Abstract
We present a noninvasive method of photoacoustic tomography (PAT) for imaging cerebral hemodynamics in awake-moving rats. The wearable PAT (wPAT) system has a size of 15 mm in height and 33 mm in diameter, and a weight of ~ 8 g (excluding cabling). The wPAT achieved an imaging rate of 3.33 frames/s with a lateral resolution of 243 μm. Animal experiments were designed to show wPAT feasibility for imaging cerebral hemodynamics on awake-moving animals. Results showed that the cerebral oxy-hemoglobin and deoxy-hemoglobin changed significantly in response to hyperoxia; and, after the injection of pentylenetetrazol (PTZ), cerebral blood volume changed faster over time and larger in amplitude for rats in awake-moving state compared with rats under anesthesia. By providing a light-weight, high-resolution technology for in vivo monitoring of cerebral hemodynamics in awake-behaving animals, it will be possible to develop a comprehensive understanding on how activity alters hemodynamics in normal and diseased states.
Keywords
INTRODUCTION
In vivo brain imaging in awake-moving animals has been a longstanding goal for neuroscience study. 1 Fluorescence optical imaging, such as two-photon microscopy, is being developed to be wearable for behaving animals and has provided high spatiotemporal resolution images. 2 Moreover, standard in vivo physiology is capable of recording neuronal action potentials as well as electroencephalography (EEG) in freely moving rats.3–5 These methods, however, are restricted to localized areas of interest and have the drawbacks of compromising the blood–brain barrier and potentially damaging the neurons and vasculatures due to the invasive procedure, thereby affecting chronic studies. However, current noninvasive brain imaging techniques, such as functional magnetic resonance imaging (fMRI), 6 positron emission tomography, 7 and diffuse optical tomography, 8 can provide whole brain functional imaging in awake-behaving animals but their spatial and/or temporal resolution is relatively low (millimeters for fMRI, positron emission tomography, and diffuse optical tomography; seconds for fMRI and positron emission tomography), limiting their use in real-time fine scale (sub millimeter resolution) brain imaging applications.
Photoacoustic (PA) tomography (PAT) is an emerging imaging modality that can provide in vivo images at sub-millimeter spatial resolution with centimeter tissue penetration.9–11 In PAT, after absorbing the incident light energy, chromophores within the tissue, such as myoglobin, hemoglobin, and melanin, will undergo a heat increase. The heat then induces an initial pressure rise, which propagates as a wideband acoustic wave in the tissue. 12
Photoacoustic tomography produces images by tomographically acquiring the acoustic signals around the object by a single ultrasonic transducer (single transducer-based PAT) or an array of transducers (transducer array-based PAT). 12 Transducer array-based PAT is capable of noninvasively imaging the animal's whole brain with a high temporal resolution (several frames per second), and possesses superior spatial resolution (~ 100 μm) as well.9,13–15 While these advantages make array-based PAT suitable for cerebral hemodynamics imaging, current array-based PAT systems are bulky, requiring subjects to be anesthetized and fixed during experiments, which might disrupt the neurochemistry and animal's perception to stimulation.9,14,16 For example, anesthetics can modify the dopamine D1 receptor binding in rat brain 17 and anesthesia also affects the neuronal excitability, vascular reactivity, and other baseline physiologies. 18 Therefore, high-speed, noninvasive PAT imaging of the whole brain in awake-behaving animals is highly desirable.
The present manuscript describes, for the first time, a miniature wearable PAT (wPAT) system that can be carried by a moving rat for noninvasive, high-speed brain imaging. The wPAT comprises a 64-element ultrasonic transducer array arranged into a miniaturized cap-shaped housing that can be mechanically mounted on the rat head. Moreover, the developed passive weight support system allows the mobility of rat by reducing the forces exerting on the rat by wPAT and the cablings, and the strategy of ultrasound detection and homogenous light illumination makes PAT achievable in a behaving rat. Despite the miniaturized size, wPAT is capable of high spatial resolution (243 μm) for fast (3.33 frames/s) brain hemodynamics imaging in awake-behaving rats. A series of experiments were conducted to show its capability. The in vivo functional imaging ability was first tested by detecting the rat cerebral deoxy-hemoglobin (HbR), oxy-hemoglobin (HbO), and total hemoglobin (HbT) changes in response to hyperoxia using three light wavelengths (710, 1,064, and 797 nm, respectively). Cerebral hemodynamic response (detected with wPAT) decreased at 710 nm but increased at 1,064 nm, while no significant deviation was observed at 797 nm when the rat was in hyperoxia. We then applied wPAT to monitor the cerebral blood volume change induced with generalized seizure after injecting pentylenetetrazol (PTZ) in awake-moving and anesthetized rats, respectively. Cerebral blood volume changed significantly faster and higher amplitude for awake-behaving rats relative to anesthetized animals. In addition, there was a significant difference in regional hemodynamics between awake-behaving and anesthetized preparations. The wPAT technique demonstrated here can be applied to a wide range of applications such as neurovascular coupling, behavior/recognition research, and metabolic related studies in awake-behaving rats, thereby providing a powerful tool for the community.
MATERIALS AND METHODS
Animal Preparation
Male Sprague Dawley rats (Harlan Labs, Indianapolis, IN, USA, 60 to 80 g) were used for the current study. After the hair was removed (but the scalp and the skull were still intact), the bottom holder of wPAT (Figure 1A) was mechanically mounted on the rats' head with two pairs of screws that were bilaterally pressed against the bone near the ear area. Mounting tapes (Scotch, 3M Inc., Maplewood, MN, USA) were placed between the screws and rat head to protect the skin. Transparent membrane (Stretch-tite Inc., Sutton, MA, USA) was glued on the bottom of the holder to keep the phantom solution before it was solidified (Figure 1A). The rat was provided with 1 hour of acclimation.
