Abstract
In stroke, there is an imperative need to develop disease-modifying drugs able to (1) induce neuroprotection and vasculoprotection, (2) modulate recovery and brain plasticity, and (3) limit the short-term motor and cognitive consequences. We hypothesized that fenofibrate, a peroxisome proliferator-activated receptor-
INTRODUCTION
Of the pharmacological targets likely to induce a pleiotropic brain effect after stroke, the three isoforms (
The three isoforms have distinct physiological functions and we particularly focused on the PPAR-
Peroxisome proliferator-activated receptor-
MATERIALS AND METHODS
Animals
All animal protocols were performed in strict accordance with the Ethical Committee in Animal Experimentation of Nord-Pas-de-Calais (C2EA-75) and the European community legislation (2010/63/UE). The experiments are reported in accordance with the ARRIVE guidelines. Male Wistar rats (mean weight 300 g) (Elevage Janvier, Le Genest Saint-Isle, France) or male C57BL/6 wild-type mice (mean weight 25 g) (Elevage Janvier) were used. Animals were housed in a light- and temperature-controlled environment with unlimited access to food and water. Animals were randomly assigned to the different groups. Experimental data were monitored by blinded investigator for group allocation. The study included all animals that underwent the whole protocol (72 hours or 7 days) and excluded from results nonischemic animals or animals with subcortical infarcts (10% to 15% per group).
Drug
Fenofibrate (F-6020; Sigma-Aldrich, Chimie Lyon, France) was dissolved in vehicle (water; 0.1% Tween-80; 0.5% carboxymethylcellulose) and orally administered poststroke by gavage twice a day during 72 hours or 7 days.
Experimental Design
Two separate protocols were performed, the first concerning the early administration of fenofibrate and the dose-response effect, the second concerning the delayed administration of fenofibrate and the short-term consequences (details of protocols in Supplementary Data). STAIR recommendations were considered to design the two protocols. 14
Protocol 1: Early 72 hours fenofibrate treatment
To assess the effect of PPAR-
Vascular reactivity was assessed
For methodological reasons (intravital microscopy limited to the mouse), we acquired additional data in mice submitted or not to MCAo, with the observation of
Protocol 2: Delayed 7 days fenofibrate treatment
We studied the effect of a delayed poststroke administration of fenofibrate on motor recovery, neurogenesis, and stroke-induced memory impairment. Fenofibrate (200 mg/kg per day) or vehicle was administered by gavage twice a day during 7 days, with the first dose given only 8 hours after onset of ischemia or sham surgery. The animals were killed 7 days after MCAo. The hepatic weight was measured at 72 hours and 7 days.
Neurologic score, grip strength test (24 hours, 72 hours, and 7 days) and infarct volume (7 days) were assessed (6 to 14 animals per group).
Neurogenesis was evaluated at 7 days (4 animals per group) and PPAR-
Stroke-induced memory impairment was assessed by cognitive test (Y-maze; 16 to 25 animals per group), immunochemistry with amyloid deposition (4 animals per group) and quantitative PCR with amyloid-related secretases (3 animals per group) at 7 days after ischemia.
Middle Cerebral Artery Occlusion Model
Cerebral infarct was induced by intraluminal MCAo as previously described.
15
Anesthesia was induced by intraperitoneal chloral hydrate administration (300 mg/kg). A rectal probe was inserted and body temperature was maintained at 37±0.5°C with a heating lamp. The caudal artery was exposed, cannulated with a 24 G polyethylene catheter and connected to a blood pressure monitor. The mean arterial blood pressure (mm Hg) was monitored throughout the experiment and blood samples were taken before, during, and after ischemia, to measure blood pH, arterial PaO2 (mm Hg), and arterial PaCO2 (mm Hg). Briefly, the right carotid arteries were exposed through a midline cervical incision and the common carotid and external carotid arteries were ligated with a silk suture. An aneurysm clip was placed across the internal carotid artery and an arteriotomy was made in the common carotid artery stump, allowing the introduction of a monofilament nylon suture with its tip rounded by flame heating. The suture was gently advanced into the internal carotid artery and passed into the intracranial circulatory system as far as in the narrow lumen at the start of the middle cerebral artery. After 30 minutes for mice or 60 minutes for rats, the suture was carefully removed until its tip was blocked by a ligature placed on the common carotid artery (to allow reperfusion). As in index of the reliability of the MCAo model,
Neurologic Assessment
Neurologic function was measured at 24 hours, 72 hours, and 7 days after MCAo using the Bederson score: 16 an animal with no apparent deficits obtained=0; the presence of forelimb flexion=1; decreased resistance to push=2; and circling=3.
Behavioral Assessment
Rotarod test
Motor coordination was evaluated 24 and 72 hours after the transient MCAo or sham procedure. The rats were placed on an accelerating rotarod. 17 The speed of the rotarod was increased from 4 to 40 rotations per minute in 2 minutes. The latency to fall off the rotarod was recorded. Each rat was tested once. All rats were trained for 5 continuous days before the formal tests.
Grip strength test
Forelimb grip strength was determined by an automated grip strength meter at 24 hours, 72 hours, and 7 days after the transient MCAo or sham procedure. The experimenter grasped the rat by the tail and suspended it above a grip ring. 18 After 3 seconds, the animal was gently lowered toward the grip ring and allowed to grasp the ring with its forepaws. The experimenter then quickly lowered the remainder of the animal's body to a horizontal position and tugged the animal's tail until its grasp of the ring was broken. The mean force in Newtons was determined with a computerized electronic pull strain gauge.
