 mins intravenously, Catapressan, Boehringer Ingelheim, Paris, France). The use of clonidine was based on its ability to potentiate the anaesthetic effects of low doses of etomidate (Bloor and Flacke, 1982; Jobert et al, 1995). After muscle relaxation was achieved with atracurium (0.5 mg/kg, intravenously bolus and thereafter 0.75 mg/kg h, intravenously, Tracrium, GlaxoSmithKline, Marly-le-Roi, France), the trachea was intubated and the animals were mechanically ventilated (Ohmeda Boyle 2000 ventilator). Anaesthesia was then maintained throughout the study by the inhalation of a humidified mixture of N2O:O2 (2:1) and a perfusion of etomidate (0.3 mg/kg h, intravenously) with sufentanil (0.2 μg/kg h, intravenously preceded by 0.1 μg/kg, intravenously bolus, Sufenta, Janssen-Cilag). The ventilation rate was fixed at 20 mins and the tidal volume was adjusted to maintain normocapnia (paCO2=39 to 41 mm Hg) (5200 CO2 monitor, Ohmeda). Percutaneous femoral arterial catheters were inserted for the continuous measurement of mean arterial pressure (MABP) and periodic control of paCO2, paO2, pH, haemoglobin (ABL 300 Radiometer), haematocrit, and plasma glucose levels (Beckman Instruments, Gagny, France). Heart rate (HR) and ECG were continuously monitored (Eagle monitor). Inspired gases were humidified at 38°C and rectal temperature was maintained within normal limits by a heating blanket system (Harvard, Les Ulis, France). Saline and Ringer's solutions were perfused at a rate of approximately 4 mL/kg h and urine output was monitored via urethral catheter (401±107 mL during the whole experiment).
Experimental Protocol
Sodium nitroprusside was infused intravenously after its extemporaneous dilution to 0.1% in a 5% glucose solution. Care was taken to protect the SNP solution from light during infusion. The rate of administration of SNP was adjusted to achieve the desired MABP response: a mean total dose of 1.8±0.9 mg/kg (equivalent to 16±3 μg/kg min) was required to reach an MABP of approximately 44 mm Hg during the hypotensive PET examination. No bleeding was required. This level of hypotension was chosen because it allowed us to cover a large range of arterial pressures and to compare these data to the TRIM study. In an attempt to limit inevitable reflex tachycardia, a short-acting and cardio-selective β-blocker, esmolol (Brevibloc, DuPont Pharma, Rueil-Malmaison, France), without reported effect on CBF (Bünemann et al, 1991), was infused (100 to 200 mg/h, intravenously) simultaneously with SNP (Khambatta et al, 1984). Recovery from anaesthesia occurred rapidly (approximately 15 mins) after discontinuation of the perfusion of etomidate and sufentanil.
In the same half-day session, each baboon underwent two successive PET examinations: for the first group (A) of 5 baboons, the initial PET scan (73±8 mins duration) was performed during normotension (MABP: 97±16 mm Hg). Ten minutes after completion of the PET scan, SNP infusion started to rapidly (14±8 mins) achieve the desired level of hypotension (MABP: 44±9 mm Hg). Mean arterial pressure was then held steady for at least 15 mins before the start of, and during the entire second PET study (70±8 mins). To compare the duration of cerebrovascular effects of SNP versus its systemic vascular effects, we proceeded in the reverse order for a second separate group (B) of 5 baboons: the first PET examination (81±15 mins) was performed once the hypotension was achieved (MABP: 44±6 mm Hg), while the second PET scan (80±16 mins) was performed 90±15 mins after cessation of the SNP infusion. However, in this group, despite the relatively long delay after SNP administration cessation, MABP failed to fully normalize: it returned rapidly to higher values (77±6 mm Hg) but remained inferior to the base-line level found in group A (97±16 mm Hg, P<0.05). By contrast with its short-acting bradycardic effects (Table 1), the reported long-lasting hypotensive effects of esmolol, because of a prolonged inhibition of the plasma renin activity (Ornstein et al, 1995), could contribute to the persistence of low MABP values despite cessation of SNP infusion. We elected therefore to exclude the ‘normotensive’ condition from Group B although maintaining the hypotensive state for the regression analyses.