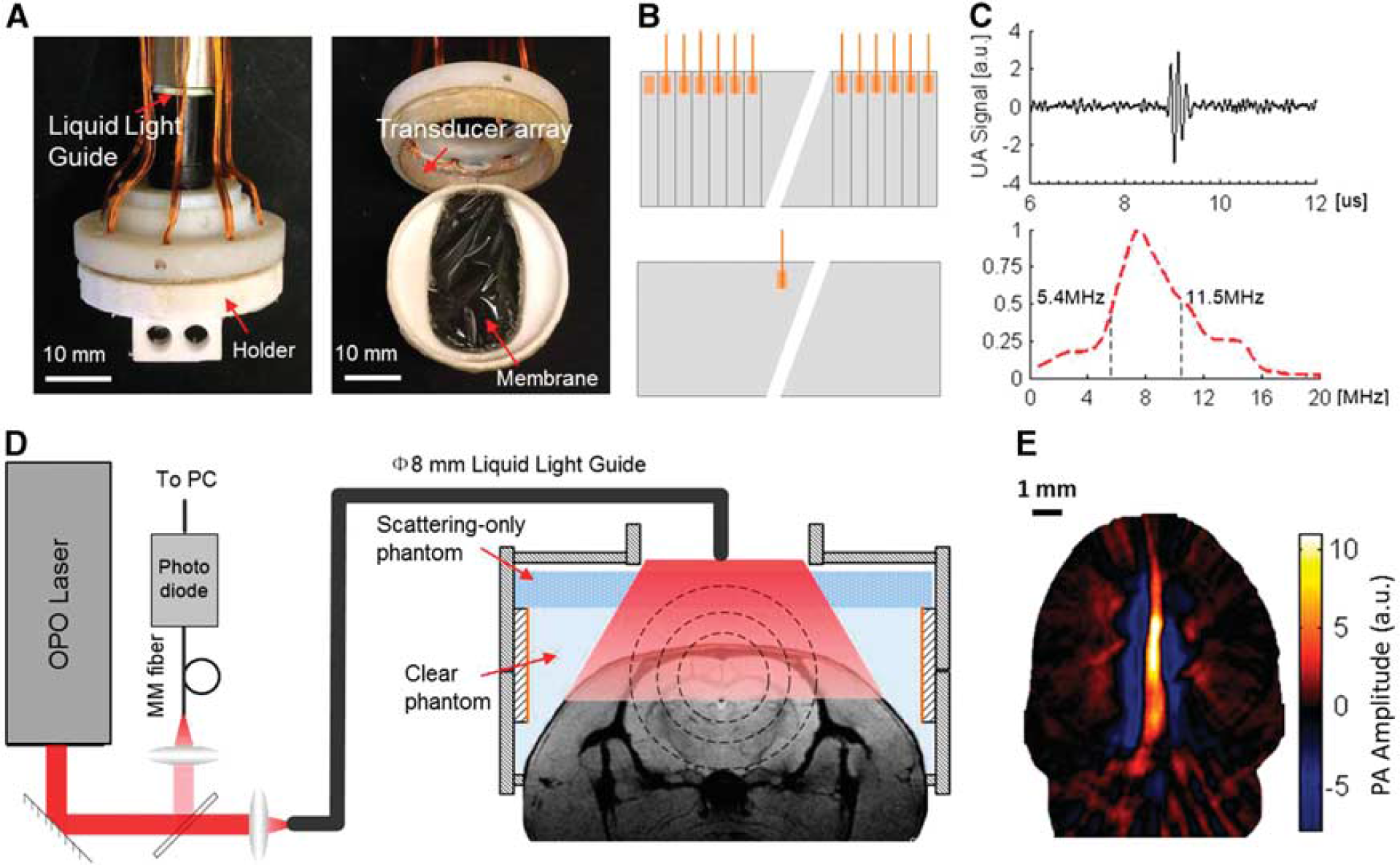
Wearable photoacoustic tomography (wPAT) imaging system and validation. (
One rat was used to test the cerebral functional imaging ability of wPAT by supplying it with 100% oxygen gas during hyperoxia. The rat was trained to immobilize condition (starting at 5 minutes and continuing up to 30 minutes) and exhibited positive reinforcement before the experiments. For each hyperoxia test, the experimental duration was less than 20 minutes. The rat was released for around 10 minutes after wPAT imaging for one wavelength and reimmobilized for another wavelength.
As there is a known interaction between hemodynamics and the underlying neurophysiology, 19 a total of 10 rats were used to test the cerebral blood volume imaging ability of wPAT. Six rats were temporarily anesthetized with isoflurane and implanted with two 300-μm diameter stainless steel screw electrodes (FHC, Bowdoin, ME, USA) within the skull (Figure 4B) for awake-behaving studies. Among these awake-behaving rats, four were used for PTZ-induced seizure experiment and two were used for saline-injection control experiment. Another group of four rats were anesthetized with 1 g/kg of body weight dose of Urethane and implanted with EEG electrodes.
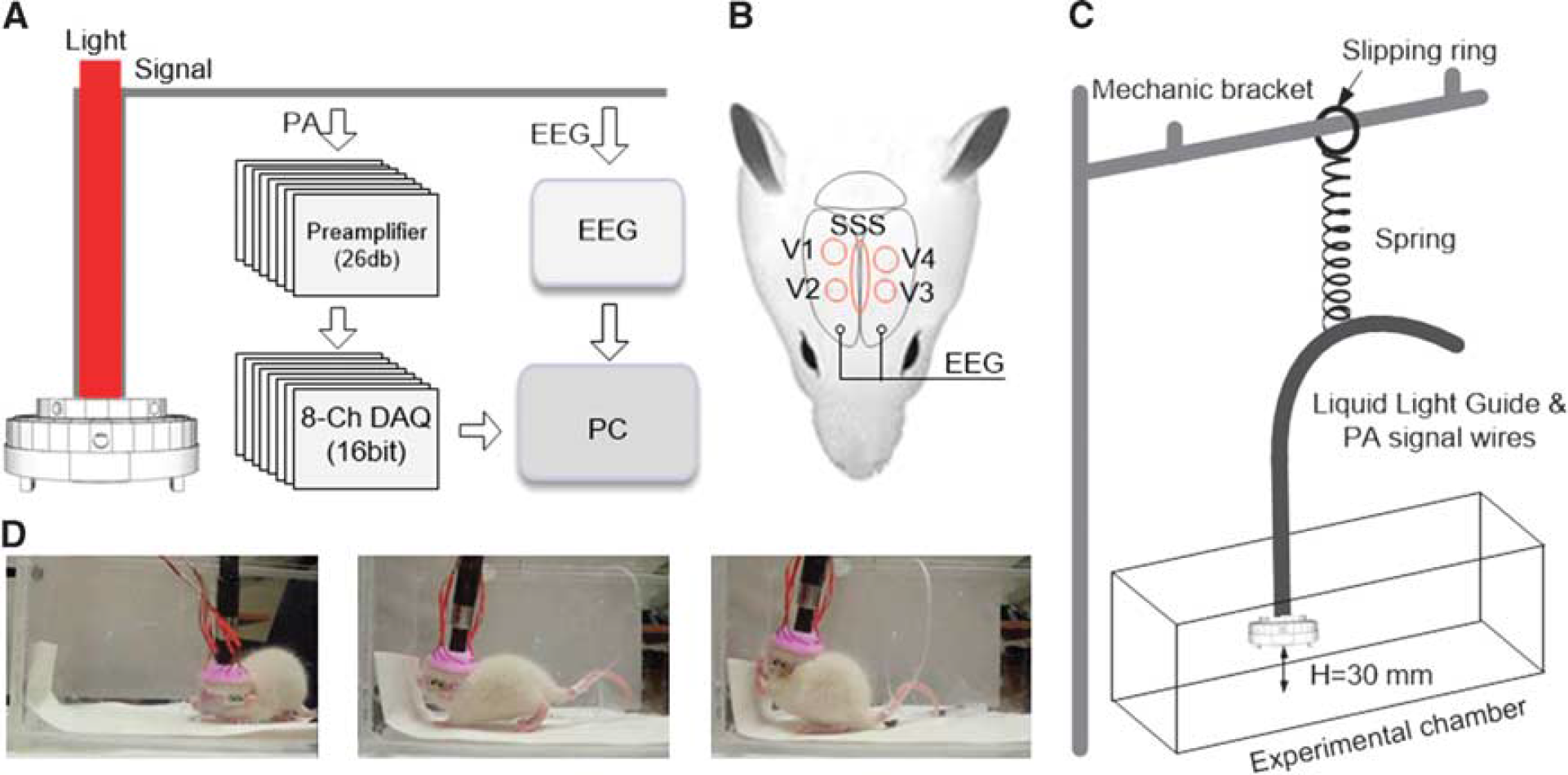
Experiment setup for pentylenetetrazol (PTZ)-induced generalized seizure. (
All procedures were approved by the University of Florida Animal Care and Use Committee and conducted in accordance with the National Institutes of Health Guide for the Care and Use of Experimental Animals.
