Abstract
In aged humans, stroke is a major cause of disability for which no neuroprotective measures are available. In animal studies of focal ischemia, short-term hypothermia often reduces infarct size. Nevertheless, efficient neuroprotection requires long-term, regulated lowering of whole-body temperature. Previously, we reported that post-stroke exposure to hydrogen sulfide (H2S) effectively lowers whole-body temperature and confers neuroprotection in aged animals. In the present study using magnetic resonance imaging, electroencephalogram recording, DNA arrays, reverse transcriptase polymerase chain reaction, western blotting and immunofluorescence, we characterized the central nervous system response to H2S-induced hypothermia and report, for the first time, that annexin A1, a major pro-inflammatory protein that is upregulated after stroke, was consistently downregulated in polymorphonuclear cells in the peri-lesional cortex of post-ischemic, aged rat brain after 48 hours of hypothermia induced by exposure to H2S. Our data suggest that long-term hypothermia may be a viable clinical approach to protecting the aged brain from cerebral injury. Our findings further suggest that, in contrast to monotherapies that have thus far uniformly failed in clinical practice, hypothermia has pleiotropic effects on brain physiology that may be necessary for effective protection of the brain after stroke.
Introduction
To minimize the incapacitating sequelae of stroke, a promising focus of research is on neuroprotective strategies that minimize functional impairment by preventing the death of neurons, which continues for days to weeks after the acute event. Despite encouraging results from experiments with young animals, human stroke trials of neuroprotectants that should act after thrombolysis to limit infarct expansion and to promote tissue recovery have not yielded satisfactory results (Sacco et al, 2007). One possible explanation for this discrepancy between laboratory and clinical outcomes is the role that age has in the recovery of the brain from insult (Popa-Wagner et al, 2010). In this regard, the aged post-acute animal model is clinically most relevant to stroke rehabilitation (Badan et al, 2003).
A viable alternative to conventional drug-based therapies is physical cooling, or hypothermia, either confined to the head or including the entire body. Mild to moderate hypothermia has been shown experimentally to reduce neuronal death after experimental ischemia (Schwab et al, 1998; Li and Wang, 2011), and prolonged, whole-body hypothermia achieved by physical methods improves motoric function and reduces infarct volume (Colbourne et al, 2000; Corbett et al, 2000). However, studies of hypothermia have been hampered by the lack of a simple method to pharmacologically induce long-term, regulated lowering of whole-body temperature. Recently, it was reported that hydrogen sulfide (H2S), a weak, reversible inhibitor of oxidative phosphorylation, induces a suspended animation-like state in mice by lowering body temperature to 15 °C during an exposure time of 6 hours at an ambient temperature of 13 °C (Blackstone et al, 2005). Using a modified version of this procedure, we previously showed that post-stroke exposure of aged rats to H2S-induced hypothermia for 48 hours results in a 50% reduction in infarct volume without causing obvious neurological or physiological side effects (Florian et al, 2008).
A prerequisite for the successful translation of neuroprotective treatments from animal studies to clinical use is a more detailed understanding of the cellular and molecular mechanisms underlying the beneficial effects of long-term hypothermia for stroke treatment. Previously, we found that the phagocytic activity of brain macrophages in the first 3 days post stroke may contribute to the early, rapid development of the infarct in aged animals (Badan et al, 2003). Dysregulation of the inflammatory response in the aged brain thus may be one reason for the severity of the damage as well as the brain's resistance to neuroprotective therapies in the elderly. To identify regulated proteins whose function is related to inflammation after stroke, we used genomic, proteomic, and cell biological approaches to analyze RNA and protein expression profiles at day 2 after focal ischemia in aged rats. Here, we identify annexin A1 (ANXA1) as one of the major upregulated pro-inflammatory proteins after stroke, and report for the first time that prolonged gaseous hypothermia lowers ANXA1 expression, and hence phagocytosis, in the aged rat brain after stroke.
Materials and methods
Animals and Experimental Design
The subjects of these experiments were aged male Sprague-Dawley rats (N=43; 17 to 18 months of age; 520 to 600 g) kept under standard laboratory conditions with free access to food and water (except under specific conditions, below). Animals were assigned to two groups: group 1 (N=17) with middle cerebral artery occlusion (MCAO); and group 2 (N=15) with MCAO plus hypothermia. Of these, seven rats in each group were used for biochemical analysis and eight rats were used for histological analysis. Six animals were used for electroencephalogram (EEG) recordings. A small group of animals served as sham controls (N=5). No animals died during the experiment, but two rats from the experimental group were removed because of failed surgical procedures. All experiments were approved by the Animal Experimentation Ethics Board of the State of Mecklenburg-Vorpommern as meeting the ethical requirements of the German National Act on the Use of Experimental Animals (approval no 7221.3-1.1-011/12).
Reversible Occlusion of the Middle Cerebral Artery
At 18 hours before surgery, the rats were deprived of food to minimize variability in ischemic damage that can result from varying plasma glucose levels. Water remained available at all times. In all cases, surgery was performed between 0900 and 1300 hours.
Animals from the control and experimental groups were randomly subjected to cerebral infarction that was induced by the focal interruption of blood flow by transiently lifting the middle cerebral artery with a tungsten hook, as previously described (Popa-Wagner et al, 2010). Throughout surgery, anesthesia was maintained by spontaneous inhalation of 1% to 1.5% isoflurane in a mixture of 75% nitrous oxide and 25% oxygen. Body temperature was maintained at 37 °C by a Homeothermic Blanket System (Harvard Apparatus, Hugo Sachs Elektronik, March-Hugstetten, Germany) and the tail artery was catheterized to allow the continuous measurement of blood pressure and the withdrawal of blood samples for determination of pH and blood gases (Blutgassystem IL 1620, Instrumentation Laboratory, Munich, Germany), as well as arterial glucose levels (Omnican7 Balance, B. Braun, Melsungen, Germany). Local changes in blood flow were monitored using a laser Doppler device (Perimed, Stockholm, Sweden), and blood gases were measured at several time points during ischemia. A decrease in the laser Doppler signal to <20% of control values was considered to indicate successful MCA occlusion. After 90 minutes, the hook was released and the common carotid arteries were reopened.
Experimental H2S-Induced Hypothermia
At 1 hour after the resumption of blood flow, individual rats from the second group were exposed for 48 hours to an atmosphere containing 70 ppm hydrogen sulfide (H2S) and 19.5% O2, achieved by mixing room air with 5000 ppm H2S-balanced nitrogen at a flow rate of 3 L/min, as previously described (Florian et al, 2008). After 2 hours, the concentration of H2S was reduced to 50 ppm (the toxicity threshold for H2S is 80 ppm). The temperature outside the experimental box was maintained at 21 °C in a well-ventilated room. Water was available during this period, although no appetitive activity was observed. Carbon dioxide, O2, and H2S were measured continuously using appropriate gas detectors (GfG, Dortmund, Germany) placed directly in the cage.