Evolution of systemic parameters during sodium nitroprusside-induced hypotension in anaesthetized baboons
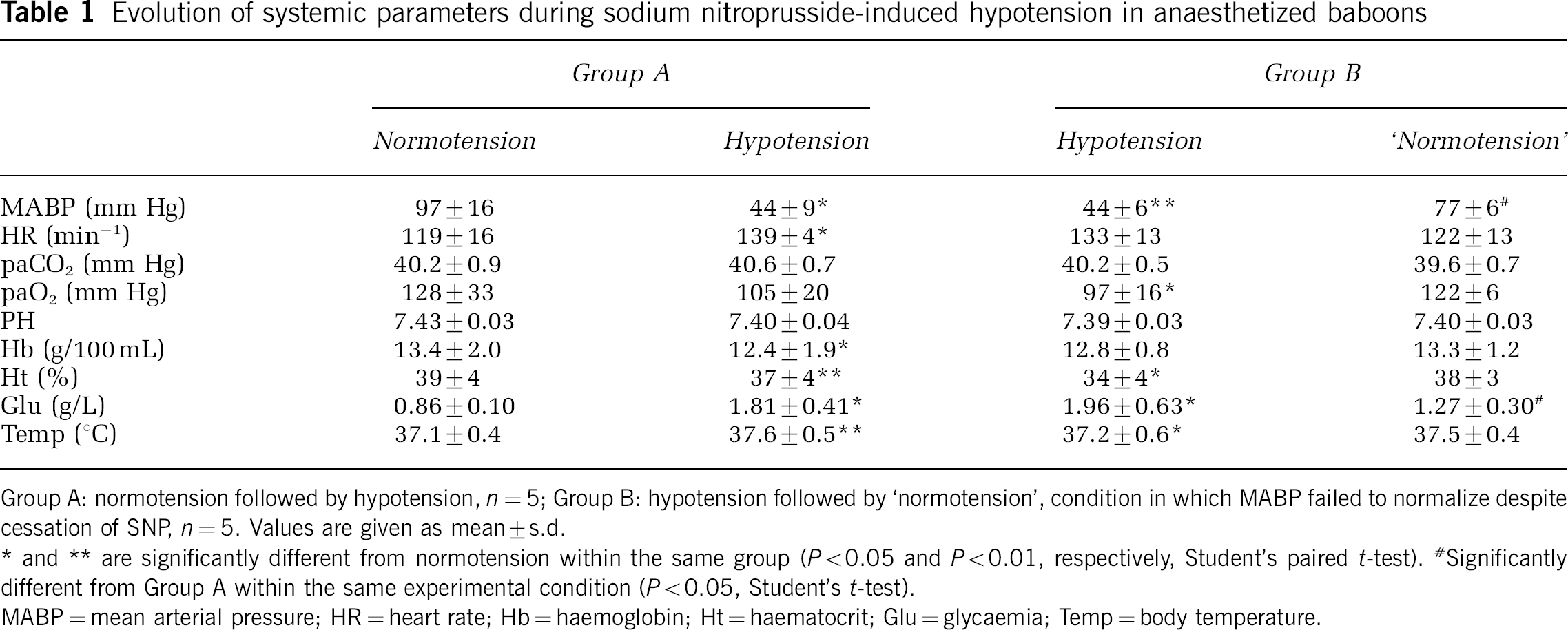
Group A: normotension followed by hypotension, n=5; Group B: hypotension followed by ‘normotension’, condition in which MABP failed to normalize despite cessation of SNP, n=5. Values are given as mean±s.d.
* and ** are significantly different from normotension within the same group (P<0.05 and P<0.01, respectively, Student's paired t-test).
Significantly different from Group A within the same experimental condition (P<0.05, Student's t-test).
MABP=mean arterial pressure; HR=heart rate; Hb=haemoglobin; Ht=haematocrit; Glu=glycaemia; Temp=body temperature.
Positron Emission Tomography
The 15O steady-state method (Frackowiak et al, 1980) with correction for intravascular tracer with C15O (Pantano et al, 1985) was used to assess local CBF, CBV, CMRO2, and the oxygen extraction fraction (OEF). The CBF/CBV ratio, a putative index of cerebral perfusion pressure (Sette et al, 1989; Schumann et al, 1998), was obtained by the pixel-by-pixel division of the CBF image by that of CBV. We used a seven-slice LETI TTV 03 PET camera with an intrinsic spatial resolution of 5.5 × 5.5 × 9 mm (x, y, z) (Mazoyer et al, 1990). To ensure reproducible interanimal head positioning, the baboon's head was fixed in a specially designed frame with ear bars placed in the bony portion of the external auditory canal and correct positioning was checked by a skull radiogram. Seven planes parallel to the canthomeatal (CM) line (−27 to +45 mm relative to this line) were selected according to an anatomical-PET atlas (Riche et al, 1988). A 68Ga-68Ge transmission scan was performed for attenuation correction before the PET session. During successive inhalations of tracer amounts of C15O, 15O2, C15O2, arterial 15O measurements were performed in 2 × 3 samples (Autogamma Cobra Packard).
Data Analyses
Regions of interest (ROIs) were determined in one plane (CM+21 mm) to avoid the large blood pools associated with venous sinuses, notably present in other planes (Riche et al, 1988). Eight elliptic ROIs (8 × 12 mm2) were disposed over the cortical mantle of one hemisphere, tangentially to each other, and thereafter mirror-copied on the opposite side in relation to interhemispheric fissure (Schumann et al, 1998). Global cortical values for the various PET parameters were obtained by averaging 16 ROIs. A Student's paired t-test was used to compare functional as well as physiological and biochemical values obtained during hypotension with those obtained during normotension. A linear regression analysis of CBF, CBV, and cerebrovascular resistance (CVR=MABP/CBF) versus MABP was performed on both SNP and TRIM data; normotensive data from both groups have been pooled. The level of significance was set at P<0.05. Results are presented as mean±s.d.
