Abstract
Currently, there is no adequate technique for intraoperative monitoring of cerebral blood flow (CBF). To evaluate laser speckle imaging (LSI) for assessment of relative CBF, LSI was performed in 30 patients who underwent direct surgical revascularization for treatment of arteriosclerotic cerebrovascular disease (ACVD), Moyamoya disease (MMD), or giant aneurysms, and in 8 control patients who underwent intracranial surgery for reasons other than hemodynamic compromise. The applicability and sensitivity of LSI was investigated through baseline perfusion and CO2 reactivity testing. The dynamics of LSI were assessed during bypass test occlusion and flow initiation procedures. Laser speckle imaging permitted robust (pseudo-) quantitative assessment of relative microcirculatory flow and standard bypass grafting resulted in significantly higher postoperative baseline perfusion values in ACVD and MMD. The applicability and sensitivity of LSI was shown by a significantly reduced CO2 reactivity in ACVD (9.6 ± 9%) and MMD (8.5 ± 8%) compared with control (31.2 ± 5%; P < 0.0001). In high- and intermediate-flow bypass patients, LSI was characterized by a dynamic real-time response to acute perfusion changes and ultimately confirmed a sufficient flow substitution through the bypass graft. Thus, LSI can be used for sensitive and continuous, non-invasive real-time visualization and measurement of relative cortical CBF in excellent spatial-temporal resolution.
Keywords
Introduction
Current intraoperative monitoring techniques lack adequate surveillance of cerebral blood flow (CBF). Monitoring of the cerebral microcirculation is crucial, because tissue viability and clinical outcome depend on restoration of blood flow to an ischemic region in a timely manner.1,2 At present, intraoperative visualization of vessel perfusion is routinely accomplished by indocyanine green (ICG) video angiography 3 and off-line transit-time calculations of the fluorescent ICG dye within the arterial, parenchymal and venous compartments have served as surrogate markers of CBF.4,5 Recently, a new software tool for color-coded mapping of this transit-time pattern became available and it was suggested that the methodology could be useful for intraoperative monitoring of regional CBF. 6 While the method is feasible and may be suitable for characterization of perfusion alterations before and after a surgical intervention, it is not an established means for CBF measurement.
The two main techniques for true non-invasive assessment of relative cortical CBF (= relative cortical perfusion) are laser doppler flowmetry (LDF) and laser doppler imaging (LDI). However, both methods lack the combination of a high spatial and high temporal resolution. While LDF is limited by registering the blood flow only at a single point, LDI does not visualize the blood flow in real time because of the time required for surface scanning and data processing. 7
Laser speckle imaging (LSI) is an experimentally validated, true full-field optical imaging technique that avoids the need for surface scanning. Light from a laser is diverged by a lens to illuminate the area under investigation and a charge-coupled device (CCD) camera images the illuminated area for real-time generation of a two-dimensional color-coded map of blood flow.7-9 Preliminary investigations by others and our group have shown the general feasibility of intraoperative LSI during neurosurgical procedures.10,11 However, both studies were limited by a small sample size (n= 3) and lacking evidence of routine clinical applicability.
The purpose of this study was to evaluate intraoperative LSI as a tool for dynamic real-time assessment of relative cortical CBF in the human brain by determining normal values of LSI-specific perfusion parameters and the change of these parameters in cases of known perfusion anomalies.
MATERIALS AND METHODS
Study Design
The study (EA2/139/12) was approved by the local ethics committee of the Charité University Hospital in Berlin, Germany, and included 38 patients who underwent 61 LSI procedures in our institution between March 2010 and April 2011. Patient data are presented in Table 1. Intraoperative LSI was performed following a fixed protocol in all patients. Twenty-three patients received superficial temporal artery to middle cerebral artery (STA-MCA) bypass grafting for treatment of single-photon emission computed tomography (SPECT)-confirmed hemodynamic compromise due to arteriosclerotic cerebrovascular disease (ACVD; n = 9 male, n = 5 female; median age 64 (range 48 to 84)) or Moyamoya disease (MMD; n = 1 male, n = 8 female; median age 35 (range 17 to 59)). Seven patients underwent high-flow (saphenous vein) or intermediate-flow (radial artery) bypass revascularization before trapping of a giant aneurysm in the anterior circulation (n = 2 male, n = 5 female; median age 48 (range 14 to 71)). Eight patients served as hemodynamically healthy controls (n = 7 male, n = 1 female; median age 47 (range 19 to 76)).
Demographic and clinical patient data.
