Abstract
During the past decades, candidate drugs that have shown neuroprotective efficacy in the preclinical setting have failed in clinical stroke trials. As a result, no treatment for stroke based on neuroprotection is available today. The activation of the glucagon-like peptide 1 receptor (GLP-1) for reducing stroke damage is a relatively novel concept that has shown neuroprotective effects in animal models. In addition, clinical studies are currently ongoing. Herein, we review this emerging research field and discuss the next milestones to be achieved to develop a novel antistroke therapy.
INTRODUCTION
Stroke is the primary cause of severe disability and the second most common cause of death according to the World Health Organization. In all, 85% of all strokes result from any major cerebral artery occlusion leading to cerebral ischemia, brain damage, and consequent neurologic impairments and disability.
Recombinant tissue plasminogen activator is the only FDA-approved pharmacological treatment for ischemic stroke. However, this treatment is not available for the majority of stroke patients due to short effective therapeutic window (up to 4.5 hours from stroke symptom onset) and increased risk of cerebral hemorrhage.
The ischemic cerebral region can be divided in two subregions: ischemic core and
Recent research has showed neuroprotective properties against stroke by drugs targeting the glucagon-like peptide-1 receptor (GLP-1R). Some of the stroke efficacy data has been achieved under preclinical conditions of clinical relevance. Furthermore, these substances are already in clinical use for the treatment of type 2 diabetes (T2D) and present a good safety profile and minimal side effects. Therefore, the potential repositioning of GLP-1R activating drugs into antistroke treatments seems promising.
THE GLUCAGON-LIKE PEPTIDE-1 RECEPTOR
Glucagon-like peptide-1 receptor is a G-protein-coupled receptor that is expressed in a wide range of tissues including pancreas, heart, and brain. 2 It is activated by GLP-1; a small peptide hormone released from intestinal L cells and exerting numerous pleiotropic effects. The best-characterized property of GLP-1 is its incretin effect, e.g., enhancing meal-stimulated insulin secretion from pancreatic β cells in a glucose-dependent manner. 2 This effect accounts for the largest part of the postprandial insulin secretion in healthy subjects. Glucagon-like peptide-1 also decreases glucagon secretion from the pancreas. Since these effects are glucose dependent, activation of the GLP-1R carries a low risk of hypoglycemia. The signal transduction pathway of GLP-1 and its analogs has been characterized in pancreatic β cell and it mainly occurs via adenylate cyclase and the cAMP/PKA pathways. 2
Endogenous GLP-1 is rapidly degraded by the enzyme dipeptidyl-peptidase 4 (DPP-4). Thus, despite its glycemic regulatory properties GLP-1 as such could not be developed clinically for the treatment of diabetes. However, there are todays several stable synthetic GLP-1R agonists resistant to DPP-4 degradation and DPP-4 inhibitors that are used clinically to treat T2D.
GLUCAGON-LIKE PEPTIDE-1 RECEPTOR ACTIVATION FOR THE TREATMENT OF STROKE
In the past few years, neuroprotection
Glucagon-Like Peptide-1 Receptor Agonists
Besides its metabolic properties, GLP-1Rs are expressed throughout the brain and Li
Exendin-4 can pass the blood brain barrier and data have recently shown that peripheral administration of this compound leads to neuroprotection against stroke. Teramoto
Type 2 diabetes is a major risk factor for stroke. Furthermore, T2D and stroke are major causes of morbidity and mortality.
9
To simulate the clinical situation of a diabetic patient experiencing a stroke, we showed that 4 weeks intraperitoneal pretreatment with Ex-4 before MCAO followed by another 4 weeks of Ex-4 treatment decreases brain damage in T2D rats.
8
The effect was already significant at the clinically T2D dose (0.1 μg/kg) and occurred independently of the regulation of glycemia. In clinical perspective, the finding has the advantage that T2D patients could receive a therapy based on GLP-1R activation primarily against their diabetes (i.e., antihyperglycemic), while at the same time improving the stroke prognosis. Briyal
Interestingly, Sato
See Table 1A for a summary of the studies above.
