Abstract
In Alzheimer's disease (AD), there is increasing evidence of blood–brain barrier (BBB) compromise, usually observed as ‘microbleeds’ correlated with amyloid plaque deposition and apoE-
Keywords
INTRODUCTION
Permeability of the blood–brain barrier (BBB) is a key requirement for the transport of exogenous molecular or particulate species between the blood and the brain parenchyma (e.g., therapeutic drugs or imaging agents). In healthy individuals, the BBB is relatively impermeable due to tightly packed endothelial cells that line the brain vasculature. However, the integrity of the BBB can be compromised by neurodegenerative disease and BBB abnormalities have been reported in Alzheimer's disease (AD) dating back to the original studies by Alzheimer. 1 It has been suggested that the neovascularization observed by Alzheimer may have actually resulted from collateralization around cerebral infarcts. 2 The incomplete endothelial layer in newly formed vessels would render these areas susceptible to increased BBB penetration. Recent work has shown BBB dysfunction in both AD and vascular dementia, where microhemmorhage has been detected in 30% of patients with AD and up to 80% of those with cerebral amyloid angiopathy (CAA).3,4 The presence of leaks in the BBB has obvious implications for the delivery of therapeutic molecules and imaging agents to the brain. In the case of AD, it suggests that enhanced permeability of the BBB may increase the transport efficiency of systemically administered drugs. It is yet unclear whether these leaks are causative of, consequential to, or irrelevant to AD.5–8 An understanding of the cerebral vascular leak process in AD is therefore of interest.
Blood–brain barrier dysfunction has also been reported in the general aging population. The Rotterdam study 9 found a high prevalence of cerebral microbleeds across the aging population, increasing from 17.8% at age 60 to 69 to 38.3% in those over 80. Microbleed prevalence in patients with symptomatic intracranial hemorrhages ranges from 20% to 70%.10,11
Microbleeds have been associated with disruption of the endothelium and vascular basement membrane,
12
and the deposition of prothrombin and hemosiderin in the extravascular space.13,14 Several causes for AD-associated microbleeds have been proposed. Amyloid deposition may promote apoptosis of vascular endothelial cells through contact with amyloid-β peptide.
15
Local Aβ levels have been correlated with the depletion of smooth muscle cells in large blood vessels and the loss of endothelium in smaller vessels.
2
In mouse models, BBB permeability increases before plaque formation.
16
In the case of CAA, deposition of Aβ plaques in the leptomeningeal arterioles has led to the suggestion that the deposits themselves cause the leak.
17
The apoE-
Of recent interest, intravenously injected nanoparticle agents have successfully labeled amyloid plaques,20–23 but their mechanism of transport across the BBB is unclear. Nanoparticle delivery to the central nervous system occurs without any permeability enhancers or active transport mechanisms, suggesting it may be mediated by passive vascular leak. The majority of past studies of vascular leak have used molecular probes to detect extravasation (e.g., Evans blue, magnetic resonance contrast agents, or a radiotracer),24,25, the largest molecules investigated have been dextrans, around 100kD. Such studies are not reflective of nanoparticle delivery for two reasons: (1) nanoparticles are typically much larger with molecular weights in the 100-MD range and (2) both BBB permeation and postextravasation transport are likely to be size dependent. We therefore chose to study the distribution of cerebral vascular leaks
We used the tet-off APP model of Alzheimer's-like amyloid neuropathology, 30 offering three advantages for the current experiments. First, because it encodes two familial mutations in the APP transgene, it develops aggressive amyloid pathology that becomes severe by the late ages studied here. Second, the mice develop CAA alongside parenchymal amyloid deposits, mimicking the vascular disruption of AD. Finally, transgene expression in this model can be controlled with oral doxycycline, which can be used to avoid side effects of APP overexpression during postnatal brain maturation. We used X-ray computed tomography (CT) to evaluate central nervous system access of peripherally administered nanoparticles with an eye toward the development of both future imaging agents and therapeutic delivery vehicles.
MATERIALS AND METHODS
Mice
All animal studies reported in this paper were conducted under study-specific protocols that were approved by the IACUC (Institutional Animal Care and Use Committee) at Baylor College of Medicine and Texas Children's Hospital. The studies reported in this paper are in accordance with the NC3RS ARRIVE guidelines.
