Abstract
Interruptions in cerebral blood flow may lead to devastating neural outcomes. Magnetic resonance has a central role in diagnosing and monitoring these insufficiencies, as well as in understanding their underlying metabolic consequences. Magnetic resonance spectroscopy (MRS) in particular can probe ischemia via the signatures of endogenous metabolites including lactic acid (Lac), N-acetylaspartate, creatine (Cre), and cholines. Typically, MRS reports on these metabolites' concentrations. This study focuses on establishing the potential occurrence of in vivo longitudinal relaxation enhancement (LRE) effects—a phenomenon involving a reduction of the apparent T1 with selective bandwidth excitations— in a rat stroke model at 21.1 T. Statistically significant reductions in Cre's apparent T1s were observed at all the examined post-ischemia time points for both ipsi- and contralateral hemispheres, thereby establishing the existence of LREs for this metabolite in vivo. Ischemia-dependent LRE trends were also noted for Lac in the ipsilateral hemisphere only 24 hours after ischemia. Metabolic T1s were also found to vary significantly as a function of post-stroke recovery time, with the most remarkable and rapid changes observed for Lac T1s. The potential of such measurements to understand stroke at a molecular level and assist in its diagnosis, is discussed.
Keywords
INTRODUCTION
The highly complex functions of the brain depend on an intricate coupling between neural activity, metabolism, and the continuous blood supply needed to meet energetic demands. 1 When cerebral blood flow is interrupted, an ‘ischemic cascade’ is triggered1,2 whereby faltering energy supplies alter cellular bioenergetics and shift ion populations between extra- and intracellular spaces. This, in turn, induces excitotoxicity, promoting further tissue injuries. As a result, structural changes to the cellular microstructure will follow, with swelling occurring owing to the osmotic modulations, and ultimately cell death ensues. 3 The overall consequence is a potentially detrimental functional impairment, with often devastating neurologic outcomes.1–3 Magnetic resonance imaging (MRI) plays a central role in characterizing these central nervous system (CNS) events in general, while providing an early diagnosis of ischemia in particular.4–6 Most notably, diffusion- and perfusion weighted MRI of water can be used for identifying the onset of stroke, and help define the treatment-oriented windows of opportunity. Magnetic resonance imaging can further assist in detecting the potentially salvageable penumbra region, and provide quantifiable information about the structural and functional alterations occurring after ischemia.4–8 Functional MRI can be used to characterize the outcome of a stroke, as well as to follow neural network reorganizations compensating for stroke-induced functional impairments.9,10
Magnetic resonance spectroscopy (MRS) is an alternative approach that, although closely related to MRI, focuses on resolving the localized spectral metabolic signatures imparted by endogenous CNS metabolites rather than reflecting the spatial distribution of water. 11 Numerous small molecules can be routinely observed by 1H MRS including bioenergetics-related metabolites (creatines (Cre), lactic acid (Lac)), osmolytes (N-acetylaspartate, (NAA)), metabolites participating in membrane synthesis (cholines, (Cho)), and neurotransmitters (glutamate, γ-aminobutyric acid).11,12 Owing to their participation in underlying metabolism and more selective compartmentation, the relative concentrations of these metabolites— as conventionally quantified from the peak intensities and line shapes—are often sought by MRS scans seeking to evaluate stroke. For example, NAA is considered a neuronal viability biomarker, 13 whereas both Lac and NAA levels correlate with stroke severity and functional outcome. 14 1H MRS also can be combined with water-based diffusion MRI to improve the clinical prediction of a stroke's repercussions; 15 diffusion16,17 and 1H MRS studies based on spin-spin (T2) and spin-lattice (T1) relaxation times have been proposed as useful complements to reveal more elaborate interactions of metabolites within the host tissues.18–24
Another potential source of insight regarding the interaction between metabolites and their host tissues, could arise from investigating longitudinal relaxation enhancement (LRE) effects. Longitudinal relaxation enhancement is a phenomenon exploited in biomolecular NMR, for examining amide groups and enhancing signal-to-noise ratios per unit time.25,26 Longitudinal relaxation enhancement involves a reduction in a resonance's apparent T1 on switching from a broadband excitation with active water suppression to a spectrally selective excitation targeting only the resonances of interest. By leaving the main solvent reservoir as well as many of the macromolecular protons magnetically unperturbed, a T1 shortening can be mediated both by chemical exchange phenomena with water, as well as via cross-relaxation effects between the targeted and unperturbed proton reservoirs. Although small molecules (such as brain metabolites) do not generally exhibit LRE effects when dissolved in solutions, 26 we recently found that on executing spectrally selective excitations in ex vivo brains, the non-labile methyl resonances of Lac, Cre, and Cho—as well as numerous other labile 1H sites—exhibited significant LREs at 9.4T. 27 These first observations of LRE probably reflect a dynamic binding of these metabolites to macromolecules, facilitating effective cross-relaxation with bound macromolecular protons, or water-related exchange effects in proximate sites of the targeted metabolite. Previous studies also noted a magnetic coupling between the water and Cre methyl resonances in vivo,28,29 as well as magnetization transfer effects between water and Lac's methyl resonance. 30 LREs, as quantified from differences in apparent T1s on changing from a broadband, watersuppressed excitation to a selective excitation that does not affect the water magnetization, could thus potentially serve as a new type of metabolic reporter.