Bibliometric overviews of preclinical studies and ongoing trials with GLP-1R agonists and DPP-4 inhibitors in stroke (A. Preclinical studies)
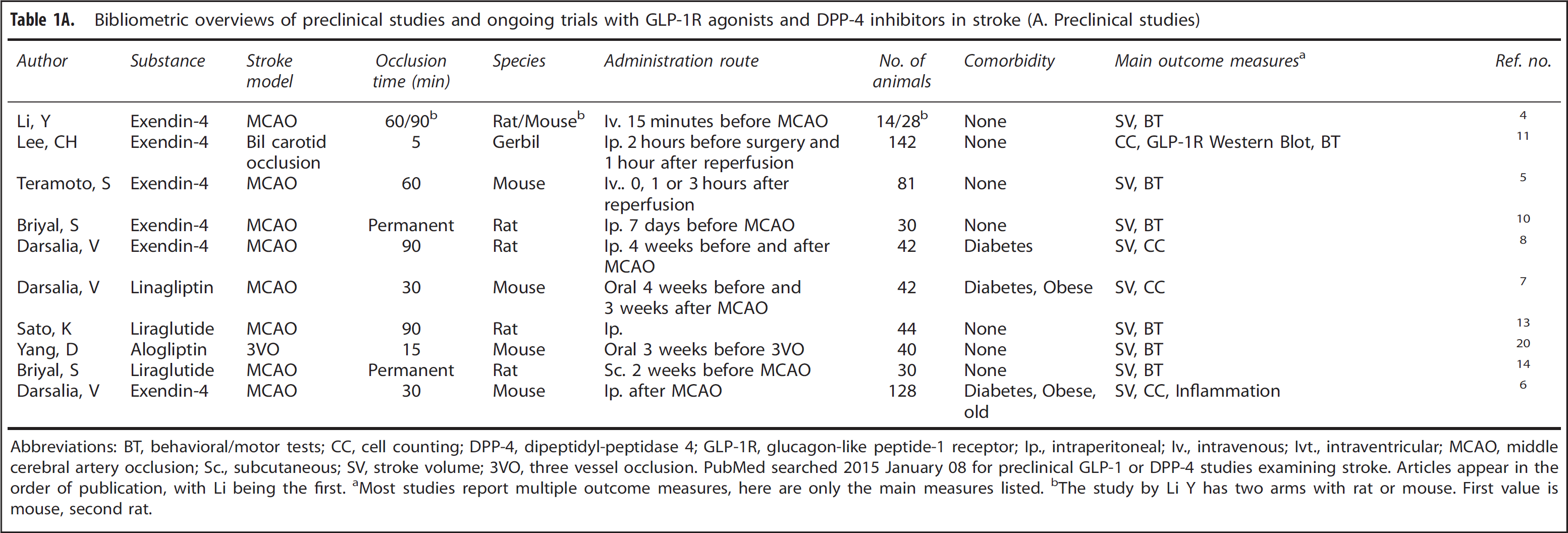
Abbreviations: BT, behavioral/motor tests; CC, cell counting; DPP-4, dipeptidyl-peptidase 4; GLP-1R, glucagon-like peptide-1 receptor; Ip., intraperitoneal; Iv., intravenous; Ivt., intraventricular; MCAO, middle cerebral artery occlusion; Sc., subcutaneous; SV, stroke volume; 3VO, three vessel occlusion.
PubMed searched 2015 January 08 for preclinical GLP-1 or DPP-4 studies examining stroke. Articles appear in the order of publication, with Li being the first.
Most studies report multiple outcome measures, here are only the main measures listed.
The study by Li Y has two arms with rat or mouse. First value is mouse, second rat.
Bibliometric overviews of preclinical studies and ongoing trials with GLP-1R agonists and DPP-4 inhibitors in stroke (B. Ongoing clinical trials)

Abbreviations: CV, cardiovascular; DPP-4, dipeptidyl-peptidase 4; GLP-1R, glucagon-like peptide-1 receptor; MRI, magnetic resonance imaging.
EU Clinical Trial Register (EU CTR) and ClinicalTrials.gov (CT.gov) databases searched with query ‘stroke'+‘substance’ on 2014 November 8. Each row constitutes a unique study. Retrospective noninterventional studies excluded.