Rat Epilepsy Model
Meduna's method of using PTZ is commonly used to stimulate absencelike seizures in rats.20,21 All four behavioral phenomena were observed in PTZ-induced seizures, including freezing, twitch, clonic seizure, and tonic-clonic generalized seizure. Although PTZ is recognized as GABAA receptor antagonist by affecting the t-butyl-bicylo-phosphorathionate site, metabolic 2-deoxyglucose studies showed that the metabolic activations also increased in the cortex after PTZ injection, indicating cerebral blood volume increase.22,23 Thus, PTZ-induced rat epilepsy model would be an appropriate example to show the cerebral blood volume imaging ability of wPAT. In our study, rats (four in awake-moving state and four under anesthesia) were received 0.1 g/kg of body weight dose of PTZ through intraperitoneal injection after 3 mins-baseline.
Data Processing
Offline analysis was performed with MATLAB (Mathworks Inc., Natick, MA, USA). The wPAT images (20 × 20 mm2) were reconstructed with Delay&Sum algorithm24,25 after a band pass filtering (0.5 to 12 MHz) of each A-line signal. Regions of interest (ROIs) were chosen according to the major blood vessels of the upper cortical regions (Figure 3A), and were analyzed for hemodynamic responses that were calculated by averaging PA signal values of pixels in each respective region and calibrated according to laser light energy monitored data. Hemodynamic response data were converted into the