Telemetric EEG Recordings
Telemetric recordings were carried out in six rats with MCAO (three exposed to H2S and three controls) using a CTA-F40 PhysioTel radiotelemetric transmitter from Data Sciences International (DSI, St Paul, MN, USA) that allowed simultaneous recording of one biopotential EEG channel (1 to 50 Hz band) together with a channel for activity (i.e., mechanical movement) and one for temperature. Cortical EEG recordings (referred to hereafter as EEG) were made from an epidural electrode placed over the ipsilateral parietal cortex (negative) against a contralateral reference electrode placed rostrally (positive), yielding a ‘pseudo-unipolar’ 1 channel montage for the affected hemisphere. The amplified signals were acquired at 250 Hz resolution using the Dataquest A.R.T.3.0 Acquisition software (DSI Inc.).
Data were exported off-line for further analysis in MATLAB (MathWorks 2011a, Natwick, MA, USA). The 10-second EEG segments were filtered through a 0.5 to 30.5-Hz band-pass third-order Butterworth digital filter using a zero-phase forward and reverse algorithm, symmetrically zero-padded to the next power of 2, and then windowed (Hamming) to reduce edge transients. The power spectral densities calculated by Fast Fourier Transformation were then combined in 1 Hz narrow bands (i.e., the 4 Hz band was the sum of Fourier power spectral densities between 3.5 and 4.5 Hz). The relative band power was calculated against the total power of the 1 to 30 Hz bands.
Magnetic Resonance Imaging
Magnetic resonance imaging (MRI) was used to visualize the volume of the lesion for both groups. Magnetic resonance imaging measurements were performed on a 7T Bruker ClinScan magnet with a 20 cm inner bore, capable of 290 mT/m in 250 μs (Bruker BioSpin MRI, Ettlingen, Germany) during the animals’ day cycle, at 50 hours (for experimental group 1) after MCAO. Images were received by a 2 × 2 phased array RF coil, designed specifically for rat brain studies, that was placed directly on the skull. The animals were anesthetized during imaging to minimize discomfort. Respiratory rate was monitored, and isoflurane concentrations were varied between 1.5% and 2.0% to keep the respiratory rate between 35/minutes and 45/minutes. After positioning the animal's head, quantitative T2 measurements were performed with a multislice spin-echo sequence with 25 slices of 0.7 mm thickness and a matrix size 640 × 640 pixels, a repetition time of 4330 ms, and an echo time of 45 ms.
Lesion Measurement using MIPAV Software
T2WI lesion volumes were determined using the image processing software MIPAV (Medical Image Processing, Analysis and Visualization, version 3.0, National Institutes of Health, Bethesda, MD, USA). After optimal adjustment of contrast, the edge of the lesions was traced manually on each of the 25 coronal slices, which covered the middle cerebral artery territory in all animals. The areas of hyperintensity were then summed and multiplied by the slice thickness to calculate lesion volumes.
Tissue, RNA and Protein Analysis
Subsequent to a survival time of 52 hours, the rats were deeply anesthetized and perfused with buffered saline followed by buffered, 4% freshly depolymerized paraformaldehyde. The brain was removed, post-fixed in buffered 4% paraformaldehyde for 24 hours, cryoprotected in 20% sucrose prepared in 10 mmol/L phosphate-buffered saline (PBS), flash-frozen in isopentane, and stored at −70 °C until sectioning. For total RNA and protein isolation, the rats were perfused with buffered saline only, and the brain was cut into 2 mm slices that were dipped in 2% 2,3,5-triphenyltetrozolium chloride solution so that the infarct core could be visualized and microdissected under a dissecting microscope as previously described (Florian et al, 2008).
RNA Isolation
Total RNA was isolated using TRIzol reagent (Invitrogen Life Technologies, Karlsruhe, Germany), as described by the manufacturer, followed by DNase 1 (Ambion, Invitrogen, Karlsruhe, Germany) digestion, and further purification using the RNeasy Mini extraction kit (Qiagen, Hilden, Germany). Purified total RNA was used for complementary DNA (cDNA) array assay and real-time PCR quantification.
Complementary DNA Array Assay
To analyze genes implicated in signal transduction, we used custom cDNA arrays containing 200 genes whose function is related to metabolic processes including phagocytosis (SuperArray, Bethesda, MD, USA) according to the manufacturer's instructions. Each individual array contains four housekeeping genes and one negative control. Housekeeping genes were included to confirm the integrity of RNA and to standardize the loading of different samples. After 1 hour prehybridization, membranes were hybridized with denatured biotin-labelled complementary RNA overnight, washed, incubated with streptavidin-alkaline phosphatase conjugate, and exposed to X-ray film. The scanned images were processed using the GEArray Expression Analysis Suite software (Superarray), the data were extracted, and gene expression profiles were analyzed. Data are presented as fold change, which is calculated as the ratio of experimental condition to control after normalization to housekeeping genes. The gene expression fold change was expressed as the mean of at least two experiments. Only those genes whose expression was equal to or greater than a 2-fold change were considered to be differentially regulated.
Real-Time Quantitative PCR
For real-time PCR, 2 μg of total RNA was reverse transcribed using random hexamers and the reverse transcription reagents supplied by Superarray. The PCR reaction was set up by mixing 10 ng of cDNA, primers, and Master mix (QuantiTect SYBR Green, Qiagen), and real-time PCR amplification was performed as follows: one cycle of 15 minutes at 95 °C and 45 cycles in three steps each (95 °C for 30 seconds, 58 °C for 30 seconds, and 72 °C for 30 seconds) using a real-time PCR cycler (MyiQ Cycler, Bio-Rad, Munich, Germany). A standard curve was generated by plotting the log10 [target dilution] of template on the x-axis against the Ct value from serial dilutions of target DNA on the y-axis. The efficiency of PCR amplification was 98%. The relative expression level of ANXA1 and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (as the housekeeping gene) was determined based on the standard curve equation generated for each individual gene (Junker et al, 2007).
To normalize, the expression level of ANXA1 was divided by the expression level of GAPDH from the same sample. Fold change= [(ANXA1/GAPDH)hypothermic conditions]/[(ANXA1/GAPDH)normothermic conditions].
Two-Dimensional Gel Electrophoresis and Western Blotting
For protein analysis, dissected tissues from the peri-infarcted area, the corresponding contralateral area, and from the sham controls were homogenized in TRIzol reagent (Invitrogen, Karlsruhe, Germany). Sample preparation was performed according to the manufacturer's protocol. Finally, the vacuum-dried protein pellet was dissolved in sample buffer containing 8 M urea, 2 M thiourea, 4% CHAPS, 65 mmol/L DTT, and 40 mmol/L Tris. Total protein was quantified by the Bradford method, and isoelectric focusing was carried out as previously described (Junker et al, 2007). The gels from at least two repeats were then scanned and processed by Delta2D (DECODON, Greifswald, Germany), a 2-DE gel analyzing and spot matching software. Using this software, the images from the peri-infarct (pi, blue) and contralateral area (cl, orange) gels were arbitrarily assigned different colors and matched. After matching, equal volumes of blue and orange spots appear black, which indicates that the protein represented by these spots has no expression change. Otherwise, the protein was considered to be differentially expressed and marked as a candidate protein, and further analyses of spot volume and identification of the spot were conducted.