Dipeptidyl-Peptidase 4 Inhibitors
Dipeptidyl-peptidase 4 is a serine aminopeptidase enzyme inactivating the incretins GLP-1 and glucose-dependent insulinotropic polypeptide through a dipeptide cleavage of the penultimate
Recent data have shown that intracerebral administration of the DPP-4 inhibitor sitagliptin reduced cortical lesions after MCAO in the rat.
19
Although the work has low clinical relevance due to the employed route of administration, it was the first to show neuroprotection by DPP-4 inhibition. We recently showed that 4 weeks pretreatment with clinical doses of the DPP-4 inhibitor linagliptin (orally) in both normal and T2D/obese mice reduced neuronal loss after MCAO.
7
As expected, the effects of linagliptin correlated with increased plasma GLP-1. However, the neuroprotective effect appeared to be independent of glycemic control. In agreement with this finding, Yang
Although these results are interesting, the responsible mechanisms remain unclear. Unlike GLP-1R agonists, DPP-4 inhibitors do not cross the blood brain barrier. Furthermore, DPP-4 inhibition does not induce an increase in plasma GLP-1 equivalent to pharmacological doses of Ex-4 or liraglutide. Finally, opposite to what has been observed for GLP-1R agonists, antistroke efficacy by alogliptin was lost when the treatment started after stroke onset. 20 We also achieved similar results to those with alogliptin by using linagliptin (unpublished). In conclusion, these data indicate that the biology at the basis of neuroprotection mediated by DPP-4 inhibitors is complex and still largely unexplored.
See Table 1A for a summary of the studies above.
Of note is that no DPP-4 inhibitor studies are primarily stroke trials.
POTENTIAL MECHANISMS AND FUTURE DIRECTIONS
During the past few years, experimental evidence has shown that GLP-1R activation is efficacious against stroke in various rodent models. However, the molecular/cellular mechanisms at the basis of GLP-1R-mediated neuroprotection are still largely unknown. Altogether the results point out direct, antiapoptotic and neuroprotective mechanisms and indirect antiinflammatory mechanisms involving microglia regulation (Figure 1). These data are also supported by the fact that GLP-1R has been found on both neurons
27
and microglia.
28
Since neuroprotective efficacy by GLP-1R activation can be achieved both after a chronic pretreatment before stroke and acutely after stroke, it will be important to determine whether the protective mechanisms are the same. Interesting, recent literature by Gejl
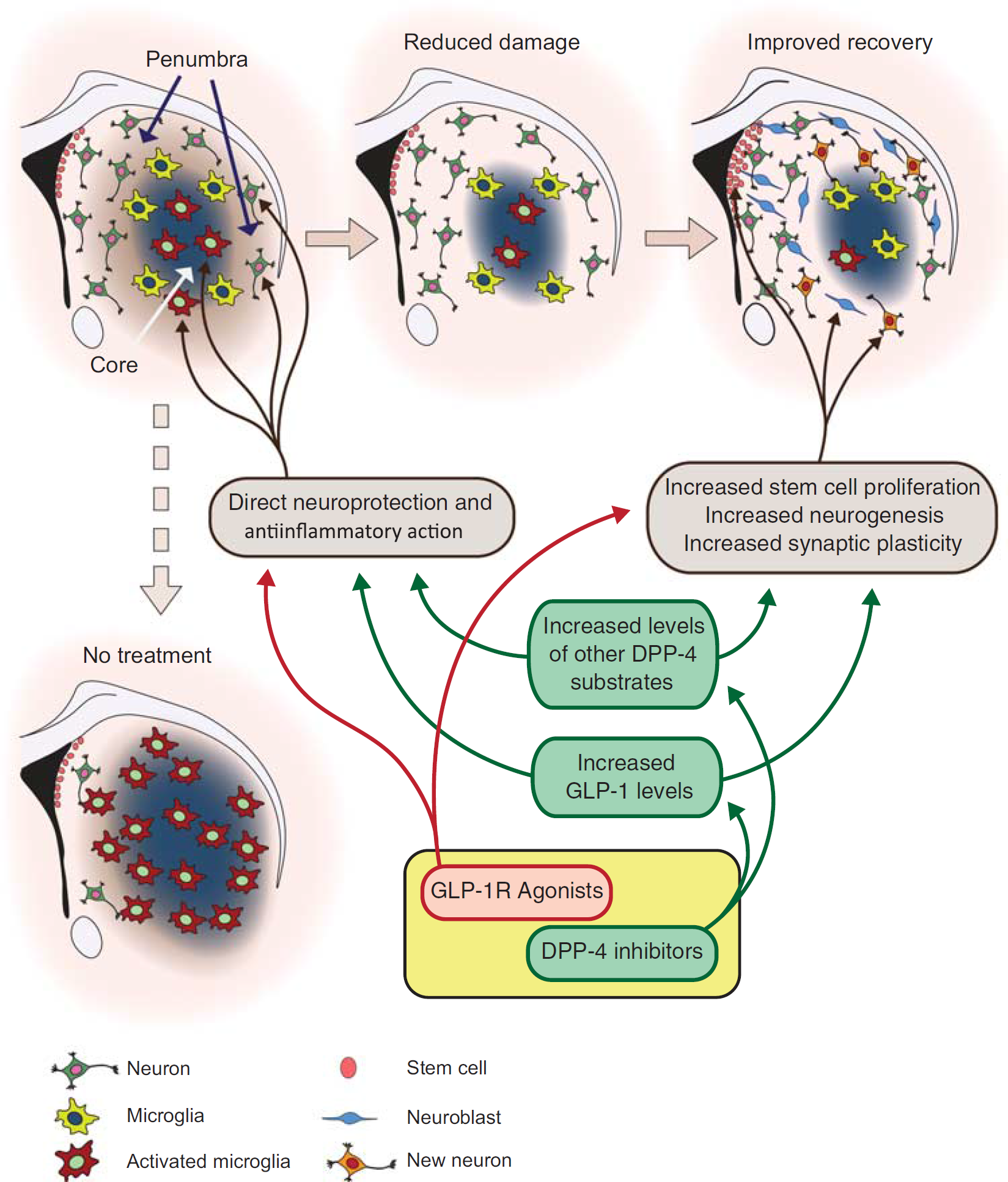
