Abstract
Pregnancy increases the risk of several complications associated with the cerebral veins, including thrombosis and hemorrhage. In contrast to the cerebral arteries and arterioles, few studies have focused on the effect of pregnancy on the cerebral venous side. Here, we investigated for the first time the effect of pregnancy on the function and structure of the cerebral vein of Galen in rats. Our major finding was that cerebral veins from late-pregnant (LP, n = 11) rats had larger lumen diameters and thinner walls than veins from nonpregnant (NP, n = 13) rats, indicating that pregnancy caused outward hypotrophic remodeling of the vein of Galen. Moreover, veins from NP animals had a small amount of myogenic tone at 10 mm Hg (3.9 ± 1.0%) that was diminished in veins during pregnancy (0.8 ± 0.3%; P < 0.01). However, endothelium-dependent and -independent vasodilation of the veins was unchanged during pregnancy. Using immunohistochemistry, we show that the vein of Galen receives perivascular innervation, and that serotonergic innervation of cerebral veins is significantly higher in veins from LP animals. Outward hypotrophic remodeling and diminished tone of cerebral veins during pregnancy may contribute to the development of venous pathology through elevated wall tension and wall stress, and possibly by promoting venous blood stasis.
INTRODUCTION
Unlike the arterial side, a substantial portion (~70% to 80%) of cerebral blood volume resides in the veins. 1 Changes in venous outflow and venous pressure result in alterations of cerebral blood volume,2,3 intracranial pressure, and cerebral perfusion pressure.4,5 Thus, cerebral veins have an important role in control of cerebral hemodynamics under normal physiologic conditions. Additionally, the cerebral veins have been implicated in the development of several pathologic states. Thrombosis of cerebral veins and/or venous sinuses can cause serious neurologic complications such as venous infarction and intracranial hemorrhage. 6 Recently, venous flow disturbances were implicated in the pathogenesis of multiple sclerosis 7 and transient global amnesia. 8 Nevertheless, the venous compartment of the cerebrovasculature has received little attention in the past decades.
Notably, pregnancy increases the risk of several complications in the brain associated with veins. Pregnancy is a major risk factor for intracranial venous thrombosis6,9 and both pregnancy and the postpartum period are associated with an increased risk of stroke and cerebral hemorrhage. 10 These findings have prompted investigations on the mechanism through which the pregnant state predisposes women to cerebrovenous pathology. A hypercoagulable state is thought to be the major contributor to the development of pathophysiology of cerebral veins in pregnancy.6,9,10 However, a hypercoagulable state does not fully explain the occurrence of events such as cerebral hemorrhage, as this is more likely to result from rupture of the vessel consequent to changes in vessel structure. 11 Hence, pregnancy may provoke changes in the structure and biomechanics of cerebral veins, underlying the pathogenesis of certain venous complications.
It is well known that pregnancy causes significant hemodynamic changes, such as increased cardiac output and decreased vascular resistance. 12 These changes are necessary to accommodate a 40% to 50% increase in plasma volume 13 and require adaptations of the maternal vasculature. Significant changes induced by pregnancy of both arteries and veins have been described in the mesenteric, uterine, and renal circulations.14–17 In recent years, considerable efforts have been made to investigate the effect of pregnancy on the cerebral vasculature. For example, we showed previously that there are changes in myogenic activity and a shift in vasodilator production during pregnancy in posterior cerebral arteries (PCAs). 18 Further, pregnancy causes outward remodeling of brain parenchymal arterioles. 19 Importantly, it was suggested that these adaptations predispose the brain to the development of neurologic complications during pregnancy when blood pressure is acutely elevated such as during preeclampsia and eclampsia. 20 To our knowledge, no studies have focused on how cerebral veins are influenced by pregnancy. Understanding of adaptations of the venous side may provide further insights into how pregnancy increases the risk for women to the development of neurologic complications.
The brain venous system consists of superficial cortical veins, deep veins, and venous sinuses. 21 In the present study, we investigated the effect of pregnancy on the cerebral vein of Galen, a vein from the deep venous system. This cerebral vein was selected because it is easily accessible and can be pressurized, which makes it a good starting point for investigations on the venous side of the cerebral circulation. It provides drainage for the medial areas of the diencephalon, the basal ganglia, the midbrain, the medial aspect of the cerebral hemisphere, and the corpus callosum 22 and thus serves a great territory of the brain. The vein of Galen has anastomoses with the basal veins 23 and consequently, only simultaneous obstruction of the vein of Galen and the basal veins will effectively obstruct venous outflow. However, obstruction of the vein of Galen alone presumably results in a rapid increase in intracranial pressure. Hence, changes in the structure of this vein may have a significant effect on venous outflow and normal brain function. Here, we studied the effect of pregnancy on the function (myogenic tone, myogenic reactivity, endothelial and smooth muscle function) and structure (remodeling and distensibility) of the vein of Galen in rats. In addition, we examined if pregnancy influenced perivascular innervation of the cerebral veins. To our knowledge, this is the first study to examine the effect of pregnancy on the venous side of the cerebral vasculature.
MATERIALS AND METHODS
Animals
Age-matched virgin female nonpregnant (NP, n = 15) and late-pregnant (LP, n = 13) Sprague-Dawley rats were used for all experiments and purchased from Charles River (Saint Constant, Canada). Animals were housed at the University of Vermont Animal Facility, an American Association for Accreditation of Laboratory Animal Care-accredited facility. Rats were maintained at a 12-hour light/dark cycle and allowed food and water ad libitum. All animal procedures were approved by the Institutional Animal Care and Use Committee at the University of Vermont and performed in accordance with the National Institutes of Health Policy on the care and use of laboratory animals. Animals were euthanized on 15 to 18 weeks of age and LP animals were examined on day 20 or 21 of a 22-day gestation. The NP animals of the same age were used as controls. On the day of an experiment, animals were anesthetized with either 3% isoflurane in oxygen or Fatal-Plus Solution (Pharmaceuticals, Dearborn, MI, USA) and decapitated.