Y-maze test
The rats were evaluated for spatial memory in one wooden Y-maze before killing at 7 days after the transient MCAo. The maze was lacquered black, and consisted of three arms with an angle of 120° between each two arms. 19 The three identical arms were randomly designated: Start arm, in which the rats started to explore (always open); Novel arm, which was blocked at the first trial, but open at the second trial; and Other arm (always open). The Y-maze test consisted of two trials separated by an intertrial interval to assess spatial recognition memory. The first trial (training) lasted 10 minutes and allowed the rat to explore only two arms (Start arm and Other arm) of the maze, with the third arm (Novel arm) being blocked. After a 1-hour intertrial interval, the second trial (retention) was conducted. The rat was placed back in the maze in the same starting arm, with free access to all three arms for 300 seconds. The first arm visited, the number and duration of explorations of each arm were recorded during the second trial.
Infarct Volume Measurement
The animals were killed with an overdose of intraperitoneal pentobarbital (200 mg/kg) 24 hours, 72 hours, or 7 days after reperfusion. Brains were removed, frozen and coronally dissected into 50-
Quantification of Plasmatic Fenofibric Acid
The major metabolite of fenofibrate is fenofibric acid. Quantification of plasmatic FFA will be performed by liquid chromatography and MS/MS method.
20
Fenofibric acid and carbamazepine (internal standard) were prepared in methanol. Calibration curve of FFA was realized at the following concentrations: 100
Vascular Reactivity
In vitro vasoreactivity analysis (in rats)
Endothelium-dependent and -independent relaxations were assessed in a Halpern arteriograph (Living Systems Instrumentation, Burlington, VT, USA). We used a proximal segment of the right middle cerebral artery perfused with oxygenated Krebs solution and maintained at 37°C and pH 7.4. The lumen diameter was measured using image analysis. 21 The relaxant dose-response curve for acetylcholine (ACh) was determined by stepwise, cumulative addition (from 10−9 mol/L to 3.10−5 mol/L ACh) after artery preconstriction with 5-hydroxytryptamine (10−6 mol/L induced 90% of the maximum constriction). To test nitric oxide-mediated smooth muscle relaxation, a single concentration of sodium nitroprusside (10−5 mol/L) was added to the bath after artery preconstriction. Relaxant responses were expressed as the percent increase in the preconstricted artery diameter.
Intravital Microscopy (In Mice)
Animals were anesthetized by intraperitoneal injection of 300 mg/kg chloral hydrate. The head of each mouse was placed in a stereotaxic frame. A craniotomy was performed with high-speed drill (S791, Bien-Air, Bienne, Switzerland) in the right parietal bone. The dura matter was retracted, and the surface of exposed brain was continuously superfused with artificial cerebrospinal fluid at 37°C and pH 7.4.
The cerebral microcirculation was observed with the use of an intravital microscope. An Eclipse 50i Nikon microscope (Nikon, Champigny-Sur Marne, France) fitted with a Xenon light source and epi-fluorescence assembly was used with filter sets for acridine orange (excitation: 470 nm, full width at half maximum 40; emission 540 nm, full width at half maximum 40). A video-camera (Model E2v, L3Vision, Bievres, France) mounted on the microscope projected the image onto a monitor.
Observation of cerebral microcirculation
Leukocyte accumulation was assessed after 24 and 72 hours of reperfusion. For each mouse, five randomly selected venular and arteriolar segments, 25 to 50
Acetylcholine-induced vasodilatation of pial arterioles
The arteriolar diameter was measured using a calibrated reticule in the Metamorph software (Molecular Imaging, Roper Scientific, Evry, France) 30 minutes after surgery of the skull (basal diameter) and 5 minutes after topical administration of ACh (10−5 mol/L) (diluted in the artificial cerebrospinal fluid). 22 The response to ACh is expressed as the percent change in diameter compared with the basal arteriolar diameter. Endothelial function is evaluated as in the four groups of mice at 72 hours of reperfusion.
Immunohistochemistry
After 24 hours, 72 hours, or 7 days of reperfusion, the rats were anesthetized and perfused through the heart with cold saline and 4% paraformaldehyde. After 24 hours of fixation in 4% paraformaldehyde at 4 °C, the brains were cryoprotected in 20% and 30% sucrose solutions in phosphate-buffered saline at 4 °C. Parallel sets of brain sections (20-
Bromodeoxyuridine Administration and Immunofluorescence
Bromodeoxyuridine (BrdU; Sigma-Aldrich) was administered intraperitoneally in two injections of 30 mg/kg per day between the third and sixteenth days preceding the week of behavioral tests, and a remainder 30 mg/kg 24 hours before killing.