The tet-responsive APP transgenic line 102 (tetO-APPswe/ind 102; MMRRC stock # 034845-JAX 30 and the tet-activator line B CaMKIIα-tTA (Jackson Laboratories #3010 31 were independently backcrossed to C57BL/6J for >25 generations before being intercrossed for these studies. The resulting double transgenic male offspring were then mated with wild-type FVB females to produce experimental cohorts on an FVBB6 F1 background. The tet-off amyloid deposition characteristics and quantitative measures of amyloid burden of this mouse line were previously studied. 32 A total of 16 APP + (6 younger than 10 months, 10 older than 14 months) and 19 APP— (6 younger than 10 months, 13 older than 14 months) animals were imaged. Of these, one of the <10Mo APP + animals died unexpectedly during the imaging procedure. Another <10Mo APP+ animal was scanned successfully but the images were found to be blurred due to unexpected animal motion during the scan. These two data sets were therefore not used, leaving a total of 14 APP + and 19 APP— animals whose data were used. Of these, 4 APP+ and 6 APP— animals were under 10 months of age, while the remaining 10 APP+ and 13 APP— animals were older than 14 months.
Doxycycline Administration
All mice used in this study were raised on doxycycline to suppress transgene expression during postnatal development. All mouse feed was obtained from Purina Mills, St Louis, MO, USA. Offspring were started on dox 1 to 3 days after birth by placing nursing mothers on medicated chow, formulated to 50mg/kg dox (Purina Mills Test Diet #5APL). At weaning, mice were maintained on dox until 6 weeks of age (Purina Mills Test Diet #5SBA). All mice were returned to regular chow to initiate transgene expression for the remainder of the experiment.
Image Acquisition
Imaging was performed on a Siemens Inveon CT system (Siemens AG, Erlangen, Germany). Animals were anesthetized (4% isoflurane in oxygen in an induction chamber for 5 to 10 minutes, 1.5% to 2% isoflurane in oxygen during imaging, delivered by face-cone). All images were reconstructed at a 35-
Image Processing
Histologic Examination
RESULTS
The Prevalence of Microbleeds
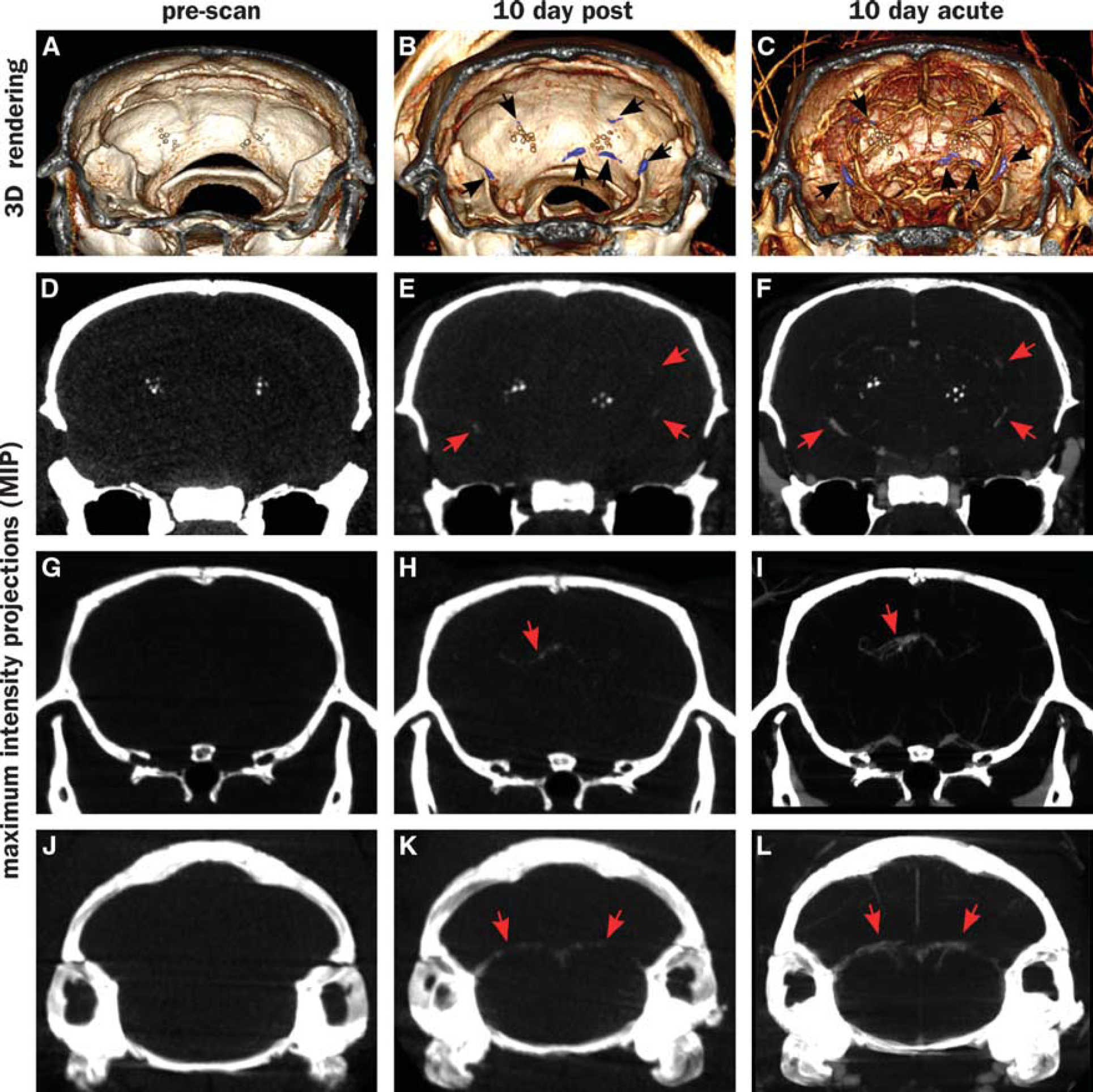
