Abstract
The interface between cerebrovascular disease (CVD) and epilepsy is complex and multifaceted. Late-onset epilepsy (LOE) is increasingly common and is often attributed to CVD, and is indeed associated with an increased risk of stroke. This relationship is easily recognizable where there is a history of stroke, particularly involving the cerebral cortex. However, the relationship with otherwise occult, subcortical CVD is currently less well established yet causality is often invoked. In this review, we consider the diagnosis of LOE in clinical practice—including its behaviour as a potential mimic of acute ischemic stroke and transient ischemic attack; evidence for an association between occult CVD and LOE; and potential mechanisms of epileptogenesis in occult CVD, including potential interrelationships between disordered cerebral metabolism and perfusion, disrupted neurovascular unit integrity, blood–brain barrier dysfunction, and inflammation. We also discuss recently recognized issues concerning antiepileptic drug treatment and vascular risk and consider a variety of less common CVD entities associated with seizures.
Keywords
INTRODUCTION
Late-onset epilepsy (LOE), or epilepsy starting in later life, is an increasingly common problem in an aging population. Common causes are considered to be cerebrovascular disease (CVD), primary neurodegenerative disorders, intracranial tumors, and traumatic brain injury. Arguably, it is relatively straightforward to rule out tumors and traumatic brain injury, and to some extent neurodegenerative disorders, but it is somewhat more difficult to definitively exclude underlying occult CVD particularly given the increasing diversity and subtlety of imaging markers for CVD. Cerebrovascular disease is easily recognized as the cause of LOE in patients with a history of stroke—particularly strokes involving the cortex, or those that are hemorrhagic, large, multiple, or associated with acute symptomatic seizures. 1 But in a currently unknown proportion of patients with LOE, otherwise regarded as cryptogenic, occult CVD may be the cause.
The interface between CVD and epilepsy is complex and multifaceted (Figure 1). In this review, we consider the evidence for an association between occult CVD and LOE, potential mechanisms and opportunities for intervention.
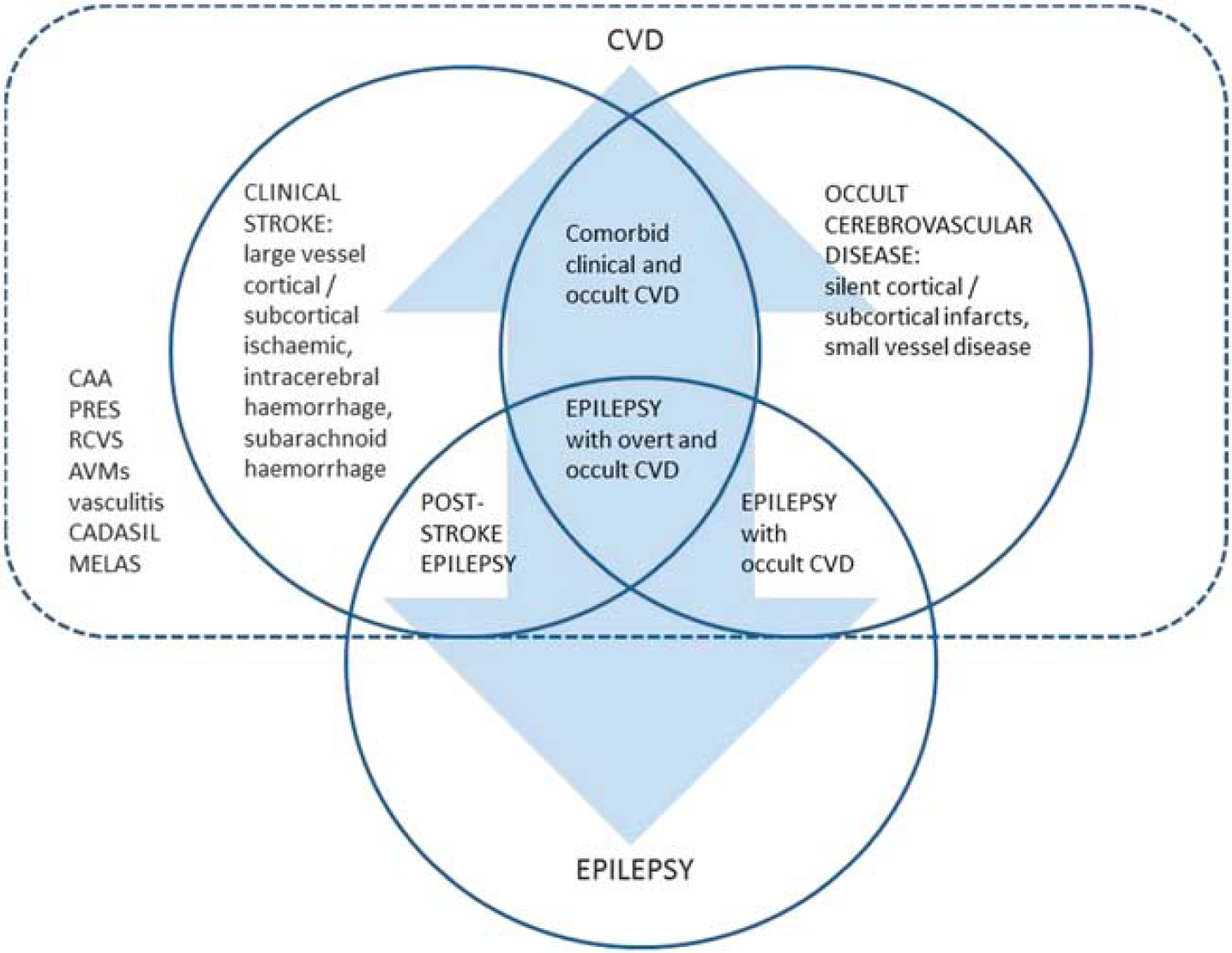
Interface between cerebrovascular disease (CVD) and epilepsy. The interrelationships between the two are complex and bidirectional, for example, stroke may lead to epilepsy, while late-onset epilepsy, potentially due to underlying occult CVD, is associated with increased stroke risk. A number of less common CVD entities are associated with seizures and epilepsy, occurring with or without clinical stroke (e.g., cerebral amyloid angiopathy (CAA); posterior reversible encephalopathy syndrome (PRES); reversible cerebral vasoconstriction syndrome (RCVS); arteriovenous malformations (AVMs); central nervous system (CNS) vasculitis; cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL); mitochondrial encephalopathy with lactic acidosis and stroke-like episodes (MELAS)). (Late-onset) epilepsy occurring in association with occult CVD is the particular focus of this review.
SEARCH STRATEGY AND SELECTION CRITERIA