24
A double labelling BrdU-NeuN or BrdU-PPAR-
Western Blotting
Western blot analysis was performed on cortex homogenates. We analyzed the expression of ICAM-1 72 hours after cerebral ischemia in control, ischemic and treated animals (anti-ICAM-1, 1:500; MCA773, Serotec). For the experiment, blots were probed with
RNA Extraction and Reverse Transcription
Total RNA was extracted from dissected brains with Extract-all (Eurobio, Les Ulis, France) according to the kit manufacturer protocol. Quantitative PCR was performed using a LightCycler system according to the manufacturer's instructions with LightCycler-FastStartDNA Master SYBR Green mix (Roche Applied Science, Meylan, France). Protocol consisted in a hot start step (8 minutes at 95 °C) followed by 50 cycles including a 10-second denaturation step (95°C), a 10-second annealing step (60°C), and a 10-second elongation step at 72°C. PCR efficiencies were optimized according to Roche Applied Science's recommendations on a standard sample expressing all studied genes. To confirm amplification specificity, PCR products were subjected to a melting curve analysis. Quantification data represent the mean of three experiments from four animals in each group. Relative quantification analyses were performed by the RelQuant 1.01 Software (Roche Applied Science). The sequences for the primers used for each of the genes analyzed are the following: Actin Up 5′-agccatgtacgtagccatcc-3′, Low 5′-ctctcagctgtggtggtgaa-3′; A Disintegrin And Metalloproteinase-10 (ADAM-10) Up 5′-gaggaaaaagcgcacaactc-3′, Low 5′-tgttacggattccggagaag-3′; Amyloid Precursor Protein (APP) Up 5′-gcggacacagactatgctga-3′, Low 5′-ctctgtggcctcttcgtagg-3′; Beta-site APP Cleaving Enzyme-1 (BACE-1) Up 5′-gctgcagtcaagtccatcaa-3′, Low 5′-attgctgaggaaggatggtg-3′, Presenilin-1 (PS-1) Up 5′-ggtccacttcgtatgctggt-3′, Low 5′-ctcccactcctcactgaagc-3′; Presenilin-2 (PS-2) Up 5′-ccctgatgctcctcttcttg-3′, Low 5′-agtgcgctgatcacaatgag-3′.
Statistical Analysis
All values were expressed as mean±standard error mean (s.e.m.). Statistical analysis was performed using the SPSS 12.0 software (IBM, Boigny-sur Bionne, France). Comparisons among multiple groups were performed using one-way analysis of variance followed by a
RESULTS
Physiological Parameters
There were no differences in physiologic parameters (weight, blood pressure, temperature, pH, PaCO2, and PaO2) between vehicle and fenofibrate-treated groups before, during, and after the surgical procedure (data not shown).
Fenofibrate Induces an Acute Neuroprotective Effect in a Dose-Dependent Manner
Fenofibrate reduced the size of the infarct (Figure 1A) in a dose-dependent manner. The decrease in infarct size was significant at 72 hours for 50 and 200 mg/kg per day of fenofibrate, with a higher neuroprotective effect at 200 mg/kg per day. On the motor level, early administration (1 hour after the onset ischemia) of fenofibrate improved postischemia performances evaluated in the rotarod test at 24 and 72 hours (Figure 1B). The functional recovery was faster at the dose of 200 mg/kg per day than 50 mg/kg per day. These effects were parallel to the FFA plasmatic concentration (Figure 1C).
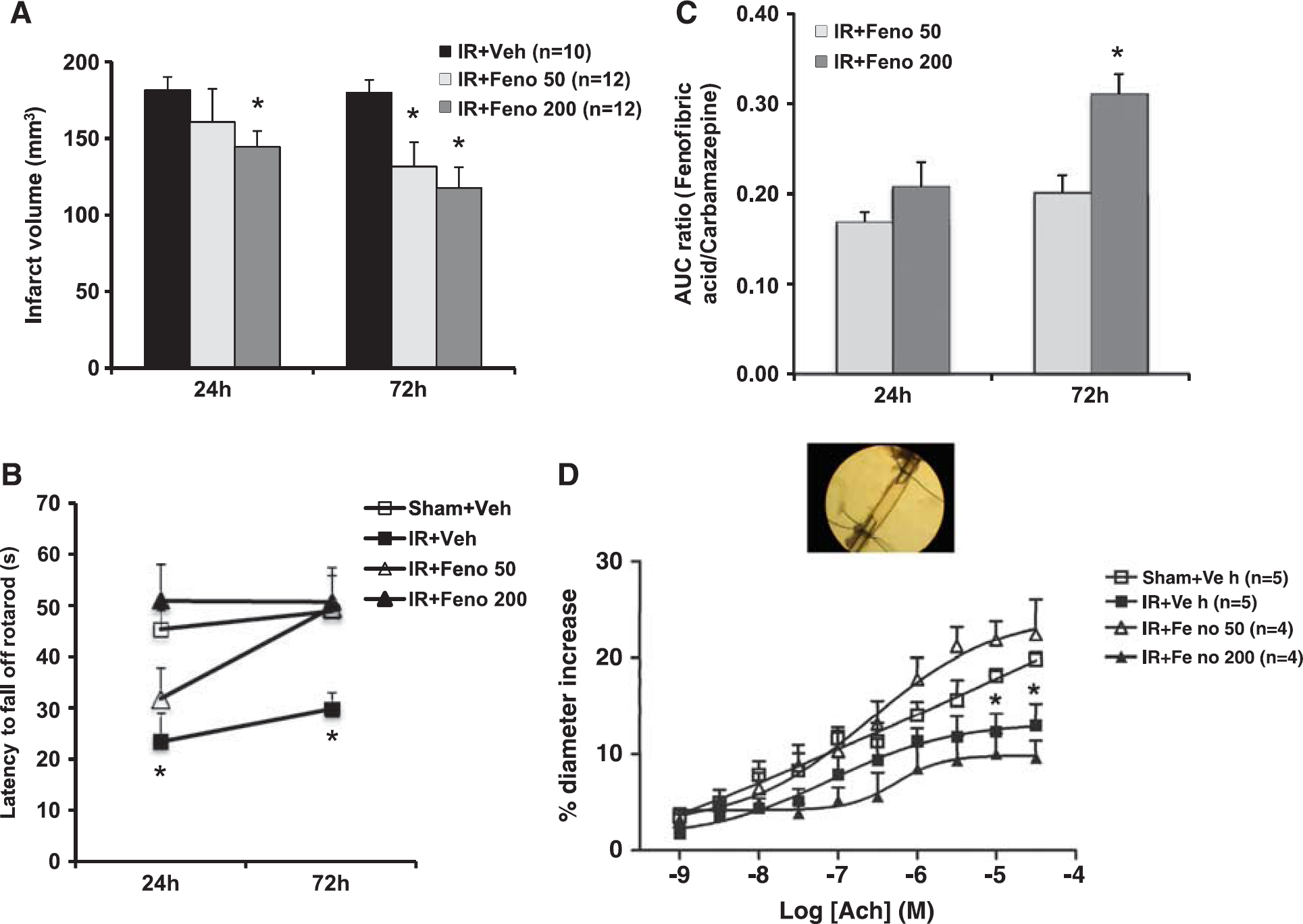
Dose-effect study of fenofibrate. (
Fenofibrate Prevents Postischemic Endothelial Dysfunction
In the aftermath of cerebral ischemia, the vascular wall could participate in the pathophysiology of postischemic impairment.