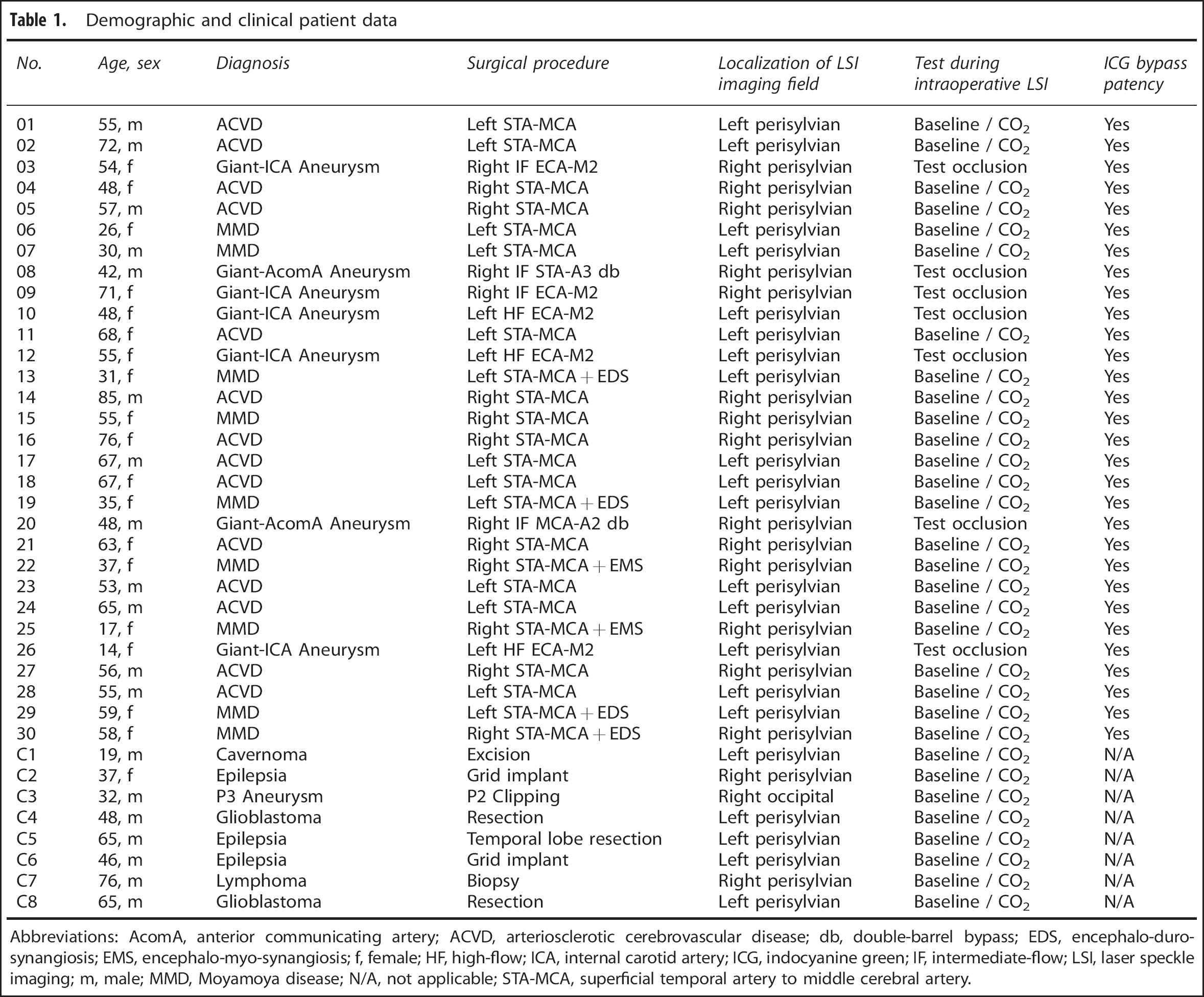
Abbreviations: AcomA, anterior communicating artery; ACVD, arteriosclerotic cerebrovascular disease; db, double-barrel bypass; EDS, encephalo-duro-synangiosis; EMS, encephalo-myo-synangiosis; f, female; HF, high-flow; ICA, internal carotid artery; ICG, indocyanine green; IF, intermediate-flow; LSI, laser speckle imaging; m, male; MMD, Moyamoya disease; N/A, not applicable; STA-MCA, superficial temporal artery to middle cerebral artery.
The following LSI-specific test parameters were evaluated:
To determine whether intraoperative LSI following a standardized protocol permits robust assessment of CBF-Flux for (pseudo-) quantitative comparison of relative CBF, we measured cortical baseline perfusion in patients with and without hemodynamic impairment.
The general applicability and sensitivity of LSI was determined by testing the cerebrovascular carbon dioxide (CO2) reactivity in patients with and without hemodynamic impairment.
The response behavior (dynamics) of LSI for real-time detection of acute perfusion changes was determined during test-occlusion and flow initiation procedures in patients undergoing high- or intermedi ate-flow bypass grafting.
The reliability of assessing bypass graft patency by LSI was evaluated by comparing graft patency assessment by LSI with graft patency assessment by ICG angiography.
Patient Management
All patients were anesthetized with propofol and remifentanil. Heart rate, arterial blood pressure, peripheral oxygen saturation and bladder temperature were registered throughout the surgery. The intraoperative mean arterial pressure (MAP) in patients undergoing bypass surgery for treatment of chronic cerebral hypoperfusion was set between 90 and 100 mm Hg. In patients not suffering from chronic cerebral hypoperfusion, an intraoperative MAP of 80 mm Hg was targeted. During all LSI measurements, MAP was maintained at a constant level and registered simultaneously with the cortical blood flow measurement. In cases where two LSI measurements were performed in one patient during the same surgery, we aimed for an equivalent MAP during each LSI measurement. By default, arterial blood gases were sampled every 30 minutes and the end-expiratory carbon dioxide (CO2) concentration during surgery was maintained at a level corresponding to an arterial partial pressure of CO2 (PaCO2) between 38 and 42 mm Hg. In cases where LSI was used to assess baseline perfusion and CO2 reactivity in one continuous measurement, additional blood gas samples were obtained during the 2-minute baseline perfusion measurement (PaCO2 low) and upon completion of the subsequent 5- to 7-minute CO2 challenge (PaCO2 high).
All patients received postoperative computerized tomography or magnetic resonance imaging within 24 hours and additionally in cases when new neurologic deficits occurred within the first 10 postoperative days to rule out procedure-related infarction and/or hemorrhage. Bypass patients received CT angiography 24 hours after surgery and digital subtraction angiography on day 7 to assess postoperative bypass patency. Intraoperative bypass patency was assessed by ICG video angiography. 12
Technical Specifications
The laser speckle imager we used is a 22 × 23 × 8 cm portable class 1 laser device (MoorFLPI, Moor Instruments Ltd., Axminster, UK) with a laser wavelength of 785 nm and power of 50 mW. An integrated CCD camera allows dynamic acquisition of two-dimensional, color-coded maps of perfusion. Physically, LSI measures the same blood flow parameter as conventional LDF in terms of relative blood flow velocity. This blood flow velocity is typically expressed in an arbitrary perfusion unit, commonly termed as Flux (CBF-Flux).