To validate such premise, the present study aims to (i) determine whether LRE effects previously observed ex vivo 27 also affect metabolic resonances in vivo; and (ii) examine whether, given the putative influence that binding processes have on these effects, LREs are affected by pathologic processes in brain tissues. To achieve such aims, this study harnesses the high quality 1H MRS spectra that can be obtained under optimized ultrahigh field conditions, and investigates the time dependencies that both metabolic T1s and LREs exhibit after ischemia. Localized LRE MRS sequences addressing solely the methyl resonances of Lac, Cre, Cho, and NAA were thus implemented, and 1H T1 behaviors in the presence and absence of water suppression were compared in a rat model subject to a transient occlusion of the middle cerebral artery (MCA). The evolution of these parameters was followed between 3 hours and 1 week after ischemia, using the sensitivity and resolution advantages afforded by the 21.1 T preclinical MRI situated at the US National High Magnetic Field Laboratory. This unique magnet permitted the in vivo detection of unequivocal LRE effects for Cre, as well as substantial changes in other metabolic T1s after ischemia and as a function of stroke recovery. The biophysical origin of these changes and their potential as novel disease biomarkers are discussed.
MATERIALS AND METHODS
All experiments were approved by the Florida State University Animal Care and Use Committee (ACUC protocol#1038). The FSU Animal Research Program has been accredited by the Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC), International. Florida State University is registered as a research facility with the United States Department of Agriculture (Registration#58-R-0001) and has an Animal Welfare Assurance number (#A3854-01) on file with the US Public Health Service. Under the direction of a veterinarian who is certified as a specialist in laboratory animal medicine by the American College of Laboratory Animal Medicine, all animal procedures were undertaken according to these regulatory bodies and AAALAC guidelines, and every effort was taken to minimize animal suffering.
Middle Cerebral Artery Occlusion
All surgical procedures in this study were performed under aseptic conditions. The MCAs of Juvenile Sprague-Dawley rats were occluded for 1.5 hours. Briefly, anesthesia was induced using 5% isoflurane in 100% medical grade O2 and further maintained with 2% isoflurane. A filament (Doccol Corp., Sharon, MA, USA) was guided 1.9 cm through the external carotid artery, whereupon it blocked blood flow proximal to the MCA. The MCA was occluded for 1.5 hours as the animal recovered in its cage. At the end of this period, the animals was reanesthetized, and the filament removed to allow for reperfusion; Stroke was induced in N=6 rats, with no mortalities throughout the entire duration of the experiments.
21.1 T Magnetic Resonance Imaging System and Experimental Timeline
All experiments in this study were performed at the NHMFL using the 21.1 T magnet, operating at 900 MHz for protons. The system is equipped with a Bruker Avance III console (Bruker BioSpin, Billerica, MA, USA) and a RRI (Resonance Research, Billerica, MA, USA) gradient system capable of producing up to 600 mT/m in all directions. Excitations and signal acquisitions were performed using a custom-built quadrature dual coil designed for optimal operation on the rat head. Anesthesia was induced using 5% isoflurane in 100% medical grade O2 and subsequently maintained with 2% isoflurane. Respiratory rates were monitored throughout the experiments, which were performed with respiratory gating. Animals were scanned at 3 hours, 24 hours, and 1 week after ischemia.
1H Longitudinal Relaxation Enhancement Magnetic Resonance Spectroscopy
The localized LRE sequence used in this study is shown in Figure 1. Its main features are spectrally selective excitation and refocusing pulses that avoid the bulk water magnetization, 27 thanks to a band-selective design based on the Shinar-LeRoux algorithm. 31 The 8-ms excitation pulse was modulated to target solely the four methyl resonances of Lac, NAA, Cre, and Cho, with ≤0.15 p.p.m. bandwidths for each; this design ensures in turn a minimal perturbation of the pools of aqueous and macromolecular protons that could partake of LRE effects, while avoiding some of the associated J-coupling evolution effects (for instance, from Lactate's methyl coupling partner), which could potentially result in spectral distortions. Under these conditions, the pulse nutation imparted on the water resonance was <0.5°, leaving 99.98% of water protons unperturbed. Because resonances during this relatively long polychromatic excitation pulse were subject to a chemical shift-induced phase evolution, they were refocused subsequently using a pair of frequency-swept adiabatic pulses (each of them 4 ms) that also avoided water perturbations. Spectral selectivity was complemented by a spatial localization imparted by a 3D-LASER module, utilizing three pairs of spatially selective adiabatic frequency-swept refocusing pulses of 5ms each. 32 To facilitate the quantification of the LRE, a comparison between the spectrally selective sequence was performed against a water-suppressed counterpart, whereby the initial excitation was preceded by a 228-ms Chemical Shift Selective water suppression module 33 (Figure 1).