Figure 1 shows an overview of the CT imaging results in this study. Computed tomography images were collected at three timepoints: precontrast, 10 days after contrast injection (‘10-day-post’) and immediately after contrast injection, on the same day as the 10-day post (‘10-day-acute’). Thus, the prescan images show only endogenous radioopaque structures (Figures 1A, 1D, 1G, and 1J), while the 10-day postinjection images show the sites of vascular leak where contrast accumulates (Figures 1B, 1E, 1H, and 1K). The 10-day-acute images show both vascular structures and leaks (Figures 1C, 1F, 1I, and 1L). Precontrast, the only visible features other than bone are hyperintense spots in the thalamus, ventral to the hippocampus (Figures 1D to 1F). Such structures have been previously attributed to amyloid overproduction. 36 In our study, these structures do not appear to be associated with any active vascular leaks as they do not exhibit emergent signal in the 10-day-post images. However, numerous new intensities appear in the 10-day-post images that can be attributed to fresh vascular leaks. Supplementary Movie M1 shows a volume rendering of all these structures in relation to each other. The observed leaks can be divided into four categories:
Spherical leaks in the cerebral cortex, not obviously associated with any major vessels in the CT images (Figures 1E and 1F). Leaks in the choroid plexus, particularly along the DMCA (Figures 1K and 1L). Leaks along other vessels (Figures 1H and 1I). Sporadic leaks that do not appear to fit into any of these categories, and with no obvious classification.
Vascular leaks along vessels in computed tomography (CT) scans of an APP+ >14Mo animal. First row (
Signal enhancement is also observed in several locations close to the surface of the skull, consistent with leaks in pial vessels. Cerebral amyloid angiopathy in pial vessels is observed in this model under histologic examination (e.g., Figures 5F and 5G). However, the proximity to the highly X-ray absorptive skull makes it difficult to unequivocally classify these as leaks. We therefore do not include them in the current analysis. If they are indeed leaks, then it would mean the BBB permeation by the probe particles is even higher than estimated in the current analysis.
A comparison between minimal X-ray dose (‘radiation control’) and the higher dose associated with repeat scanning showed no consistent relationship between the radiation dose and leak prevalence. Changes observed between these conditions (both increases and decreases) were not significant at the 95% confidence level (Supplementary Figures S1 and S2). Animals that underwent the precontrast scan were therefore pooled with those that did not undergo precontrast scanning to quantify the dependence of leak on genotype.