25
The ischemia-reperfusion process altered ACh-induced endothelium-dependent relaxation at 72 hours
Fenofibrate Induces an Acute Protective Effect on Leukocyte Rolling and Adhesion
To explore the protective effect of fenofibrate on vascular compartment at 50 mg/kg per day, we studied
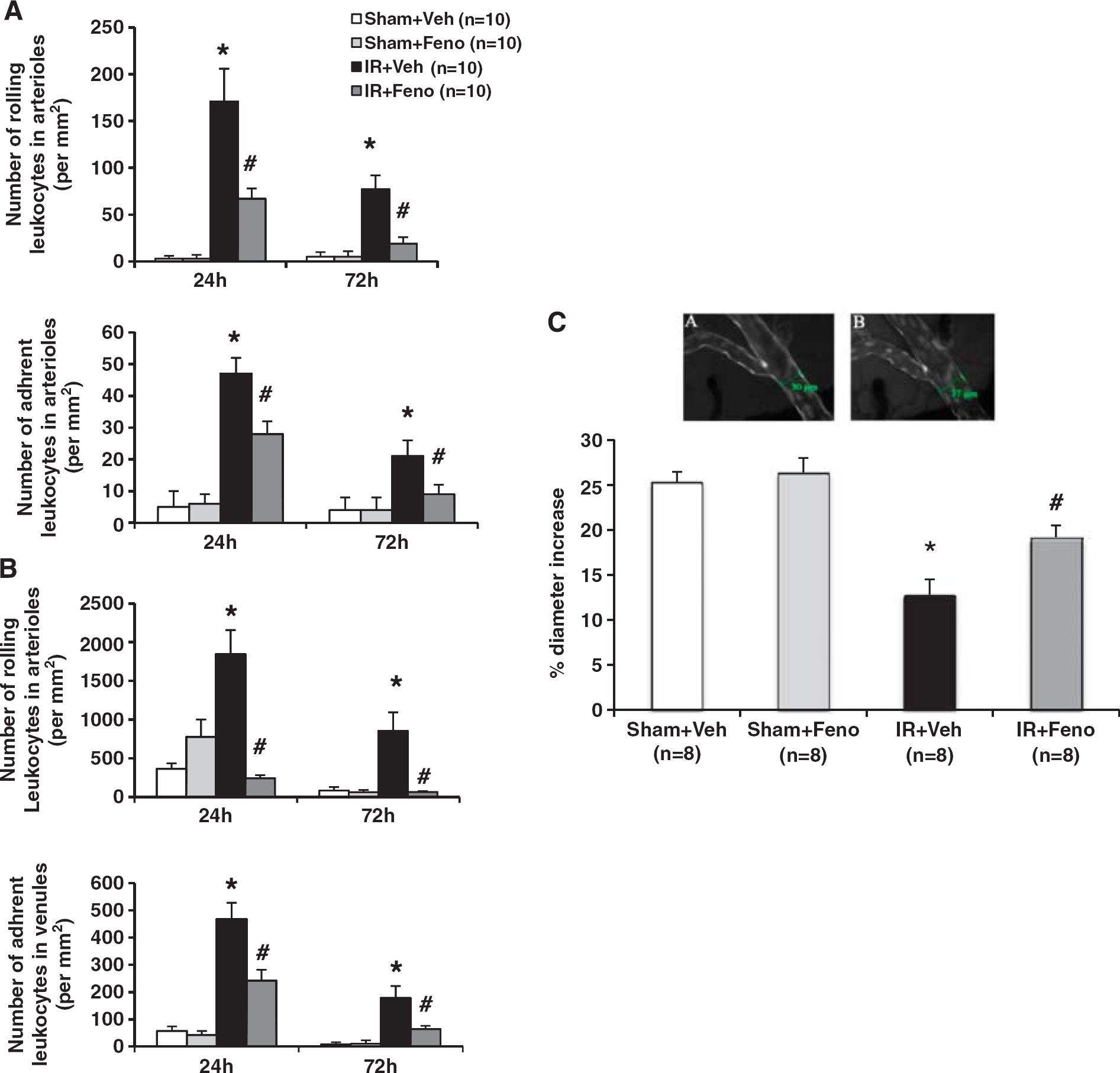
Effects of treatment with the peroxisome proliferator-activated receptor-
Fenofibrate Prevents Both Parenchymatous and Vascular Inflammation
On the tissue level, postischemic inflammation resulted in tissue expression of MPO (a marker of polymorphonuclear leukocytes (PMNs) infiltration) and Ox-42 (a marker of microglial activation). Ischemia induced an increase in PMNs infiltration in the ischemic area (Figure 3A) as well as microglial activation (Figure 3B) at 24 and 72 hours. Fenofibrate administration at 50 mg/kg per day significantly reduced parenchymatous infiltration by circulating PMNs (Figure 3A) and