Comparative Study
As stated above, the changes in cerebral haemodynamics under SNP can be related either to a direct or to an indirect effect of the drug. In an attempt to analyse separately the direct cerebral vasodilatatory effect of SNP from the indirect effect because of systemic hypotension, we compared our current results with those obtained in one of our previous studies (Schumann et al, 1998) in which we assessed the cerebrovascular changes during ‘pure’ systemic hypotension. Moderate hypotension was induced by an intravenous injection of TRIM, a ganglioplegic agent devoid of major effects on cerebral vasculature, and profound hypotension was achieved by the combined effect of TRIM and lower-body negative pressure. To simplify matters, we will refer to these previous data in the present paper by the term ‘TRIM’. For this TRIM study, the non-human primate species, anaesthesic protocol (especially the use of etomidate, clonidine and sufentanil), PET scanner, and image processing were strictly identical to those used in the present investigation.
Results
Physiological and Biochemical Data
Table 1 summarizes the values of systemic variables obtained during the experimental conditions in both Groups A and B. In Group A, MABP decreased significantly from 97±16 to 44±9 mm Hg after SNP administration (−54%, P<0.05). Administration of SNP resulted in an increase in both HR and plasma glucose levels (+17% and +110%, respectively, P<0.05), which reflects the sympathetic response to hypotension. Esmolol did not completely prevent this reaction but limited the severe tachycardia that is commonly observed after SNP administration (Khambatta et al, 1984). In addition, the increase in plasma glucose levels might be, in part, enhanced by the administration of the 5% glucose solution, the vehicle of the SNP perfusion. The various biochemical and haematological parameters remained physiologically and globally stable despite minimal decreases in haemoglobin and haematocrit. The same observations were made in Group B, where MABP decreased to 44±6 mm Hg during SNP-induced hypotension. As already mentioned, MABP did not return to its normal values after cessation of SNP (return to ‘normotension’: 77±6 mm Hg, being different from the normotension achieved in Group A, P<0.05). Systemic parameters measured during hypotension in Groups A and B were, however, not different from each other.
Positron Emission Tomography Data
Normotension/hypotension: Group A
Both CBF and CBV demonstrated a significant and marked increase (+30% and +37%, respectively, P<0.05) during SNP-induced hypotension (Table 2, Figure 1). No significant change in the CBF/CBV ratio was noted. The CVR was significantly reduced (−64%, P<0.05) during the hypotensive period. Because the CMRO2 remained stable between both experimental conditions, the OEF presented a slight, although not statistically significant, decrease in its value (−17%) during the hypotensive period. There was no particular regional pattern of change in haemodynamic and metabolic parameters, suggesting that the watershed areas were equally affected by SNP-induced hypotension. Because CMRO2 was not compromised in any of the brain regions investigated, no neuropathological study was undertaken.
Representative parametric images from a single plane (parallel to and 21 mm above the canthomeatal line) obtained in one anaesthetized baboon (Group A), showing the increase in cerebral blood flow (CBF) and blood volume (CBV) and the lack of change in cerebral oxygen metabolism (CMRO2) during sodium nitroprusside-induced hypotension (OEF=oxygen extraction fraction). The pseudocolour representation is shown according to the scale depicted on the right.
Evolution of cerebral haemodynamic and metabolic parameters during sodium nitroprusside-induced hypotension in anaesthetized baboons
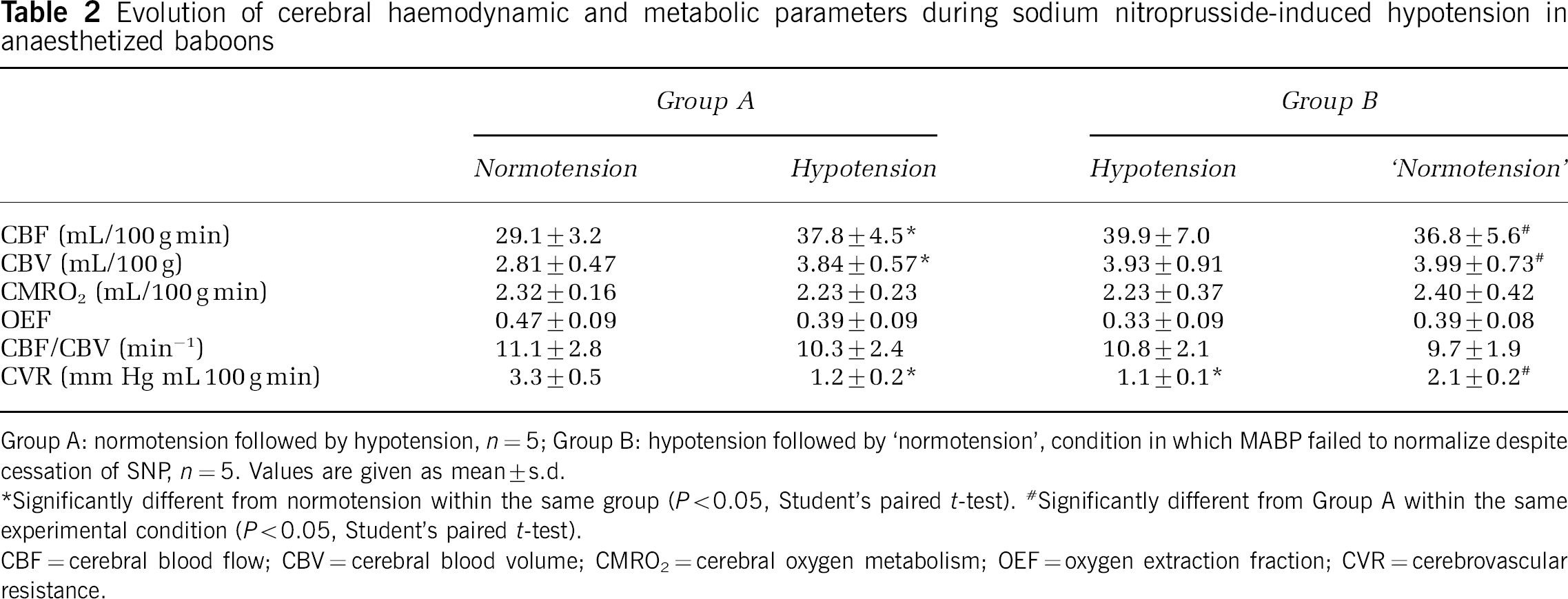
Group A: normotension followed by hypotension, n=5; Group B: hypotension followed by ‘normotension’, condition in which MABP failed to normalize despite cessation of SNP, n=5. Values are given as mean±s.d.