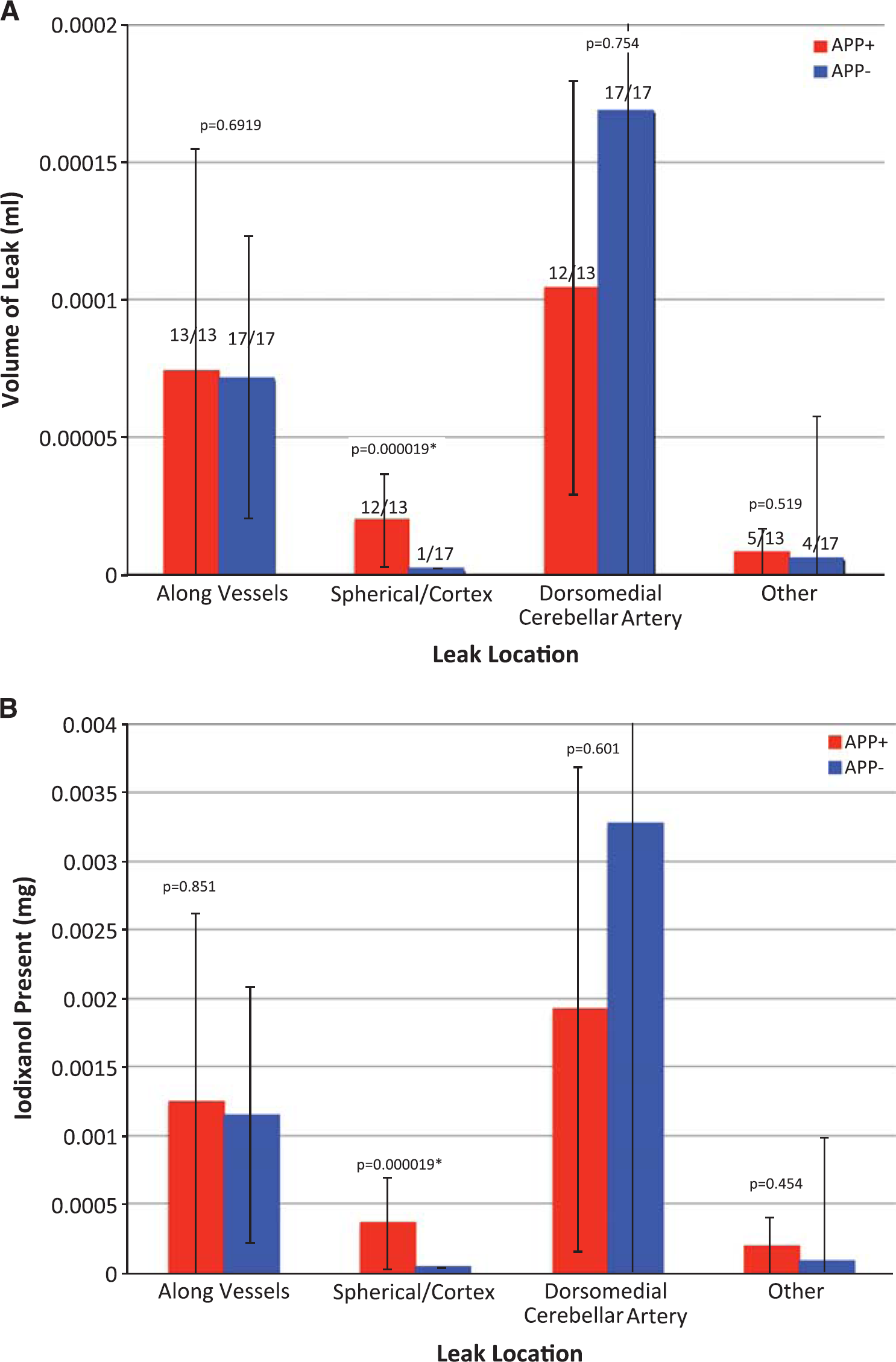
Leaks were quantified by the leak volume and total payload delivered (Figure 2). (For the purposes of this study, payload was defined as the encapsulated contrast agent: iodixanol.) As noted in Figure 2A, all animals (13/13 APP + and 17/17 APP —) exhibited vascular leaks. Similarly, 12/13 APP+ and 17/17 APP— animals showed leaks along the DMCA, suggesting that these leaks were independent of genotype. Spherical leaks in the cortex were present in practically all (12/13) APP+ animals, but only in one (1/17) APP — animals. Diffuse or miscellaneous other leaks were a minor contribution to the overall leak, and were sporadically present (5/13 APP+ and 4/17 APP —). The leaks in the DMCA were the largest, followed by those along other vessels, spherical leaks in the cortex and other miscellaneous leaks, in that order.