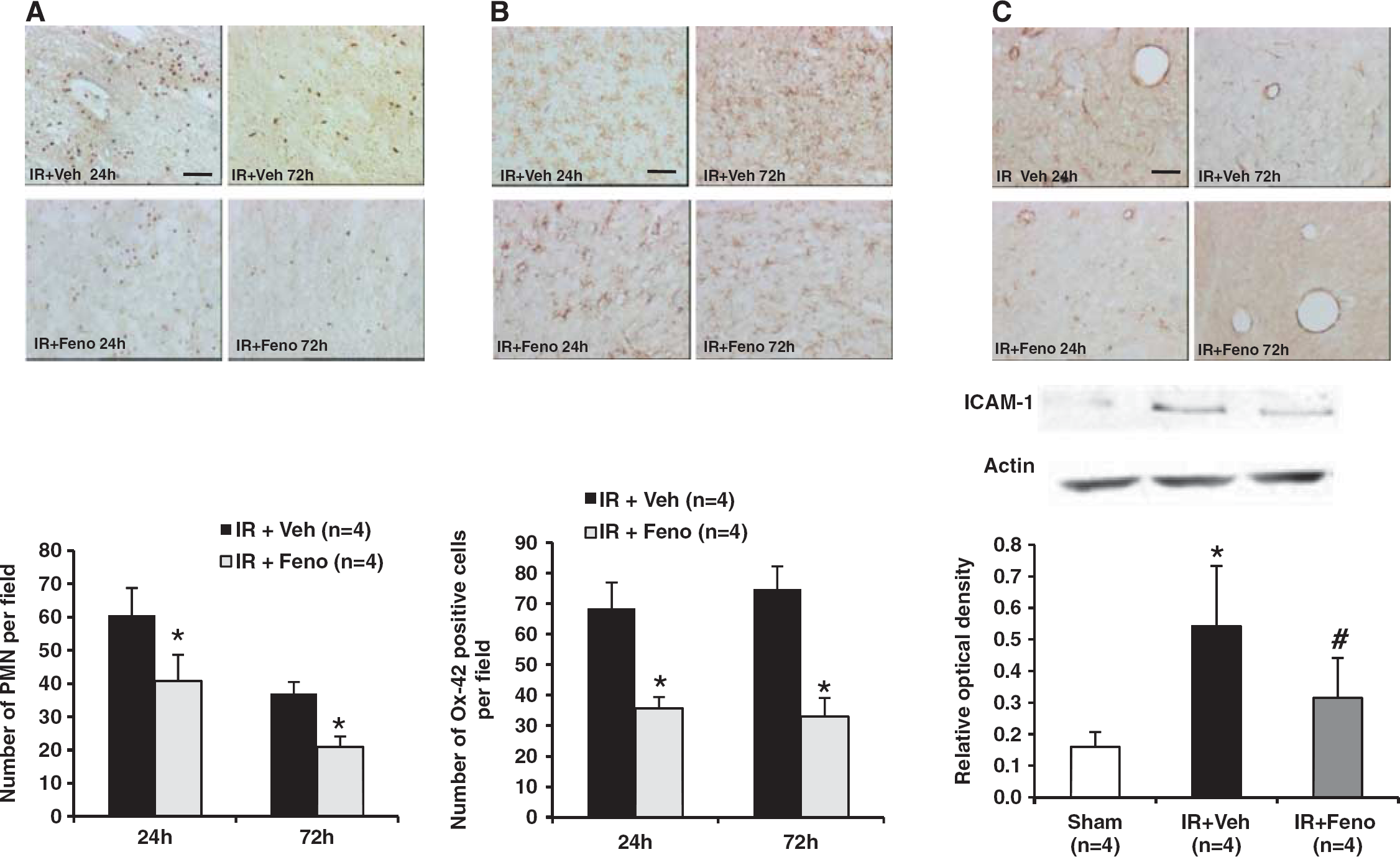
Effects of treatment with the peroxisome proliferator-activated receptor-
Fenofibrate Induces a Delayed Protective Effect on Infarct Volume, Motor Function, Neurologic Deficit, and Memory Recovery
Delayed administration of fenofibrate 200 mg/kg per day (8 hour after the start of reperfusion) also enabled a reduction in infarct size after 7 days of treatment (Figure 4A). Fenofibrate increased gradually liver weights of animals at 72 hours and 7 days compared with vehicle-treated animals. At 24 hours, mortality was more important in vehicle-treated group as compared with fenofibrate-treated group. Then, at 72 hours and 7 days, this difference was not observed between the treated animals and the control group (data not shown).