For western blotting, 20 μg of protein were separated by 12% sodium dodecyl sulfate-PAGE and transferred to a polyvinylidene difluoride membrane by semi-dry blotting. Annexin A1 was detected with a rabbit polyclonal antibody raised against amino acids 235 to 299 of annexin I of human origin (H65) (1:100 dilution, Santa Cruz, Heidelberg, Germany) for 2 hours, followed by washing and binding of secondary peroxidase-conjugated donkey anti-rabbit IgG (H+L) (Jackson Immunoresearch Laboratories, West Grove, PA, USA) for 1 hour. Finally, the antigen–antibody complex was visualized using the ChemiDoc XRS from BioRad.
Mass Spectrometry
In-gel digestion was performed as previously described (Junker et al, 2007).
The resulting MS/MS data were analyzed with the Bioanalyst Software (Applied Biosystems, Foster City, CA, USA) and the integrated Mascot script. Peak lists were compared with the SwissProt database using the Mascot search engine (Matrix Science, London, UK). Peptide mixtures that yielded a Mowse score of at least 59 were regarded as positive identifications.
Immunohistochemistry
Sections (25 μm thick) were cut on a freezing microtome and processed for immunohistochemistry as free-floating material, as previously described (Florian et al, 2008). Briefly, after incubation with blocking solutions containing 3% donkey serum/10 mmol/L PBS/0.3% Tween 20, tissue sections were exposed overnight at 4 °C either to mouse anti-NeuN (1:1000, Millipore, Schwalbach, Germany) or to rabbit polyclonal antibody to ANXA1 (1: 200, Santa Cruz) diluted in PBS containing 3% normal donkey serum and 0.3% Tween 20. After extensive washing in PBS containing 0.3% Tween, sections were incubated overnight at 4 °C with biotinylated donkey anti-mouse IgG or donkey anti-guinea pig IgG (Jackson ImmunoResearch Laboratories) diluted 1:4000 in PBS containing 1% normal donkey serum and 0.3% Tween 20. After washing in PBS, sections were incubated for 4 hours at room temperature in ABC Elite reagent (Vectastain Elite Kit, Vector, Linaris, Wertheim, Germany) diluted 1:100 in PBS containing 0.3% Tween 20. The antibody complex was then visualized with 0.025% 3′,3′ diaminobenzidine and 0.005% hydrogen peroxide in 100 mmol/L Tris buffer (pH 7.5).
Cellular Phenotyping by Immunofluorescence
For phenotyping, sections were blocked and incubated overnight at 4 °C with the following antibodies: rabbit polyclonal antibody raised against amino acids 235 to 299 of annexin I of human origin (H65) (1:100 dilution, Santa Cruz); goat anti-annexin A1 (1:100 dilution, Santa Cruz), mouse anti-ED1-FITC, a marker for macrophages (1:100, Serotec, Duesseldorf, Germany); rabbit anti-PMN-FITC (1:500; BD Pharmingen, Heidelberg, Germany); or mouse anti-rat CD11b (1:100; Serotec). Cells were visualized by adding Cy2-conjugated donkey anti-rabbit IgG (H+L) (1:4000), Cy5-conjugated donkey anti-goat IgG (H+L) (1:4000), or Cy5-conjugated donkey anti-mouse IgG (H+L) (1:3000). Fluorescence signal was assessed on a Leica confocal microscope TCS SP5 as described previously (Popa-Wagner et al, 2010).
Statistical Analysis
The main effect of treatment (hypothermia) was evaluated by using the Mann–Whitney U test. The level of significance (two-tailed threshold) was set at P≤0.05. Data are presented as means s.e.m. for the EEG data and as mean±s.d. for all other data, as indicated for each figure.
Results
Post-Stroke Hypothermia
Exposure of aged rats to an atmosphere containing a low dose of H2S after stroke led to a gradual decrease in whole-body temperature, which stabilized at 31±0 5 °C after 8 hours (Figure 1B, filled circles). After 48 hours, the animals were returned to normal atmospheric conditions. The animals recovered within minutes and did not show any signs of neurological or physiological deficits related to the H2S-induced hypothermia.
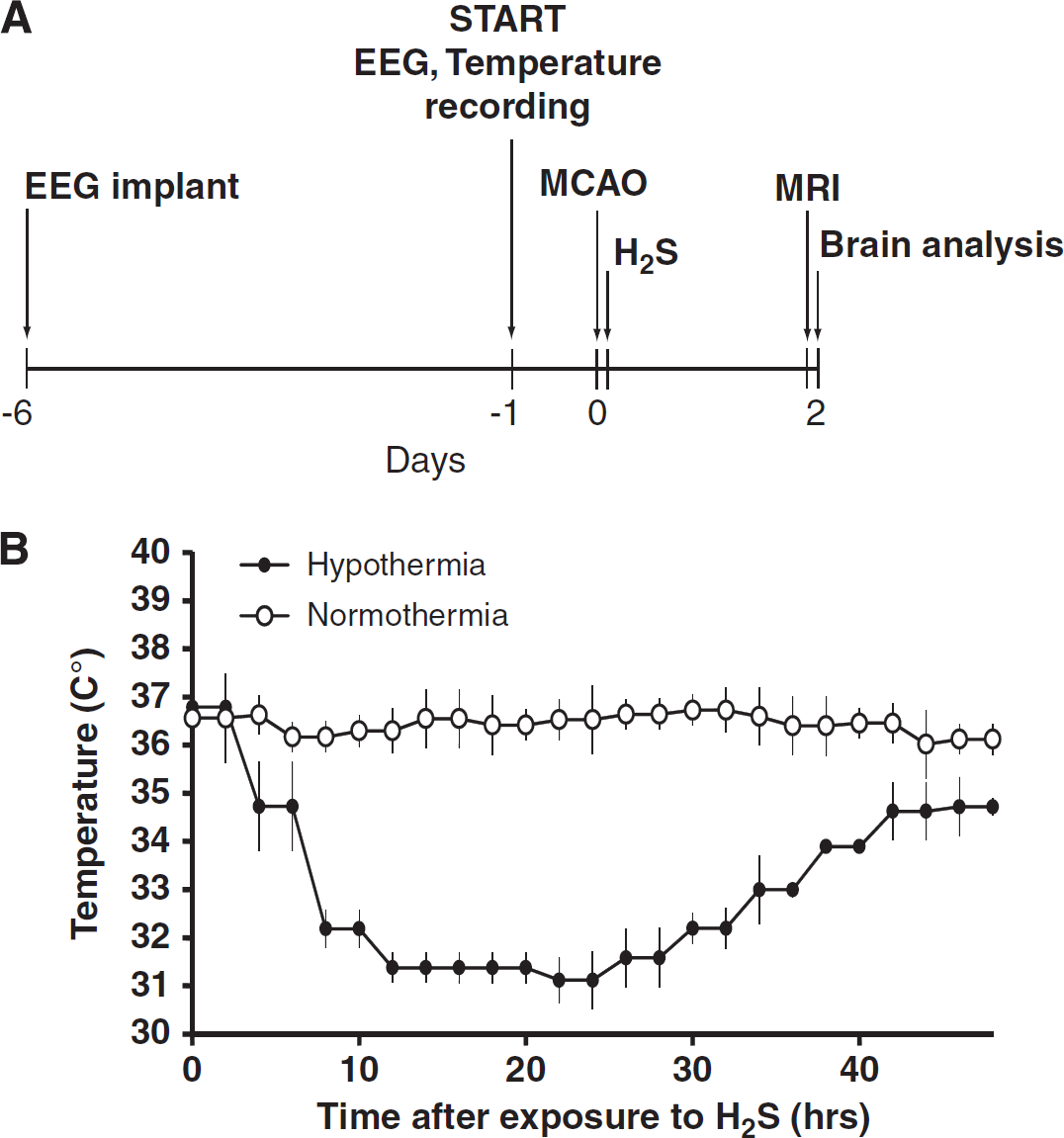
(
Power-Down of Background EEG During H2S-Induced Cooling
Telemetric recordings were started 15 to 30 minutes after the completion of MCAO surgery to allow the effects of isoflurane anesthesia to wear off. A representative recording for a rat subjected to H2S is presented in Figures 2A–F and the corresponding group means are shown in Figures 2G–J.