(
Comparison of leak volume between the APP + (amyloid-bearing,
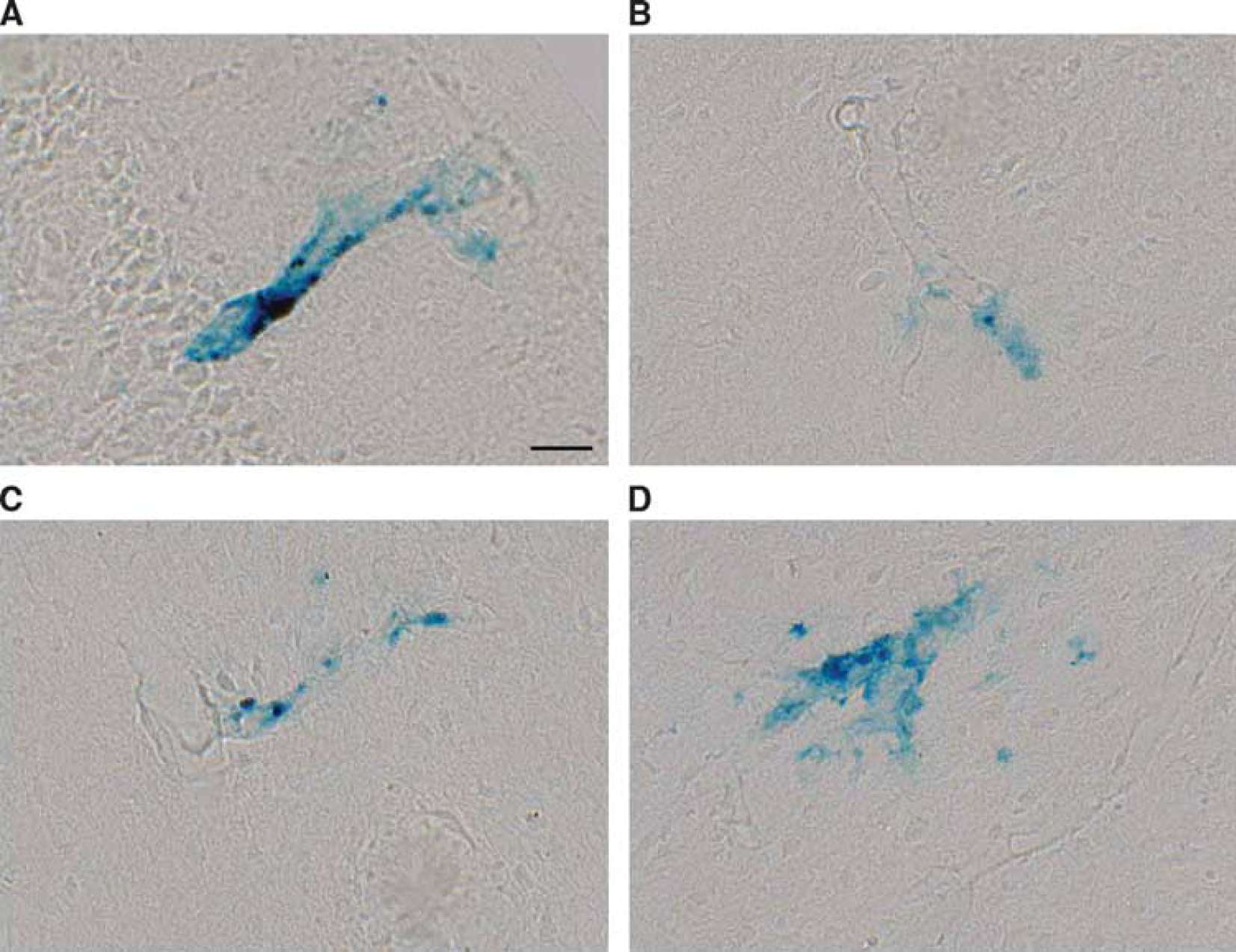
Cortical microhemorrhages were detected by Perls Prussian blue staining. Representative vascular leaks identified in insular (
Stratification of the mice by age (Supplementary Figure S3) reveals that the leaks along vessels (excluding those along the DMCA) increase significantly with age in the APP+ mice alone. APP+ mice >14 months of age have significantly more leaks than APP + mice<10 months of age. However, there is no difference between APP + and APP — mice older than 14 months, suggesting that the increase in leak may be part of the aging process, and not necessarily related to amyloid deposition.