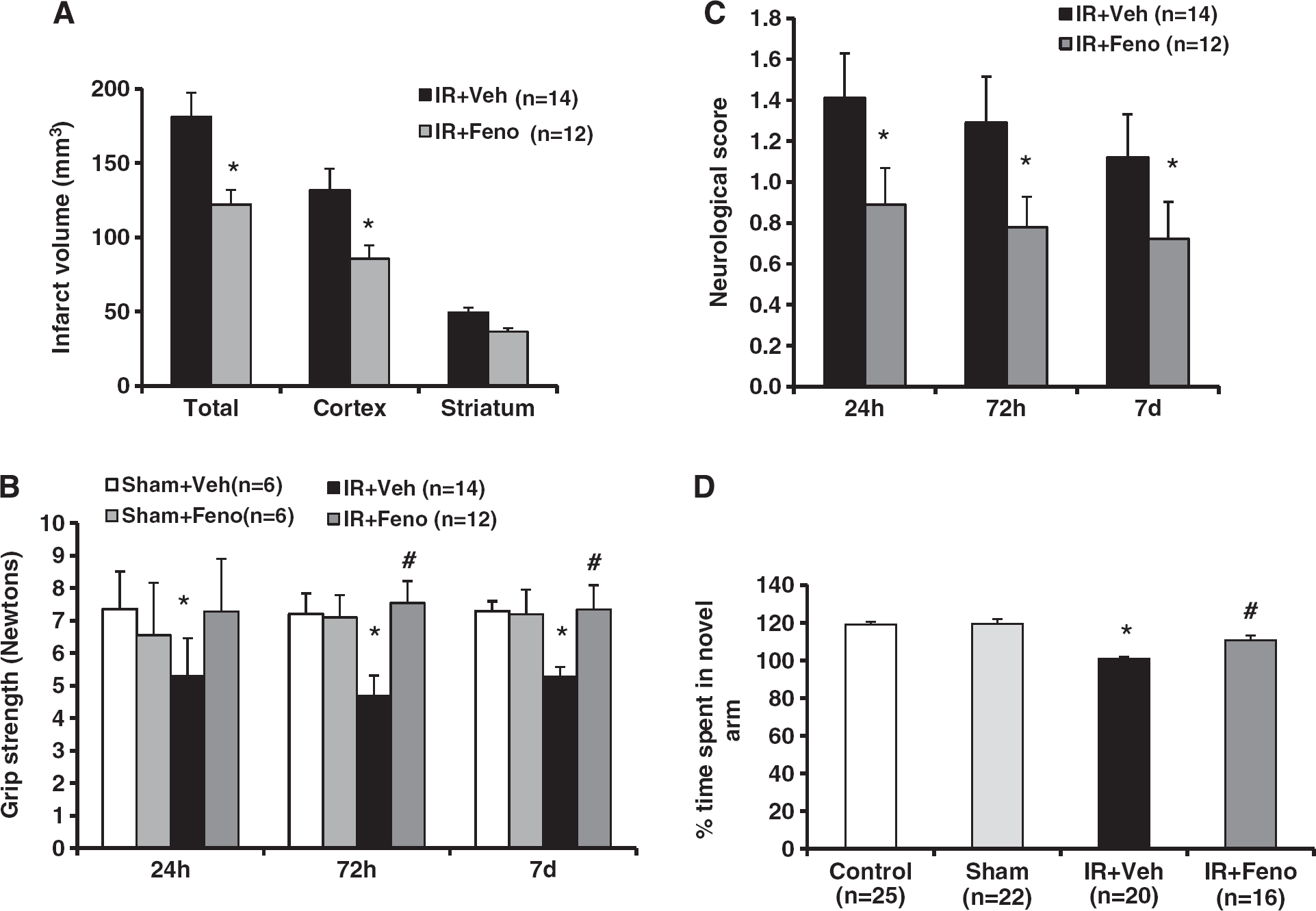
Effects of delayed treatment with the peroxisome proliferator-activated receptor-
The neurologic deficit at 24 hours, 72 hours, and 7 days after ischemia onset was decreased in the fenofibrate-treated group (Figure 4C). Delayed and prolonged administration of fenofibrate produced motor improvement until 7 days after ischemia in the grip strength test (Figure 4B).
In the Y-maze test, the total number of visits in the three arms did not differ between the sham and ischemic groups (data not shown). Differences in the time spent in the new arm of the Y-maze observed among sham and ischemic groups were due to ischemic memory deficits and not locomotor deficits. On the cognitive level, a 7-day course of fenofibrate prevented the postischemic visuo-spatial memory impairment. Postischemic rats experienced a memory deficiency in the Y-maze test since they spend as much time in the new arm of the labyrinth as in the arm already explored. This defect in memory-based learning is partially prevented by fenofibrate administration (Figure 4D).