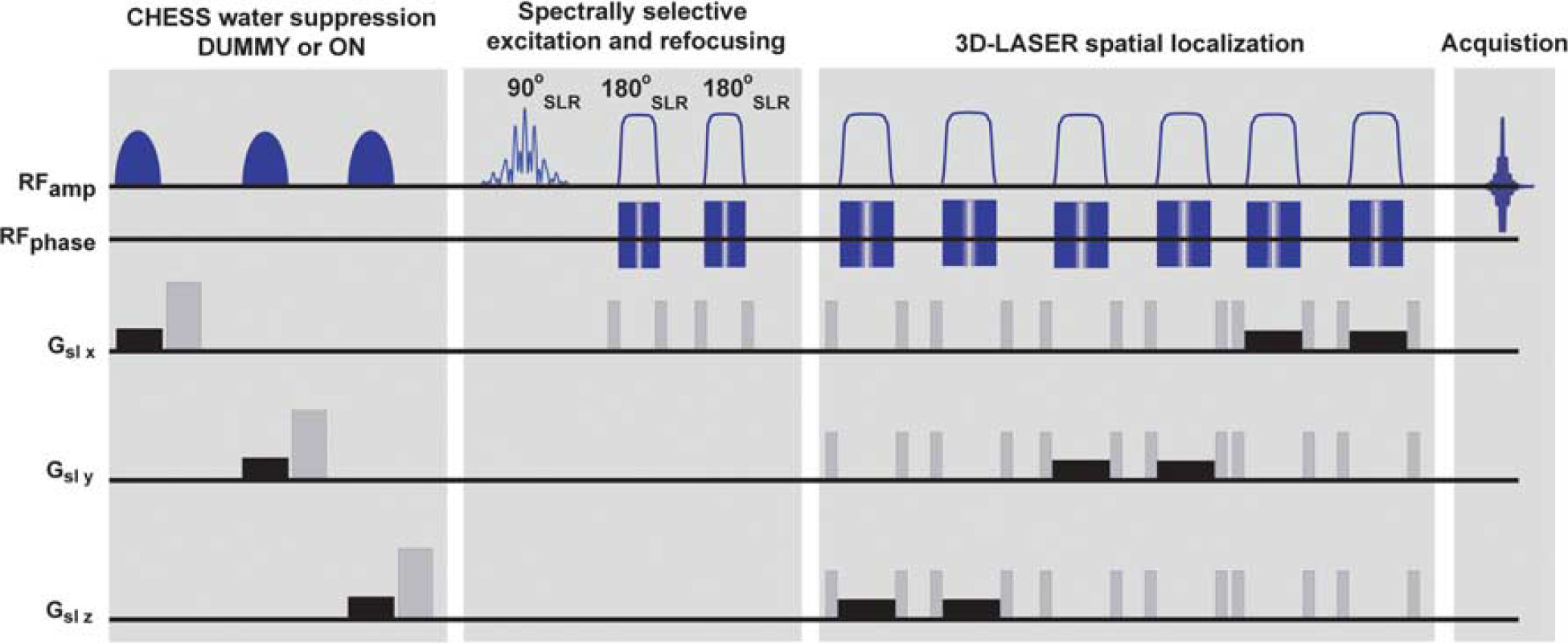
Localized longitudinal relaxation enhancement (LRE) magnetic resonance spectroscopy sequence used to probe apparent metabolic T1s. The sequence incorporates a spectrally selective module exciting and refocusing only four resonances of interest: lactic acid, N-acetylaspartate, creatine, and cholines. The majority of remaining spins in the sample remain unperturbed, thereby facilitating the quantification of LRE effects. Magnetization thus excited is then localized using a 3D-LASER module. 32 To compare water suppressed and nonwater suppressed preparations, a chemical shift selective (CHESS) module is played out either in DUMMY mode (i.e., with CHESS pulses off and hence no active form of water suppression), or it is turned ON to observe water-suppressed spectra. A series of eight dummy scans preceded the entire sequence to achieve steady-state magnetization. Crusher gradients are shown in gray, whereas slice-selective gradients are shown in black.
Magnetic Resonance Imaging Scouts and Voxel Localization
Before the MRS experiments, T2-weighted images of the rat brain were acquired at all the stroke recovery time points. These MRI scans used a spin-echo Rapid Acquisition with Relaxation Enhancement (RARE) sequence with TR/TE = 5,000/35 ms, RARE factor 8, spatial resolution 230 × 230 × 1000 (μm) 3 , and 12 slices. The stroke-affected region was determined from these images, and a (4.8-mm) 3 voxel in the stroke ipsilateral hemisphere was selected. A second, symmetrically placed voxel of the same size was selected in the contralateral hemisphere in a subsequent experiment. The accurate spatial localization of these sharpedged voxels was confirmed using MRI by concatenating a LASER module identical to that employed in the MRS sequences with a nonselective spin echo imaging sequence. These three-dimensional T2-weighted images were also used to align the location of the targeted voxels across the different time points of this longitudinal study, and care was taken to ensure the robust placement of these voxels in identical positions throughout the measurements. Under the LASER localization conditions that were used, we estimate the maximum chemical shift-driven displacement between the voxels associated to the different metabolites, to span under 1 mm. Each voxel was selectively shimmed before T1 measurements using the broadband-excited water signal, whose typical line widths were 25 to 40 Hz.
In Vivo T1 Metabolic Measurements
After ipsi- and contralateral voxels were selected, apparent metabolic T1s were measured for the rats by a progressive saturation approach
22
. This entailed eight dummy scans and 48 signal averages at a constant echo time of 84 ms, with repetition times incremented from 0.7 to 7.1 s in 15 steps. The order of the repetition times was randomized to avoid any consistency-related bias. For each voxel (ipsi- and contralateral), two sets of apparent T1 measurements were performed: first, with a LRE sequence leaving all water spins unperturbed (i.e., the chemical shift selective module operated in DUMMY mode), and second, with the LRE sequence preceded by active water excitation and crushing via the chemical shift selective module. Progressive saturation data were fitted to an exponential recovery curve
RESULTS
Figure 2A shows representative T2-weighted images from a strokeaffected rat 24 hours after ischemia at 21.1 T. Strong hyperintense contrast is evidentproximal to the MCA, clearly delineating the stroke. A representative localization of the ipsi- and contralateral voxels chosen in this study is shown in Figure 2B, overlaid on a dimmed T2-weighted image of the brain. Notice the sharp spatial localization profiles arising from the LASER module, with magnetization confined to the stroke-affected and healthy regions in the ipsi- and contralateral hemispheres, respectively. Along the third (not explicitly plotted) dimension, these voxels spanned ca. 4.8 mm, corresponding approximately to the slices outlined in a yellow frame in Figure 2A; thus, it was ensured that the ipsilateral voxel contained mostly impaired brain tissue, with minimal partial volume effects from undamaged tissue. The contralateral voxel was symmetrically placed using similar criteria, as shown in Figure 2B. Having achieved the desired spatial specificity, the spectral profiles arising from the addressed voxels was examined via the localized 1H MRS sequence in Figure 1. Representative one-dimensional traces of these spectra are shown in Figure 2B. Features worth highlighting in these data are: the clean, undistorted baselines obtained from the selective echoed excitations; the high spectral specificity reached, with only the four resonances of interest excited (along with another hitherto unassigned peak resonating next to lactate); the nearly complete absence of the water resonance despite the absence of any active form of water suppression; and an excellent signal-to-noise ratios that for NAA in the contralateral hemisphere exceeds 250 in only 48 scans.