Isolated Vein Experiments and Arteriograph System
After decapitation, the brain was quickly removed and placed in cold physiologic salt solution (PSS). The vein of Galen was carefully dissected and cleared of connective tissue. A segment of the dissected vein (~300 to 350 μm in length) was mounted at both ends on glass cannulas within an arteriograph chamber and secured with two strands of nylon suture. The proximal cannula of the arteriograph was connected to an in-line pressure transducer and servo controller (Living Systems Instrumentation, Burlington, VT, USA) that maintained intravascular pressure at a set pressure or changed at a variable rate. The distal cannula was closed off to prevent flow through the vein during the experiment as to avoid flow-mediated responses. The veins were perfused with PSS and maintained at pH 7.40 ± 0.05 by aeration with a blood-gas mixture (5% CO2, 10% O2, and balanced N2) and heated to 37°C for the entire experiment. Isolated veins under these conditions were viable throughout the experiment which lasted ~5 hours. The arteriograph contained an optical window on the bottom of the chamber and was placed on an inverted microscope connected to a video dimension analyzer (Living Systems Instrumentation) to allow for measurement of lumen diameter and wall thickness.
Experimental Protocol
Veins (NP: n = 13, LP: n = 11) were equilibrated for 1 hour at 10 mm Hg after which pressure was increased stepwise to 20 mm Hg and active lumen diameter and wall thickness were recorded at each pressure once stable. After the active pressure versus diameter curves were completed, pressure was returned to 10 mm Hg for the remainder of the experiment. Once the lumen diameter was stable again at 10 mm Hg, increasing concentrations of 5-hydroxytryptamine (serotonin, 5-HT) were cumulatively added to the organ bath in the concentration range from 10−8 to 10−5 mol/L to obtain a concentration-response curve. Lumen diameter and wall thickness were recorded at each concentration once stable. 5-Hydroxytryptamine was washed out of the bath with fresh PSS (10 to 15 minutes) and veins were preconstricted to 30% to 50% of the maximal diameter with either 5-HT or increasing and cumulative concentrations of the thromboxane A2 agonist U46619 (percent preconstriction for NP = 34.4 ± 3.3 and LP = 38.4 ± 3.2). Some veins did not preconstrict well and were excluded from the analysis. This resulted in a comparison of n = 12 for the NP group and n = 8 for LP group. A concentration-response curve to the small- and intermediate Ca2
Perivascular Staining and Determination of Nerve Density
Whole-mount immunohistochemical staining of perivascular nerves and quantification of nerve density was performed on fixed veins of Galen. Veins were incubated for 30 minutes in blocking media consisting of 4% normal goat serum and 0.5% Triton X-100 in 0.1 mol/L PBS. After blocking, veins were incubated overnight at 4°C on a rotary mixer with the pan-neuronal antibody rabbit anti-protein gene product 9.5 (PGP 9.5, EMD Millipore, Billerica, MA, USA) at a dilution of 1:500 in blocking media, or with mouse anti-5-HT (Abcam, Cambridge, MA, USA) at a dilution of 1:40 in blocking media. After 24 hours, veins were rinsed three times in 0.1 mol/L PBS for 5 minutes, followed by incubation in the secondary antibody goat anti-rabbit Alexa Fluor-488 (Invitrogen, Grand Island, NY, USA) for PGP 9.5 or goat anti-mouse Alexa Fluor-555 (Invitrogen) for 5-HT at 1:500 in blocking media. After washing with 0.1 mol/L PBS, veins were quickly rinsed in double distilled water before mounting on glass slides with Aqua Polymount (Polysciences, Warrington, PA, USA). Imaging of the vessels was performed on an Olympus BX50 microscope at × 20 magnification using a filter for Alexa Fluor −488 or −555 and captured using Q Capture Pro software (QImaging, Surrey, BC, Canada). Nerve density and the area of each vessel was determined using MetaMorph software (Molecular Devices, LLC, Sunnyvale, CA, USA) with a 10 × 10 μm grid to count the number of intersect points where nerves crossed the grid per area. Nerve density was assessed by two unbiased, independent reviewers blinded to the identity of the specimens. A PCA and a mesenteric artery (MA) were used as positive controls for the PGP 9.5 and 5-HT staining, respectively. The PCA and MA underwent the same staining procedure as the veins.
Ultrastructure of the Veins using Transmission Electron Microscopy
A separate set of veins (NP: n = 2, LP: n = 2) were fixed for transmission electron microscopy. Veins of Galen were mounted and pressurized to 10 mm Hg and allowed to equilibrate at 10 mm Hg for 45 minutes, after which they were fixed in 2.5% glutaraldehyde. After fixation, veins were embedded in 2% SeaPrep agarose for 15 minutes at 4°C, followed by fixation in Karnovksy's solution for another 15 minutes at 4°C. Veins were rinsed with cacodylate buffer, postfixed in 1% osmium tetraoxide (OsO4) in buffer at 4°C for 30 minutes and finally prepared for transmission electron microscopy as previously described. 24 Imaging of the veins was performed on the JEOL 1400 Transmission Electron Microscope (Japan) and all electron micrographs were taken at a magnification of × 2,500.
Data Calculations
Percent tone was calculated as a percent decrease in diameter from the passive diameter at each pressure by the equation: (1 – [Φtone/Φpassive]) × 100%; where Φtone is the active diameter of the vessel at a particular pressure and Φpassive is the passive diameter of the vessel at a particular pressure. The slope (m) of the active and passive pressure versus diameter curves was determined from the diameters at pressures from 10 to 20 mm Hg and calculated by the equation: m = ΔΦ/Δpressure, where ΔΦ is the difference in diameter between 20 and 10 mm Hg, and Δpressure is the difference in pressure. Constriction to 5-HT was calculated at each concentration as a percent decrease in vessel diameter from baseline by the equation: (Φconcentration – Φbaseline]/Φbaseline) × 100%, where Φbaseline is the initial diameter of the vein at 10 mm Hg and Φconcentration is the diameter of the vein in 5-HT. Reactivity of the veins to NS-309 and SNP was calculated by the equation ([Φconcentration – Φbaseline]/[Φpassive – Φbaseline]) × 100%, where Φconcentration is the diameter of the vessel at each concentration of NS-309 or SNP, Φbaseline is the baseline diameter of the vein before adding the drug and Φpassive is the passive diameter of the vessel at 10 mm Hg. Passive distensibility was calculated by the equation ([Φpassive – Φ5 mm Hg]/Φ5 mm Hg) × 100%, where Φpassive is the passive diameter at a particular pressure and Φ5 mm Hg is the passive diameter of the vessel at 5 mm Hg. Wall-to-lumen ratio was determined by dividing the passive wall thickness at a particular pressure by the passive lumen diameter of the vessel. Outer diameter was determined from measured lumen diameter and wall thickness by the equation: outer diameter = lumen diameter + (2 × wall thickness). Circumferential wall tension (T) was calculated by the equation: T = pressure × r, after pressure in mm Hg was converted to pressure in dynes per square centimeter (1 mm Hg = 1,333.2 dynes/cm2) and radius (r) of the lumen in micrometers (μm) was converted to centimeters (cm). Circumferential wall stress was then calculated by the equation: T/wall thickness.