Device Setup
The imager was fixed to a clamped joint and attached to a mobile stand to permit versatile positioning over a desired imaging field. For purposes of data acquisition, the imager was connected to a laptop computer (Dell Computer Cooperation, Round Rock, TX, USA) via standard universal serial bus (USB) and FireWire (IEEE 1394) interfaces (Figure 1). After craniotomy and durotomy, the laser speckle imager was positioned 300 mm above and perpendicular to the exposed cortical area as described previously. 10 Identical focal and zoom settings were maintained within a 5 × 5 cm2 imaging field in control and STA-MCA bypass patients and a 7 × 7 cm2 imaging field in patients undergoing high- and intermediate-flow bypass revascularization. Direct illumination of the surgical field by light sources other than the laser light was avoided. An adjustable polarizing filter was used to eliminate specular reflection from shiny tissue. Blood clots on the cortical surface were cleared by sterile saline irrigation.
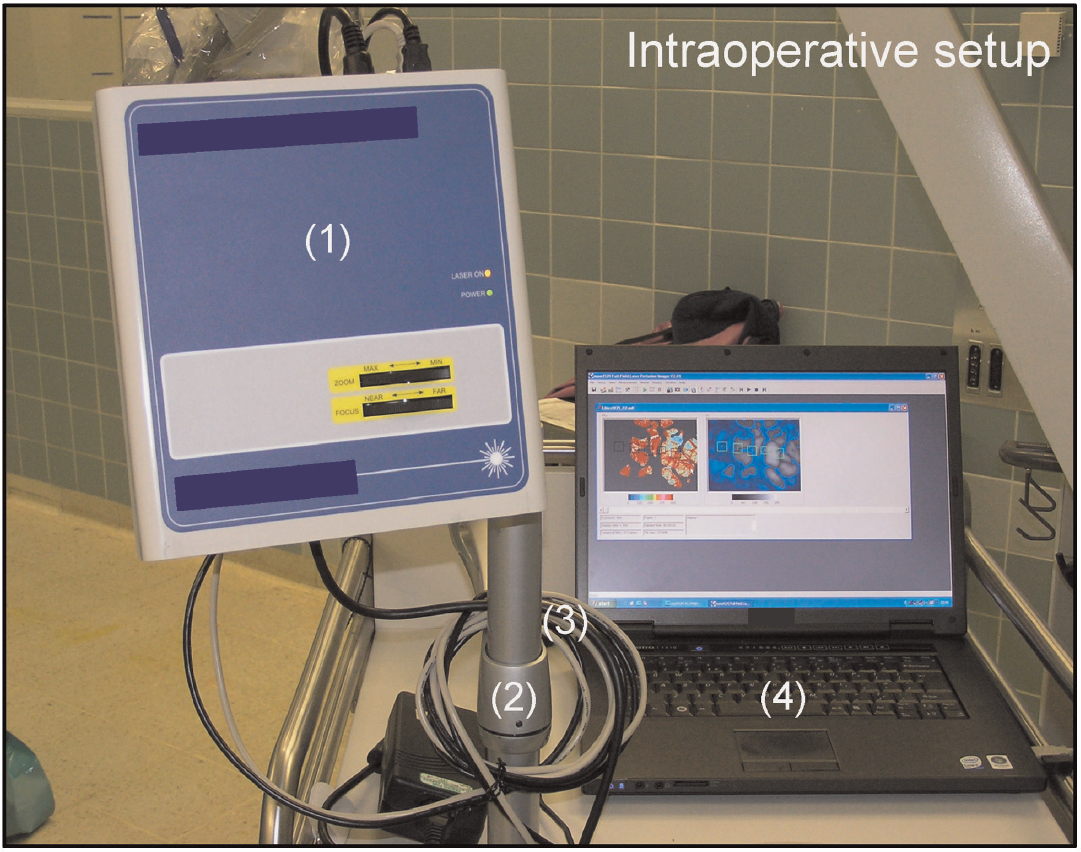
Device setup for intraoperative laser speckle imaging. Intraoperative setup of the laser speckle device, (1) fixed to a clamped joint (2) and attached to a mobile trolley, which permits free positioning over the surgical field. Standard USB and FireWire interfaces (3) enable a connection to a laptop computer (4) for data recording.
System Configuration
To optimize the measurable perfusion range, sensitivity and signal-to-noise ratio of the LSI measurement, two different exposure times of the CCD camera and two different temporal filters were selected. For measurements of baseline perfusion and CO2 reactivity in control and STA-MCA bypass patients, relative CBF was recorded at 25 Hz using a temporal filter of 100 frames per image, resulting in a scan rate of 0.25 Hz (768 × 576 pixel) at an exposure time of 8.4 ms for each image. For detection of acute perfusion changes in patients undergoing high- or intermediate-flow bypass grafting, the temporal filter was set to 25 frames per image, resulting in a scan rate of 1 Hz (768 × 576 pixel) at an exposure time of 4 ms.
Baseline Perfusion and CO2 Reactivity in Control and STA-MCA Bypass Patients
After surgical exposure, the LSI device was positioned over the imaging field and a 2-minute baseline perfusion measurement was obtained. For CO2 reactivity assessment, the end-expiratory CO2 concentration was gradually elevated by 10 mm Hg and the cortical blood flow response was continuously recorded over a time period of 5 to 7 minutes. After the measurement, normoventilation was restored and surgery resumed as planned. In patients undergoing STA-MCA bypass grafting, an end-to-side anastomosis between the frontal or parietal branch of the STA and a cortical M4 segment of the MCA was performed. After confirmation of bypass patency by ICG angiography, LSI was repeated at the same area of the cortex with a second baseline perfusion and CO2 reactivity measurement.
Bypass Test Occlusion in High- and Intermediate-Flow Bypass Patients
After completion of the high- or intermediate-flow bypass, graft patency was controlled by ICG angiography. The LSI device was positioned over the exposed cortical area and baseline perfusion was continuously recorded. During continuous LSI, the aneurysm-carrying vessel and the bypass were simultaneously clipped for 10 to 12 seconds before flow was initiated through the bypass. If baseline perfusion provided through the bypass was not lower than the perfusion provided by the main vessel, the main vessel was permanently occluded to trap the aneurysm.