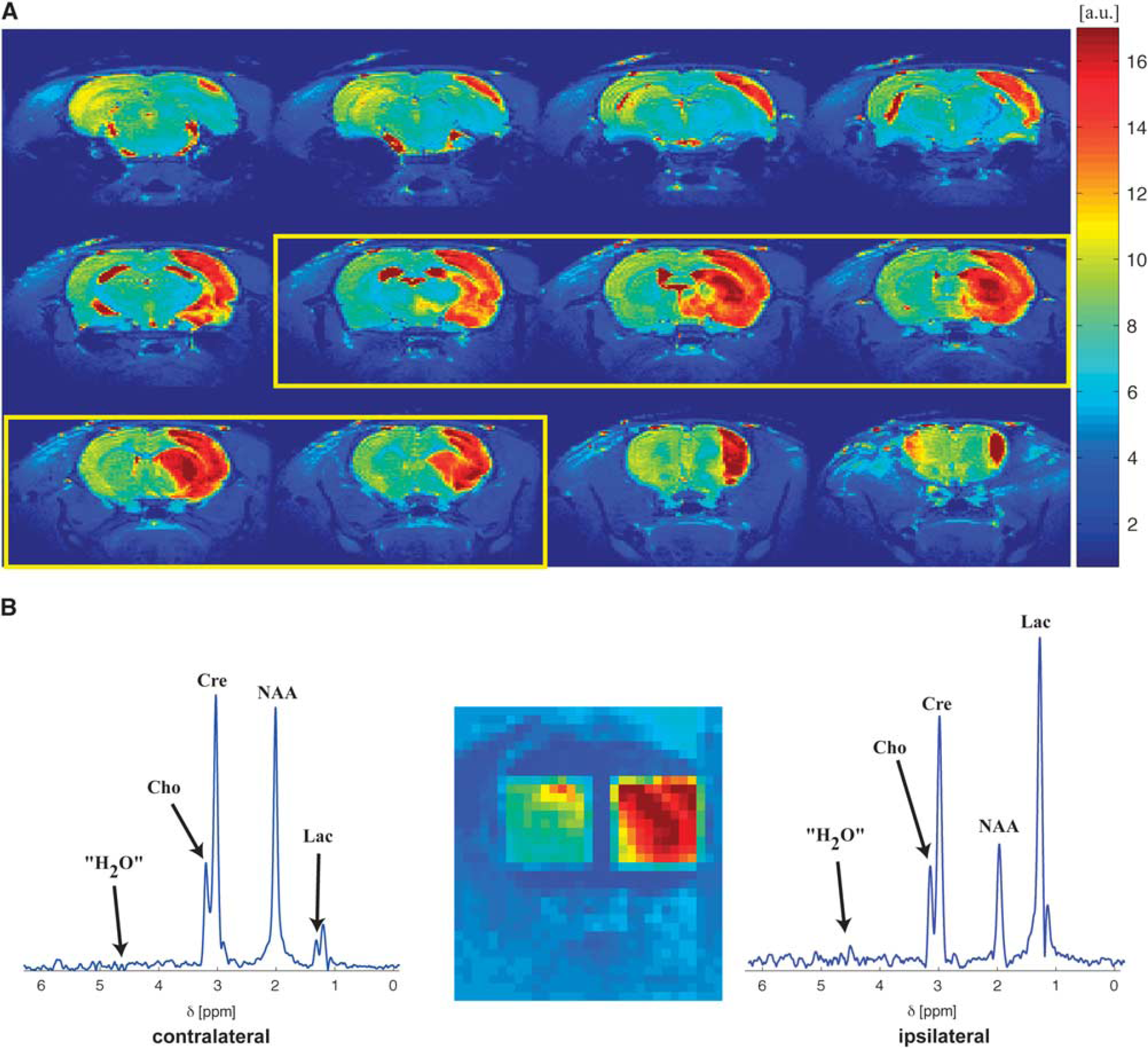
Voxel localization and ensuing 1H magentic resonance spectroscopy (MRS) 21.1 T spectra representative of the data collected in this study. (
Given the high quality of these MRS spectra, robust apparent T1 measurements in the presence and absence of water suppression were performed. Figure 3 shows characteristic data from progressive saturation experiments, performed in vivo on the voxels shown in Figure 2B. In all these experiments, a monotonic magnetization recovery as a function of TR could be clearly observed, with little or no differences in the residual water resonance regardless of whether water suppression was or was not used. The right-hand graphs in Figure 3 show representative examples of fittings performed for data arising from the Cre peak, demonstrating the robustness of these apparent T1-value quantification. The good quality of these data facilitated repetition of these measurements over a cohort of animals, and for several post-ischemia recovery times. The apparent T1s obtained in non-water-suppressed and from actively water-suppressed experiments were compared and used to deduce the existence of significant LRE effects in both hemispheres.