Drugs and Solutions
All isolated vein experiments were performed in PSS with the following composition (in mmol/L): NaCl 119.0, MgSO4 1.17, KCl 4.70, NaHCO3 24.0, KH2PO4 1.18, EDTA 0.026, CaCl2 1.6. Physiologic salt solution was stored without glucose and glucose was added daily before an experiment (5.5 mmol/L). In PSS without Ca2
5-Hydroxytryptamine, SNP, and NS-309 were purchased from Sigma-Aldrich (St Louis, MO, USA) and U46619 was purchased from Enzo Life Sciences (Farmingdale, NY, USA). 5-Hydroxytryptamine and SNP were made fresh each day as a 10−3 and 10−4 mol/L stock solution in double distilled water. NS-309 and U46619 were dissolved in dimethyl sulfoxide to 10−3 mol/L stock solution aliquots and kept at −20°C until use. Aliquots were thawed before an experiment and used as 10−3 or 10−4 mol/L stock (diluted in double distilled water) solutions.
Statistical Analysis
Data are depicted as mean ± standard error of the mean (s.e.m.). Differences between NP and LP groups were determined using an unpaired Student's t-test and a Welch's correction for unequal variances where appropriate. A paired t-test was used to determine the differences between active and passive slopes within the groups. Repeated measures analysis of variance followed by a post hoc Bonferroni test for multiple comparisons was used to determine the differences in percent tone at different pressures within the groups. Differences were considered significant when P ≤ 0.05. Statistical analyses and graphing were performed using GraphPad Prism 5 software (La Jolla, CA, USA).
RESULTS
Cerebral Veins have Decreased Myogenic Tone and Larger Active Lumen Diameters during Pregnancy
To determine if the vein of Galen had basal tone and responded to pressure with myogenic reactivity, and if the pregnant state influenced this, an active pressure versus diameter curve for the veins from NP (n = 13) and LP (n = 11) animals was obtained. Figure 1A shows that in both NP and LP animals, the vessel diameter increased with increasing pressure and the slope of the active pressure versus diameter curves was positive (NP: m = 0.95 ± 0.12, LP: m = 1.19 ± 0.11). The slope of the active pressure versus diameter curves of cerebral veins was less than the slope of the respective passive pressure versus diameter curves in both groups (passive m NP: 1.20 ± 0.13, P = 0.08 and passive m LP: 1.34 ± 0.12, P < 0.05), suggesting that these veins possess some myogenic reactivity. Importantly, veins from LP animals had significantly larger active diameters at all pressures studied compared with veins from NP animals. This increased lumen diameter was at least partly because of a decrease in myogenic tone. Figure 1B shows that veins from LP animals had significantly less tone at all pressures studied compared with veins from NP