Image and Data Processing of Laser Speckle Recordings
Cortical perfusion was recorded with the purpose-designed data acquisition software (MoorFLPI measurement software, Version V3.0, Moor Instruments, Axminster, UK) in the arbitrary perfusion unit CBF-Flux. For the purpose of providing the surgeon with simultaneous real-time visual and (pseudo-)quantitative information on blood flow, the data acquisition software permits a user-defined positioning of up to 16 variable regions of interest (ROIs) within the live speckle image. Within these ROIs, the mean CBF-Flux is calculated in real time and traced as a line graph. For study purposes, CBF-Flux was calculated off-line with the purpose-designed review software (MoorFLPI review software) on the basis of the intraoperatively recorded laser speckle blood flow maps. A user-defined polygonal ROI was placed within the imaging field and the mean cortical CBF-Flux within the ROI was calculated. In cases where the imaging field was partially obstructed (i.e. by a bypass graft), two separate ROIs were placed over the unobstructed cortical area and the mean CBF-Flux was calculated from both ROIs. For repeated measurements before and after STA-MCA bypass grafting, identical ROIs were placed in the same cortical area. Finally, graph traces from all 61 data sets were generated for the time course of each measurement.
In control and STA-MCA bypass patients, baseline perfusion was calculated as the mean CBF-Flux during a 1-minute plateau of the 2-minute baseline perfusion measurement. CO2 reactivity was expressed as the percent change in CBF-Flux between the baseline plateau and a 1-minute perfusion plateau upon completion of the CO2 challenge. Graph traces of cortical perfusion during test occlusion and flow initiation in patients undergoing high- or intermediate-flow bypass grafting were expressed as the percent change in perfusion compared with 100% of baseline perfusion.
Statistical Analysis
Data are presented as median and range or mean ± standard deviation. Statistics were generated by GraphPad Prism for Mac (Version 5.0f, GraphPad Software, San Diego, CA, USA). For comparison of physiologic variables (MAP and PaCO2), baseline perfusion and CO2 reactivity between control, ACVD, and MMD patients, and for comparison of baseline perfusion before, during, and after test occlusion, a one-way analysis of variance (ANOVA) was performed. For comparison of physiologic variables before and after elevation of PaCO2 and for comparison of baseline perfusion and CO2 reactivity before and after STA-MCA bypass grafting, a two-tailed paired t-test was used. Statistical significance was set at P < 0.05.
RESULTS
(Pseudo-)Quantitative Assessment of Relative CBF
We hypothesized that LSI following a fixed protocol should allow (pseudo-)quantitative comparison of CBF-Flux. In our study, LSI permitted robust visualization and measurement of macro- and microcirculatory flow in all control and STA-MCA bypass patients. Mean arterial pressure and PaCO2 did not differ between groups (Table 2). No difference was found in baseline perfusion between patients with and without hemodynamic compromise (Control: 1994 ± 493 Flux;ACVD: 1936 ± 487 Flux;MMD: 1938 ± 396 Flux (Figure 2B)). In patients who received STA-MCA bypass grafting, LSI detected a significant perisylvian CBF-Flux increase around the site of the anastomosis (ACVD: 2208 ± 631 Flux, *P < 0.05 vs before;MMD: 2549 ± 609 Flux, *P < 0.01 vs before (Figures 2A and 2C)). The higher relative increase of baseline perfusion after bypass grafting in patients with MMD is illustrated in Figure 2D.
PaCO2 and MAP before and after elevation of end-expiratory CO2.
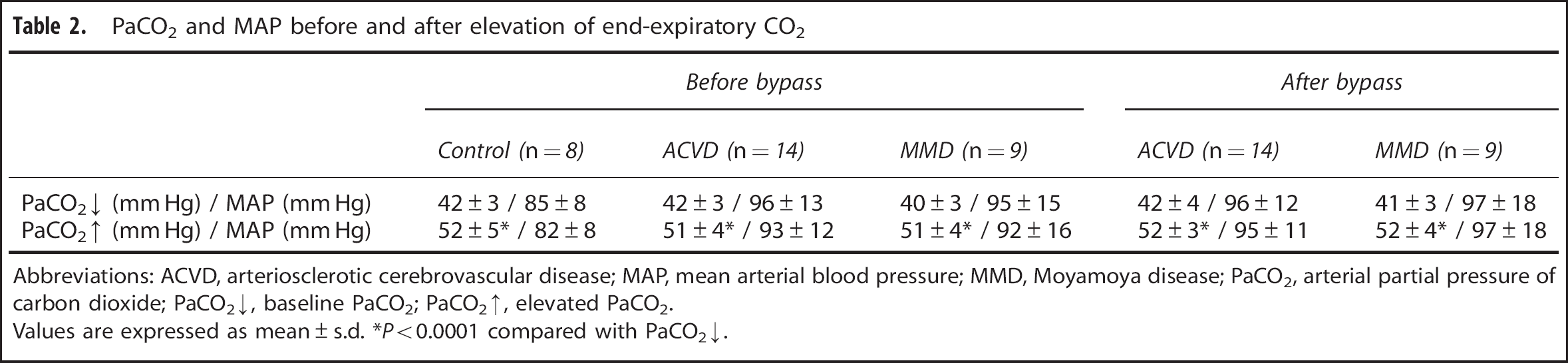
Abbreviations: ACVD, arteriosclerotic cerebrovascular disease; MAP, mean arterial blood pressure; MMD, Moyamoya disease; PaCO2, arterial partial pressure of carbon dioxide; PaCO2 ↓, baseline PaCO2; PaCO2 ↑, elevated PaCO2.
Values are expressed as mean ± s.d. *P < 0.0001 compared with PaCO2 ↓.