Significantly different from normotension within the same group (P<0.05, Student's paired t-test).
Significantly different from Group A within the same experimental condition (P<0.05, Student's paired t-test).
CBF=cerebral blood flow; CBV=cerebral blood volume; CMRO2=cerebral oxygen metabolism; OEF=oxygen extraction fraction; CVR=cerebrovascular resistance.
Hypotension/‘normotension’: Group B
None of the PET parameters measured during hypotension in Group B were different from those measured during hypotension in Group A (Table 2). During the ‘normotensive’ state, the CBF and CBV values remained elevated, suggesting a long-lasting effect of SNP on cerebral blood vessels. They were in addition higher than the normotensive values measured in Group A (Table 2). Because the normal relationships between CBF and MABP that are expected from a baseline state are clearly modified by the long-lasting effects of SNP on cerebral blood vessels and presumably by the prolonged systemic effects of esmolol on the plasma renin activity (Ornstein et al, 1995), the ‘normotensive’ data of Group B have not been taken into account for the regression analyses.
Regression analyses
We applied linear regression analyses on the scatter plots of CBF, CBV, and CVR against MABP during the administration of SNP (Figures 2–4). All regressions were statistically significant. We applied the same type of regression analysis to our TRIM data. Again, all regressions were also statistically significant. To compare the SNP and TRIM effects on cerebral haemodynamics, we superimposed the SNP and TRIM scatter plots (Figures 2–4). These graphs show dissimilar profiles for CBF and CVR between SNP and TRIM. Whereas CBF is roughly maintained or even starts to decrease for severe reduction in MABP for the TRIM data (Figure 2), this parameter clearly increases under SNP-induced hypotension. This particular finding would argue for a direct dilatatory action of SNP on cerebral blood vessels.
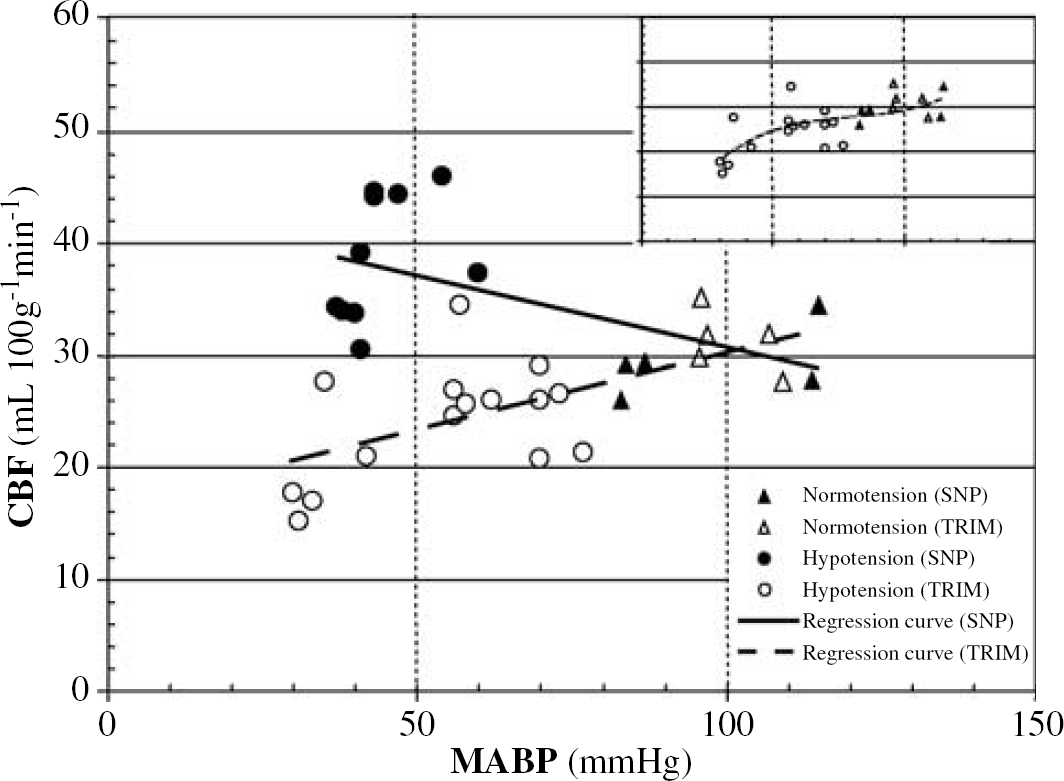
Linear regression curve of cerebral blood flow (CBF) versus mean arterial pressure (MABP) during sodium nitroprusside (SNP) (solid line)- and trimetaphan (TRIM) (dashed line)-induced hypotension. The equation and r2 determination coefficients are, respectively: y=−0.13x+43.62, r2=0.36 (P<0.05) for SNP, and y=0.14x+16.51, r2=0.47 (P<0.05) for TRIM. Inset: Nonlinear (third order polynomial) regression curve of CBF versus MABP during TRIM-induced hypotension: y=5.47 × 10−5x3−1.32 × 10−2x2+1.11x−4.85, r2=0.52 (P<0.01).