animals. Compared with cerebral arteries, veins from NP animals had little tone (3.9 ± 1.0% at 10 mm Hg) whereas veins from LP animals had virtually no tone (0.8 ± 0.3% at 10 mm Hg, P < 0.01 versus NP). In addition, myogenic tone of veins in the LP group was significantly higher at 20 mm Hg compared with 10 mm Hg (1.7 ± 0.4% versus 0.8 ± 0.3%, P < 0.05), while this was not the case in the NP group (3.9 ± 1.0% at 10 mm Hg and 5.3 ± 1.2% at 20 mm Hg, P = 0.22).
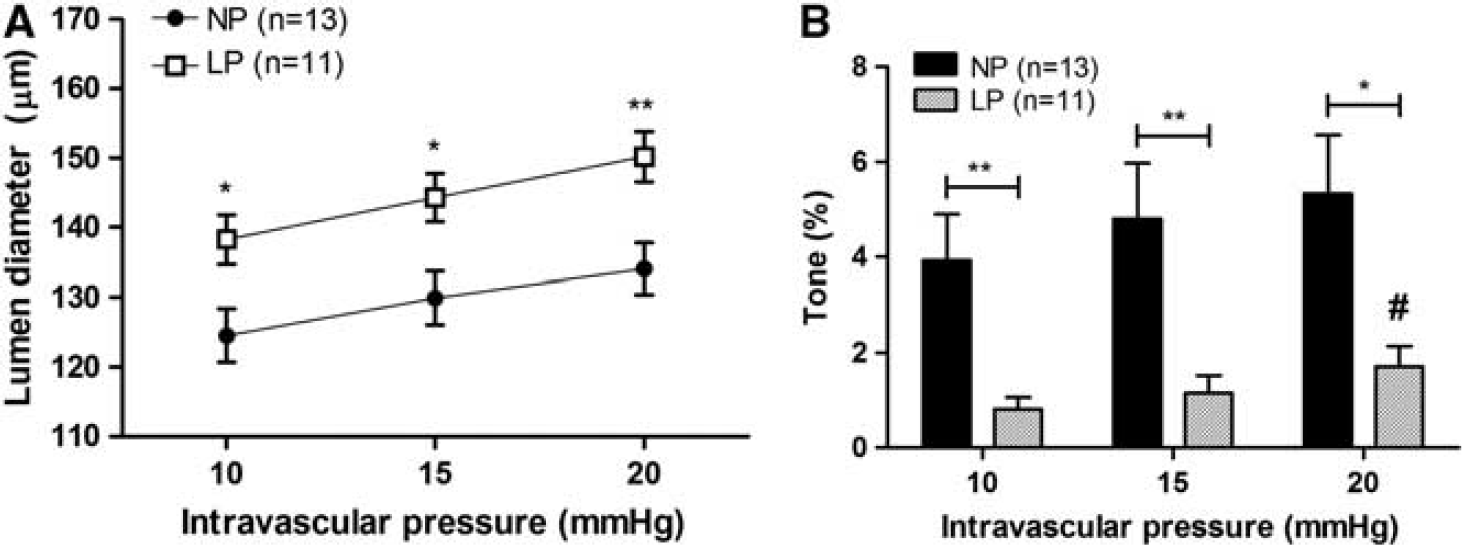
Effect of pregnancy on active diameters and tone of the vein of Galen. (
Pregnancy Causes Outward Hypotrophic Remodeling of Cerebral Veins
To determine the effects of pregnancy on the structure of the vein of Galen, diameters and wall thicknesses were recorded under passive conditions in the pressure range from 5 to 25 mm Hg. Passive lumen diameters were greater in veins from LP animals (n = 11) compared with veins from NP animals (n = 13) at all pressures studied (Figure 2A), although these differences only reached significance at 20 and 25 mm Hg. There was a trend for veins from LP animals to be larger in lumen diameter at 10 and 15 mm Hg (P = 0.08 versus NP at both pressures). Notably, the outer diameters of veins from NP and LP animals were not significantly different (Figure 2B), whereas passive wall thickness of the veins was significantly smaller in LP animals (Figure 2C). The difference in passive lumen diameters was not because of an increase in distensibility, since passive distensibility of the veins was similar between NP and LP animals at all pressures (Figure 2D). Thus, pregnancy caused outward remodeling of the cerebral veins. As a consequence of the increased lumen and decreased wall thickness, the wall-to-lumen ratio was significantly decreased in veins from LP animals (P < 0.01, data not shown). Together, these data show that the increased vessel lumen of the veins from LP animals occurred at the expense of the vessel wall. Thus, outward remodeling of the vein of Galen in pregnancy was hypotrophic in nature.
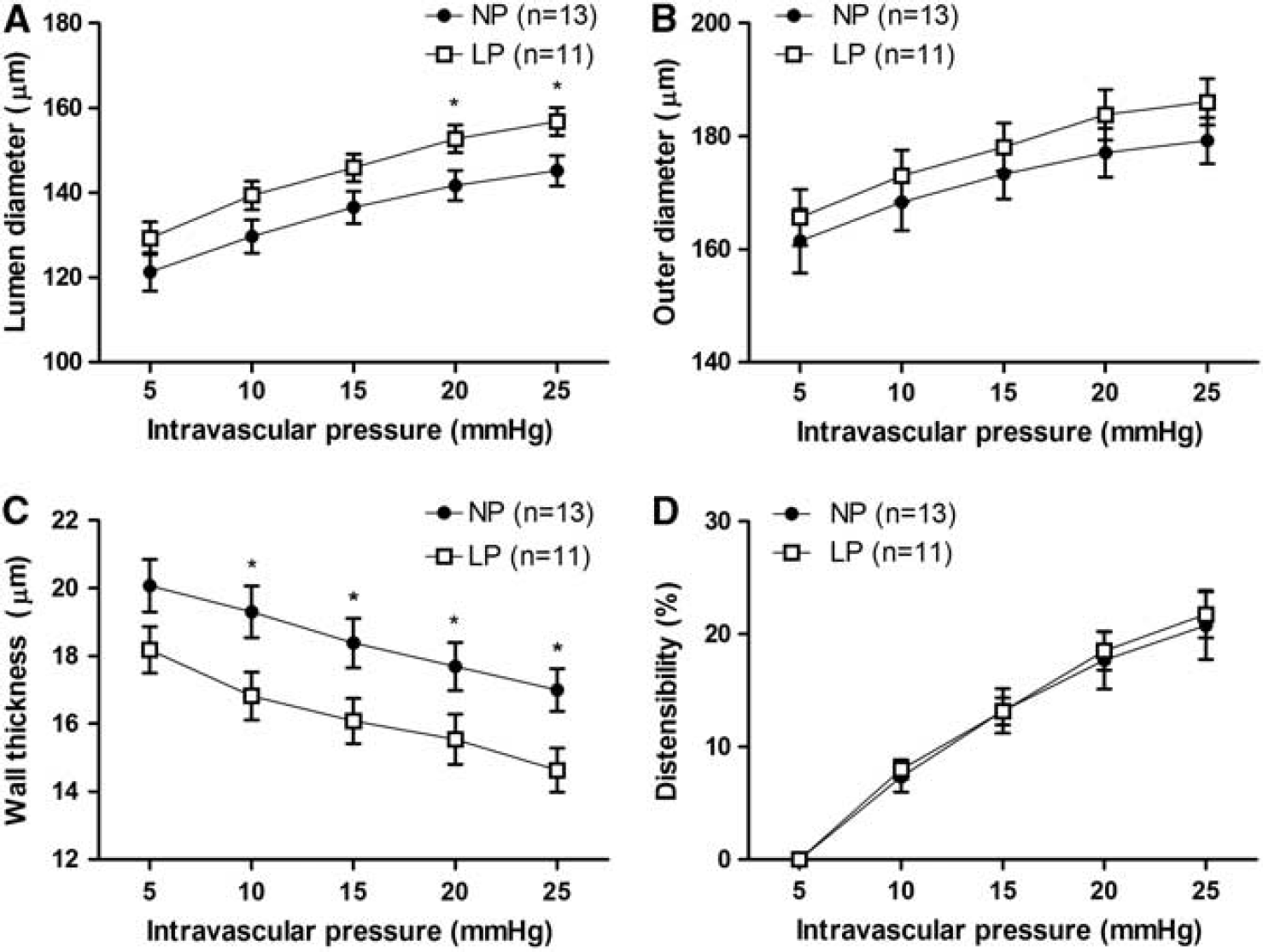
Effect of pregnancy on the passive structure of the vein of Galen. (
Cerebral Veins Experience Greater Wall Tension and Wall Stress During Pregnancy
To examine the effect of pregnancy on the biomechanics of the vein of Galen, we calculated passive wall tension and wall stress in the veins. Passive wall tension in the cerebral veins was greater in LP animals compared with NP animals (Table 1). This increase in wall tension during pregnancy was significant at the higher pressures but was also present at 10 and 15 mm Hg (P = 0.08). In addition, veins from LP animals were subject to increased wall stress because of the thinner walls and larger lumen compared with veins from NP animals at all pressures studied (Table 1).