References for this review were identified by searches of PubMed and OVID Medline from 1948 until December 2013 with the terms ‘cerebral ischemia,’ ‘cerebral hemorrhage,’ ‘cerebral infarct,’ ‘cerebral microbleed,’ ‘cerebrovascular disease,’ ‘blood brain barrier,’ ‘epilepsy,’ ‘seizure,’ ‘elderly,’ and ‘late-onset.’ Articles were also identified through searches of the authors' own files. Only papers published in English were reviewed.
LATE-ONSET EPILEPSY AND OCCULT CEREBROVASCULAR DISEASE: CLINICAL ASPECTS
Late-Onset Epilepsy in Clinical Practice
The diagnosis of LOE is often not straightforward, especially when one considers the interface with CVD, whether occult or overt. For example, the extent to which epilepsy may be underdiagnosed (or overdiagnosed) among patients presenting with suspected stroke or transient ischemic attack (TIA) is uncertain. This clearly hampers estimates of the true prevalence of LOE. Potential reasons for the misdiagnosis of epilepsy in older patients have been explored in detail previously 2 and therefore it is not necessary to repeat discussion of all clinical features, but seizure presentations are varied and can include falls, confusional states, amnesia, and focal neurologic symptoms. With respect to the latter, there is particular potential for misdiagnoses between stroke/TIA and late-onset seizures/epilepsy. This issue is particularly pertinent now that there is increased emphasis on emergency assessment of patients with suspected TIA or stroke. In one recent series of 350 clinical presentations of suspected stroke, the final diagnosis was a stroke mimic in 109 (31%). Seizures accounted for 21% of all stroke mimics and 29% of stroke mimics presenting within 6 hours. 3
The ‘borderlands' of TIA, seizures, and other transient neurologic deficits have recently been expertly discussed 4 in relation to a retrospective study comparing the characteristics of patients with inhibitory seizures and TIA. 5 In particular, short, repeated episodes of speech disturbances—especially when accompanied by confusion or amnesia—were suggestive of inhibitory seizures. Several difficulties exist in clinical diagnosis. First, seizures may simulate TIA, with some patients presenting transient focal deficit (Todd's paralysis); second, hemodynamic TIAs in the context of severe carotid stenosis, for example, can be associated with limb jerking, while shaking movements can be mistaken for seizure activity in brainstem ischemia; third, seizures may be the only manifestation of otherwise occult CVD, with seizures arguably sometimes heralding stroke; fourth, some causes of CVD (e.g., intracranial venous thrombosis) often provoke both ischemia (and hemorrhage) and seizures; finally, investigations can be inconclusive or misleading, for example, diffusion-weighted imaging is frequently normal in TIA, and normal by definition according to updated TIA diagnostic criteria, 6 while seizures can cause diffusion-weighted imaging abnormalities, and electroencephalography (EEG) frequently shows abnormalities in the context of CVD in the absence of seizures.
It is somewhat surprising, given that occult CVD is likely a common underlying cause of epilepsy, particularly in older adults, that this whole field has received relatively little attention. There is a clear need for further research to establish prevalence and better describe natural history, as well as to identify potential mechanisms of epileptogenesis, and opportunities for intervention.