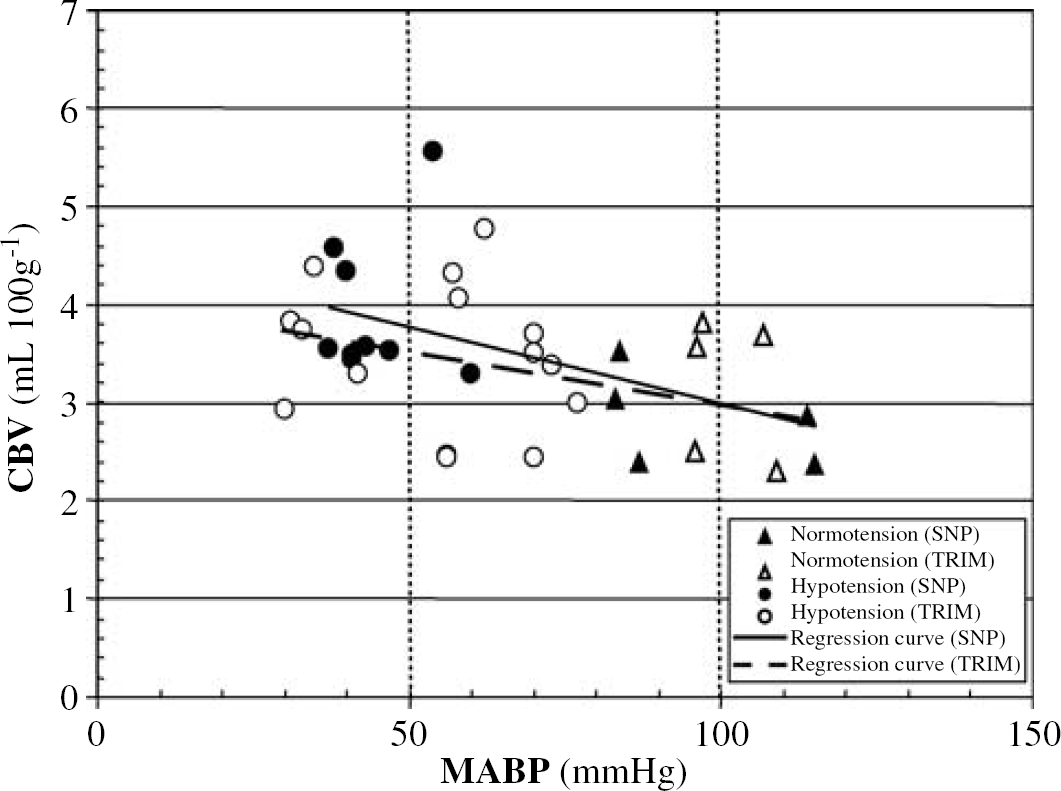
Linear regression curve of cerebral blood volume (CBV) versus mean arterial pressure (MABP) during sodium nitroprusside (SNP) (solid line)- and trimetaphan (TRIM) (dashed line)-induced hypotension. The equation and r2 determination coefficients are, respectively: y=−0.02x+4.55, r2=0.34 (P<0.05) for SNP, and y=−0.01x+4.05, r2=0.16 (P<0.05) for TRIM.