percent intensity change over time by subtracting and dividing the mean value of baseline (0 to 180 seconds, 599 images). Corresponding trend of time course for each ROI (Figure 5B, Supplementary Figures 2 and 3) was further obtained using a moving window average method (window length 5 seconds). Significant hemodynamic events were defined as epochs when the corresponding trend value was 200% larger than the standard deviation of the baseline.
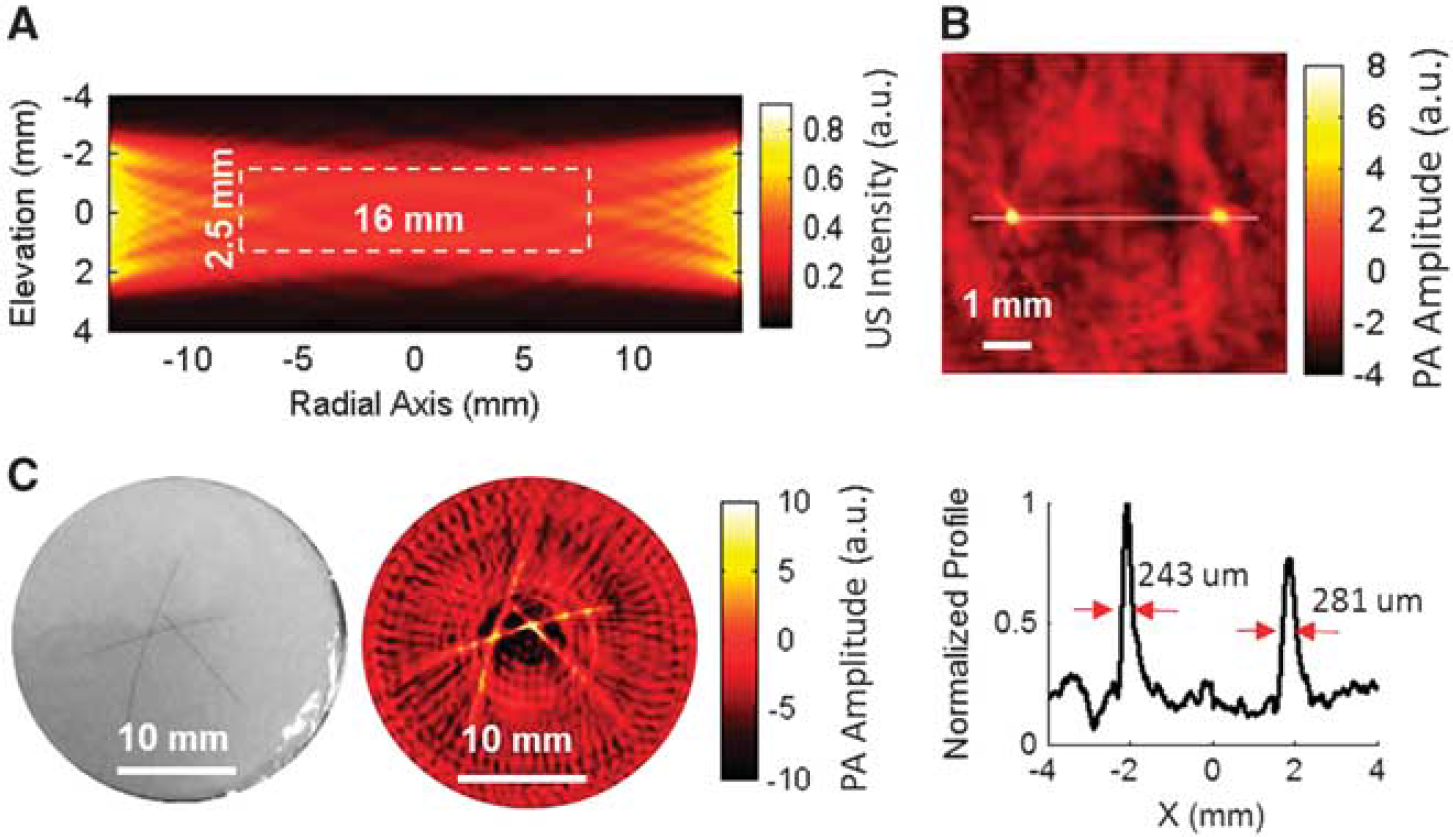
The spatial resolution evaluation. (
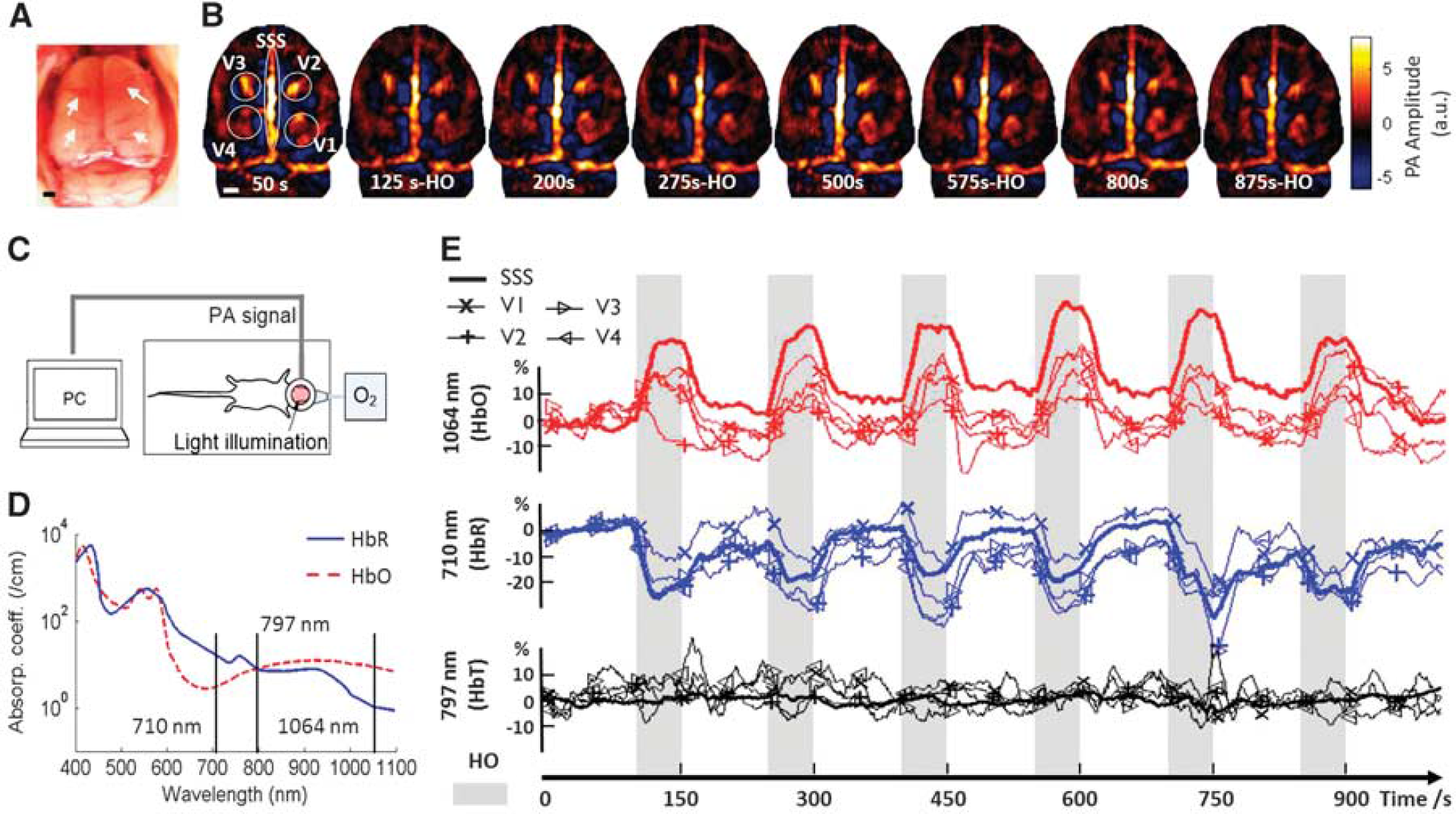
Functional imaging ability assessment of wearable photoacoustic (PA) tomography (wPAT). (
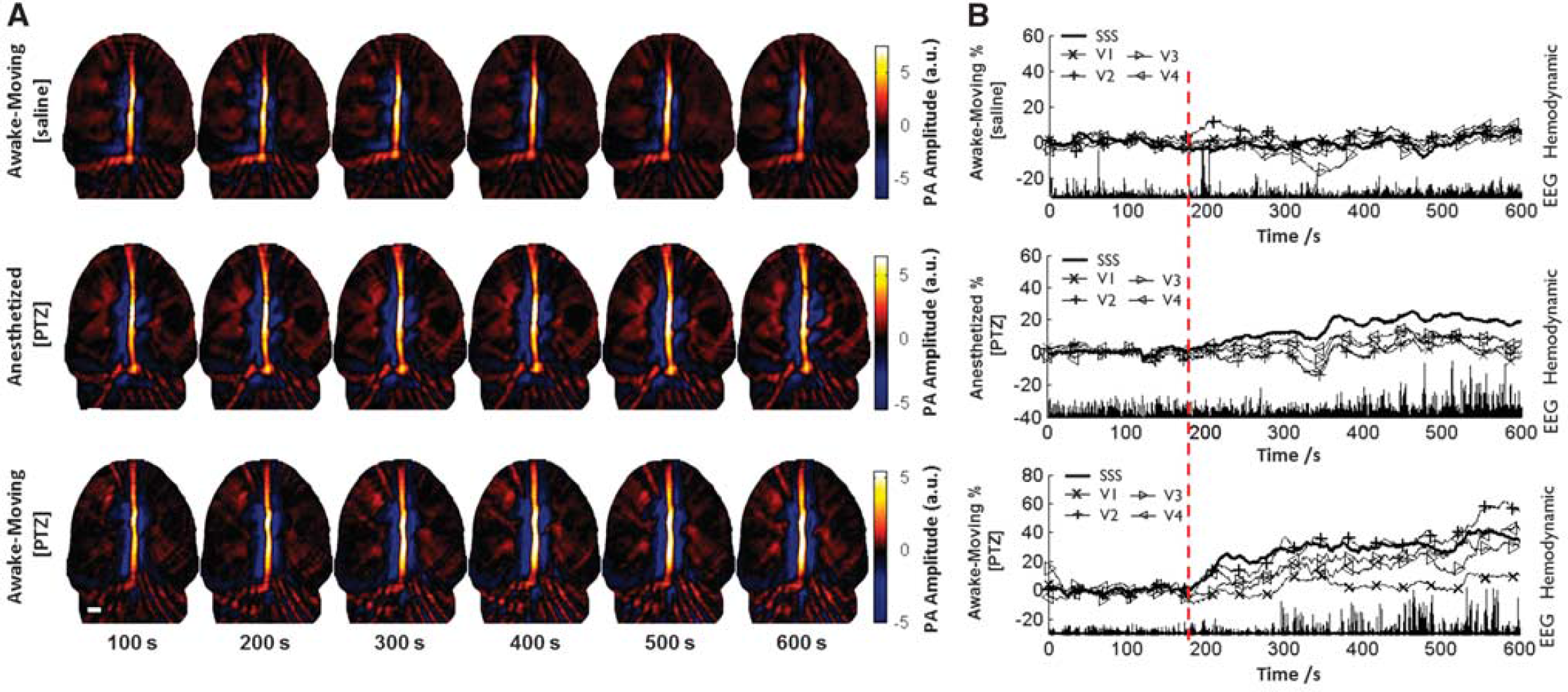
Cerebral blood volume responses for rats under different states. (
Electroencephalography signals were acquired by a Tucker Davis Pentusa neural recording system (Tucker Davis Technologies, Alachua, FL, USA) at a sampling rate of 6 kHz and a resolution of 16-bit. Electroencephalography data were band-pass filtered from 2 to 200 Hz, and the short-time Fourier transform was performed for time-frequency power spectrum density (PSD) analysis using Welch's method. The PSD value was calculated in the Gamma range (30 to 80 Hz, which is considered relate closely with the initiation of seizures 26 ) by integrating power spectrum within this range. Latency time of EEG recording was defined as the time between PTZ injection and occurrence of electric discharge.