Delayed Treatment by Fenofibrate Modulates Postischemic Neurogenesis
We showed that some of BrdU-labelled cells co-express PPAR-
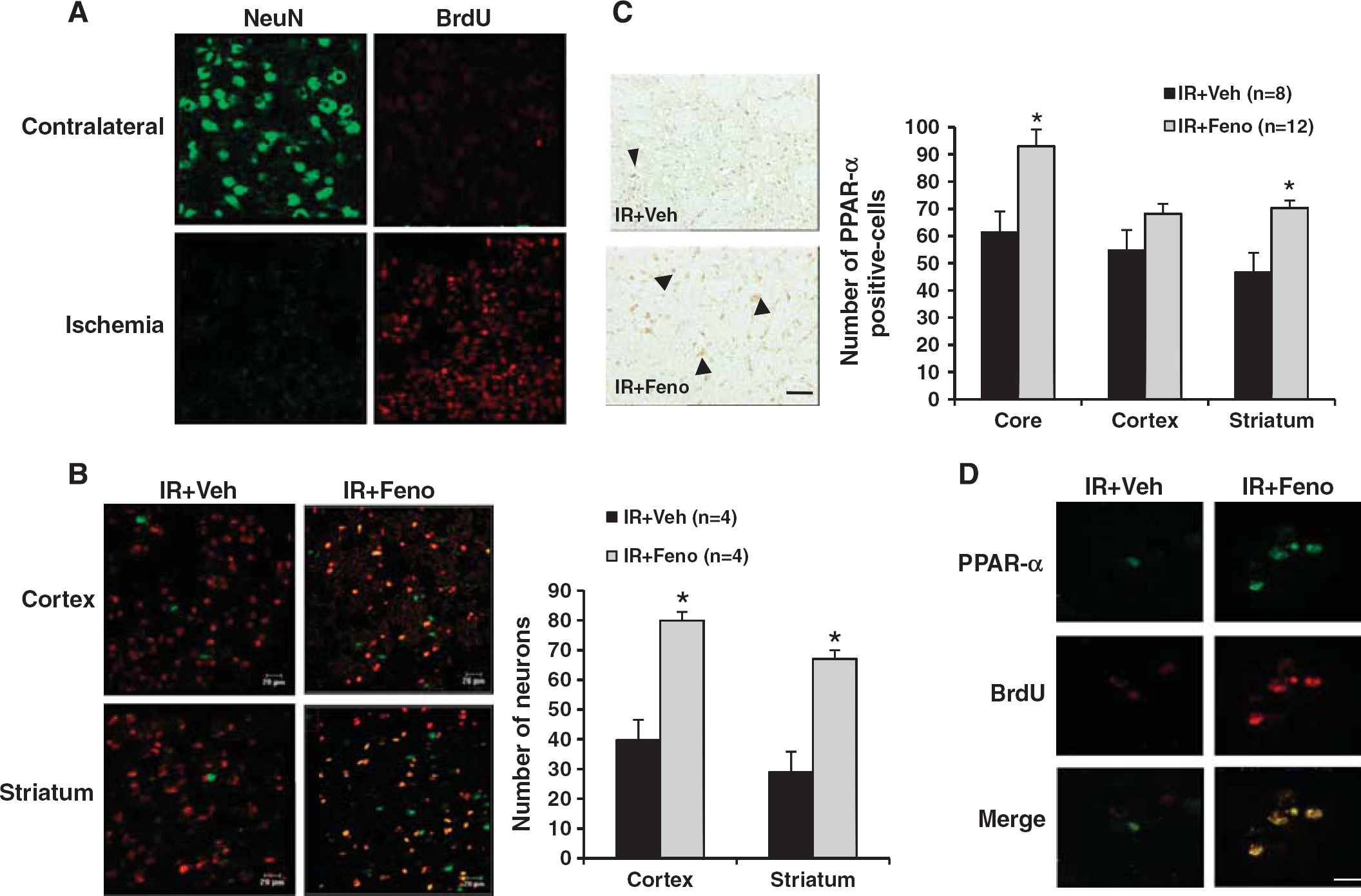
Evaluation 7 days after the induction of cerebral ischemia of the peroxisome proliferator-activated receptor-
Fenofibrate Prevents the Postischemic Memory Expression of Amyloid-β Peptide
During the 7 days after brain ischemia, depositions of amyloid-
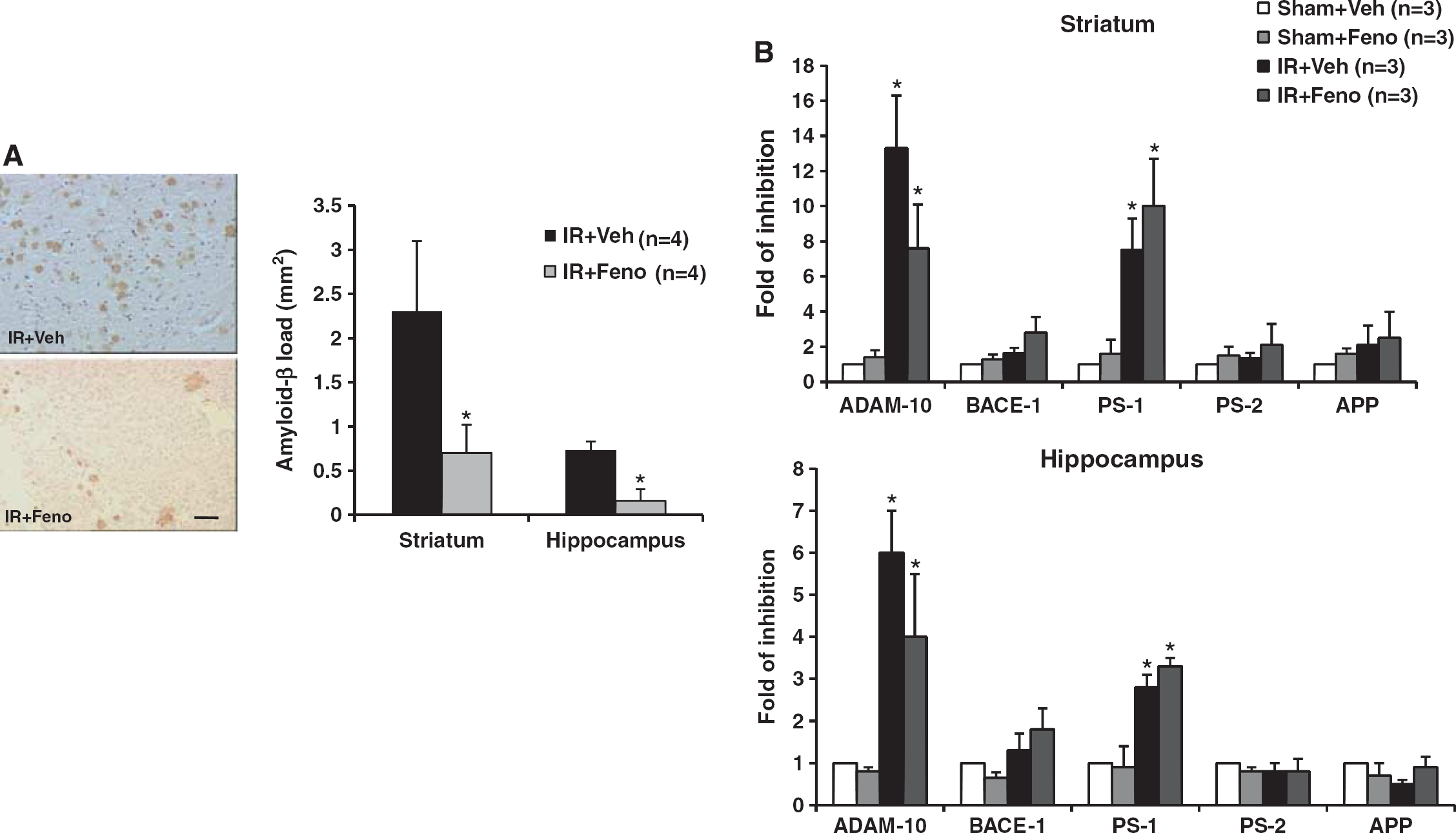
Effects of treatment with the peroxisome proliferator-activated receptor-
DISCUSSION
We show for the first time that fenofibrate administration during the acute phase of experimentally induced brain ischemia has beneficial immediate and short-term effects. Early and delayed administration of fenofibrate (relative to the onset of ischemia) induces a significant decrease in the infarct size and countered functional impairments in brain ischemia model. This effect appears to be dependent on the dose of fenofibrate. In parallel, and according to the dose of fenofibrate, we observe an associated vasculoprotective effect. The prevention of the endothelial dysfunction observed in the first 72 hours is accompanied by (1) the prevention of anomalies in leukocyte-endothelium interactions induced by the ischemic process, and (2) a decrease in the tissue inflammation resulting from microglial activation and parenchymatous infiltration by polynuclear neutrophils. Short-term monitoring of ischemic animals shows that fenofibrate increases postischemic neurogenesis, favors functional recovery, prevents the appearance of postischemic amyloid deposition, and has a beneficial effect on ischemia-induced learning impairments.
The study of dose-effect response to fenofibrate treatment showed different actions on neuronal and vascular compartments. We observed a dose-dependent effect on infarct volume in parallel to an improvement in motor function. Preventive neuroprotective effect of the fenofibrate has already been shown (i.e., administration preceding the induction of brain ischemia).12, 26, 27 These effects of fibrates are completely absent in PPAR
Fenofibrate's neuroprotective effect is associated with modulation of the vascular consequences of brain ischemia, in terms of endothelial function anomalies and leukocyte–endothelium interactions. This modulation of the vascular consequences of ischemia fits with the known vascular expression of PPAR-
The mobilization of neutrophils actively participates in the deleterious effects of systemic inflammation in cerebral ischemia.