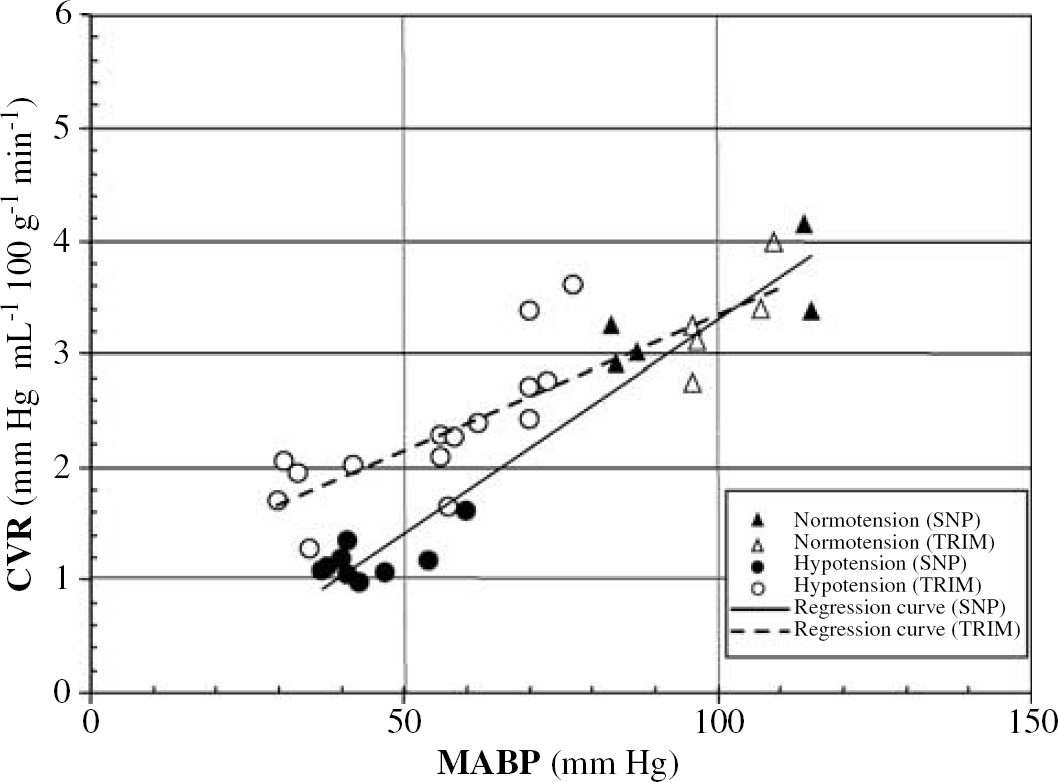
Linear regression curve of cerebrovascular resistance (CVR) versus mean arterial pressure (MABP) during sodium nitroprusside (SNP) (solid line)- and trimetaphan (TRIM) (dashed line)-induced hypotension. The equation and r2 determination coefficients are, respectively: y=0.04x−0.48, r2=0.94 (P<0.05) for SNP, and y=0.02x+0.92, r2=0.76 (P<0.05) for TRIM.
Discussion
The main finding of our present study is that SNP, associated with a partial sympathetic blockade, induces a marked increase in CBF and CBV in the anaesthetized baboon; these increases were seen despite an important reduction in MABP.
Pharmacological and Methodological Issues
Pharmacological issues are of primordial importance in the interpretation of any experimental study of cerebrovascular physiology. The responses of the cerebral vasculature to pharmacological agents, including SNP, are indeed dependent on the conditions of anaesthesia that determine the preservation or alteration of autoregulation and the basal tone of the blood vessels (Michenfelder and Milde, 1988). Two potential points arise in our study: the use of partial sympathetic blockade (clonidine and esmolol), and the choice of the anaesthetic agents (etomidate, sufentanil and nitrous oxide).
To limit the SNP hypotension-induced tachycardia, we elected to partially block the sympathetic pathways by the use of a β-blocker (esmolol). A major contribution of esmolol to the increase in CBF during SNP-induced hypotension seems, however, unlikely as this agent neither blocked the ketamine-induced increase of CBF velocity in humans (Strebel et al, 1995) nor altered CBF in the dog (Bunegin et al, 1987). Although its pharmacokinetic profile may be different from esmolol, especially on the rennin–angiotensin system, metoprolol, another selective β-blocker has been reported not to alter CBF (Bünemann et al, 1991). Clonidine, used to potentiate the anaesthetic effects of low doses of etomidate, also contributed to the partial sympathetic blockade. This drug inhibits the release of brain noradrenalin with a potential effect on perivascular sympathetic fibres that innervate pial cerebral arteries. Neither dilatation of pial arteries nor an increase in CBF has, however, been noted after systemic administration of clonidine at a five-fold higher dose (Kanawati et al, 1986).
Studies concerning the vasodilatatory effects of sufentanil (a potent opioid analgesic) are often contradictory, depending mainly on species. In healthy human volunteers, the intravenous administration of 0.5 μg/kg of sufentanil (five-fold the dose used in our study) had, however, no significant effect on CBF (Mayer et al, 1990). Because high doses of etomidate are known to decrease both CBF and CMRO2 (Renou et al, 1978), one may hypothesize that the relative state of vasoconstriction induced by etomidate could have amplified the vasodilatatory effects of SNP. The CBF response to vasoactive agents is indeed known to be influenced by basal CBF (McPherson et al, 1989), CMRO2 (Fujishima et al, 1971) and MABP (Harper and Glass, 1965). The cerebrovascular responsiveness to hypercapnia is, for example, decreased when the basal CBF is elevated by halogenous anaesthetics, probably because of the preexisting vasodilatation that limits the vasodilatatory reserve (McPherson et al, 1989). Conversely, when basal CMRO2 and/or CBF are low, the CBF response to vasodilatatory agents is expected to be higher. It is therefore likely that our regimen of anaesthesia, leading to relative low CBF and metabolism levels, might have amplified the vasodilatatory effects of SNP, in comparison with other studies that did not report any change in CBF by SNP under halogenous anaesthesia (Joshi et al, 2002a; Hamaguchi et al, 1992; Tsutsui et al, 1995). The potential state of vasoconstriction elicited by our regimen of anaesthesia is, however, relative and can therefore not completely explain the increase in CBF observed during SNP administration. The time delay between induction of the anaesthesia with etomidate and the subsequent PET examinations was indeed long (approximately 2 and 4 h, respectively, for the first and second PET scans) with regard to the short half-life of etomidate, which is approximately 20 mins (Levron and Assoune, 1990). The concentration of etomidate used in our study was also reduced through the use of clonidine (Jobert et al, 1995). Furthermore, the use of nitrous oxide, known to increase CBF and cerebral metabolism in animals (Baughman et al, 1990) and humans (Deutsch and Samra, 1990), might have limited the vasoconstriction induced by etomidate. Finally, the same anaesthetic regimen was used for the TRIM group (Schumann et al, 1998), in which no increase in CBF was observed during hypotension (Figure 2).