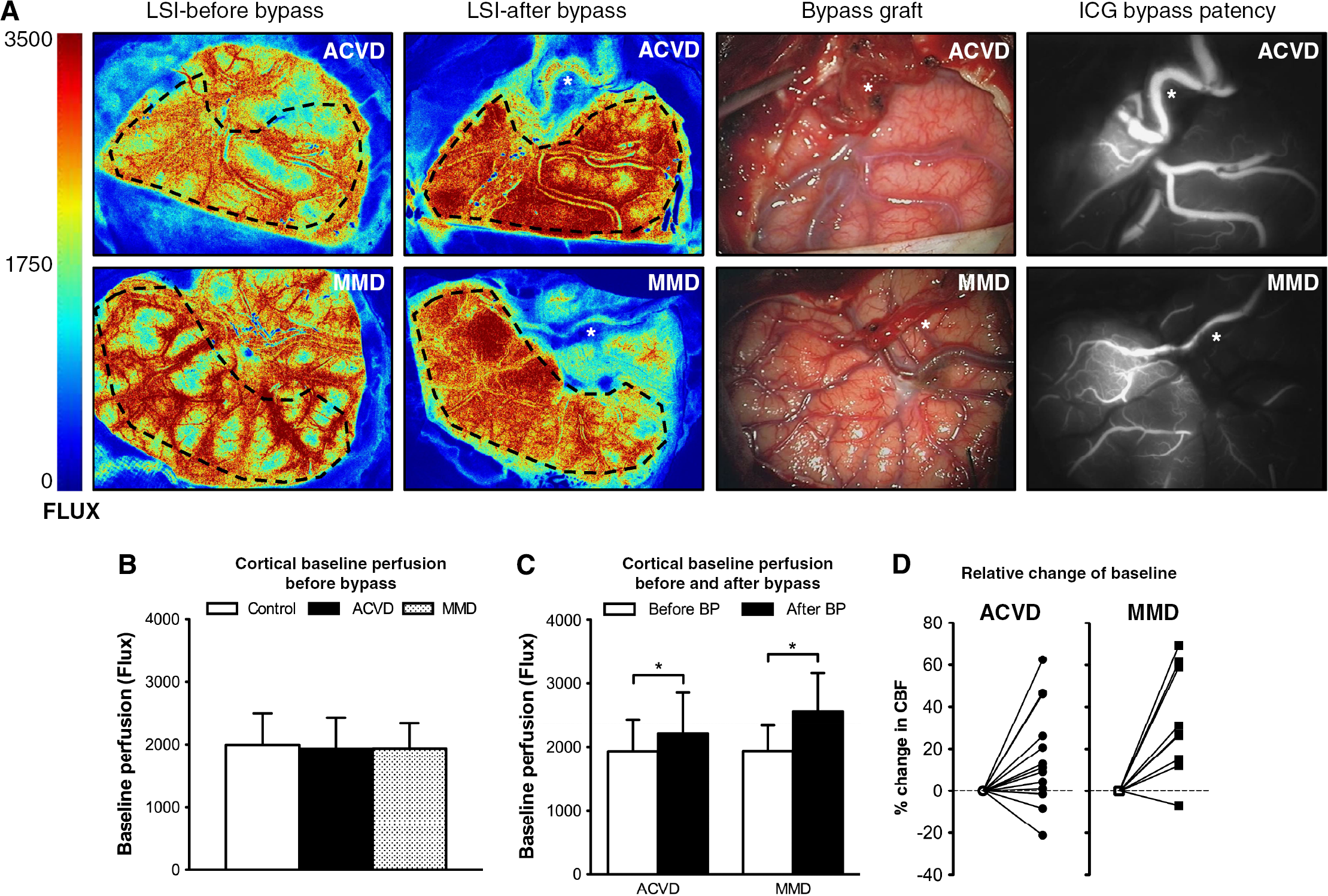
(Pseudo-)quantitative assessment of relative cerebral blood flow(CBF). (
Applicability and Sensitivity of LSI During Continuous Perfusion Assessment
To evaluate the applicability of intraoperative LSI and characterize the LSI-specific sensitivity for the detection of slight changes in cortical perfusion, we compared CO2 reactivity in patients with and without hemodynamic impairment, because non-ischemic cerebral hypoperfusion typically implies an impaired vascular reserve. Graph traces of a continuous LSI measurement during elevation of PaCO2 are shown in Figure 3A. In control patients, elevation of PaCO2 resulted in a mean cortical perfusion increase of 31.2% ±5%. In patients with SPECT-confirmed hemodynamic compromise, LSI detected a significantly impaired CO2 reactivity (ACVD: 9.6% ± 9%;MMD: 8.5% ± 8%;*P < 0.0001 for ACVD and MMD vs control (Figures 3B and 3C)), which was confirmed by a second CO2 reactivity testing after completion of the bypass graft (ACVD: 9.9% ± 13%;MMD: 2.9% ±5%;*P < 0.05 for MMD after bypass vs MMD before bypass (Figures 3B and 3D)). The relative decrease of the CO2 reactivity after bypass grafting in patients with MMD is shown in Figure 3E.