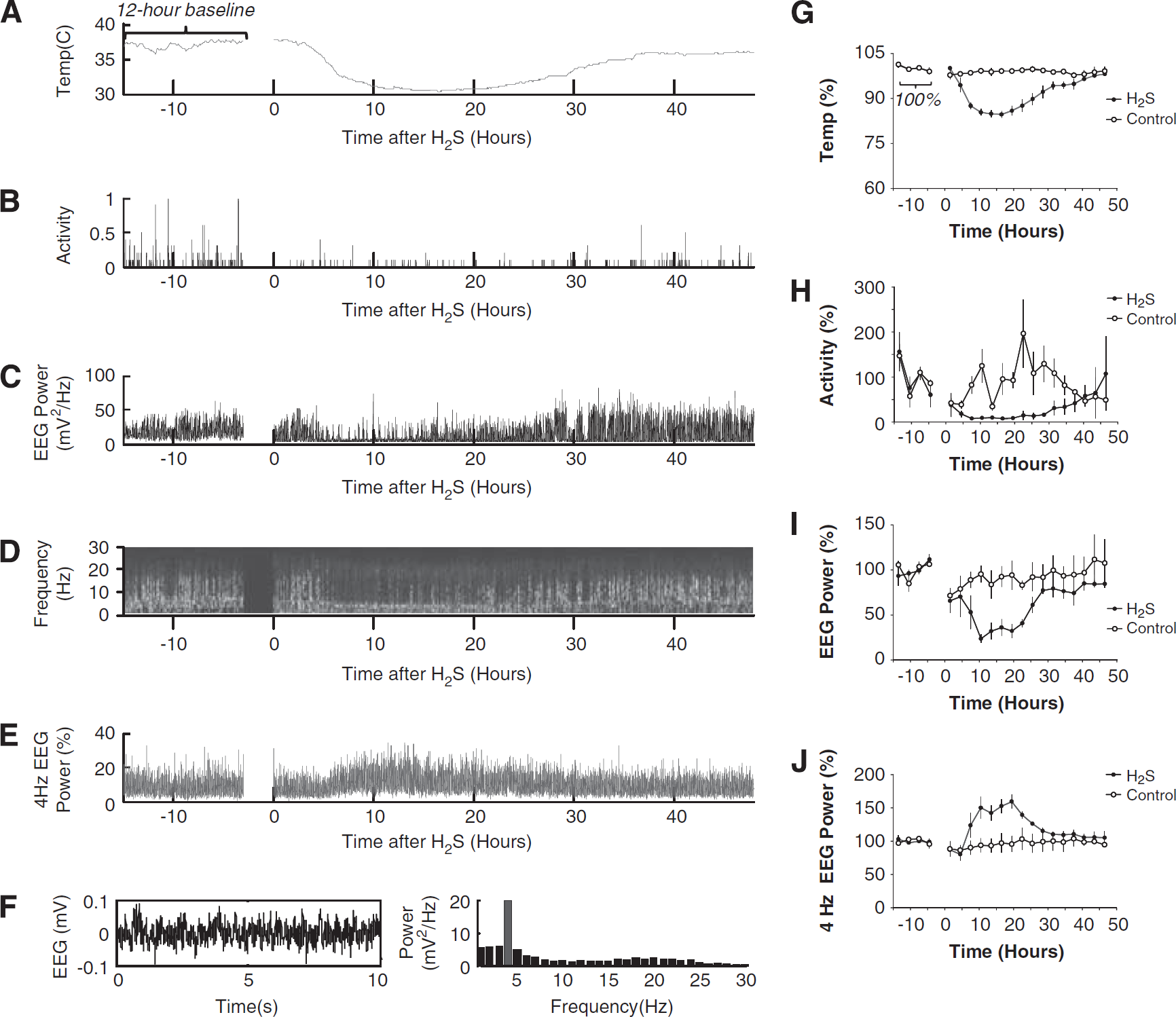
Electroencephalogram (EEG) telemetry data. A representative telemetric recording for a rat subjected to H2S is presented in
Within the first 3 hours, the EEG power over the ischemic hemisphere of control rats showed a progressive but incomplete recovery, attaining 70% of the pre-MCAO baseline values (Figures 2C, 2D, and 2I). This electroencephalographic recovery was associated with a recovery in activity up to 50% of the pre-MCAO baseline (Figures 2B and 2H).
In the rats exposed to H2S, the initial post-MCAO recovery was only transient. The fall in temperature that started after 3 hours of H2S exposure was followed by a marked reduction in EEG power and activity. After 12 hours of H2S exposure, the reduction in temperature by 15% (Figures 2A and 2G) was paralleled by a reduction in EEG power by as much as 80% (Figures 2C and 2I) and a reduction in activity to nearly 0 (Figures 2B and 2H). During this ‘power-down’ state, the normal sleep–wake EEG cycles were replaced by a rhythmic, low-amplitude oscillatory activity of ∼4 Hz (Figures 2D-F). This 4 Hz activity was not observed during slow–wave sleep episodes before MCAO or after MCAO in rats not exposed to H2S, and was distinct from the previously reported ‘post-ischemic EEG slowing’ variants in rats (Hartings et al, 2003). Furthermore, the 4 Hz activity appeared to be continuous over several hours and could not be explained by the seizure activity that typically occurs after MCAO in rats as isolated 1 to 3 Hz, high-amplitude spike episodes lasting ∼1 minute (Hartings et al, 2003). The 4 Hz activity was, however, remarkably similar to that recorded in hibernating animals during torpor (Deboer, 1998). Thus, it is reasonable to infer that the ‘power-down’ state induced by H2S in rats was similar to hibernation.