Passive wall tension and wall stress in the vein of Galen at different pressures
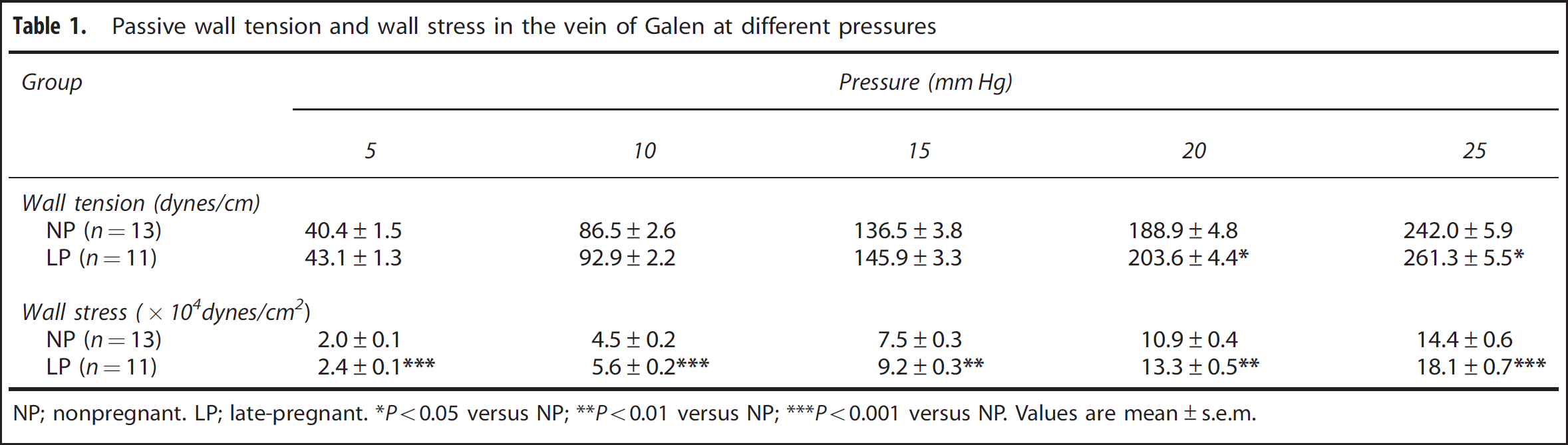
NP; nonpregnant. LP; late-pregnant.
P<0.05 versus NP;
P<0.01 versus NP;
P<0.001 versus NP. Values are mean±s.e.m.
Wall Ultrastructure of the Vein of Galen in Nonpregnant and Late-pregnant Rats
To further investigate the structural characteristics of the vein of Galen in NP and LP rats, transmission electron microscopy was performed on pressurized vein segments to evaluate venous ultrastructure. In Figure 3, electron micrographs of a longitudinal section through the wall of the vein of Galen from an NP (Figure 3A) and an LP (Figure 3B) animal are shown. No remarkable differences in morphology of the venous wall were noted between the groups (n = 2 for both groups). A continuous single layer of endothelial cells was observed, separated from the tunica media—consisting of one to three layers of vascular smooth muscle—by the internal elastic lamina. Wall thickness was variable within the venous segments and mostly dependent on size and number of smooth muscle cells. Interesting to note was that other cell types resembling perivascular nerves and pericytes were observed in close association with the adventitial layer of the veins.
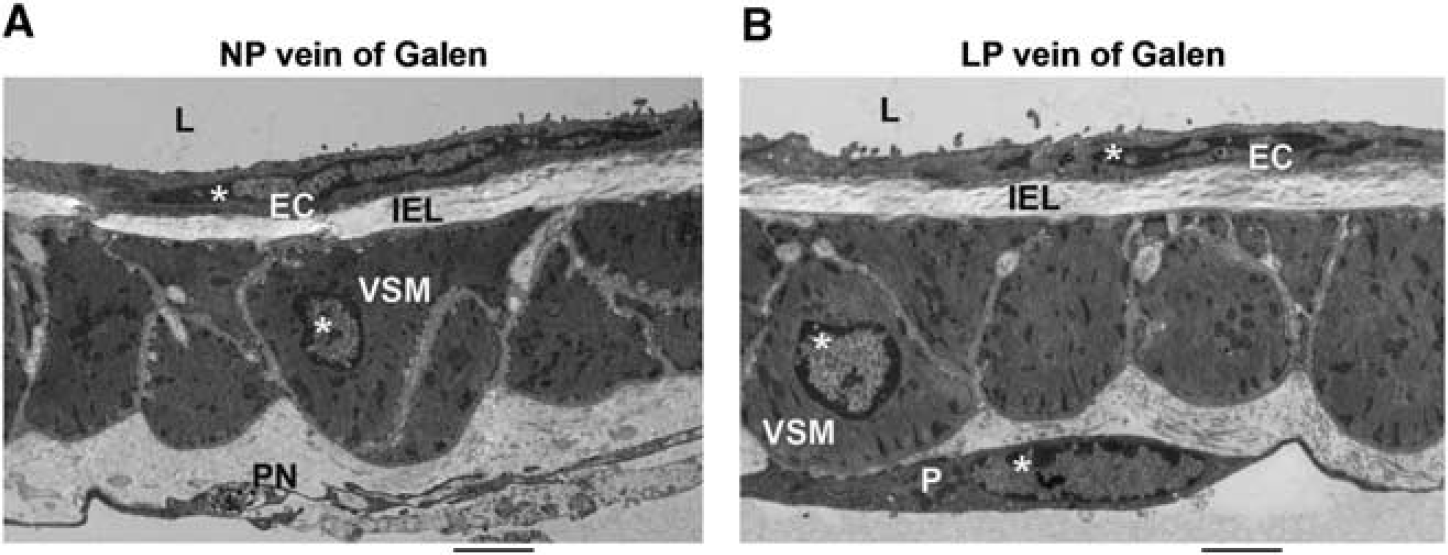
Ultrastructure of the wall of the vein of Galen from nonpregnant (NP) and late-pregnant (LP) animals. Transmission electron micrograph (× 2,500) of a longitudinal section of the vein of Galen fixed pressurized at 10 mm Hg (
Endothelium-Dependent and -Independent Vasodilator Responses Are Not Changed by Pregnancy
Reactivity of the vein of Galen from NP and LP animals to pharmacological agonists was determined to investigate the effect of pregnancy on venous function. The SKCa and IKCa channel agonist NS-309 caused veins from NP (n = 12) and LP (n = 8) animals to dilate in a concentration-dependent manner. Reactivity of the cerebral veins to NS-309 was similar in NP and LP animals at all concentrations (Figure 4A), as was the maximal dilatory response (79.4 ± 3.8% versus 79.7 ± 4.2%, respectively). This is one of the first reports to show that SKCa and IKCa channels are present on venous endothelium and the first to show that activation of the SKCa and IKCa channels in cerebral veins causes significant dilation. Similarly, the NO donor SNP caused the cerebral veins from NP (n = 9) and LP (n = 5) animals to dilate in a concentration-dependent manner (Figure 4B), and maximal reactivity was not different in veins from NP and LP animals (72.5 ± 6.3% versus 63.2 ± 9.2%, respectively). These results show that endothelium-independent vasodilation of cerebral veins to NO was not changed in pregnancy.