Seizures as Stroke or Transient Ischemic Attack Mimics—Two Illustrative Case Studies
Traditionally, it has been said that the neurologist had little to offer the patient except a diagnosis—but in the emergency setting accurate clinical diagnosis of acute focal neurologic dysfunction is obviously crucial when treatments such as intravenous thrombolysis, neurointerventional treatment, or carotid surgery are likely to be considered. Investigations cannot replace clinical assessment in this regard. Arguably, involvement of the neurologist, or at least clinicians with an appropriate level of neurologic expertise, in the acute setting has never been more important. The point is perhaps best illustrated with case studies, as detailed below:
Patient 1
An 81-year-old female presented to her local hospital as an emergency with the abrupt onset of aphasia. An immediate computed tomography (CT) brain scan showed changes in keeping with cerebral small vessel disease (cSVD) (Figure 2). She was managed as an acute ischemic stroke and received thrombolysis with intravenous recombinant tissue plasminogen activator. She was noted to have a rapid recovery to her normal baseline and was discharged. Three months later she presented again with a further episode of isolated, transient, expressive aphasia, lasting several minutes. Further questioning revealed a history of repetitive gulping immediately before the onset of aphasia. Her husband recalled that this had also happened with the previous episode 3 months earlier. Given the stereotyped nature of the events including the motor automatism (gulping), the episodes were diagnosed as focal seizures and the patient was commenced on an antiepileptic drug (AED) in addition to vascular secondary prevention medication. She remained free of further episodes 2 years later.
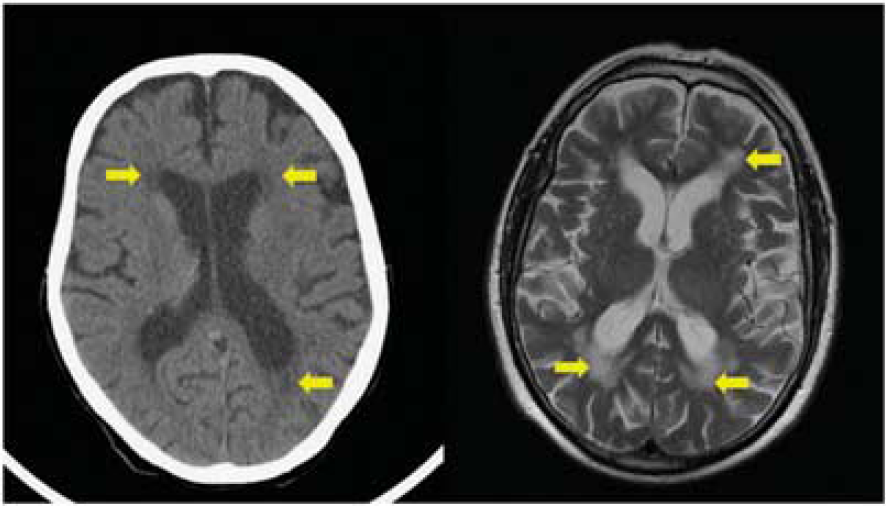
Cerebral small vessel disease on computed tomography (CT) and magnetic resonance imaging (MRI). Left-hand panel (patient 1): CT image showing hypoattenuation adjacent to lateral ventricles (arrowed); right-hand panel (patient 2): T2 axial MRI image showing high signal changes in white matter (arrowed).
Patient 2
An 83-year-old male presented with recurrent episodes of speech disturbance managed as TIAs following attendances at TIA clinics at two other teaching hospitals. Review of his clinical history, including description by a witness, revealed him to be having stereotyped episodes, lasting several minutes, of mutism followed by recovery of speech, involving rapidly improving expressive dysphasia. During these episodes he would appear vacant and be plucking at his clothes, with subsequent confused behaviour. He had no recollection of the episodes. Such episodes were consistent with complex partial seizures and AED treatment was advised but the patient was initially reluctant to accept the diagnosis of epilepsy having previously been given a diagnosis of TIAs, and in view of the implications for driving. A magnetic resonance imaging (MRI) brain scan showed diffuse white-matter high signal changes in keeping with cSVD, involutional change, but no other structural abnormality (Figure 2). A routine scalp EEG captured a clinical seizure with epileptiform changes arising from the left temporal region. Electroencephalography changes comprised rhythmic 4 to 5 Hz theta activity throughout the left hemisphere, maximal in the temporal region, spreading after a further 20 seconds to the right frontocentral region and then slowing further to rhythmic 2 to 3 Hz delta activity throughout the left hemisphere with sharpened slow wave activity seen particularly in the posterior quadrant. This further slowed to 1 to 2 Hz delta activity seen diffusely throughout both hemispheres. The whole electrical event lasted ∼50 seconds, during which time the patient vocalized, then became agitated, startled to fiddle with clothing and was unresponsive. The temporal lobe onset is in keeping with classic views on anatomic substrates of complex partial seizures and would suggest that the MRI visualized distribution of subcortical CVD is merely a marker of more widespread changes structurally or functionally disrupting neuronal circuits involving the temporal lobe and relevant in epileptogenesis.