In spite of continuing H2S exposure, the ∼31 °C cooling plateau could not be maintained, as the temperature started to increase after ∼24 hours. (Figures 2A and 2G). This was an unexpected observation; however, it was apparent to a various extent in all recordings, and possible technical errors related to H2S exposure dose were carefully excluded in methodological control experiments (data not shown). The magnitude of the temperature variations during H2S administration was much larger than the circadian temperature variability measured pre-MCAO (Figure 2A). Furthermore, the increase in temperature was followed by the recovery of EEG power (Figures 2C and 2I), with disappearance of the dominant 4 Hz activity (Figures 2E and 2J) and emergence of normal sleep–wake cycles (Figure 2D) and activity (Figure 2H). Taken together, these findings indicate that the efficacy of metabolic inhibition by H2S decreased with time.
Infarct Size
Representative MRI data for the two rats closest to the mean for each group are shown in Figures 3A and 3B. Cortical lesion, as defined by the region of T2 hyperintensity, was reduced by hypothermic treatment (Figure 3B) compared with the normothermic group (Figure 3A). Prolonged H2S-induced hypothermia reduced lesion size by ∼57% (Figure 3C; P=0.001).
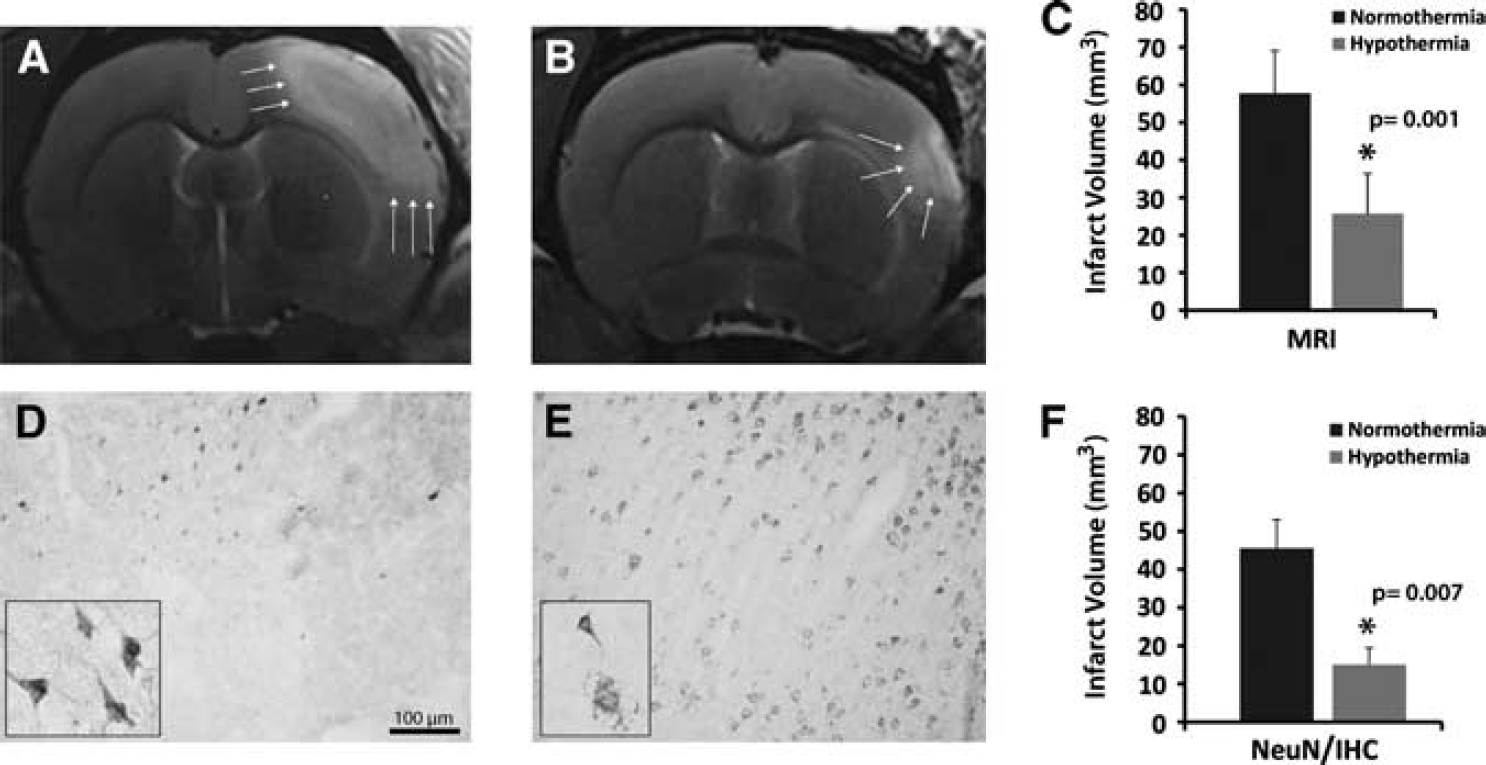
(
By immunohistochemistry with NeuN, a marker of neuronal nuclei that is used to reveal details of the infarcted area (Popa-Wagner et al, 2010), the infarct core appeared to be well-preserved in animals kept under hypothermia (Figure 3E) as compared to control animals, in which the infarct core was practically devoid of viable neurons (Figure 3D). By immunohistochemical analysis, the infarct volume was reduced by ∼67% (Figure 3F; P=0.007).
Post-Stroke Hypothermia Downregulates Annexin A1 in the Peri-Infarct Area
Overlapping of chemiluminescent images obtained by cDNA hybridization of complementary RNA from aged rats subjected to MCA occlusion revealed that hypothermia caused a strong downregulation of ANXA1 mRNA in the peri-infarct area of rats (Figure 4A; reddish spot in white square). To validate these results, we performed quantitative reverse transcriptase polymerase chain reaction. By quantitative reverse transcriptase polymerase chain reaction, we found a threefold upregulation in the expression of ANXA1 mRNA expression in untreated stroke animals (Figure 4B, gray column). This upregulation was abolished upon exposure to H2S (Figure 4B, black column; P=0.001).