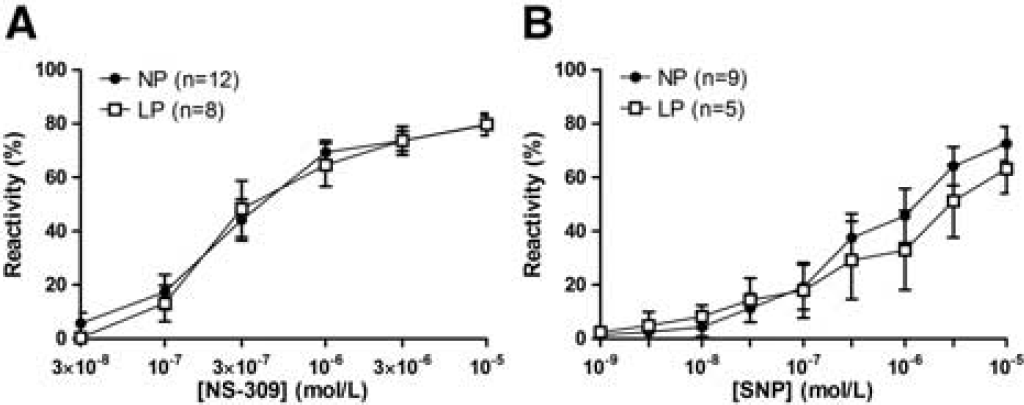
Effect of pregnancy on the reactivity of the vein of Galen to NS-309 and sodium nitroprusside (SNP). (
Effect of Pregnancy on 5-Hydroxytryptamine Reactivity and Serotonergic Innervation of Cerebral Veins
To investigate if pregnancy influenced perivascular innervation of the vein of Galen, nerve density was determined with immunohistochemistry. In Figures 5A and 5B, photomicrographs of segments of the vein of Galen, stained for perivascular nerves with the pan-neuronal antibody PGP 9.5, from an NP and an LP animal are shown. Veins from both NP (n = 4) and LP (n = 4) animals had scarce perivascular innervation. In most venous segments, only one or a few nerve fibers were observed. This was in contrast to the PCA that contained significant PGP 9.5-positive staining (Figure 5C). Nerve density was higher in veins from NP animals, although this was not significant (P = 0.14, Figure 5D). Photomicrographs of segments of the vein of Galen stained for 5-HT from an NP and an LP animal are shown in Figures 5E and 5F, respectively. In the NP group (n = 5), only one vein showed positive staining for 5-HT, whereas in the LP group (n = 6) serotonergic innervation was observed in all venous segments, albeit in variable levels. This was in contrast to the MA shown in Figure 5G that had a considerable number of 5-HT-positive nerves. When quantified morphometrically, nerve density of 5-HT-containing nerves in the veins was significantly higher in the LP group compared with the NP group (Figure 5H, P < 0.05), indicating a trophic effect of pregnancy on serotonergic innervation of cerebral veins.