Identical trends were observed in the amount of iodixanol that was deposited in the tissue (Figure 2B; Supplementary Figure 4). The data suggest that at the time of observation (10 days after injection), a total of 4 to 5
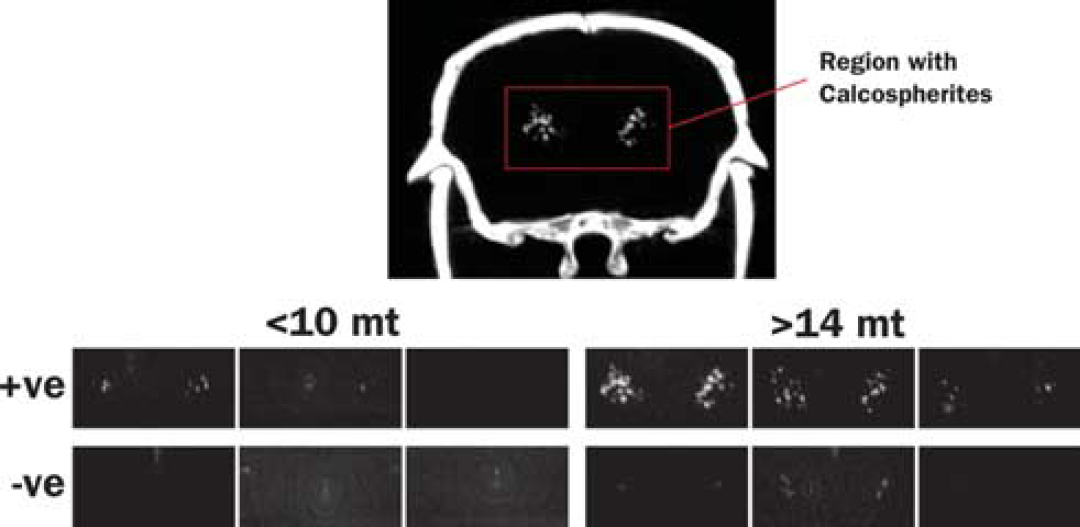
Maximum intensity projections of computed tomography (CT) images showing the regions where calcospherites are typically observed. Test animals are grouped as younger (<11 months) and older (>14 months), and by APP transgene status (+ve and — ve). Calcospherites were observed in 3/4 APP+ <11Mo, 9/9 APP + >14Mo, 0/6 APP— <11Mo, and 9/11 APP — > 14 Mo mice, and are obviously more intense in older, APP+ mice.
Persistent Computed Tomography Hyperintensities Are Identified as Calcospherite Deposits Rather than Amyloid
Thalamic hyperintensities were detected by CT with the following frequencies: 3/4 APP+ <10Mo, 9/9 APP+ >14Mo, 0/6 APP — <10Mo, 9/11 APP — > 14 Mo. Thalamic signal was more intense in older, APP+ animals (Figure 4). Although past studies have suggested that this thalamic signal is caused by deposited amyloid, its presence in APP— animals, and indeed, its visibility on CT, indicates the presence of calcium rather than protein. Histologic staining indicated that the deposits are mineral calcospherites rather than peptide fibrils. Unlike amyloid plaques, which become pink when stained with cresyl violet (Figures 5A to 5C), the thalamic deposits appeared darkly stained (Figures 5A, 5B, and 5D). Further, the thalamic deposits were globular in shape, whereas amyloid deposits were distinguished by a characteristic starburst-like appearance. We tested additional sections with von Kossa stain to confirm mineralization in the thalamus. As predicted, these deposits were stained dark by the von Kossa stain (Figure 5E). Counterstaining of the von Kossa sections with thioflavine-S clearly distinguished amyloid plaques from calcospherites (Figures 5F and 5G). In sum, these