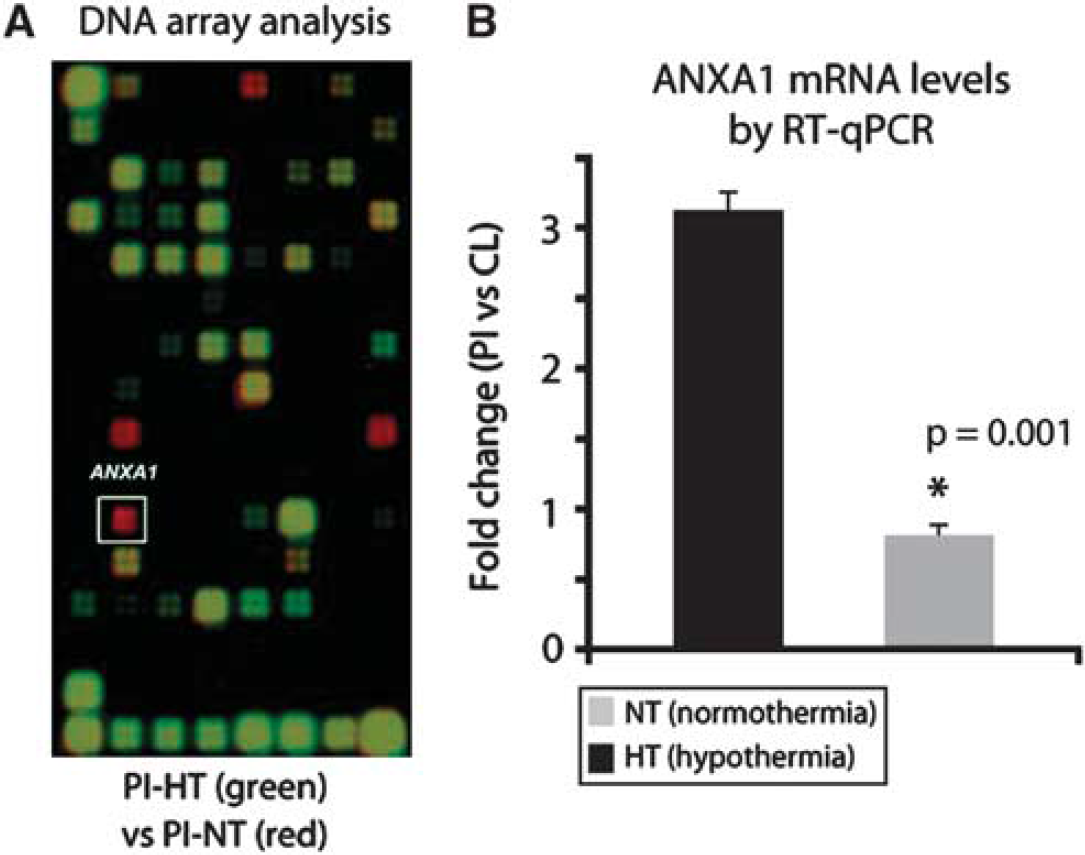
(
Early Upregulation of Annexin A1 in the Peri-Infarct Area After Stroke
To identify regulated proteins whose function is related to inflammation after stroke, we used proteomic methods to analyze protein expression profiles at day 2 after MCAO. An enlarged region of matched two-dimensional gel images is shown in Figure 5A. At day 2, one upregulated spot in the peri-infarcted area of post-stroke rats was identified as annexin 1 (ANXA1). No significant changes were detected in the contralateral hemisphere as compared with the sham-operated controls.
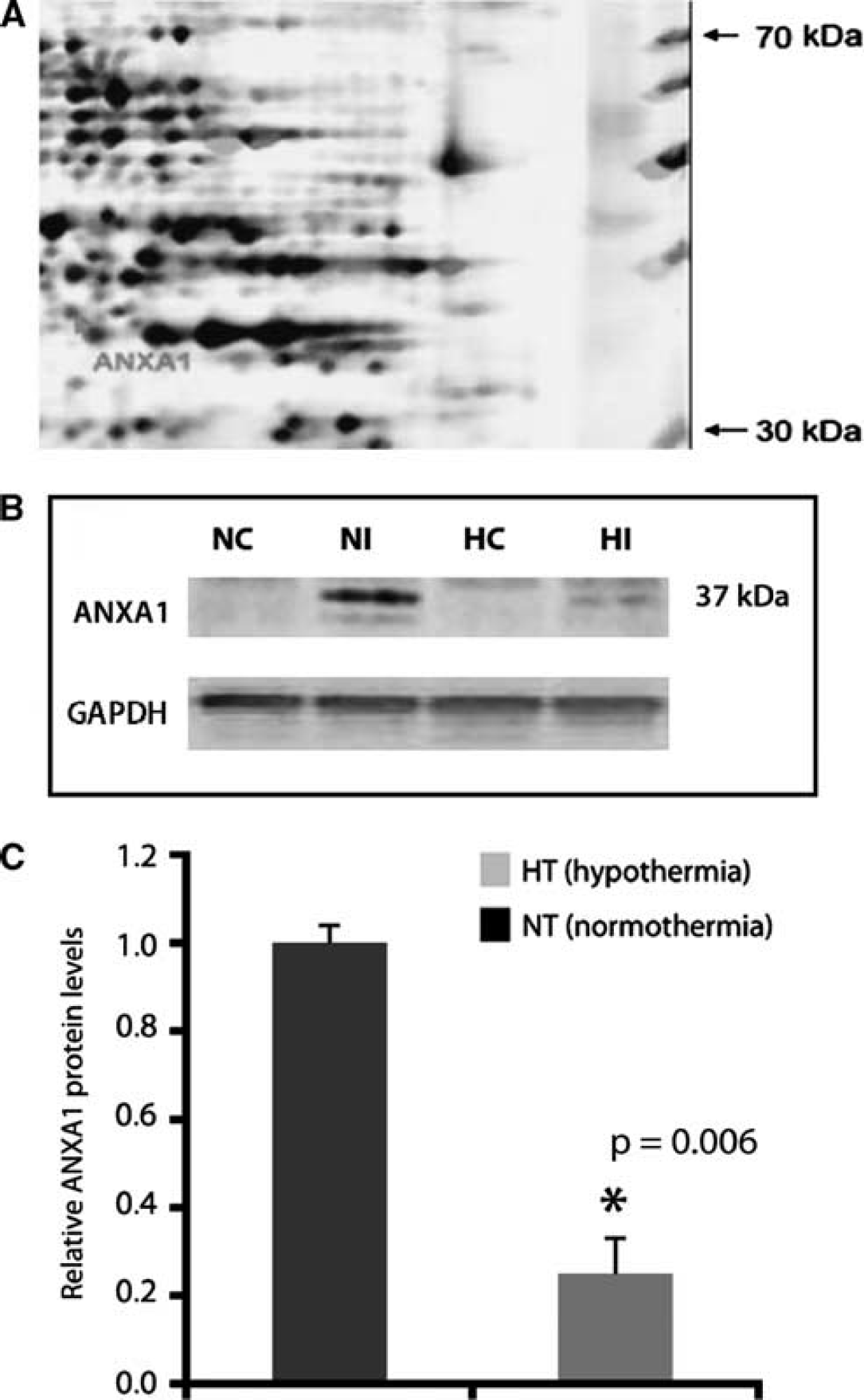
(
By western blot, one band located around 37 kDa was detected using a rabbit antibody raised against ANXA1 (Figure 5B). The major band corresponds to the native, biologically active 37 kDa protein. Quantification of ANXA1 protein expression indicated that hypothermia caused a strong downregulation of ANXA1 (Figure 5C, P=0.006).
The results from western blots were further substantiated at the cellular level. Using a rabbit polyclonal antibody to ANXA1, we found that immunostaining in the peri-infarcted area was associated with immune-like cells (Figure 5A, arrows). After 2 days of hypothermia, their number was greatly reduced (Figure 5B). The contralateral side was practically devoid of immunostaining (not shown). Quantification of ANXA1-positive cells indicates that hypothermia reduced the number of ANXA1-positive cells by 42% (Figure 5C; P=0.02).