OCCULT CEREBROVASCULAR DISEASE AND LATE-ONSET EPILEPSY: EVIDENCE FOR AN ASSOCIATION
In a cross-sectional, community-based, case–control study comprising 4,944 subjects, 65 of whom had epilepsy, a history of stroke was strongly associated with lifetime epilepsy (odds ratio (OR) 3.3; 95% confidence interval (CI) 1.3 to 8.5), as well as with LOE (OR 3.1; 95% CI 0.9 to 10.6). 7 History of stroke or myocardial infarction, peripheral vascular disease, hypertension, serum total cholesterol, and left ventricular hypertrophy were significantly associated with lifetime epilepsy and LOE, and even when stroke patients were excluded, ORs remained statistically significant for the relationships between total cholesterol and LOE (OR 1.3, 95% CI 1.0 to 1.6) and left ventricular hypertrophy and LOE (OR 2.9, 95% CI 1.0 to 8.6). The results suggested that there may be a relationship between vascular factors and the risk of LOE, apart from the relationship that exists through clinically overt stroke. It is perhaps surprising that even in this study, with a relatively low age threshold of 40 years for a definition of LOE, that relationships were noted between vascular determinants and LOE.
The relationship between hypertension and seizures through brain damage with or without manifest stroke has been investigated. In a case–control study of 227 patients admitted for a first unprovoked seizure and 294 acute surgical controls, hypertension was significantly associated with unprovoked seizures, even after adjustment for antecedent stroke and other potential confounders (adjusted OR=1.57; 95% confidence limit 1.0 to 2.44). 8
Prevalence of Occult Cerebrovascular Disease in Patients with Late-Onset Epilepsy
One previous study 9 included outpatients who had developed single or multiple seizures after the age of 40. Age-matched controls with nonvascular neurologic disease were recruited from the authors' outpatient clinics. None of the participants had evidence of CVD. Patients presenting with epilepsy for the first time after the age of 40 had significantly more occult, previously unsuspected, infarcts than controls. Of 56 patients aged over 60 at the time of seizure development, 12 had infarcts on CT scan compared with one of the 57 controls aged over 60, giving a prevalence of occult CVD in patients with LOE of 21.4%.
In our own case–control study of 105 patients with LOE or late-onset seizures and 105 age and sex-matched controls, radiologic CVD was significantly more prevalent in cases (66%) compared with controls (33%) (P<0.0001), with cSVD changes in 50% cases and 32% controls (P<0.05). 10 Cerebral SVD showed a nonsignificant trend toward greater severity among cases.
The Occurrence of Stroke in Patients with Late-Onset Epilepsy
Using the Office for National Statistics GP Research Database 4709 patients without a history of CVD who developed seizures after the age of 60 and 4709 age, sex and GP practice matched controls were identified. 11 Searching these patients' records from the date of diagnosis of first seizure for subsequent diagnoses of stroke enabled them to calculate an OR for the risk of clinical stroke in LOE patients of 2.89 (95% CI 2.45 to 3.41).
A cohort of 14 patients who developed seizures after the age of 40, and who had evidence of CVD on CT scan not suspected clinically, was followed up in one study. 12 In addition, 15 patients who developed seizures after the age of 40 who did not have occult CVD were also followed up. The two groups were similar in age distribution and were followed up for a mean of 4.9 years. One patient with occult CVD had a stroke compared with no patients in the nonoccult CVD group. In another study of 88 patients aged 22 to 80 with completed stroke confirmed on CT, 7 patients had experienced heralding seizures that occurred between 20 days and 5 years before their stroke. 13 All seven had cerebral infarcts on CT. Three patients were aged over 60 at seizure onset (range 24 to 71). One further study that included 176 patients with their first acute stroke, 8 were found to have a history of epilepsy, of whom 2 were aged over 60 at the time of seizure development (range 11 to 64). 14
Summary of Evidence
The limited available evidence, summarized in Table 1, suggests (1) a relationship between vascular risk factors and the risk of LOE, apart from the relationship that exists through clinically overt stroke, in patients older than 40 years, (2) an excess of clinically unsuspected, radiologic CVD, particularly cortical infarction but apparently also excess SVD changes, and (3) an excess (almost threefold) risk of stroke in patients with LOE.