findings suggest that mineralized deposits visible by CT are not uncommon in aged animals, but unlike amyloid, are localized primarily to the thalamus and present in both transgenic and control mice. Amyloid deposits are clearly more prevalent than the calcospherites, consistent with our previous observations of amyloid deposition in this mouse line. 32
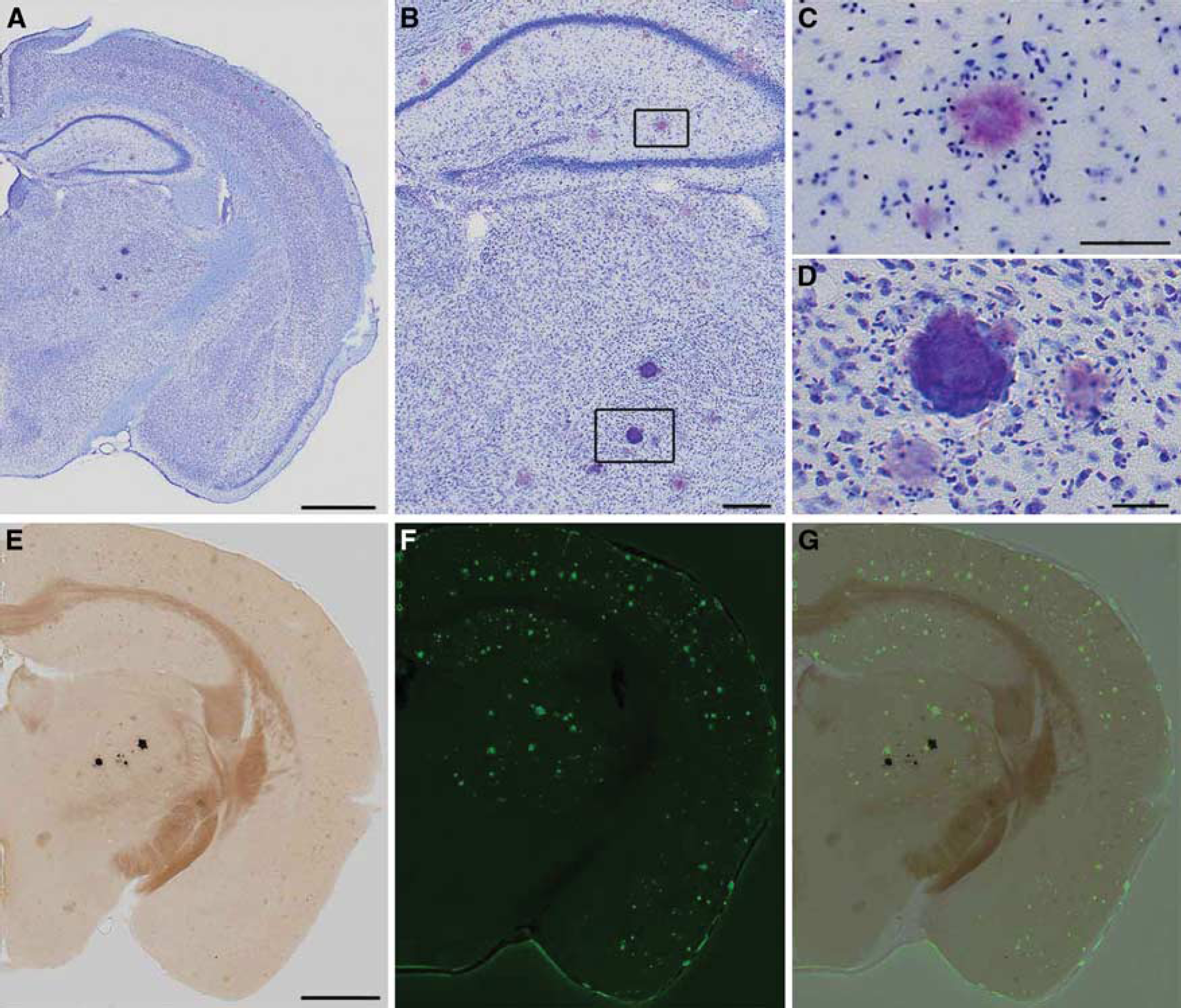
(
DISCUSSION
The transport of small molecules and nanoparticles from the bloodstream into the brain is of paramount importance for delivery of drugs and diagnostic contrast agents. Past studies in this area have focused primarily on the delivery of chemother-apeutic agents for the treatment of brain cancers, or on small molecule probes for visualization of BBB leak, typically in younger mice. Our own studies of cerebral vasculature using nanoparticle agents, e.g. 38 , also studied younger mice. However, with the development of novel therapeutic and diagnostic imaging agents for AD, there is a significant interest in understanding the BBB permeability of nanoparticles, in older animals with amyloid deposition. Here, we have set out to bridge this gap by investigating the BBB penetration of a liposomal nanoparticle probe in a mouse model for AD, at older ages. It is apparent from this study that the BBB is leaky in this model, in four distinct locations: (1) in vessels of the choroid plexus particularly the DMCA, (2) around other vessels throughout the brain, (3) nodular leaks in the cerebral cortex of APP+ animals, and (4) sporadic, diffuse leaks throughout the brain. On the basis of our observations of hemosiderin deposition, and the broadly accepted appearance of ‘microbleeds’ in human AD patients, we believe that these are physical leaks, where the vascular wall is sufficiently compromised that blood and its constituents can flow through the wall, driven by hydrodynamic forces, without the need for any active transport mechanism.