Annexin A1 is Expressed by Polymorphonuclear Cells
To determine the phenotype of ANXA1-positive cells and to gain insight into the cellular function of the protein, we performed double-immunofluorescence staining using a rabbit antibody against polymorphonuclear cells (PMN, green) and a goat antibody raised against ANXA1 (red). We found that ANXA1 colocalized with PMN-positive cells (Figure 6D). To exclude other major cell phenotypes, additional double-immunofluorescence experiments using microglial markers ED1 and Iba1, as well as CD11b, were conducted. None of these markers colocalized with ANXA-1 (not shown).
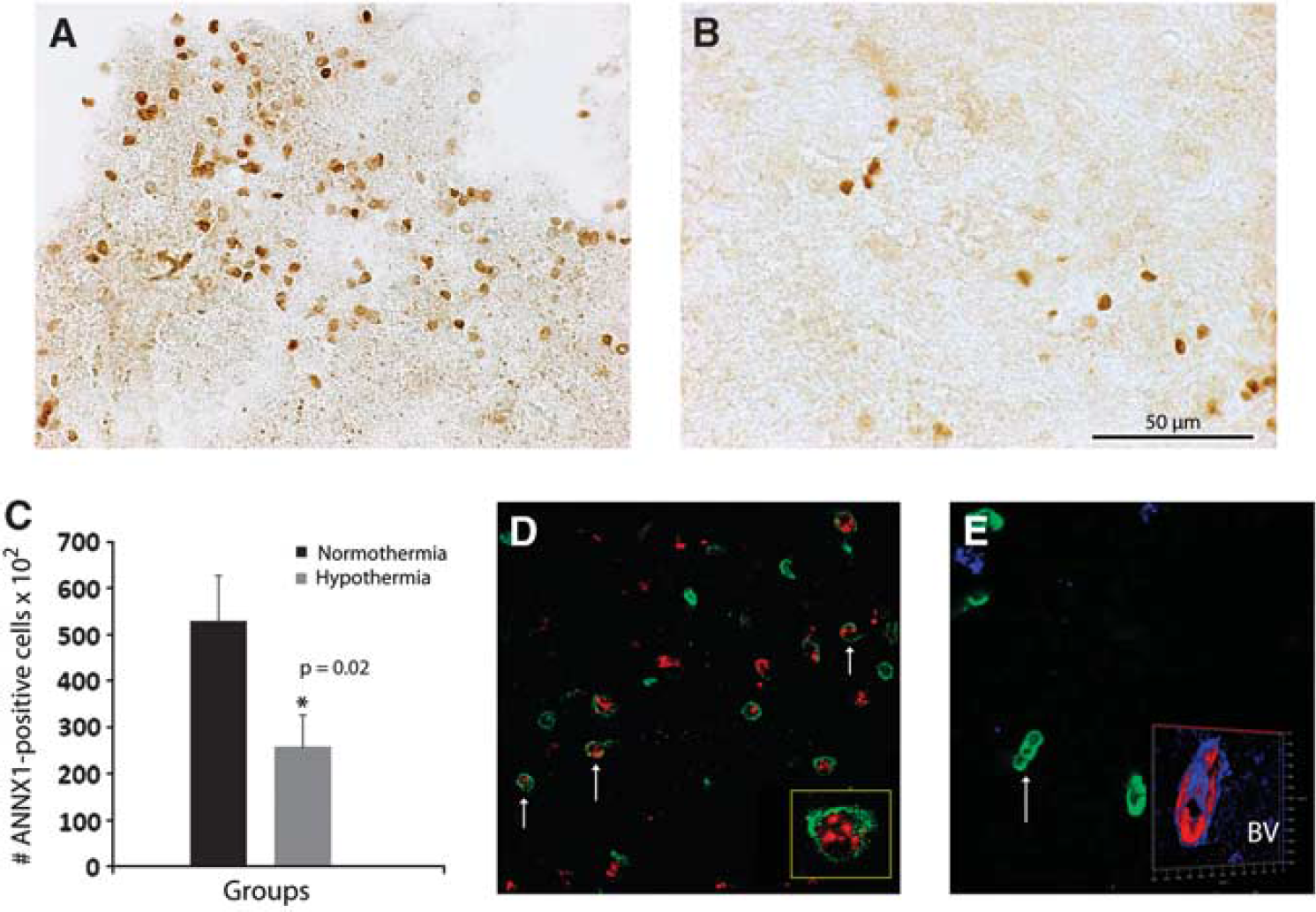
Phenotype and function of annexin A1 (ANXA1) in the rat brain after stroke. In control, normothermic rats, ANXA1 is expressed by immune-like cells (
Two days of hypothermia greatly diminished the number of colocalized PMN-ANXA1 cells. In fact, hypothermia was associated with a different cell morphology, i.e., the ANXA1-positive cells had an elongated appearance and sometimes the cells were clustered (Figure 6E, arrow). Occasionally, ANXA1 colocalized with blood vessels (Figure 6E, inset).
Discussion
In animal studies of focal ischemia, short-term hypothermia often reduces infarct size. However, efficient neuroprotection requires long-term, regulated lowering of whole-body temperature, and a detailed mechanistic analysis is needed to optimize the therapeutic benefits of this approach. In the present study, we undertook a comprehensive physiological, biochemical, immunohistochemical, and gene expression characterization of the brain response to long-term gaseous hypothermia, and identified ANXA1 as a prominent target of the anti-inflammatory actions of H2S-induced hypothermia in the peri-infarcted area of aged rat brains.
Physical brain cooling has been reported to improve recovery from traumatic brain injury in rats (Kuo et al, 2010) and from focal cerebral ischemia in baboons (Schwartz et al, 2011), but these methods are difficult to translate to the clinical situation. More recently, a 48-hours brain-selective cooling of the ischemic territory significantly mitigated injury and behavioral impairment (Clark et al, 2009). Short-term (2 hours) post-stroke extraluminal cooling of bilateral common carotid arteries also exhibited neuroprotective effects (Wei et al, 2008). As an alternative approach, gaseous hypothermia enables precise temperature control that is achieved by the simple inspiration of a mixture of air and H2S. The disagreeable odor of H2S can be circumvented by using injectable formulations that yield therapeutic doses of the gas; a number of these procedures are already in clinical trials (Jha et al, 2008).
Our findings indicate that the organism adapts to prolonged exposure to H2S and increasingly counteracts the pharmacological effects of the gas by gradually raising both temperature and blood pressure. One confounding factor regarding the effect of H2S on lowering both body temperature and blood pressure is the superimposition of the effects of the normal circadian rhythm, which is abolished in the first 10 to 15 hours of exposure by an as yet unknown mechanism, but which thereafter regains its rhythmicity. In addition, H2S is a physiological vasodilator that can influence blood pressure (Zhao et al, 2001; Cheng et al, 2004).
Power-Down of Background EEG During H2S-Induced Cooling
Although the first description of the hypometabolic state induced by H2S was referred to as a state of ‘suspended-animation’, the effects on electrocortical activity were not investigated (Blackstone et al, 2005). Here we report that, after exposure to H2S, the normal sleep–wake oscillations are replaced by a low-amplitude EEG dominated by a 4-Hz rhythmic activity, reminiscent of EEG recordings in hibernating animals (Deboer, 1998).