Clinical studies investigating relationship between cerebrovascular disease and late-onset epilepsy
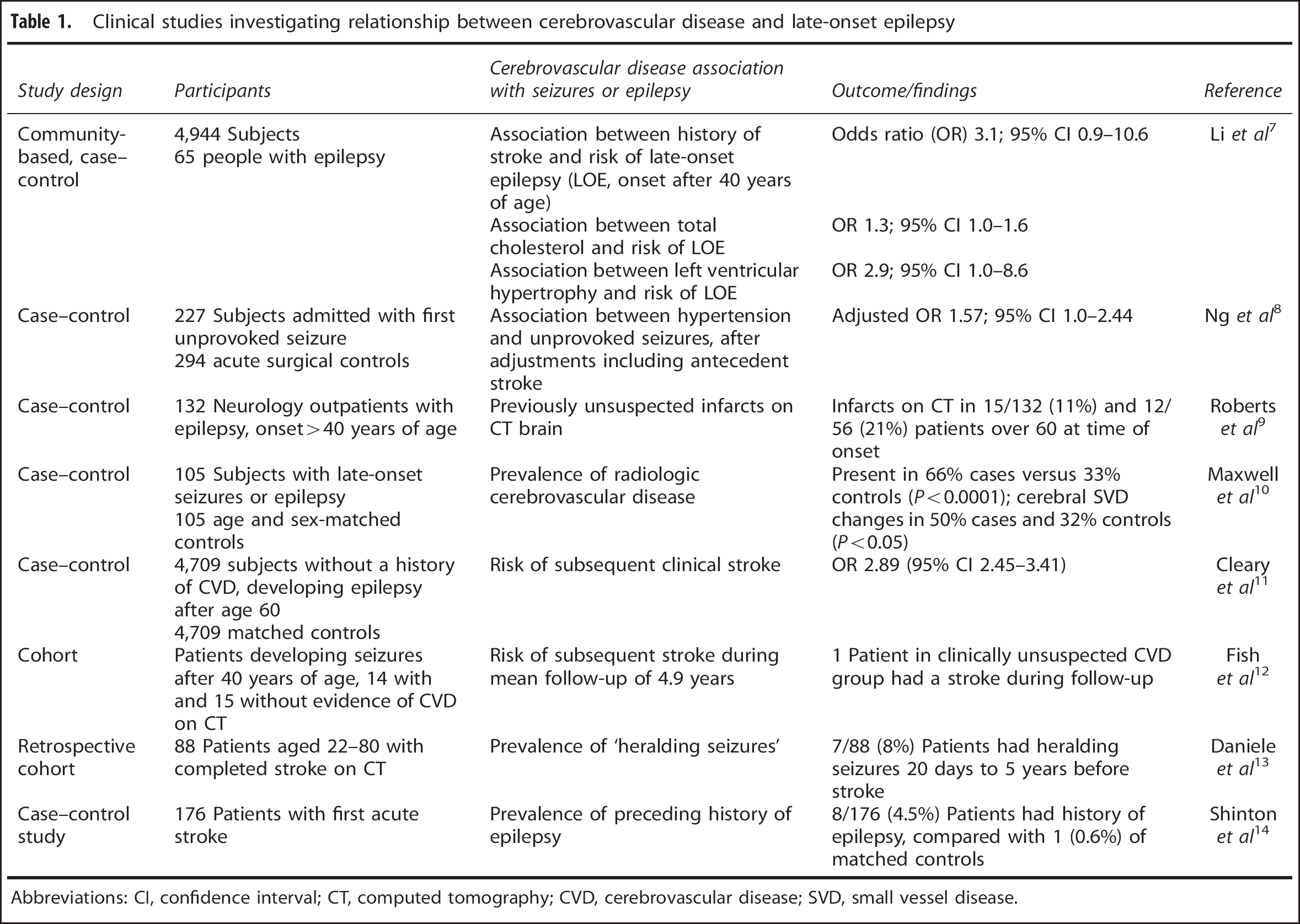
Abbreviations: CI, confidence interval; CT, computed tomography; CVD, cerebrovascular disease; SVD, small vessel disease.
This has implications for clinical practice in that patients presenting with LOE should have any other cerebrovascular risk factors addressed. Furthermore, research is needed to determine the prevalence of occult CVD in LOE, and to investigate whether addressing cerebrovascular risk factors in this cohort of patients can reduce the incidence of stroke.