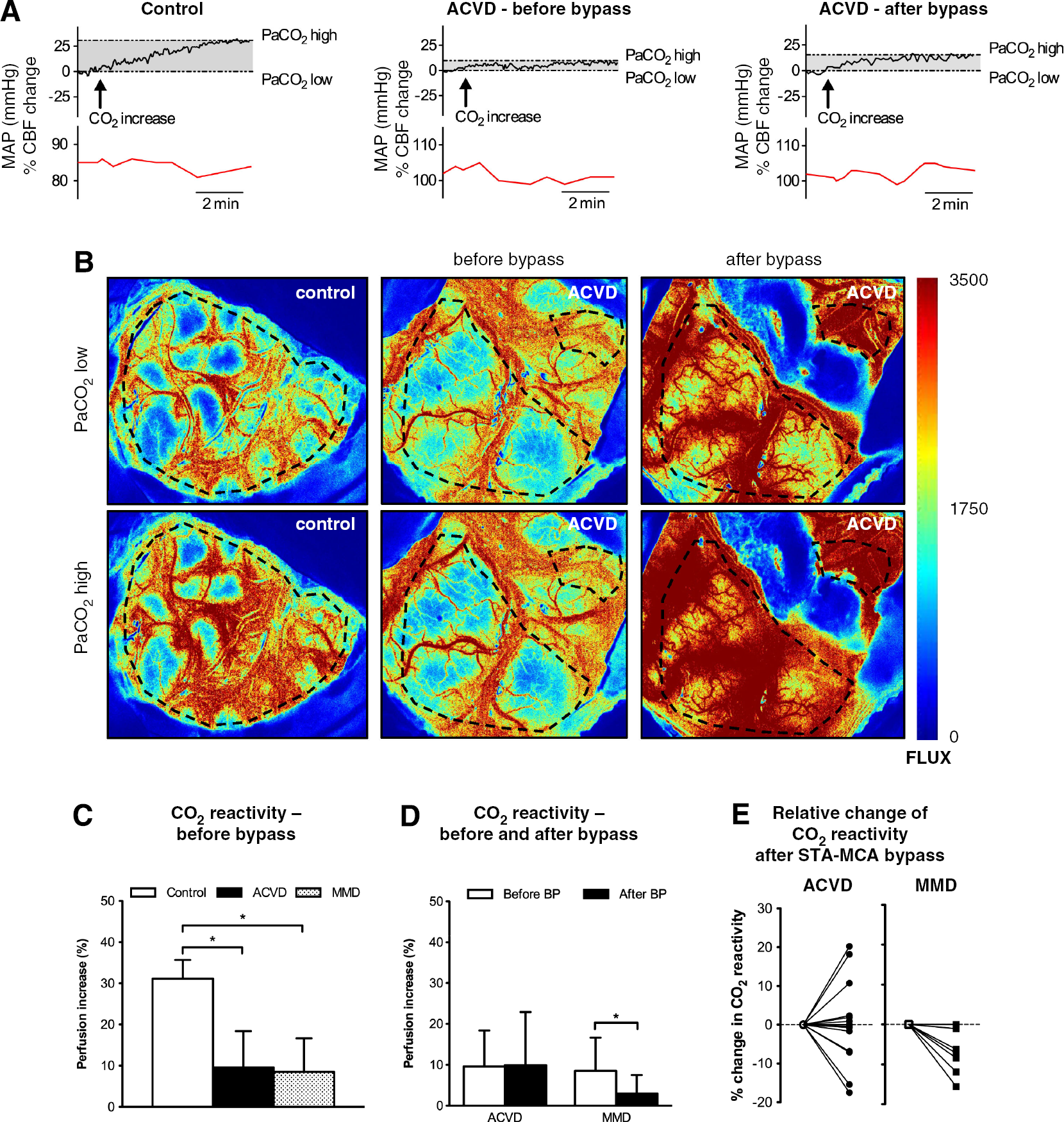
Applicability and sensitivity of intraoperative laser speckle imaging. (
Dynamics of LSI During Acute Perfusion Changes
The temporal response pattern of LSI in regard to acute changes in cortical perfusion was analyzed in a separate group of patients. Laser speckle images of patients undergoing high- or intermediate-flow bypass revascularization are shown in Figure 4A. In all cases, LSI permitted continuous real-time assessment of relative CBF. After test occlusion, LSI visualized and measured an immediate cortical perfusion drop to 56.5% ± 13% of baseline perfusion, which remained at a consistent level for the duration of the occlusion time. After flow initiation through the bypass, cortical perfusion promptly returned to 102.3% ±3% of baseline, indicating a sufficient flow substitution by the bypass (baseline: 3327 ± 569 Flux; occlusion: 1927 ±716 Flux; bypass open: 3408 ± 600 Flux; *P < 0.0001 vs occlusion (Figures 4A and 4B)). Individual graph traces of the temporal response pattern and the degree of flow reduction during the test-occlusion procedure are shown in Figure 4C.
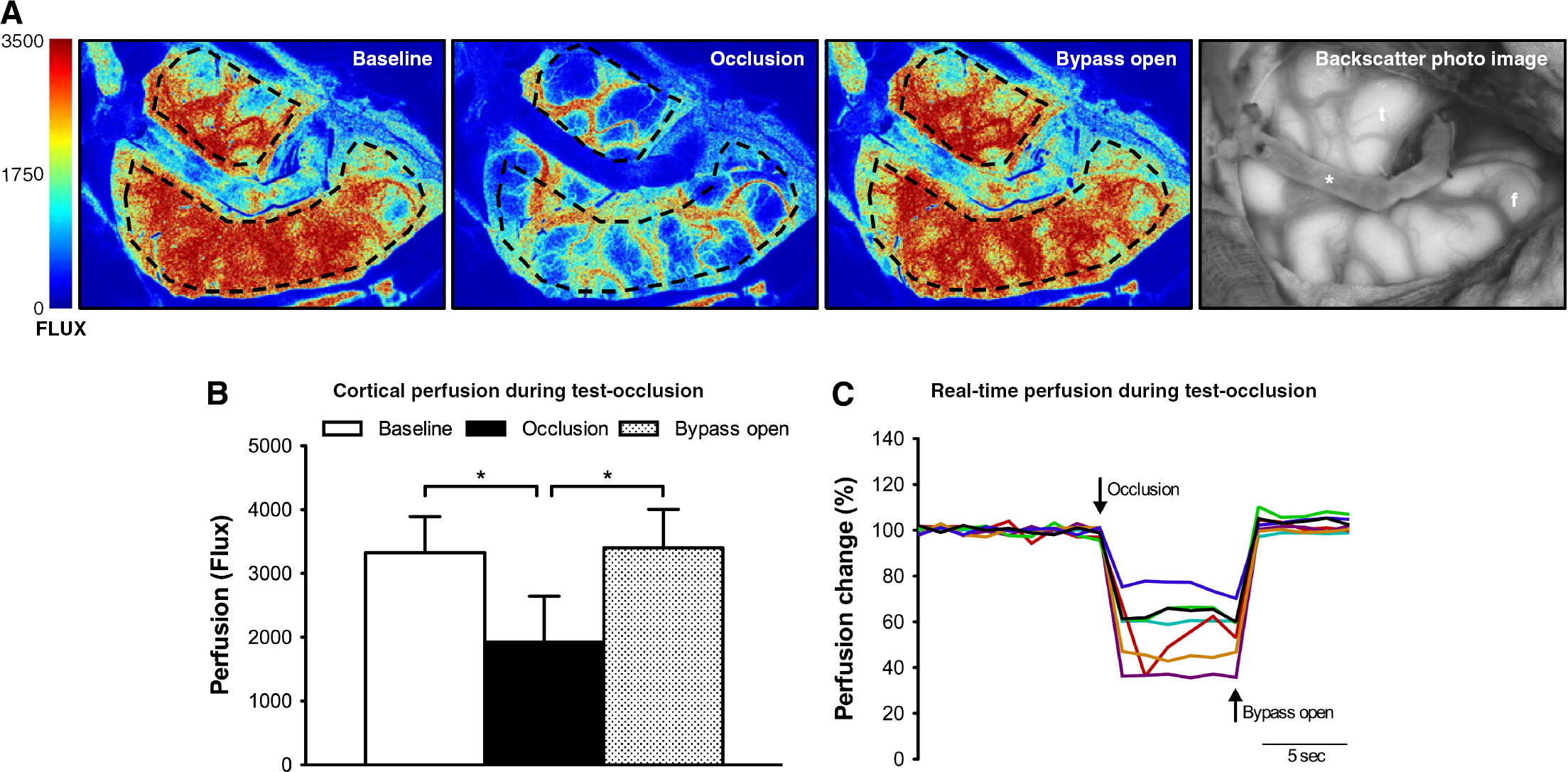
Temporal response properties (dynamics) of laser speckle imaging. (
Graft Patency Assessment Through LSI
Laser speckle imaging visualized positive perfusion within all STA-MCA, high-, and intermediate-flow bypass grafts (30/30), which was later confirmed by ICG angiography. In patients undergoing high- or intermediate-flow bypass grafting, visual assessment of a patent or non-patent bypass graft corresponded with cortical hypo- or normoperfusion during test occlusion and flow initiation, respectively.
DISCUSSION
In this study, we demonstrated the general applicability of LSI for continuous, non-invasive real-time imaging of relative CBF during a series of standardized CBF variations in patients undergoing neurovascular procedures. Laser speckle imaging proved to be easy-to-use, robust and was characterized by excellent image quality in high spatial resolution. All patients tolerated the procedure well and side effects were not observed. Repeated measurements did not pose any problems in regard to patient safety and image quality. Under standardized conditions, LSI permitted quantitative assessment of relative CBF. The system featured a high temporal resolution and a high sensitivity to even minor flow changes, which is mandatory for monitoring CBF under physiologic and pathologic conditions.
Intraoperative Imaging of CBF