Disturbance of the sleep–wake cycle (i.e., by sleep deprivation) was found to have dual effects on ischemic neuroprotection. Although sleep deprivation for 12 hours post MCAO was found to aggravate ischemic damage in rats (Gao et al, 2010), brief sleep deprivation could also be neuroprotective (Moldovan et al, 2010).
A certain degree of sleep deprivation also accompanies other cooling methods. In an early study using the ‘automated fan and water spray system’ it was found that the procedural stress did not affect the post-ischemic outcome when cooling was counteracted (Colbourne and Corbett, 1995). Although the extent of sleep deprivation was not directly addressed, that study seems to indicate that procedural sleep deprivation does not have an important role in the neuroprotective effect of cooling. Nevertheless, this does not exclude the possibility that the decrease in neuronal activity induced by H2S is not in itself neuroprotective.
Various transient EEG patterns, including nonconvulsive seizures, periodic epileptiform discharges, and intermittent rhythmic delta activity in rat focal ischemia were found to occur after MCAO in rats (Hartings et al, 2003). Nevertheless, the 4 Hz activity observed in the present study appeared to be continuous during H2S exposure (Figure 2D and 2E), and did not appear in control animals (Figure 2F); hence, it could not be simply attributed to such transient patterns. Intriguingly, an increase in cortical oscillations close to 4 Hz was recently found to precede epileptic seizures in a rat model of absence epilepsy (Sitnikova and van Luijtelaar, 2009), raising the possibility that neocortical power-down by H2S in our study arrested the development of seizures with an additional neuroprotective effect. Nevertheless, occasional seizure discharges could still be identified after MCAO in rats exposed to H2S (data not shown), indicating that seizure activity was not abolished. Whether the amount of seizure activity was reduced during H2S exposure to an extent that may have been neuroprotective cannot be reliably answered by our current data. More detailed electrophysiological studies should be carried out to investigate the effects of H2S on seizure threshold, also in the absence of MCAO.
Prolonged Exposure to Hydrogen Sulfide-Induced Hypothermia Reduces Infarct Volume After Stroke
Several years ago, we introduced NeuN immunohistochemistry as a sensitive means of measuring infarct volume (Badan et al, 2003). In the current study, we were able to confirm the validity of this approach by MRI measurements that yielded highly similar values for the infarct volume, particularly if we take into account the fact that MRI is also measuring the volume of edema, which immunohistochemistry does not detect. As the infarct core had disintegrated in most aged control animals, we hypothesize that hypothermia delays neuronal death and promotes the preservation of the infarct core.
In spite of the obvious functional benefit and neuroprotective effect of reperfusion under mild hypothermic conditions, the mechanism of this treatment is not yet fully understood. The beneficial effects of hypothermia have been attributed to diminished excitotoxicty, neuroinflammation, apoptosis, free-radical production, seizure activity, blood–brain barrier disruption, blood vessel leakage, and/or cerebral thermopooling (Polderman, 2009).
There are two distinct windows of opportunity for the clinical use of hypothermia. In the early intra-ischemia period, hypothermia modulates abnormal cellular free-radical production, poor calcium management, and poor pH management. In the more delayed post-reperfusion period, hypothermia modulates the downstream necrotic, apoptotic, and inflammatory pathways that cause delayed cell death (Lampe and Becker, 2011). Recent studies have reported that targeting the inflammatory response to ischemic injury limits the expansion of the lesion and increases the survivability of neurons after stroke (Ajmo et al, 2008; Shichita et al, 2009). In this study, we have shown that H2S-induced hypothermia significantly reduces the number of PMN cells expressing ANXA1. Recently, it has been shown that downregulation of ANXA1 expression results in impaired phagocytosis and transient actin accumulation (actin flashing). These data identify ANXA1 as an important component of phagocytosis that appears to link actin accumulation to different stages of phagosome formation (Patel et al, 2011). The anti-inflammatory role of ANXA1 has been explained by its ability to prevent the infiltration of neutrophils when they adhere to inflamed endothelium. In stroke, the leakage of neutrophils into the infarct site could follow two pathways: (i) directly from the bloodstream via the disrupted blood–brain barrier; or (ii) infiltration of neutrophils that bind to the inflamed endothelium. Both mechanisms may be operating (Oliani et al, 2001; Popa-Wagner et al, 2010). Yet a third mechanism could be that hypothermia itself reduces the number of circulating leukocytes and may diminish their phagocytic capacity (Bouma et al, 2010).
ANXA1 is a small, glucocorticoid-inducible protein (37KDa), previously known as lipocortin 1, that has a role in several anti-inflammatory processes such as macrophage phagocytosis and neutrophil migration (Fan et al, 2004; Lim et al, 1998). Some studies also suggest a protective role for ANXA1 in central nervous system after ischemia and in neurodegeneration (Gavins et al, 2007; Rothwell et al, 1997). In the brain, ANXA1 is expressed in the ependymal cells lining the ventricular system of the healthy brain in adult rats and in the cells of the innate immune system (Solito et al, 2008). Microglial cells also express ANXA1 in the normal adult human and rodent brain (McArthur et al, 2010; Buckingham et al, 2006). Except for the spurious vascular localization of ANXA1 (due most likely to the adhesion of ANXA1-expressing cells), we could not confirm the colocalization of ANXA1 with the macrophage marker ED1. Other studies have suggested that, under normal physiological conditions, ANXA1 has a role in phagocytosis and the engulfment of apoptotic cells in the periphery (Arur et al, 2003; Parente and Solito, 2004). In our aged rat stroke model, the expression of ANXA1 was mostly restricted to PMN cells and, in remote areas relative to the infarct core, to the inflamed endothelium of the vasculature.
It thus appears that H2S-induced hypothermia has a pleiotropic effect by: (i) reducing the metabolic rate; (ii) inducing a hibernation-like state; (iii) reducing the epileptic forms of EEG activity; and (iv) inducing a sleep-deprivation state. Therefore, the ability of ANXA1 to control and contain inflammation may have a pivotal role in post-ischemic recovery.
Conclusions
To date, all monotherapeutic attempts to prevent or minimize brain damage after stroke have failed. In view of our findings that stroke disrupts the physiology, biochemistry, and gene expression of multiple central nervous systems in an age-dependent manner, the failure of therapies aimed at a single target system is perhaps inevitable. Our present results suggest that H2S-induced hypothermia, by simultaneously targeting multiple points of intervention, could have a higher probability of success in treating stroke. However, many questions still must be answered regarding the use of therapeutic hypothermia for ischemia in clinical practice, such as the H2S concentration, optimal target temperature and duration, the therapeutic window in humans, and cost effectiveness. In our experiments, the concentration of H2S was well below the toxicity limit for humans of 80 ppm. A more serious problem is the translation of these findings in rats to animals with a larger body size. Recently, it has been reported that inhaled H2S did not induce hypothermia in pigs (Drabek et al, 2011). However, as we have shown in this work, the beneficial effects of H2S may extend beyond the cooling effect. Therefore, a better understanding of the pathophysiology of the ischemic injury processes on which hypothermia and H2S act will serve to further promote the use of this promising method to reduce the mortality and morbidity caused by stroke.