Statistical Analysis
Comparisons (hemodynamic percent intensity change and latency time, Figure 6) between groups were performed based on one-way ANOVA. Correlation analysis between total hemodynamic response and EEG PSD was calculated using Spearmans' rank correlation analysis, where 30 periods' (20 seconds each, selected with equal intervals) mean hemodynamic percent intensity changes and PSD in the gamma range were employed for the analysis. All statistical results were expressed as mean ± s.e.m.
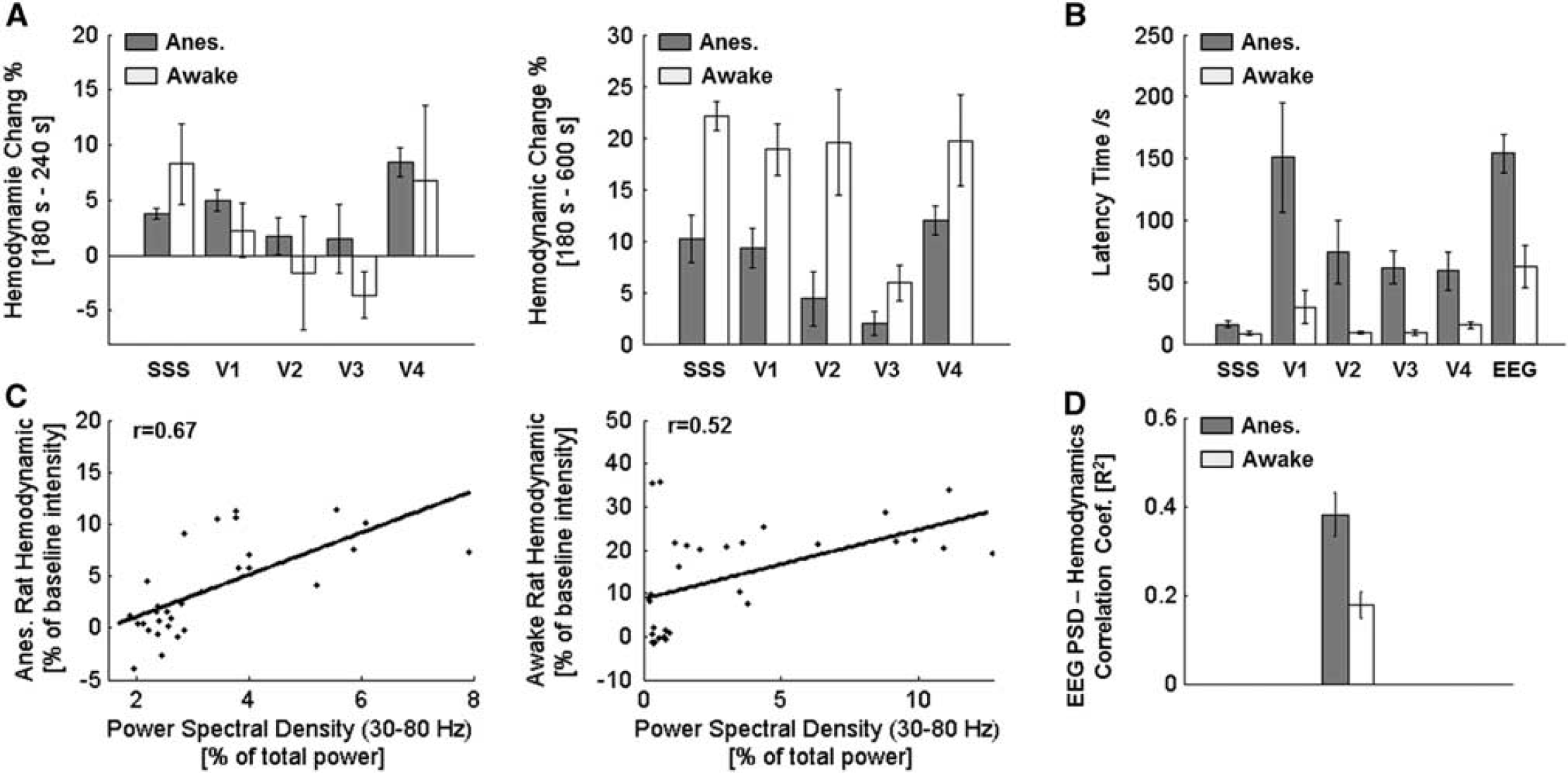
(
All data were reported following the ARRIVE (Animal Research: Reporting in Vivo Experiment) guidelines.
Wearable Photoacoustic Tomography System
The wPAT is a miniaturized cap-shaped, fully functional PAT system (Figure 1A). It was designed with the aid of SketchUp (Trimble Navigation, Ltd., Sunnyvale, CA, USA) to have an inner diameter of 30 mm, an outer diameter of 33 mm, and an overall height of 15 mm (Figure 1A). The main enclosure of wPAT was produced on a 3-D printer using polyamide material (online 3D Printing Service, I.materialise.com). The entire cap had a weight of 8 g (excluding cablings).