Intraoperative functional diagnostics have increased neurosurgical efficiency and patient safety.13-15 However, the key determinant of tissue viability remains CBF, which to date cannot be adequately assessed intraoperatively. Although numerous imaging modalities, such as positron emission tomography (PET), SPECT, xenon-enhanced CT, dynamic perfusion CT, or magnetic resonance imaging (MRI) perfusion studies are used to study cerebral hemodynamics, intraoperative application of this technology remains limited by immobility, technical requirements, long data acquisition and processing times.
Non-invasive optical imaging has the advantage of reduced cost and infrastructure requirements, which facilitate intraoperative application. In general, the ideal technique should be sensitive enough to detect harmful events, quantitative, continuous, non-invasive, robust, user-friendly, and cost-effective and have a high spatial-temporal resolution. However, none of the available optical imaging techniques for intraoperative assessment of cerebral perfusion meet these criteria.3,6,16–19 The current gold standard for intraoperative visualization of perfusion within the vasculature is ICG angiography.3,12,20 Although the spatial-temporal resolution is high, the methodology requires injection of an exogenous substance and does not permit direct and continuous real-time assessment of relative CBF. For this purpose, LSI serves as an optical blood flow visualization and measurement tool based on dynamic light scattering off moving particles (erythrocytes).7,21 In this study, we evaluated the general applicability of intraoperative LSI because, at present, no data exist on direct cortical blood flow assessment in the human brain and information on procedure-related blood flow changes is of leading interest during neurosurgery. Moreover, robustness, comparability, and (pseudo-)quantitative assessment of relative CBF by LSI must be addressed in the clinical setting to allow meaningful data interpretation.
(Pseudo-)Quantitative Assessment of Relative CBF
Arbitrary blood flow velocity measurements by LSI are naturally influenced by confounding factors such as the distance and angle of the laser to the imaging surface, the size of the imaging field, ambient lighting, polarizing reflexes, the exposure time and the temporal resolution of image acquisition, among others. Against this background, recent evidence suggests that under standardized conditions, LSI may provide (pseudo-) quantitative information on flow.21-24 In our study, standardized patient management, device setup and system configuration permitted robust assessment of relative CBF and our findings were in line with the normal or at best mild reduction in resting CBF that is typically noted in patients with hemodynamic impairment.