The present study is the first to examine the effects of SNP on both cerebrovascular and metabolic parameters as measured by PET. This is a significant methodological advance since the PET technique offers the unique possibility to assess reliably and quantitatively both CBF and CMRO2. We infer from our data that the SNP-induced increase in CBF cannot be ascribed to an increase in cerebral metabolism because the cerebral oxygen consumption remained unchanged (Table 2, Group A).
Sodium nitroprusside-induced hypotension leads to a significant decrease in haematocrit, −5% in Group A and −11% in Group B, probably because of the persistent sympathetic reaction under SNP-induced hypotension. Indeed, an increased sympathetic activity is known to constrict the afferent renal arterioles, activate plasma renin activity and could therefore lead to a decrease in urine output. Such an effect of SNP-induced hypotension has already been reported in humans (Nielsen et al, 1993) and could contribute to a certain haemodilution. We did not measure urine output regularly, and therefore cannot conclude on the effect of hypotension and its attendant sympathetic activation on this parameter. We instead evaluated the total urine output at the end of the experiment. This volume is usually slightly lower than the total fluid administered during the entire experiment, suggesting the existence of a limited haemodilution. Because haematocrit is inversely correlated to CBF (Thomas et al, 1977), one might suppose that the reduction in haematocrit could contribute to the increase in CBF found during SNP-induced hypotension. However, analysis of the relationships between CBF and haematocrit reported in the literature would predict that such a decrease in the haematocrit would induce only 6% increase in CBF in Group A (Thomas et al, 1977; Hino et al, 1992) and 12% in Group B (von Kummer et al, 1988). In conclusion, we cannot exclude a possible contribution of the haematocrit changes to the cerebrovascular modifications observed during SNP-induced hypotension but, according to the literature, this contribution should be minimal, in regard with the important (30%) increase in CBF measured during SNP administration.
Analysis of the Results and Physiological Hypotheses
Effects of sodium nitroprusside on cerebral blood flow
Our PET data show that SNP induces a significant increase in CBF, as predicted by the linear regression analysis. They suggest that SNP produces a prominent cerebral vasodilatation that surpasses the cerebrovascular effects of systemic hypotension, resulting in an increase in CBF. The comparison between the data obtained under SNP and TRIM (Figure 2) also underscores the possibility of a powerful direct dilatatory effect of SNP on cerebral circulation. Indeed, TRIM produces a systemic hypotension with an essentially preserved CBF by means of adaptive cerebrovascular changes (sigmoid curve, inset Figure 2). Around an arterial pressure of 50 mm Hg, autoregulation is exhausted, and further reductions of MABP produce a decrease in CBF.
In Group B, we measured CBF at first during SNP-induced hypotension. As already discussed, MABP values obtained at the return to ‘baseline’ state were lower than the normotensive level of Group A. Cerebral blood flow measured during hypotension in Group B was not different from CBF measured during ‘normotension’, showing that CBF remained elevated despite a return of MABP to higher levels. A similar discrepancy in a prolonged increased cerebral perfusion despite a return of MABP to higher levels after cessation of the infusion of SNP has already been described in the rat (Gustafson, 1984) and the pig (Stånge et al, 1991). Results from Group B further suggest that these long-lasting changes in CBF seem to be mainly related to the direct cerebrovascular effects of SNP, regardless of MABP.
Influence of sodium nitroprusside on the status of autoregulation
Preservation, or impairment, of autoregulation during SNP-induced hypotension may constitute a confounding factor for the interpretation of the effects of the drug on CBF. The effects of SNP administration on CBF are the net results of both autoregulatory vasodilatation and SNP-mediated vasodilatation. Autoregulation has been reported to be reasonably preserved in some studies (McDowall et al, 1974; Grubb and Raichle, 1982) though abolished in another study (Stånge et al, 1991). We did not assess the autoregulatory capacity of the baboons under SNP administration in the present study and cannot therefore draw any specific conclusion. Two possibilities can be evoked: if autoregulation was preserved under SNP, CBF should then remain essentially unchanged during hypotension. In the alternative case of an altered autoregulatory capacity induced by SNP itself, one would expect a decrease, and not an increase, in CBF. Therefore, whatever the autoregulatory state, one might assume that the increase in CBF observed in our study is intimately related to a direct effect of SNP on cerebral blood vessels. This conclusion is in accordance with the results of Ivankovich et al (1976), who showed that SNP, injected intraarterially, induces an increase in CBF although MABP does not change.