ANTIEPILEPTIC DRUG TREATMENT AND VASCULAR RISK
Recently, attention has been drawn to the potential deleterious effects of certain AEDs on vascular risk profile. This concern probably relates most to enzyme inducing AEDs. Several commonly prescribed AEDs, including phenobarbitone, phenytoin, and carbamazepine, stimulate the synthesis of a number of monooxygenase and conjugating enzymes. This may tend to reduce the action of various drugs, including the widely used lipid-lowering HMG-CoA reductase inhibitors (statins). Induction of enzymes involved in metabolic pathways can also occur and may alter lipid profile. Thus, potential exists for interaction between these AEDs and vascular risk. 15
The risk of myocardial infarction, stroke, and death associated with epilepsy and its modification by treatment with AEDs has been investigated. 16 In a Danish cohort followed from 1997 to 2006, in patients without previous stroke, AED-treated epilepsy was associated with increased risk of myocardial infarction, stroke, and all-cause death. The hazard ratio for stroke (HR, 2.22; 95% CI, 2.09 to 2.36) did exceed those for myocardial infarction (HR, 1.09; 95% CI, 1.00 to 1.19) and all-cause death (HR, 1.92; 95% CI, 1.86 to 1.97) even in this cohort study of all citizens aged 10 or older. Another study investigated 160 adult patients receiving AED monotherapy, including two enzyme inducers (carbamazepine and phenytoin), an enzyme inhibitor (valproic acid) and a noninducer (lamotrigine), and 60 controls. 17 Long-term monotherapy with older generation AEDs, including carbamazepine, phenytoin, and valproic acid, caused significantly increased carotid intima medial thickness (a marker of atherosclerosis), while carbamazepine and phenytoin treatment was associated with alterations in cholesterol, homocysteine, folate, and high sensitivity C-reactive protein. Elevated high sensitivity C-reactive protein, reflecting chronic low-grade systemic inflammation, has been linked to vascular risk. 18 No such changes were seen in patients receiving long-term lamotrigine monotherapy. The relevance of changes in homocysteine metabolism in terms of vascular risk, the contribution of methylenetetrahydrofolate reductase polymorphisms and the place of vitamin B supplementation in the context of AED use to reduce homocysteine are also unclear. 19
These recent observations relating to AEDs raise important considerations relevant to the relationships between LOE, occult CVD, and stroke. However, currently only a proportion of patients diagnosed with LOE will be commenced on vascular secondary prevention medication. Thus, stroke risk may currently not be substantially affected by any reduction in action of statins potentially associated with enzyme inducing AEDs. Furthermore, given that the relative hazard of stroke after the onset of LOE was noted to be constant over time, 11 it seems unlikely that incident stroke risk in LOE is prominently related to the duration of AED exposure. In addition, as discussed above, radiologic CVD is known to be more prevalent in patients with LOE by comparison with age-matched controls before AED exposure.
Thus, the extent to which AED exposure may itself be causal in respect of CVD and stroke, either independently from any potential relationship between occult CVD and LOE, or in addition to occult CVD and LOE, is currently unknown. But in the context of LOE where an association with occult CVD is suspected (and perhaps in the wider epilepsy population), it may be preferable to consider treatment with modern nonenzyme inducing AEDs to minimize the potential for exacerbation of vascular risk.
OCCURRENCE OF SEIZURES WITH OTHER CEREBROVASCULAR DISEASE ENTITIES
Certain other CVD entities exist in which seizures and epilepsy may occur, or indeed may be part of the initial presentation. Cerebral amyloid angiopathy (CAA), which predominantly occurs in the elderly and most frequently manifests clinically as intracerebral hemorrhage, is probably underrecognized. It is associated with β-amyloid deposition in the adventitia and media of small and mid-sized arteries of the cerebral cortex and leptomeninges. It is recognized that the presentation in a subset of patients includes seizures, cognitive symptoms, predominantly white-matter hyperintensities on T2 or fluid-attenuated inversion recovery MRI, and inflammation of CAA-affected vessels. 20 It may also account for some cases of so-called ‘heraldic seizure’ where seizures, perhaps arising due to concurrent cerebral ischemia, precede stroke due to intracerebral hemorrhage. 21 Episodes of focal, transient, neurologic dysfunction potentially mimicking seizures can also be associated with focal cortical hemosiderin deposition potentially due to underlying CAA, likely to be increasingly identified with the use of gradient-echo MRI.22, 23
While CAA is a very specific entity and may only account for a minority of late-onset seizures overall, its association with cerebrovascular lesions in Alzheimer's disease or in mixed Alzheimer's disease/vascular dementia is likely to be extremely important, 24 although the mechanisms clearly require further investigation. Seizures have been reported in 7% of clinically probable Alzheimer's disease cases, and may accompany the onset of dementia, perhaps reflecting a shared pathogenesis. 25 Seizures can also occur in the context of posterior reversible encephalopathy syndrome, 26 reversible cerebral vasoconstriction syndrome, 27 and in the context of cerebral arteriovenous or cavernous malformations, and subarachnoid hemorrhage or their treatment. Other specific but rarer CVD entities potentially manifesting first with epileptic seizures include central nervous system vasculitis, 28 cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy 29 and mitochondrial encephalopathy with lactic acidosis and stroke-like episodes. 30
LATE-ONSET EPILEPSY AND OCCULT CEREBROVASCULAR DISEASE: POTENTIAL MECHANISMS
Mechanisms of epileptogenesis in the setting of any form of central nervous system injury actually remain relatively obscure. When otherwise clinically occult CVD is deemed the likely cause of LOE, radiologic markers of cSVD may well be found, with or without large vessel infarcts or evidence of prior intracerebral hemorrhage. But given that epilepsy typically derives from the cerebral cortex, how can this be reconciled with the distribution of occult cSVD being principally subcortical, and often in the absence of any obvious cortical abnormality? It is possible that abnormalities, due to CVD, causing structural or functional disruption of corticocortical or subcorticocortical circuits are not identified either on account of the choice of imaging modality or sequence (e.g., subtle lesions missed on CT, hemosiderin not identified in the absence of a gradient-echo MRI sequence), or because such abnormalities may be below the resolution of current conventional neuroimaging techniques (e.g., microinfarcts, disrupted white-matter integrity). Some of these abnormalities are only currently identifiable histopathologically. 31 Furthermore, even widely accepted MRI ‘visible’ expressions of cSVD such as white-matter hyperintensities show relatively weak clinicoradiologic association, partly due to the heterogeneous pathologic substrates of white-matter hyperintensities. More recently, the spectrum of ‘visible’ cSVD markers has been expanding, and is considered to include enlarged perivascular spaces 32 and reduced brain volume, at least as a surrogate marker. 33 Previous studies reporting radiologic CVD in LOE have largely not included more recently recognized markers of CVD.