31
Inhibition of leukocyte–endothelium interactions also accounts for some of the parenchymatous effects of fenofibrate—particularly the infiltration of the ischemic area by the PMNs responsible for local inflammation or oxidant release. However, fenofibrate seems also to exert a direct effect on the microglial activation in the brain parenchyma, related to glial expression of PPAR-
Early administration of fenofibrate improves postischemia motor performances. This improvement is maintained even after a delayed and prolonged administration of fenofibrate. Moreover on the cognitive level, treatment by fenofibrate during 7 days after ischemia prevents the postischemic memory impairment. Functional recovery is associated with an infarct volume decrease. The potential value of fenofibrate is further reinforced by our demonstration of the drug's short-term beneficial effects. In addition to effects in the early phases of ischemia, fenofibrate is able to upmodulate the brain tissue reorganization responsible for functional recovery and brain repair. It is known that spontaneous, postischemic neurogenesis results from the activation and differentiation of stem cells of neural or hematopoietic cells. This process is partly mediated by neurovascular interactions but has a low yield. Fenofibrate increases the neurogenesis observed well after ischemia. A PPAR-
In summary, we have shown for the first time that acute administration of fenofibrate exerts several different effects in the aftermath of brain ischemia. In the early phase, fenofibrate reduces infarct size. At later time points, the drug favors brain repair and minimizes the impact of brain ischemia on the amyloid cascade that contributes to the occurrence of poststroke dementia. All these properties suggest that fenofibrate and PPAR-
Footnotes
The authors declare no conflict of interest.
ACKNOWLEDGMENTS
The authors thank the platform of Imagerie et Biophotonique Cellulaire Fonctionnelle (Interdisciplinary Research Institute, Parc de la Haute Borne, Villeneuve d'Ascq, France) and Dave Trinel. The authors are grateful to the imaging platform of the Institut de Medecine Predictive et de Recherche Thérapeutique (IFR114) and to Florent Auger. The authors thank Dr Luc Humbert and Dr Jean-François Wiart (engineers from the Laboratoire de Toxicologie and Génopathies, Centre de Biologie, Bd du Professor J Leclercq 59037 LILLE) for the fenofibric acid assays.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