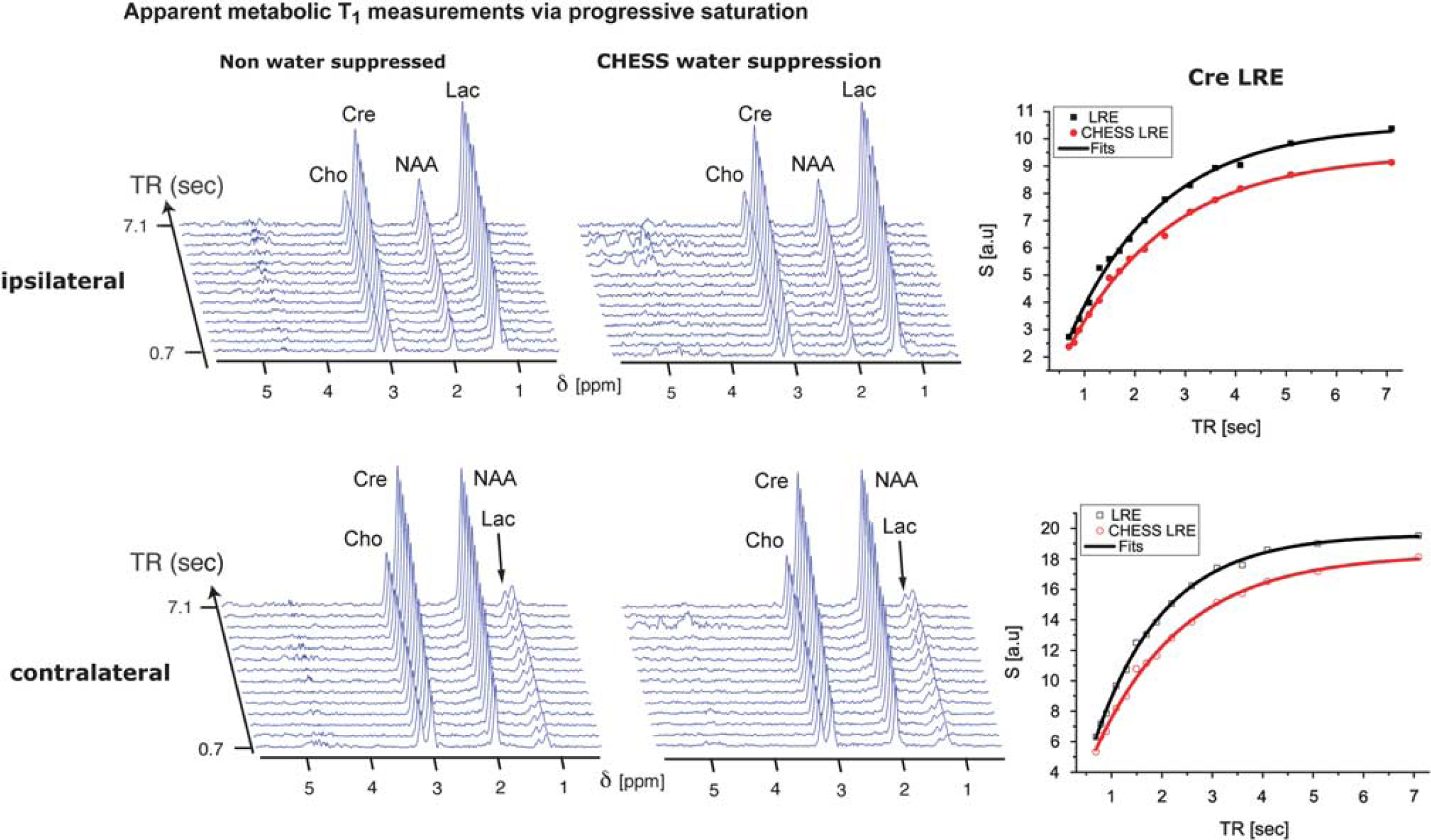
Representative data sets of apparent metabolic T1 measurements obtained 24hours post ischemia using a progressive saturation (i.e., a variable TR) sequence, arising from the ipsilateral (top panel) and contralateral (bottom panel) hemispheres. Both the non-watersuppressed and water-suppressed one-dimensional traces arose from signal averaging 48 scans over the voxels delineated in Figure 2. Spectra such as these were used for estimating the apparent T1s in N=6 rats at three different post-ischemic time recovery points. A single T1-recovery data set took less than 30 minutes to acquire. The right-hand graphs show the fitting of these data for the Cre resonance according to
Figure 4 summarizes the apparent T1s obtained for the four metabolites targeted in these studies. These measurements reveal a series of new, metabolite-specific and statistically significant results. First, there is convincing evidence for the presence of LRE effects for Cre at all time points, and for both ipsi- and contralateral voxels. At 3 hours after ischemia and in the ipsilateral hemisphere for instance, the apparent T1 of Cre increased from 1.5 ± 0.2 to 1.9 ± 0.1 s on performing chemical shift selective water suppression. These effects were statistically significant (P< 0.0051, Fisher post hoc test), and marked a 27% increase in apparent T1 on applying active water suppression, which strongly perturbs the water resonance from its Mz equilibrium state. For later time points (24 hours and 1 week after ischemia), ipsilateral Cre LRE was still statistically significant with 16% increases (P< 0.0048 and P<0.04, respectively). However, these LREs did not evidence a statistically significant temporal variation between the 3 hours, 24 hours, and 1 week post-ischemia time points. In the contralateral hemisphere, Cre's LRE was also highly consistent and statistically significant (P<0.006) across the different recovery stages, with 21%, 19%, and 21% reductions in the apparent T1s respectively, on switching from water suppressed to non-water suppressed excitation.