A striking feature in the brains of these animals is the presence of radioopaque particulate structures in the thalamus, ventral to the hippocampus (Figures 1D to 1F and 2). These structures have been described previously, 36 and were present in mice with mutant APP and PSEN1 genes, but not in mice with mutant PSEN1 alone. They were therefore attributed to APP overexpression, a conclusion that was supported by the identification of Aβ plaques within the structures. Our results show that these structures are present even in the wild-type siblings of the transgenic mutants, and most prevalent in older animals. Most importantly, the structures appear to be highly calcified and while Aβ deposits may be present close by, we see no evidence for Aβ within these structures. Finally, and most relevant to the current study, these structures do not appear to be associated with vascular leakage.
Active vascular leaks were found in multiple other locations, with the most prominent ones appearing in the choroid plexus (12/13 APP+ animals and 17/17 APP— animals). Focal leaks within the cerebral cortex (where amyloid plaque burden was substantial) were associated with small blood vessels that were not visible on CT, most likely due to limited spatial and contrast resolution. They occurred in 12/13 APP+ animals, and only 1/17 of the APP −. Further, the APP+ animals exhibit significantly larger leak volumes and payload transport in the cerebral cortex than the control animals, suggesting that spherical cortical leaks are a consequence of amyloid deposition. Other vascular leaks comprised the group with the second largest leak volume and payload delivery. They appeared to be equally prevalent in all mice regardless of genotype (13/13 APP+ and 17/17 APP—), although when stratified by animal age, these vascular leaks appeared to be significantly more prevalent in older APP+ mice than in younger ones.
Because our measurements of leak volume and iodixanol localization were based on a single time point 10 days after injection, it is not possible to accurately estimate the actual delivery of iodixanol to the brain. The residual amount remaining at 10 days after injection result from a balance between the passive leak into and active clearance away from the brain over this time period. However, assuming a positive clearance rate away from the brain, the total amount of iodixanol delivered must exceed the amount present at the 10-day point. The measured amounts are in the order of 4 to 5
Our findings are consistent with past work suggesting that the mouse BBB becomes permeable to peripherally delivered nanoparticles at ages above ~11 months. In earlier studies using an APP/PS1 model of AD, we showed that amyloid-targeted nanoparticles penetrated the BBB and localized to plaques after intravenous injection
20
Interestingly, the nanoparticles did not preferentially target plaques in any particular location in the brain; plaques throughout the hippocampus and cerebral cortex were uniformly labeled. In the present work, we have shown in an independent model of AD that (1) nanoparticles injected intravenously penetrate the BBB and (2) the majority of leak takes place from major blood vessels, particularly in the choroid plexus. This raises the question of how such particles in our earlier study could have transported throughout the brain to uniformly label amyloid plaques in locations far removed from the leak. The diffusion of nanoparticles in the brain parenchyma is not expected to result in more than about 10 to 50
As in the aged human brain, aged mice also show a significant cerebral vascular leak, but only a fraction of the overall leaks appear to be dependent on the presence of amyloid pathology. This suggests that agents injected into the systemic circulation should have ready access to the brain, contrary to the general assumption of an impenetrable BBB. Isolated spherical leaks in the cerebral cortex of APP+ animals were associated with small blood vessels, while leaks found along major vessels in the brain, particularly in the choroid plexus, were not clearly associated with amyloid deposition. A number of calcified, radioopaque deposits in the thalamus appear to have no hemosiderin content, suggesting that they do not contribute to nanoparticle distribution. Our findings have important implications for drug delivery to the brain of aged animals, especially those with amyloid neuropathology: liposomal nanoparticles of the type tested here, i.e., pegylated liposomes, should have ready access to the brain
Footnotes
AVA is a founder and stockholder of Marval Pharma Inc. (Netanya, Israel), Alzeca LLC (Houston, TX), and Sensulin LLC (Oklahoma City, OK). ET is a founder and stockholder of Alzeca LLC (Houston, TX). The authors are grateful to Clay Goodman for neuropathological identification of calcospherite deposits.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