The interior of the wPAT casing was lined with a 64-element transducer array manufactured out of polyvinylidene fluoride (PVDF). Although PVDF transducers have a lower sensitivity than piezoelectric ceramics (PZT) and single crystal (PMN-PT) based transducers, it has the unique merit of flexibility, allowing it to be easily fabricated in donut-like or many other shapes. 27 Moreover, PVDF is light in weight and has a high mechanical damping ability and low acoustic impedance (PVDF, Z0 = 2.7 [kg/(m2·second)] × 106; PZT, Z0 = 33.7 [kg/(m2·second)] × 106; tissue-like phantom, ~ 2 [kg/(m2·second)] × 106; brain tissue, Z0 = 1.6 [kg/(m2·second)] × 106; skin, Z0 = 1.99 [kg/(m2·second)]× 106; bone axial (longitudinal waves), Z0 = 7.75 [kg/(m2·second)] × 106)28,29 resulting in high bandwidth and high coupling efficiency to tissue.27,29 The transducer array was manufactured from PVDF film by removing the silver layer between elements from one side and leaving the other side as a shared negative face. A 110-μm-thick PVDF film with a size of 5 × 94.2 mm2 was divided into 64 cells (element size, 1.47 × 5 mm2; etching width, ~ 100 μm). Each division served as an individual element's positive face, and all the transducer elements shared the negative face on the other side (Figure 1B). This innovative design not only satisfied all the requirements for behaving imaging, but also maximally reduced the differences between elements. The transducer array was then glued to the housing and isolated with 5-minute Epoxy gel (Devcon Inc., Danvers, MA, USA). Testing of the transducer array acoustic properties provided a central frequency of 8.1 ± 0.07 MHz (mean ± s.e.m., n = 64) with a bandwidth of 75.4 ± 1.19% (mean ± s.e.m., n = 64, −6 dB level) (Figure 1C shows the ultrasound signal (top) and frequency response (bottom) of one element transducer).
A liquid light guide (core diameter = 8 mm, NA = 0.59, model# 77631, Newport Inc., Irvine, CA, USA) was incorporated into the wPAT system for light delivery (Figures 1A and 1D). Pulsed laser light (repetition rate: 20 Hz, pulse duration: 10 ns, wavelength range: 680 to 2,550 nm, model: Surelite OPO Plus, Continuum Inc., San Jose, CA, USA) was split into two beams; one was used for light energy monitoring and the other was coupled into the proximal end of the liquid light guide whose distal end was placed at the top of the wPAT probe. The measured maximum laser energy at the rat head surface (~ 1.5 cm2) was approximately 6 mJ/cm2 per pulse, which was below the suggested maximum exposure of 20 mJ/cm2 specified by the American National Standards Institute. 30 Clear phantom solution (i.e., distilled water +1% agar power) was used to fill the 6- to 7-mm-high space between the probe and the rat head. It served as impedance matching medium to ensure high efficiency delivery of PA signal to the transducer array. In addition, a layer of 3-mm-thick scattering-only phantom (reduced scattering coefficient μs′ = 1/mm) was attached to the top of the clear phantom. This layer of phantom served as an optical diffuser to achieve a homogenous light distribution/excitation (Figure 1D). The phantom solution was solidified within 3 to 5 minutes in the room temperature.
All signals were recorded for offline processing via a custom-built 64-channel signal amplifying (26 dB) and data acquisition (10 bit, 50 Ms/s) module that can acquire 64 channels' signal at a time enabling a high-speed imaging ability. This module had in total 192 input amplifying channels (AD8099) that were multiplexed (MAX 4051) into 64 data acquisition channels (PCIAD 850, US Ultratek Inc., Concord, CA, USA) (see Wang et al 16 for detailed discussion about these amplifiers and data acquisition boards). Since wPAT had only 64 transducer elements, the signals from all transducers could be amplified and acquired without multiplexing. For each laser pulse, a trigger was sent to the amplifying and DAQ module to acquire the 64-channel PA signal, and the data acquisition length for each channel was 1,000 points (which was ~ 30 mm in distance for each channel, calculated by multiplying the DAQ time and velocity of ultrasound in tissue: [1,000/(50 × 106/s) × 103 μs] × [1.495 mm/μs]). Therefore, each laser pulse could produce a wPAT image with a field-of-view of ~ 30 mm in diameter. The high-speed, noninvasive imaging ability of wPAT in awake-moving rats was demonstrated by continuously obtaining wPAT images over time, which show overall good quality and sensitivity for cerebral hemodynamic studies (Figure 1E).
The Spatial Resolution of Wearable Photoacoustic Tomography
The wPAT was a 2D imaging modality as the transducer ring had only one element in the elevation dimension. Each element was a flat rectangle acoustic transducer (1.47 × 5 mm2) (Figure 1B). Therefore, the wPAT did not have resolution capability in the elevation dimension. Simulation on the ultrasound detection domain of the transducer array was performed using Ultrasim simulation MATLAB toolbox. 31 Due to the symmetry of the transducer array, the calculation of ultrasound detection domain was simplified to calculate the ultrasound intensity distribution of two opposite placed transducer elements (e.g., element #1 and element #33). As shown in Figure 2A, the simulated result indicated that wPAT had a disk-shaped imaging ‘plane’ with a diameter of 16 mm and a thickness of 2.5 mm.
To measure the lateral resolution of wPAT, two identical copper rods (diameter: 50 μm; center-to-center distance: ~ 4 mm) were vertically buried in a cylindrical tissue-like phantom (absorption coefficient μa = 0.007/mm; reduced scattering coefficient μs′ = 1/mm; height = 5 mm) at a depth of 2 mm. Figure 2B shows the wPAT image (top) and normalized profile crossing the centers of the two copper rods (bottom). The FWHM (full width at half maximum) for each copper rod was measured to be 243 and 281 μm, respectively, indicating that wPAT has a spatial resolution of 243 μm. We further applied wPAT to image a phantom sample (absorption coefficient μa = 0.007/mm and reduced scattering coefficient μs′ = 1/mm for the background) with three human hairs (diameter: ~ 70 μm each) embedded at a depth of 2 mm. The wPAT image (right, Figure 2C) showed a good agreement with the phantom sample (left, Figure 2C) in terms of target shape and position. Three transects' values of FWHM for each hair were measured based on the wPAT image, resulting in a statistic result of 280 ± 34 μm (mean ± s.e.m., n = 9) with a maximum of 380 μm in the peripheral region.
RESULTS
Wearable Photoacoustic Tomography for Cerebral Functional Imaging
As an initial test, the functional imaging ability of wPAT was evaluated on an awake rat applied with hyperoxia. To repeatedly apply oxygen gas to the rat, the rat was restrained but left in awake state as shown in Figure 3C, and the illumination laser was delivered with mirrors (Thorlabs Inc., Newton, NJ, USA). According to the optical absorption coefficient of HbO and HbR (Figure 3D), three wavelengths (710, 1,064, and 797 nm) were used to study HbR, HbO, and HbT responses, respectively. The wPAT images were obtained with six times averaging, i.e., the imaging rate was 3.33 frames/s. For each wavelength experiment, the rat was first in resting state for 100 seconds and then followed by six trials of hyperoxia-normoxia (i.e., normal oxygen level) where each trial consisted of 50 seconds of hyperoxia and 100 seconds of normoxia. The total testing time for one wavelength experiment was 1,000 seconds, and the rat was released for ~ 10 minutes and the laser was set to the next wavelength after each run. The rat was reimmobilized for the next wavelength experiment.