Effects of sodium nitroprusside on cerebral blood volume
Sodium nitroprusside produces a significant increase in CBV. The graph of CBV versus MABP (Figure 3) indicates that changes in CBV are similar after either SNP or TRIM administration. The comparison of the SNP and TRIM data for CBV (Figure 3) reveals a large dispersion of the data, rendering any interpretation delicate. Although the increase in CBV was significant after either SNP or TRIM, SNP would appear to increase CBV more than TRIM. This observation suggests that for the same range of decreased MABP, SNP yields a greater cerebral vasodilatation than that strictly related to autoregulation (i.e., the intrinsic vasodilatatory mechanisms). In Group B, CBV measured during hypotension was not statistically different from that measured during ‘normotension’, underlying the prominent non-pressure-dependent and long-lasting cerebral vasodilatatory property of SNP. There are some difficulties in defining the cerebrovascular compartments involved in CBV measurement with PET. It has been estimated that cerebro-cortical CBV comprises ∼30% in the arterial and ∼70% in the venous compartment (Lammertsma and Jones 1983). Accordingly, the increase in CBV observed in our study could be attributed to arteriolar and/or venous dilatation, even if the major venous sinuses were carefully avoided in our choice of ROIs. In vivo, SNP has been reported to produce both arteriolar and venous vasodilatation, but with a preponderant effect on the arteriolar vessels (Auer, 1978; Endrich et al, 1987).
Effects of sodium nitroprusside on cerebrovascular resistance
Sodium nitroprusside produces a significant decrease in CVR. The graph of CVR versus MABP (Figure 4) indicates a near linear decrease of CVR under SNP, which attains a plateau at extreme values of hypotension. In the TRIM group, this collapse occurs at lower absolute levels of MABP than those seen with SNP. The comparative graph (Figure 4) illustrates the sharper decrease of CVR during SNP compared with TRIM, an observation that underlines the powerful vasodilatatory properties of SNP.
Analysis of the Literature Concerning Sodium Nitroprusside
The literature concerning the cerebrovascular effects of SNP is rather controversial. Most studies failed to show any change in CBF (Fitch et al, 1976; Akerman et al, 1976; Sivarajan et al, 1985; Joshi et al, 2002a) while some have shown an increase in CBF after SNP administration (Candia et al, 1978; Fitch et al, 1988; Hartmann et al, 1989). More generally, when CBF is clearly reduced, either the level of anaesthesia is light and the subject is hypertensive at the basal state (Grubb and Raichle, 1982; Griffiths et al, 1974) or the level of SNP-induced hypotension is remarkably severe (McDowall et al, 1974). In the former study, MABP falls with SNP from 132 to 62 mm Hg, yielding a 10% reduction of CBF (Grubb and Raichle, 1982), which could be the consequence of an artificially increased CBF at rest. In the latter case, MABP decreases from 92 to 34 mm Hg and CBF is decreased by 20% (McDowall et al, 1974); the intense decrease of the average MABP (10 mm Hg lower than that achieved in our present investigation) is probably responsible for the significant decrease in CBF. When CBF is increased (Candia et al, 1978; Fitch et al, 1988; Hartmann et al, 1989), most of the studies display a biphasic pattern: an initial, moderate but significant increase in CBF, and then for higher doses of SNP (i.e., severe hypotension), a decrease in CBF. This biphasic response of CBF after SNP administration, could explain why CBF could increase or decrease as a function of the part of the curve that is under investigation. The doses of SNP used in our study (16 μg/kg min, intravenously) are in a high range in comparison with several studies (Bünemann et al, 1991; Griffiths et al, 1974) that showed no change in CBF under SNP administration (5 and 3 μg/kg min intravenously, respectively). The use of a relatively high dose of SNP in our experiments, based on its ability to induce an important decrease in MABP (−54%), could partly explain the differences with these studies.
Since the recommended clinical dose of SNP is somewhat lower than the one we used in this work, its dilatatory effect on the cerebrovascular bed should be of less importance in clinical practice. It should, nonetheless, be taken into consideration. On the one hand, this direct cerebrovascular effect of SNP emphasizes the major limitation of systemic SNP use, that is, its deleterious effect on intracranial pressure (see also Thiagarajah et al, 1987), rendering its utilization difficult in patients with a dysfunctional cerebrovascular compliance. However, SNP has been used over the last decade, in an attempt to treat local vasospasm, a narrowing of the lumen of major cerebral arteries after subarachnoid haemorrhage. Experimental and clinical studies suggest that intracisternal administration of SNP increases CBF (Vajkoczy et al, 2000; Raabe et al, 2002) but this finding is not always the case (MacDonald et al, 2002). Our results, showing an increase in CBF by SNP, speak for the existence of such a dilatatory effect of SNP on cerebral resistance blood vessels and therefore for the intrathecal administration of SNP to treat cerebral vasospasm.
Conclusions
The present study demonstrates that the systemic administration of hypotensive doses of SNP leads to an increase in both CBF and CBV in the anaesthetized baboon. These haemodynamic changes are not related to changes in cerebral metabolism. Under SNP administration, modifications of cerebral haemodynamics are mainly related to the direct cerebrovascular effect of the drug, rather than to systemic changes.
Footnotes
Acknowledgements
The authors thank Dr Aram TerMinassian (Departement d'Anesthésie Réanimation, CHU, Angers, France) for his helpful comments on the manuscript. The authors acknowledge the technical support of Laurent Chazalviel, Claudine Madeleine in the animal care and preparation of the investigations and the cyclotron and radiochemistry staff of Cyceron.