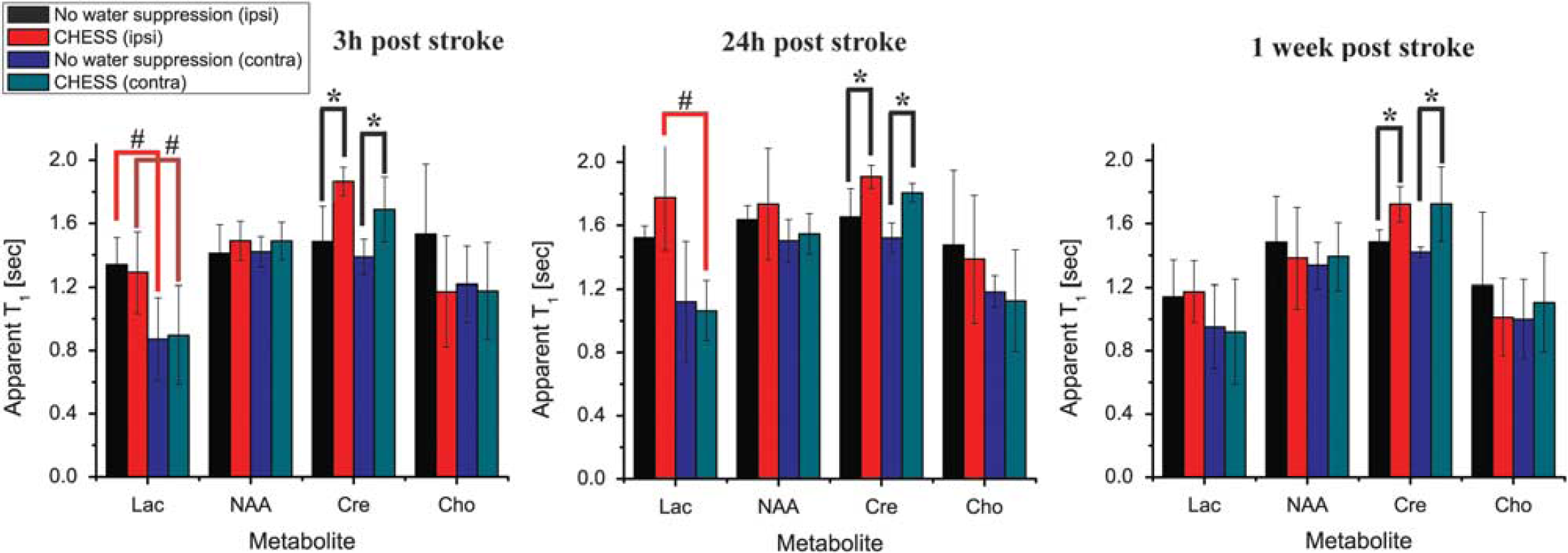
Quantification of the apparent metabolic T1s extracted from N=6 rats monitored 3 hours, 24 hours, and 1 week after ischemia. Graphs quantify the apparent T1s observed for voxels in the ipsi- and contralateral hemispheres obtained at each time point, without and with the use of chemical shift selective water suppression. Statistically significant longitudinal relaxation enhancement (LRE) is detected for creatine (Cre) for all time points in both ipsi- and contralateral hemispheres; lactic acid (Lac) shows a LRE trend only 24 hours post ischemia; neither choline (Cho) nor N-acetylaspartate (NAA) exhibit definite LRE effects. Symbols represent as follows: *P<0.05 compared with the other method in the same hemisphere; #P<0.05 compared with the opposite hemisphere with the same method.
Whereas Cre was the only metabolite showing such consistent LRE effects, a trend was also exhibited by the Lac resonance in the ipsilateral hemisphere: At the 24 hours time point, Lac's apparent T1 increased from 1.51 ± 0.07 to 1.8 ± 0.3 s on suppressing the water. This LRE was observed in the ipsilateral hemisphere at the 24 hours time point; at 3 hours or 1 week after ischemia LRE effects were not detected, nor were they observed in the contralateral hemisphere. Likewise, the NAA and Cho resonances exhibited no discernible LRE effects at any time point, and their apparent T1s showed no statistically significant dependencies on their mode of excitation for either of the analyzed voxels.
It is also instructive to follow the temporal evolutions of apparent metabolic T1s after the ischemia. Figure 5 summarizes these T1 variations as a function of the post-stroke time elapsed, revealing some common traits but also clear distinctions between the metabolites. The apparent T1 of Lac responds most dramatically to the onset of ischemia, with a 450% increase within only 3 hours. By contrast, NAA, Cre, and Cho show no significant modulations at this early time point. At 24 hours, the Lac-apparent T1 further increases, as do the apparent T1s of Cre and NAA, which, as mentioned above, were prolonged in a statistically significant fashion. These increases, however, are transient. At the latest time point (1 week after ischemia), NAA, and Cre T1s returned to baseline levels, exhibiting no statistically significant differences compared with their contralateral counterparts. Interestingly, although reduced compared with the 24-hour post-ischemia time point, Lac's T1 still has not reverted back to their original baseline even at this last time point. Although on average the Cho peak also exhibited a similar dynamic T1 history, its effects were statistically less reliable (Figure 5).