We analyzed hemodynamic response for five ROIs, including superior sagittal sinus (SSS) and four major cortical blood vessel regions 1, 2, 3, and 4 (V1, V2, V3, and V4, respectively) as marked in Figure 3A, which was a photograph of a rat brain with skull and scalp removed after the imaging experiment. Figure 3B shows eight wPAT images obtained at 710 nm (mostly sensitive to HbR); the images at the 50th second, 200th second, 500th second, and 800th second were obtained under normoxia, while the images at 125th second, 275th second, 575th second, and 875th second were obtained under hyperoxia (Supplementary Figure 1 shows all three wavelengths). Comparing these wPAT images with the photograph given in Figure 3A, the SSS, transverse sinus, and other major cortical blood vessel regions (V1, V2, V3, and V4) were readily evident. Moreover, these wPAT images showed that image intensities in hyperoxia were weaker than that in normoxia (Figure 3B). Figure 3E shows the hemodynamic response time courses of SSS, V1, V2, V3, and V4 obtained at 1,064 nm (HbO), 710 nm (HbR), and 797 nm (HbT). The gray shaded background marks the period when the rat was in hyperoxia. These time courses showed that, when the rat was applied with oxygen gas, the hemodynamic response decreased at 710 nm but increased at 1,064 nm, while there was no significant change at 797 nm for all ROIs. According to the optical absorption coefficient of HbO and HbR (Figure 3D), these results indicated decreased concentration of HbR but increased concentration of HbO when the rat was under hyperoxia. The fact that little hemodynamic change was detected at 797 nm, suggested that the cerebral blood volume did not change much in response to hyperoxia.
Wearable Photoacoustic Tomography for Cerebral Blood Volume Imaging
To evaluate the potential of wPAT for cerebral blood volume imaging in awake-moving rats, we next applied wPAT to study cerebral hemodynamics with a rat epilepsy model (PTZ-induced generalized seizure) using 797 nm wavelength light at which the absorption coefficient of HbO and HbR is identical. To concurrently record the cerebral hemodynamics, EEG and behavior activity of the rats, an experimental setup combining wPAT imaging, EEG recording, and video monitoring was constructed (Figures 4A and 4D). The EEG system was set in a waiting state until received a trigger signal from the wPAT system when the wPAT system finished initialization and was about to acquire data. To minimize the influence of EEG recording to wPAT imaging, EEG electrodes were implanted in the acentric imaging area (Figure 4B). In addition, we designed a passive weight support system to let wPAT wearable on an awake-moving rat. As shown in Figure 4C, all cables were suspended on the passive weight support system, which included a low elasticity spring (k = 10 N/m), a slipping ring, and a mechanic bracket. Wearable PAT was lifted to a height around 30 mm with transformed weights of ± 20 g at the heights of 30 ± 20 mm, respectively, allowing the rat to move around in the experimental chamber (generally, the vertical movement of rat head was within ± 10 mm).
Acute generalized tonic-clonic seizure was induced by intraperitoneal injection of 0.1 g/kg of body weight PTZ for two groups of rats (four each; one group in awake-moving state; the other was anesthetized with 1 g/kg of body weight Urethane). Control experiments were also performed on two resting rats (awake-moving with injection of saline). In this experiment, wPAT, EEG, and video recordings were synchronously recorded for the duration of the experiment (600 seconds), and intraperitoneal injection of PTZ/saline was administered after 180 seconds of baseline data collection. While multiple regions can be evaluated by wPAT, the present study focused on the major cerebral vasculatures, including SSS, V1, V2, V3, and V4 (Figure 4B). After the injection of PTZ at the 180th second, wPAT image intensity increased consistently and EEG spike wave epileptiform discharges occurred under both anesthetized and awake-moving states (middle and bottom panels, Figure 5). However, according to the recorded videos, seizure activities, such as freezing, twitch, and tonic-clonic seizure, were observed in all awake-moving rats within the testing period, while no seizure activity was observed in the four anesthetized rats within the testing period (Supplementary Movies 2 and 3).
To further quantify the response, we examined time courses of hemodynamic response percent intensity change for the five ROIs relative to the 180-second baseline for all states (Figure 5B, Supplementary Figures 2 and 3). No significant hemodynamic deviation was observed for the resting rats after the injection of saline as shown in the top panel of Figure 5 (Supplementary Movie 1). A significant hemodynamic response increase was observed in both anesthetized (Figure 5, middle; Supplementary Figure 3A) and awake-moving (Figure 5, bottom; Supplementary Figure 3B) states after the injection of PTZ (statistic results were shown in Figure 6A). Differences were also noticed between the two states after the injection of PTZ that cerebral hemodynamics started to change shortly after the PTZ injection in awake-moving rats (a dip response for two out of the four rats and immediate increase for the other two rats, Supplementary Figure 2B); in contrast, the cerebral hemodynamics increased slowly and consistently for all four anesthetized rats (Supplementary Figure 2A). Moreover, for the rats under anesthesia, a much longer latency time (~ 50 seconds longer for hemodynamic and ~ 100 seconds longer for EEG discharge) was observed compared with the awake-moving state. Interestingly, for awake-moving state, the five ROIs' hemodynamic latency time were comparable, while, for the anesthetized group, the four major cortical blood vessel regions (V1, V2, V3, and V4) showed a much longer latency time than SSS (~ 40 seconds, statistic results were shown in Figure 6B). Further, preceded hemodynamic changes over EEG discharges were noted for both seizure-induced conditions (~ 50 seconds for awake-moving state and ~ 90 seconds for anesthetized state, Figure 6B, mean ± s.e.m., n = 4), which was also observed in fMRI,32,33 near-infrared spectroscopy, 34 diffuse optical tomography, 35 and intrinsic optical imaging36,37 -based studies.