After completion of the STA-MCA bypass, LSI detected a significant perfusion increase in the area surrounding the bypass anastomosis. This phenomenon has previously been reported after flow augmentation in patients with hemodynamic compromise, 25 where the chronic critical reduction in cerebral perfusion may induce a persistent maximal dilation of the cerebral arterioles, causing postoperative hyperperfusion in response to the rapid perfusion recovery, which may or may not be associated with transient neurologic deficits. In case of flow augmentation through STA-MCA bypass grafting, the postoperative perfusion increase is typically confined to the perisylvian area around the bypass anastomosis and the clinical presentation of symptomatic hyperperfusion may mimic that of ischemic stroke with sudden onset, upper extremity (hand) motor dysfunction, motor or sensory aphasia, dysarthria, and seizure activity.26-28 Therefore, a tool for early and easy distinction of hyperperfusion from ischemic stroke is desirable. At present, the only positive risk factors for development of symptomatic hyperperfusion after STA-MCA bypass grafting are reported as (1) a preoperatively increased oxygen extraction fraction 27 and (2) a cerebral blood volume increase in adults. 28 However, routine assessment of CBF and metabolism via PET poses high logistical and financial challenges and is not readily available. For this purpose, LSI could serve as a noninvasive, cost-effective, and easy-to-use method to analyze the local perfusion change after successful STA-MCA bypass grafting. Controlled and prospective studies with larger patient numbers for comparison of LSI-specific perfusion changes with quantitative CBF measurements are needed for further validation of the methodology and could help establish precision rates (positive and negative predictive values) of perfusion thresholds; phase-specific treatment strategies could further reduce surgical complications and improve long-term outcome.
Applicability and Sensitivity
The applicability and sensitivity of intraoperative LSI was characterized by analyzing standardized LSI-specific responses to PaCO2 elevation because consistent results have been reported on cerebral vasodilatation to hypercapnia29,30 and CO2 reactivity is impaired in patients with hemodynamic compromise. 31 The CBF increase of 31% after a PaCO2 increase of 10 mm Hg in the control group of our study was in line with the 30% to 40% increase of CBF per 10 mm Hg increase of PaCO2 in patients undergoing general anesthesia with propofol and remifentanil.17,32 Although propofol has been shown to lower the cerebral metabolic rate, neuronal activity, cerebral blood volume and CBF,33,34 CO2 reactivity is considered to remain intact compared with awake patients.32,33,35–37 Because propofol and remifentanil are the anesthetic agents of choice for intracranial surgery, 33 intraoperative CO2 reactivity testing could therefore permit a more robust characterization of perfusion abnormalities compared with baseline perfusion measurements alone. Further assessment of cerebrovascular CO2 reactivity is relevant because baseline CBF measurements alone may not sufficiently predict the risk of ischemia, as demonstrated in patients with hemodynamic compromise. 38
LSI sensitively detected cortical perfusion changes during continuous assessment of relative CBF by registering a limited CO2 reactivity in patients with hemodynamic impairment. Although we did not expect an immediate recovery of the CO2 reactivity after STA-MCA bypass grafting, further reduction of the CO2 reactivity in patients with MMD was an unexpected finding. The following hypothesis could explain these results: First, cerebrovascular reactivity after STA-MCA bypass grafting may not immediately improve, because changes in PaCO2 do not regulate the net flow though the STA donor vessel. Second, the fixed dilation of the cerebral resistance vessels in chronic cerebral hypoperfusion could require some time to adjust to the new hemodynamic situation and may hamper autoregulatory vasoconstriction after external flow augmentation. This hypothesis is in line with the postoperative perfusion increase in ACVD and MMD patients, despite a similar MAP before and after completion of the bypass graft. In addition, surgical manipulation could lead to a greater postoperative impairment of CO2 reactivity in patients with MMD due to the altered morphologic structure of the vascular wall 39 and increased microvascular density in these patients. 4
Temporal Dynamics
A suitable technology for continuous intraoperative CBF assessment must allow real-time detection of acute perfusion changes. LSI blood flow maps should be easy to interpret in a way such that brief visual inspection provides useful information in regard to tissue integrity and perfusion. In our experience, this was given by implementation of a temporal filter, which reduced the temporal resolution but enhanced the spatial detail resolution and improved the signal-to-noise ratio. To test whether the response properties of the system were still suitable for real-time perfusion assessment after implementation of the temporal filter, we tested the dynamics of the system during acute changes in cortical perfusion. Our results show that a scan rate of 1 Hz permitted detection of acute perfusion changes in a dynamic fashion while visualizing cortical perfusion in excellent detail resolution. The temporal filter additionally reduced the measurement variability associated with motion artifacts due to pulsation of the brain and facilitated comparison of the LSI and the macroscopic view of the investigated area. While it was not our intention to evaluate LSI as a tool for intraoperative functional testing, this remains an approach that could be addressed in future studies. For this purpose, however, the scan rates that we selected would be too low and could be increased to 25 Hz in order to maximize temporal resolution.
Bypass Graft Patency Assessment
Intraoperative monitoring of cerebral perfusion in the macro- and microvasculature requires selection of the most appropriate technique. For the purpose of visualizing and measuring relative CBF in the living human brain, we demonstrated the validity and applicability of LSI in a routine setting. For the purpose of graft patency assessment, however, it should be noted that LSI was limited by an obstruction of the imaging field (particularly through the adventitial cuff surrounding an STA-MCA bypass graft) and a lower spatial detail resolution compared with ICG angiography. Although it was ultimately possible to confirm positive flow within at least some part of the bypass graft in all cases, LSI did not permit interpretation of flow direction, vessel stenosis or patency of the bypass anastomosis. For the purpose of perfusion visualization within the vasculature, ICG angiography should therefore remain the method of choice.
Limitations
A main technical limitation of the presented methodology is that LSI can only assess blood flow in superficial vascular territories that are exposed surgically. Further, the system we used is currently not integrated into the surgical microscope. The clear benefits of microscope integration are its greater ease-of-use and better incorporation into the surgical workflow. To minimize this disadvantage, a separate surgeon performed assembly of the system without interrupting the surgical workflow and mostly the system remained assembled between measurements. The same surgeon was also responsible for operating the LSI device during each measurement, which increased the standardization and robustness of the procedure. As an advantage, the versatility of our setup permitted its use in different operating theaters for the purpose of back-to-back measurements in different patients.
DISCLOSURE/CONFLICT OF INTEREST
The authors declare no conflict of interest.