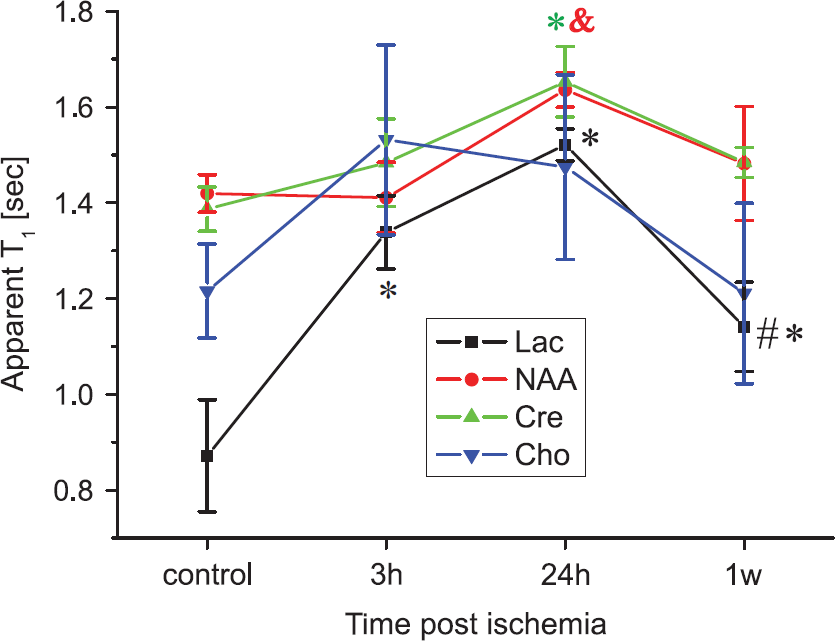
Apparent metabolic T1 variations at different time points after ischemia revealed by progressive saturation longitudinal relaxation enhancement magnetic resonance spectroscopy. Symbols represent as follows: *P<0.05 compared with contralateral; &P<0.05 compared with 3 hours; #P<0.05, compared with 24 hours. Error bars represent the s.e.m.
DISCUSSION
Magnetic resonance spectroscopy can be highly informative, conveying the status of numerous endogenous metabolites within the healthy and diseased tissues.11,12,34–36 Most MRS measurements report on ratiometric metabolic levels, revealing relative depletions or elevations of certain metabolites as markers of the underlying physiologic processes. 35 Within the context of CNS diseases, the most notable biomarkers are arguably the NAA and Lac peak intensities; depletions in the former are thought to reflect impairments in neuronal viability,13,35 and elevations in the latter reflect altered bioenergetics and compromised metabolism.5,14,19 Given a sufficiently good sensitivity, MRS can complement this in vivo quantitative insight with more elaborate sources of information: diffusion-weighted MRS, for instance, can potentially convey cellular-specific information on the tissue's micro-architecture and its response to an ischemic insult,16,37 and relaxation rates22–24 can inform about the interactions between metabolites and their environment. Despite this potential, relaxation-based MRS experiments have been rather scarce in the context of disease, especially in animal models,12,18,24 probably owing to the low sensitivity associated to such measurements. A major goal of this study was to exploit the excellent sensitivity that ultrahigh field operation and spectrally selective sequences can endow to 1H MRS, to investigate T1 relaxation and the potential existence of LRE effects for in vivo models of ischemia. In this regard, it should be stressed that LRE provides a different contrast from standard, broadband T1 measurements, as it measures the changes in apparent T1 with and without cross-relaxation contributions from other spins.25,26
The existence of such LRE effects—that are widely known to influence labile amide but also side chain groups in biomolecular NMR 38 —for the non-labile methyl resonances of metabolites in tissues, can be rationalized by a number of complementary mechanisms. One rests on a process whereby free metabolites exchange with a significant—even if spectrally invisible—population that effectively possesses longer correlation times owing to association with macromolecules; this in turn would affect the relaxation times of the observable metabolic signals differentially, depending on whether these potential cross-relaxation sources are excited or not. Similarly, concurrent macromolecular association of the metabolites and of water molecules could lead to mutual cross-relaxation effects, which do not arise when the metabolites are simply dissolved in solutions. Either of these effects could lead to apparent metabolic relaxation time measurements that depend on whether the spectral excitations are broadband or selective, thereby highlighting the connection between the observed LREs, and the interactions between metabolites and their environment.
A major goal of this study was to investigate whether metabolic LREs could exist in vivo. Out of the four metabolic peaks that were targeted, statistically significant in vivo LREs were documented for the Cre resonance at 21.1 T; this was apparent in both the ipsi- and contralateral hemispheres of stroke-affected rats, for all recovery times examined after ischemia. These LREs are consistent with previous in vivo reports of ‘magnetic couplings’ between water and the Cre methyl resonance:28,29 qualitative observations of signal differences in the Cre resonance on water inversion, can now be quantified and explained via an effective modulation of the resonance's apparent T1. As direct proton exchanges between the Cre methyl group and water is impossible, and as LREs are not observed in aqueous Cre solutions, Cre's in vivo LRE mechanism could arise from either an intermolecular H2OCre crossrelaxation facilitated as described above; or from rapid exchanges between the water and labile protons in the Cre carbamimidoyl group that are affected by the tissue, and which can impact the methyl proton's relaxation via intramolecular Overhauser effects. A similar source of potential LRE effects could be postulated for the overlapping phosphocreatine peak, but based on additional exchanges between water and the phosphate hydroxyl groups. Further studies of these potential mechanisms are required to discern the exact mechanism leading to LREs in vivo.