Neuroprotective mechanisms against stroke induced by glucagon-like peptide-1 receptor (GLP-1R) agonists and dipeptidylpeptidase 4 (DPP-4) inhibitors. GLP-1R agonists and DPP-4 inhibitors could exert neuroprotection via common or independent mechanisms. Evidence is accumulating that direct antiapoptotic survival effects on neurons and indirect effects through the regulation of microglia could be at the basis of acute neuroprotection by GLP-1R agonists. In addition to stimulate neuroprotection by increased plasma GLP-1 levels, DPP-4 inhibitors could also modulate the activity of other neuroprotective factors that will need to be identified. Other potential mechanisms ranging from effects on adult neurogenesis and on synaptic plasticity will need to be investigated to understand whether the effect of both GLP-1R agonists and DPP-4 inhibitors can have a role in late stroke recovery.
The GLP-1R activation has been reported to increase neurologic recovery and to improve learning and memory in different animal models of neurodegenerative disorders. 3 In addition, GLP-1R activation promotes synaptic plasticity and neurite outgrow 3 and can stimulate adult neurogenesis (Figure 1). 30 The findings could form the bases for potential regenerative therapy for chronic stroke patients. At this regard, experimental data and future work are highly needed.
CONCLUSIONS
Preclinical efficacy studies and clinical results indicate that an antistroke therapy based on GLP-1R activation might be translated into the clinical practice. A number of these molecules are already in clinical use for the treatment of T2D, making this potential translation easier since these molecules are rather safe. If translation is possible, then the preclinical data indicate that diabetic/high-risk stroke patients receiving GLP-1R therapy before stroke would benefit mostly from this type of treatment. Whether a poststroke acute treatment can lead to neuroprotection in a clinical relevant manner remains to be investigated since the protective effect of GLP-1R agonists in rodents decreases quickly after stroke onset. If so, it would require that the drug administration occurs soon after a stroke event; possibly already during the ambulance transport. Finally, safety/feasibility studies assessing whether this type of therapeutic strategy is possible will need to be completed and analyzed in the near future.
DISCLOSURE/CONFLICT OF INTEREST
Work in CP's laboratory is supported by Boehringer Ingelheim Pharma GmbH & Co. TK is an employee of Boehringer Ingelheim Pharma GmbH & Co. TN is on the national advisory board of Eli Lilly, Novonordisk, and Sanofi.