Neurovascular Unit Integrity, Occult Cerebrovascular Disease, and Late-Onset Epilepsy
The structural and functional integrity of the central nervous system depends on coupling between neural activity and cerebral blood flow (CBF), and regulation of transport across the blood–brain barrier (BBB). These two critical processes rely on the coordinated activity of a ‘neurovascular unit’ comprising the microvascular endothelium, and neuronal and glial cell elements in close proximity. In the normal healthy state, the increase in CBF produced by brain activity, or functional hyperemia, is an example of the close interaction between neurons, glia, and vascular cells. Neurovascular coupling can be defined as this relationship between the vascular response, that is, change in CBF and the associated change in regional cerebral oxygen metabolism in response to neural activation, which can be measured with the MRI technique of arterial spin labelling to allow estimation of CBF and the blood-oxygenation-level-dependent signal. 34 We have recently explored the concept of the disordered neurovascular unit contributing to LOE. 35
Disordered Cerebral Metabolism and Perfusion
Patients with leukoaraiosis and LOE had significantly reduced regional cerebral oxygen metabolism in all cortices (frontal, temporal, parietal, and occipital) compared with controls, indicating hypometabolism in these regions (P<0.05). 36 These patients also had significantly reduced regional CBF (rCBF) in all cortices compared with controls (P<0.05). 36 However, whether such reductions in rCBF and regional cerebral oxygen metabolism indicate an underlying degenerative or vascular process, or a combination of the two, especially in the elderly, is difficult to ascertain. Degenerating cortical neurons have a diminished oxygen demand, resulting in hypometabolism and reduced blood flow, while damage to blood vessels may reduce blood flow thereby reducing oxygen delivery and inducing hypometabolism. By contrast, increases in rCBF are described during epileptic seizures. A study of 40 patients with complex partial seizures using 133Xe CT imaging at rest and during a light stimulation procedure found a significant increase in rCBF in the region of the suspected epileptic focus in nonlesional patients. 37 Another study using perfusion CT to measure rCBF with EEG correlation found increased rCBF during subtle status epilepticus but regional hypoperfusion in postictal patients. 38 Middle cerebral artery blood flow velocity assessed with transcranial Doppler ultrasonography during simultaneous EEG recording reveals asymmetric perfusion increases closely related to onset and cessation of EEG seizure activity during simple partial motor seizures. 39 Seizures accompanying the onset of acute ischemic stroke and in relation to reperfusion/hyperperfusion associated with thrombolytic treatment are intriguing. 40 Although seizures have been reported with thrombolytic treatment to herald a dramatic neurologic recovery, 41 in another recent study, early-onset seizures occurred mainly in patients with severe neurologic impairment at stroke onset and severe remaining disability, arguably making it less likely that seizures were attributable to cerebral hyperperfusion. 42 The relationship between seizures and cerebral perfusion is undoubtedly complex and incompletely understood.