Despite this clear observation of Cre LRE in vivo, our experiments also suggest that these effects are not significantly altered by the onset of ischemia: the magnitudes of Cre's LREs were statistically identical for ipsi- and contralateral hemispheres at 21.1 T, and did not change as a function of recovery time. Therefore, it appears that at 21.1 T Cre LREs are not good candidates as stroke biomarkers. Better potential diagnostic value appears to stem from the Lac resonance, which showed an increased LRE trend 24 h after ischemia for the ipsilateral hemisphere, but exhibited no such effects at other time points or in the contralateral hemisphere. Interestingly, Lac underwent highly statistically significant LRE when brains were subject to the most extreme ischemic conditions at 9.4 T, 27 with LRE measurements performed ex vivo at ∼ 3 hours post mortem. Combined, these findings suggest that Lac LREs may become—pending further elucidations of the origins of this phenomenon—an interesting candidate for gaining insights into biochemical processes underlying stroke. Further studies testing larger animal cohorts, stroke outcomes, and different field strengths are required to establish these links.
In agreement with findings presented in a previous 9.4 T ex vivo report, 27 NAA does not exhibit any significant LRE in vivo—neither in the contralateral nor ipsilateral hemisphere. Interestingly, the NAA resonance's intensity—which correlates with its concentration in the tissue—drops as early as 3 hours after ischemia,13,35,39 and is generally considered an important biomarker for neuronal viability. The absence of LRE for the NAA methyl resonance may be indicative of either a weak binding to macromolecules (which would be consistent with its negatively charged nature at physiologic pH) or an absence of easily exchangeable protons that would participate in an intramolecular Overhauser relaxation. By contrast with what had been observed in a previous ex vivo report 27 , Cho also fails to reveal statistically significant LRE in vivo at 21.1 T. This could arise from intrinsic differences between in and ex vivo tissues such as pH; further examinations of this difference are also in progress.
As part of the LRE measurements involved in this study, the in vivo evolutions of metabolic T1s for the targeted methyl peaks were also followed at 21.1 T. A number of features are worth highlighting from this longitudinal post-ischemia study. First, it appears that metabolic T1s for the targeted resonances do not increase with increasing fields. Indeed, a previous study comparing metabolic T1s in normal in vivo rat brains at 4, 9.4, and 11.7 T suggested an increase in T1s up to 7 T and a leveling off thereafter; 23 subsequent studies extended these findings to 14.1 T. 22 The present work finds that the metabolic relaxation times for the contralateral NAA, Cre and to a certain extent Cho peaks at 21.1 T, closely resemble those found previously at 9.4, 11.7 and 14.1 T in normal animals. This finding is suggestive of intricate interactions between the metabolites and their host tissue, as this behavior significantly departs from the one exhibited, for example, by the water resonance. Another feature that emerges from these metabolic T1 measurements is a clear longitudinal evolution after ischemia. Whereas NAA, Cre, and Cho T1s are known to increase only 24h after ischemia18,21 (findings that were herein also confirmed at 21.1 T), we find much more rapid dynamics for the Lac resonance. Lac's apparent T1 increases by more than 50% from its baseline value as early as 3 hours after ischemia; interestingly, this timescale parallels the onset of cytotoxic edema arising from anaerobic glycolysis and the ensuing ischemic cascade 3 . It follows that Lac T1s are highly sensitive probes of the tissue's response to ischemia, especially in comparison with the slow T1 dynamics exhibited by the other metabolic signals, which are more indicative of the onset of vasogenic edema at the 24-h time point 18 . Although the mechanisms of these fast Lac T1 dynamics remain to be elucidated, the absence of similar LRE effects in free solutions suggests a role for this metabolite's binding to tissue macromolecules. Understanding such T1 changes and potential LRE effects for these upfield resonances 27 —as well as for downfield peaks27,40—may help in understanding the processes underlying ischemia from the point of insult to recovery, as well as in studying novel biomarkers for the ischemic penumbra and functional outcomes of stroke.
CONCLUSIONS
This paper examined, for the first time, in vivo evidence for metabolic LRE effects and changes in the apparent metabolic T1s in stroke-affected rats at 21.1 T. Differential LRE effects were observed for specific metabolites on switching from water-suppressed acquisitions to non-water-suppressed excitation modes, shedding light on the complex interactions of metabolites within ischemic and non-ischemic tissues. Cre's in vivo LRE appeared to be stroke independent; by contrast Lac's LRE exhibited an ischemia-dependent evolution. Furthermore, the apparent T1 of Lac exhibited rapid changes, suggesting this metabolite's sensitivity to the environment created by cytotoxic edema. These aspects of Cre's and Lac's T1 and LRE effects deserve further systematic studies under normal and diseased conditions to better understand the molecular details associated with stroke and recovery. From a methodological standpoint, it is also worth stressing the remarkably clean and undistorted baselines and the excellent sensitivity that characterized the selectively excited 1H MR spectra shown. These positive characteristics are the result of multiple, mutually reinforcing effects derived from executing this kind of experiment at very high fields, including favorable relaxation properties (which enhance metabolic and suppress water signals), and of course the inherent high sensitivity endowed by recordings at ultrahigh fields. These factors enable rapid multiscan experiments leading to good spatiotemporal resolution, no water signal contamination, excellent signal-tonoise ratios per unit time, as well as very clean baselines. Numerous avenues in the research and diagnosis of the CNS can thus open by this synergy.
Footnotes
The authors declare no conflict of interest.