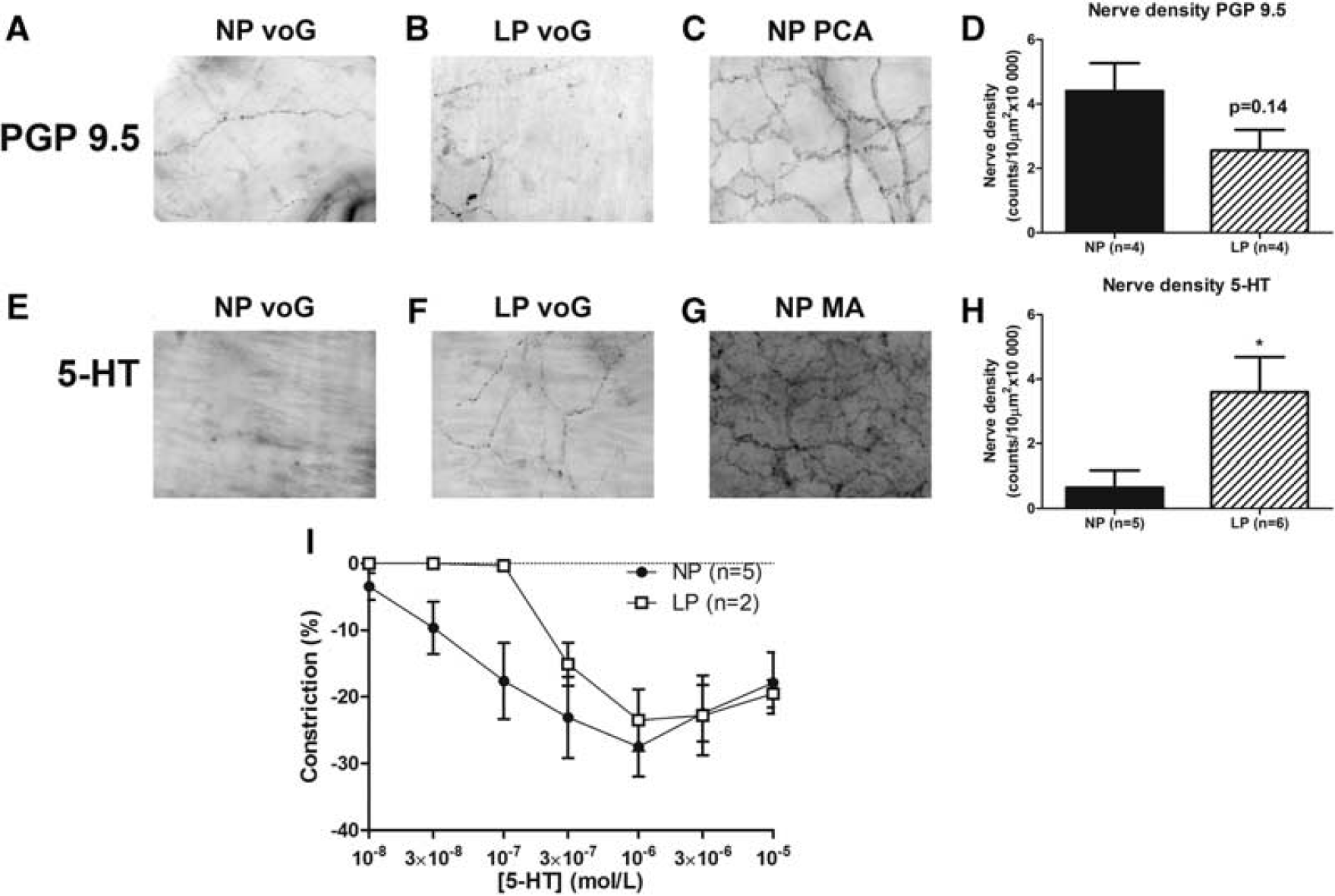
Perivascular innervation of the vein of Galen during pregnancy. Photomicrographs (× 20) of whole mount segments of (
In addition, the effect of pregnancy on reactivity of cerebral veins to 5-HT was determined. Administration of 5-HT caused constriction in 5 out of 13 veins from the NP group, whereas in the LP group 2 out of 11 veins constricted to 5-HT. In Figure 5I, the effect of 5-HT on the veins that did show a response is shown. In both groups, the veins constricted to 5-HT with a maximum constriction at 10−6 mol/L (NP: 27.5 ± 4.5% and LP: 23.5 ± 4.6%). The effect of 5-HT on the cerebral veins appeared to be biphasic, as the veins dilated at the higher concentrations. The two veins from the animals in the LP group that responded, constricted at a higher concentration 5-HT than the five veins from the animals in the NP group that responded, suggesting that pregnancy may decrease sensitivity of the veins to 5-HT. However, because so few veins responded to 5-HT, this was difficult to assess. Important to note is that all the veins that did not respond to 5-HT, did show constriction to U46619, indicating that the contractile apparatus of the veins was intact.
DISCUSSION
Pregnancy is a remarkable physiologic state, characterized by a 40% to 50% increase in plasma volume and substantial cardiovascular adaptation necessary to accommodate this large increase in plasma volume. 13 In addition, considerable changes in maternal hemodynamics occur, including an increase in cardiac output, stroke volume, and heart rate, while peripheral vascular resistance is decreased. 12 Uterine and renal circulations also undergo significant adaptation to pregnancy, including an ~10 fold increase in uterine blood flow 25 and a 50% increase in renal blood flow and glomerular filtration rate. 14 In contrast, cerebral hemodynamics appear to remain relatively constant over the course of gestation. Investigations of cerebral hemodynamics in pregnant women and animal models of pregnancy have shown that there are no or only small changes in global cerebral blood flow and cerebral vascular resistance19,26 compared with the NP state. However, although global cerebral hemodynamics may be unaffected during pregnancy, local changes in the structure and function of cerebral vessels may occur. Understanding the influence of pregnancy on local changes in the cerebral circulation may be potentially important to understand venous pathology that occurs during gestation, including edema, hemorrhage, and thrombosis.
The major finding of the present study was that the vein of Galen from LP animals was significantly larger in lumen diameter compared with the NP state. This was because of pregnancy causing outward remodeling of the vein of Galen and decreased basal tone. Since vessel diameter is the most powerful determinant of vascular resistance as it is inversely proportional to the fourth power of the vessel radius and directly related to blood viscosity, 27 the increase in lumen diameter observed in this study may serve to decrease cerebral venous resistance during pregnancy. The effect of the larger lumen diameter on venous resistance in the brain may be enhanced by the fact that blood viscosity is reduced in pregnancy because of the large increase in plasma volume. 13 This type of remodeling is similar to that shown to occur in brain parenchymal arterioles during pregnancy. 19 This same study showed that pregnancy had no effect on the lumen diameter of large pial arteries. 19 Thus, pregnancy appears to selectively affect certain vessels within the brain that may have effects on local vascular resistance while not changing overall cerebral vascular resistance.
Remodeling of the vein of Galen observed in the present study was characterized by an increase in lumen diameter at the expense of the vessel wall, i.e., the vessel wall became thinner during pregnancy without a change in outer diameter (hypotrophic remodeling). The mechanism through which remodeling occurred is not clear from this study. Notably, pregnancy was shown to cause outward hypotrophic remodeling of brain parenchymal arterioles, 19 similar to our findings in cerebral veins. Outward remodeling of the arterioles during pregnancy was because of activation of the transcription factor peroxisome proliferated-activated receptor-γ, 19 possibly mediated by relaxin, 28 a peptide hormone produced in high levels during pregnancy. It is possible that remodeling of the cerebral veins during pregnancy occurred through the same mechanism, although no studies have been performed yet to investigate whether there are peroxisome proliferated-activated receptor-γ and relaxin receptors present on the vein of Galen. Future studies are needed to determine the mechanism by which remodeling of the vein of Galen occurs during pregnancy.