Statistical results (mean ± s.e.m., n = 4) showed that, in the first 60-second period after PTZ injection (left panel of Figure 6A), the hemodynamic response percent intensity change in the anesthetized state increased consistently compared with the awake-moving state that had negative mean value change for ROIs of V2 and V3, and much bigger s.e.m. values for all ROIs. This may indicate the complex response process for awake-behaving animals that each individual may respond differently to the stimulations. However, in the period from PTZ injection to the end of experiment (right panel of Figure 6A), there was a mean ~ 20% hemodynamic increase in the awake-moving state compared with an ~ 10% increase in the anesthetized state. This difference may indicate the depression effect of anesthetic agent to animals' physiologic function. Correlation analysis (Figures 6C and 6D) between hemodynamic responses and PSD of EEG in the gamma range (30 to 80 Hz 26 ) revealed a moderate correlation for both anesthetized and awake-moving states. Other studies also reported similar positive correlation between hemodynamic signal and EEG PSD within gamma range.35,38
DISCUSSION AND CONCLUSION
We have conducted experiments based on the developed wPAT system using hyperoxia and acute PTZ-evoked seizure rat models. Results show that wPAT successfully detected cerebral HbO, HbR, and HbT changes of a rat when under hyperoxia by using 1,064, 710, and 797 nm wavelengths, respectively. The findings suggested that wPAT can detect HbO and HbR changes within the brain vasculatures; therefore, it would be applicable to study pathologic events such as stroke, brain ischemia, and seizure. Simultaneous recordings of cerebral blood volume change, brain electric potential discharge, and behavior activity were performed in awake-moving and anesthetized rats. As anticipated, significant increases in blood flow and neural dynamics were observed after PTZ administration relative to saline controls. Compared with the anesthetized state, rats in awake-moving state showed: (1) a higher hemodynamic intensity increment and (2) a shorter latency time for both hemodynamic and EEG responses after PTZ administration. Moreover, based on the pattern of blood flow in the brain, the SSS should reflect hemodynamic changes in a later time compared with other regions as it is the major vein receiving blood flow from the cerebral blood vessels. Results from awake-behaving rats were in good agreement with this expectation, while the four major cortical blood vessel regions' response (V1, V2, V3, and V4) showed a much longer latency time than SSS in the anesthetized results. This potentially suggests that rat's neural activity is understandably depressed under anesthesia, indicating an inherent drawback of anesthesia-based animal models, and the importance and advantage for using awake-moving animal models. In addition, preceded hemodynamic responses to EEG discharges were observed for all rats at both states. Several hypotheses attempting to explain this phenomenon have been proposed and discussed in a review. 39 From a technical point of view, one possibility for this phenomenon may be that the EEG recording suffers from low spatial resolution and that certain brain regions' potential change cannot be detected due to the limited electrodes positioning. 40 However, the concurrent hemodynamic responses could be captured by whole-brain high spatial-temporal resolution imaging modalities, such as wPAT. This observation is in agreement with fMRI, near-infrared spectroscopy, and diffuse optical tomography-based results,33–35,41 the wPAT images, in addition, showed a much higher spatial and temporal resolution than such modalities.
Tremendous efforts have been dedicated to advance functional brain imaging techniques to resolve the enigma of neurovascular communications between neural activity and subsequent cerebral blood vessel conditions.42,43 Importantly, our results suggest that wPAT is capable of noninvasive, high spatiotemporal imaging in awake-behaving rodents and is therefore optimal for understanding the interaction between both neural- and hemodynamics. For example, by leaving animal in awake-moving state, more realistic hemodynamic parameters (vasodilation, intensity, latency time, and recover time) of cerebral vasculature could be obtained in response to stimulation, such as forepaw stimulation, 44 whisker stimulation, 45 and hypoxia challenge.46,47
Moreover, employing multi-wavelength excitation, wPAT can be used to reconstruct the oxy-saturation of the cerebral hemodynamics in an awake-moving rat as we demonstrated in this study. In addition, wPAT can be applied for cerebral metabolic research and cerebral drug delivery or targeted molecular imaging by choosing appropriate laser wavelengths and employing exogenous optical contrast agents. 48 Finally, wPAT can be used for preclinical studies by leaving animals in a conscious state. For example, it can be used to evaluate the effects of optogenetics49,50 and to study stroke, and epilepsy as demonstrated in this work.
In summary, this study suggests that wPAT can noninvasively acquire high spatial resolution brain images at a high speed from awake-moving rats. It can also be used to image cerebral hemodynamics in an awake-behaving mice by designing an appropriate wPAT holder that can be mounted on the mice head. Due to the reduced number of transducer element, wPAT's spatial resolution is little worse than the current array-based PAT systems.9,15 However, cerebral hemodynamic responses of different regions were successfully detected and imaged by wPAT. The image quality (X-Y spatial resolution) of wPAT can be improved by increasing the number of elements (~ 100 μm radial resolution was reported by using a 512-element transducer array 51 ) or using transducers with higher central frequency and wide bandwidth (30 μm image resolution was reported with a 20-MHz central frequency transducer having a bandwidth ranging from 50% to 80% 52 ). To improve the resolution in the elevation dimension, one approach is to use an elevation-focused transducer array (an elevation resolution of 0.6 to 2 mm within the imaging ‘disk’ was reported in Gamelin et al 9 ). An alternative method is to reduce the size of transducer element along the elevation dimension (e.g., to 1 mm), but this may reduce the detection sensitivity. Although several-times averaging was applied to enhance signal-to-noise ratio, it is worth to note that the primary limitation of temporal resolution of wPAT is the repetition rate of pulsed laser. Much faster imaging rate (tens of frames per second) is achievable by using higher repetition rate pulsed lasers that are commercially available. Micro-blood vessels/chromophores within the scalp may have a confounding effect to the detected signals since they can also generate PA signal when light propagating through the scalp. However, the dominate PA signal should be owing to the absorption by major cerebral blood vessels (SSS, V1, V2, V3, and V4) since they have much bigger size and absorb most light energy. References10,15,51 have shown that different regions' cerebral hemodynamics responses differ in response to whisker, focus seizure, or forepaw stimulation, indicating that the scalp has a small effect on the detected PA signal. However, if minimal invasive procedure (i.e., removal of the scalp) is acceptable, much shorter wavelength light (e.g., 560 nm for HbO, 578 nm for HbR, and 570 nm for Hbt) could be used for illumination. In this case (1) confounding factors due to excitation of chromophores within the scalp could be eliminated; (2) higher signal-to-noise ratio signal could be obtained since hemoglobin has a much stronger absorption in shorter wavelengths; and (3) the signal generation layer could be confined to the upper cortical regions since short wavelength light has a small penetration depth, which could improve the elevation resolution of the system.
In the future, wPAT could be improved for fast 3D imaging by building a donut-like 2D transducer array to resolve hemodynamic responses from different layers of rat brain. To achieve this goal, the ultrasonic transducer material must have a higher sensitivity as the size of the transducer element may be reduced to 1 × 1 mm2. It can also be developed to record and display cerebral activities in real time by developing a GPU-based fast data processing module. 53 Driven by the goal of revealing natural process of brain activity, wPAT will be a powerful tool for the community as it permits studies in behaving rats that may challenge current anesthesia-based models but ultimately improve our understanding of the process involved in animal's real life.
AUTHOR CONTRIBUTIONS
JT developed the wPAT system, designed and performed the experiments, analyzed the data and wrote the paper; LX developed the wPAT system; JZ performed the animal experiments; HH and TZ developed the wPAT system; PC analyzed the data and wrote the paper; HJ conceived the concept of wPAT, directed the study, and wrote the paper.
DISCLOSURE/CONFLICT OF INTEREST
The authors declare no conflict of interest.
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