Blood–Brain Barrier Dysfunction
The microvasculature is abnormal in cSVD, with loss of smooth muscle cells, vessel wall thickening, luminal narrowing, and increased vessel stiffness. These changes may contribute to the attenuation of vasomotor reactivity in response to hypercapnia or acetazolamide seen in cSVD, 43 and consequently lead to an impairment of neurovascular coupling. Such impairment of neurovascular coupling appears to be specific for cSVD. 44 Recently, BBB breakdown has received considerable attention as a cause of cSVD. Blood–brain barrier breakdown may lead to ischemia, leukoaraiosis, and lacunar infarcts. 45 Mild chronic hypertension may damage cerebrovascular endothelium in small vessels leading to thickening of the arterial wall and narrowing of the lumen, resulting in ischemia (leukoaraiosis if ischemia occurs in white matter) or infarction. Alternatively, the damage to the arterial wall may progress, causing disintegration and a leak of blood. 45 Blood–brain barrier leakage may be visualized as an increase in vessel permeability using dynamic contrast enhanced MRI 46 or as small areas of low signal on T2∗-weighted magnetic resonance images that may be deposits of hemosiderin, a breakdown product of hemoglobin, usually indicating a brain microbleed. 47 Blood–brain barrier dysfunction is certainly described as a consequence of seizures, but given that BBB opening is also recognized to promote seizures in humans, and the association of BBB dysfunction with CVD, this is likely to represent an area worthy of further investigation in the clinical context. 48
Cerebrovascular Disease, Inflammation and Epileptogenesis
There is increasing evidence that inflammation is an integral part of diseased hyperexcitable brain tissue from which spontaneous and recurrent seizures may originate. The relative contributions of specific proinflammatory pathways, such as interleukin-1/Toll-like receptor signalling, are currently unclear, but the case for brain inflammation and associated BBB damage contributing to epileptogenesis is becoming stronger. 49 Cerebrovascular determinants of epilepsy, including the role of inflammation and BBB dysfunction, have recently been discussed elsewhere. 48 These authors posited that the BBB dysfunction sits at a crossroad of multiple pathophysiologic pathways involving changes in brain milieu, altered neuroglial physiology, brain inflammation, leukocyte–endothelial interactions, disordered angiogenesis, and hemodynamic changes leading to energy mismatch. Potential novel treatment strategies may even include therapeutics promoting BBB repair. It is however acknowledged that considerable gaps in knowledge hamper our understanding of this area. Figure 3 depicts possible interrelationships leading to epileptogenesis.
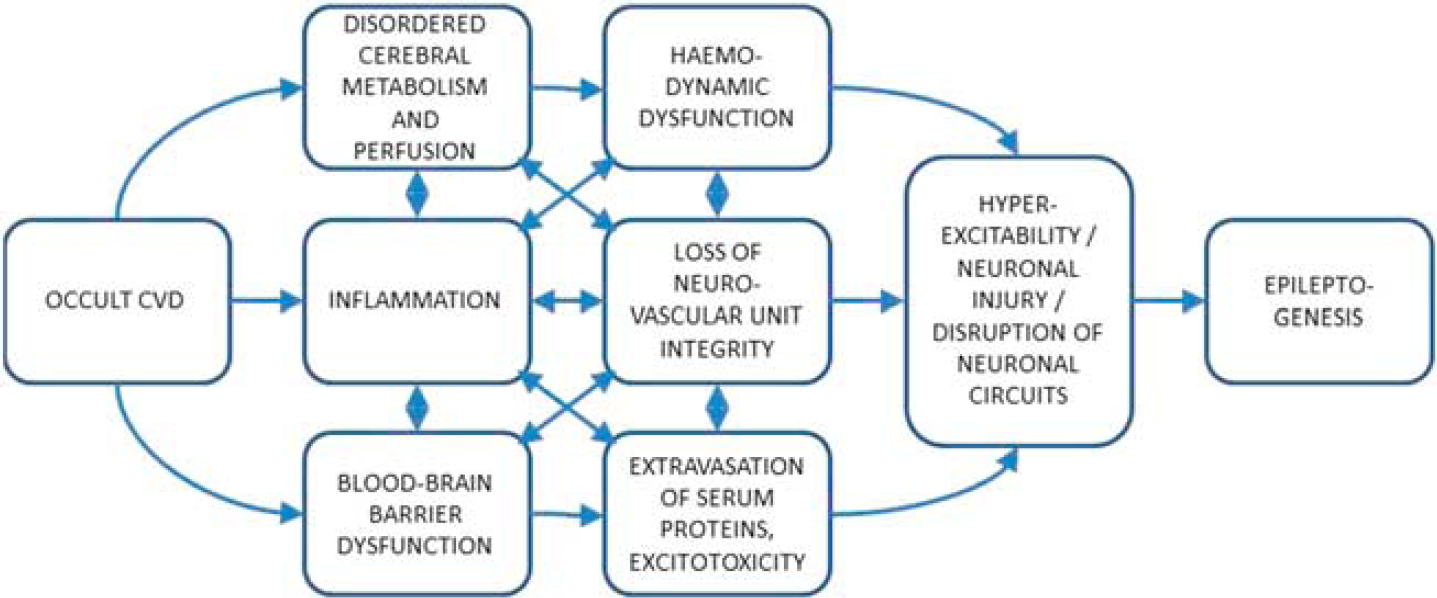
Schematic outlining possible interrelationships in the pathophysiology of epileptogenesis associated with occult cerebrovascular disease (CVD).
FUTURE DIRECTIONS
Considering the evidence relating to elevated stroke risk after LOE, it is likely that potential exists for improved cerebrovascular risk modification in this population. Currently, the evidence relating to the burden of occult CVD in patients with LOE is weak, and our understanding of potential underlying mechanisms is limited. Further studies are needed, employing current CVD imaging markers. The potential influence of occult CVD upon the structural/functional integrity of corticocortical and subcorticocortical circuits and hence epileptogenesis requires greater elucidation. Identification of suitable imaging markers for occult CVD-related epilepsy will likely be valuable for future trials.
Footnotes
The authors declare no conflict of interest.