Pregnancy increases the risk of several cerebrovascular complications, including venous thrombosis and hemorrhage.6,9,10 An important finding here is that because of hypotrophic outward remodeling, cerebral veins experience greater wall tension and wall stress during pregnancy. Elevated wall stress in pregnancy may make the cerebral veins more vulnerable to rupture and thereby predispose the veins to hemorrhage. Furthermore, enlargement of the cerebral veins may lead to venous pooling or stasis. This, in combination with a hypercoagulable state in pregnancy, can pose a substantial risk of hemorrhage and venous thrombosis. 29 To our knowledge, however, no studies have focused on structural differences of cerebral veins as a contributing factor in the pathogenesis of cerebral venous thrombosis in human subjects, and in pregnancy in particular, because of difficulties investigating brain hemodynamics in pregnant women. In a study on deep leg veins in humans, an increase in vessel diameter combined with a decrease in flow velocity over the course of gestation, is a likely cause leading to venous pooling and stasis. 30 These findings are consistent with an observed incidence and pattern of deep venous thrombosis in pregnancy 30 and may relate to the triad of Virchow that postulates thrombosis is caused by changes in blood flow, state of the vessel wall, and composition of blood (e.g., coagulation factors). 29 Together, this suggests that the changes caused by pregnancy in the cerebral veins, if they also occur in humans, may predispose to cerebral venous thrombosis.
Our results show that cerebral veins possess some myogenic tone and reactivity to pressure, although this was minimal, possibly because the cerebral venous side operates at low pressures. Regardless, myogenic tone was significantly decreased in cerebral veins from LP animals which contributed partly to larger active lumen diameters. Several studies have described myogenic tone in veins of peripheral circulations, the amount of which varies between veins and the function of the vascular bed.31–33 However, this is the first study we are aware of to describe myogenic tone and reactivity in cerebral veins in the normal physiologic and pregnant state. The mechanism by which pregnancy decreased tone in the vein of Galen is not clear. Decreased myogenic tone could be caused by changes in the vasodilator influence of the endothelium, which mitigates tone. In the present study, we found that activation of SKCa and IKCa channels caused vasodilation which suggests that there are functional SKCa and IKCa channels present on cerebral veins. This finding is supported by a recent study in which the involvement of SKCa and IKCa channels in relaxation of rabbit jugular vein was shown 34 and importantly, these channels have been implicated as key players in endothelium-derived hyperpolarizing factor-mediated vasodilation. 35 Since reactivity of the cerebral veins to SKCa and IKCa channel stimulation was similar in NP and LP animals, this mechanism is not likely to be responsible for decreased tone in pregnancy. Nitric Oxide is another important vasodilator that inhibits tone and thus may be involved in decreasing tone of the vein of Galen during pregnancy, as has been shown in peripheral vessels. 36 We showed here that the reactivity of smooth muscle to NO in the cerebral veins was not changed in pregnancy. In addition, veins did not respond to acetylcholine (data not shown) and thus increased production of NO by the endothelium is also not likely responsible for the decrease in venous tone in pregnancy. It is possible that there are fundamental changes in smooth muscle function or vasodilator prostacyclin production that diminishes tone in veins during pregnancy.
We showed that the vein of Galen is innervated by a modest amount of perivascular nerves. Total perivascular innervation, reflected by PGP9.5-containing nerves, was higher in veins from NP rats, although this was not significant likely because of the small sample size. However, we also show the presence of 5-HT containing nerve fibers on the cerebral veins that was significantly increased in veins from LP animals. 5-Hydroxytryptamine has been reported to cause constriction37,38 and dilation37,39 in cerebral veins, depending on species and vessel size. Here, we found little response of veins to 5-HT and the vessels that did respond constricted upon administration of 5-HT at the lower concentrations (with a small dilatory response at the highest concentrations). Considering only the responding veins, it appears that pregnancy caused a blunted response to 5-HT and that reactivity to 5-HT was not related to level of serotonergic innervation. A few other studies have reported unresponsive veins to 5-HT. Hardebo et al 38 found that the majority of veins (9 out of 14) contracted upon exposure to 5-HT in human pial veins. Leber et al 37 found that topically administered 5-HT caused dilation of feline small pial veins (< 150 μm) and tendency of large pial veins (> 150 μm) to remain unreactive or even to constrict. The latter finding is in agreement with our observations, although we only investigated the vein of Galen which has characteristics of a large pial vein, e.g., it is extrinsically innervated and has a large amount of smooth muscle cells. Importantly, Ishine et al 39 showed in porcine pial veins that these veins received vasodilator and constrictor nerves (adrenergic, cholinergic, peptidergic, and nitric oxidergic), but that transmural nerve stimulation-induced relaxation was not blocked by receptor antagonists of the corresponding transmitters. This suggests that although the veins received vasodilator and constrictor nerves, these nerves are unlikely to have an influence on venous tone. In addition, this study showed that 5-HT fibers were present only after 30 minutes incubation with 5-HT before fixation and that 5-HT was taken up into sympathetic adrenergic nerve fibers on the veins rather than having authentic serotonergic innervation. 39 In the present study, the vessels were fixed after reactivity experiments. Therefore, the differences in serotonergic innervation between veins from NP and LP animals may reflect differences in uptake of 5-HT in other nerve fibers.
In summary, we show for the first time that cerebral veins undergo significant changes in structure and function during pregnancy in an animal model. Pregnancy caused outward hypotrophic remodeling and diminished tone in the vein of Galen which may cause a local decrease in cerebral vascular resistance. These structural changes may contribute to the development of venous pathology in pregnancy, including venous thrombosis and hemorrhage. The mechanisms through which pregnancy causes remodeling and changes in tone of the cerebral veins are not clear and deserve further investigation.
DISCLOSURE/CONFLICT OF INTEREST
The authors declare no conflict of interest.
Footnotes
ACKNOWLEDGEMENTS
The authors are grateful to Nicole M Bishop for help with the immunohistochemistry and the transmission electron microscopy and to Julie Sweet for technical assistance. The authors gratefully acknowledge the support of the Marco Polo Foundation, JK de Cock Foundation, University of Groningen Foundation, and Dr Christine Buisman Foundation.
