Abstract
The aim of the study was to better understand blood-flow changes in large arteries and microvessels during the first 15 minutes of reflow in a P7 rat model of arterial occlusion. Blood-flow changes were monitored by using ultrasound imaging with sequential Doppler recordings in internal carotid arteries (ICAs) and basilar trunk. Relative cerebral blood flow (rCBF) changes were obtained by using laser speckle Doppler monitoring. Tissue perfusion was measured with [14C]-iodoantipyrine autoradiography. Cerebral energy metabolism was evaluated by mitochondrial oxygen consumption. Gradual increase in mean blood-flow velocities illustrated a gradual perfusion during early reflow in both ICAs. On ischemia, the middle cerebral artery (MCA) territory presented a residual perfusion, whereas the caudal territory remained normally perfused. On reflow, speckle images showed a caudorostral propagation of reperfusion through anastomotic connections, and a reduced perfusion in the MCA territory. Autoradiography highlighted the caudorostral gradient, and persistent perfusion in ventral and medial regions. These blood-flow changes were accompanied by mitochondrial respiration impairment in the ipsilateral cortex. Collectively, these data indicate the presence of a primary collateral pathway through the circle of Willis, providing an immediate diversion of blood flow toward ischemic regions, and secondary efficient cortical anastomoses in the immature rat brain.
INTRODUCTION
Perinatal arterial stroke is a cerebrovascular event occurring around the time of birth, with pathologic or radiologic evidence of focal arterial infarction mainly affecting the middle cerebral arterial (MCA) territory, with an incidence of 1 in 2,500 term births. 1 Perinatal brain injury has been associated with impaired cerebral blood flow (CBF) autoregulation to maintain blood flow with respect to arterial blood pressure. Impairment of this homeostatic mechanism has a key role in the pathophysiology of the ischemic and hemorrhagic insults in newborn brains. 2 Few studies have characterized CBF after neonatal or pediatric ischemia and hypoxia, and there is variability in reperfusion patterns reported according to the species, the developmental stage and ischemia duration. 3
In the adult rat brain, major blood-flow reduction was observed in the ischemic core territory, when collaterals fail to provide adequate perfusion, 4 while this reduction was less marked in the surrounding penumbra. 5 Restoration of blood flow—reperfusion—to the ischemic territory was characterized by a significant hyperemia occurring immediately after occlusion release, which may be seen as a mechanism to counteract the metabolic shortage. This was followed by a postischemic hypoperfusion lasting for hours, described as the ‘no-reflow phenomenon’. 6 This finding raises the question whether the relative or absolute hyperemia could also reflect blood-flow changes that occur in large vessels and in deep brain regions. To approach these questions, tissue perfusion measured by [ 14 C]-iodoantipyrine autoradiography indicated that tissues that are heavily compromised by the 2-hour period of ischemia and are destined to incur infarction show a ‘relative hyperemia’ during recirculation. 7
Brain tissue perfusion has also been quantified by autoradiography in the P7 hypoxic-ischemic rat, and showed CBF in the opposite cortex remained at normal levels despite a ‘hypoxia only’ insult, whereas flow to the brainstem and cerebellum nearly doubled or tripled after hypoxia–ischemia (HI) in rat, respectively.8–10 Conflicting data were reported using laser-Doppler flowmetry under normoxic resuscitation after HI in rat with increased, 11 decreased 12 and none modifications 13 of regional CBF. More recently, the spatial and temporal profiles of cortical surface CBF were analyzed in P8 mouse and P7 rat pups subjected to HI using laser speckle imaging. These authors reported that after the end of the hypoxic insult, the mean CBF level gradually increased and nearly attained its presurgical level in the ipsilateral hemisphere. 14
To better understand whether variability in blood-flow changes evoked above were because of methodological limitations, we choose to characterize hemodynamic responses in large arteries and microvessels during early reflow in a P7 rat stroke model using a large panel of techniques. We evaluated the spatiotemporal profiles of CBF changes using 2D color-coded pulsed Doppler ultrasound, laser speckle contrast (LSC) imaging, and [ 14 C]-iodoantipyrine autoradiography. As decrease in blood supply leads to shortage of oxygen and mitochondrial dysfunction, we then evaluated the respiratory chain activity as an indicator of tissue metabolic state. Detailed results provided by these experiments might further our understanding on blood-flow regulation in the P7 rat brain.
MATERIALS AND METHODS
Neonatal Ischemia–Reperfusion
All experiments of this study complied with ethical guidelines of Robert Debré Hospital Research Council Review Board (A75-19-01), INSERM, and the ARRIVE guidelines (http://www.nc3rs.org/ARRIVE) and have been approved by the local ethic committee (Paris, France). Ischemia was induced in Wistar P7 rat pups (16 to 18 g; Janvier, Le Genest St-Isle, France) 15 and was adapted to isoflurane anesthesia. 16 Briefly, thermoregulated (37.0 ± 0.5°C) and anesthetized pups (1% isoflurane in O2/N2O (1:3)) were exposed to left middle cerebral artery electrocoagulation (MCAo) combined with a transient (50 minutes), concomitant occlusion of both common carotid arteries (CCAs). Carotid blood-flow restoration was assessed by release of the carotid clips. Sham-operated animals were only subjected to carotid artery isolation and did not receive MCAo or transient CCA occlusion (CCAo).
Physiologic Parameters
Arterial blood was collected by a PE10 catheter introduced in the femoral artery and gases (pH, PaO2, Pa
Magnetic Resonance Imaging Acquisition
Pups were anesthetized by isoflurane 0.8% mixed in 30% O2, 70% N2 and monitored after ischemia and 10 minutes and 2 hours reperfusion (
Ultrasound Imaging
Thermoregulated rat pups (
Laser Speckle Contrast Imaging
Imaging was performed using the full-field laser perfusion imager (Moor FLPI V3.0; Moor Instruments Ltd, Axminster, UK). Briefly, thermoregulated and anesthetized (1% isoflurane) rat pups (
Quantitative Measurement of Multilocal Cerebral Blood Flow
Local CBF was measured in isoflurane anesthetized P7 rat pups with the quantitative autoradiographic technique using [
14
C]-iodoantipyrine as a diffusible tracer. Measurements were performed in sham (
Preparation of Isolated Mitochondria
Sham and ischemic P7 rats were killed by decapitation at the end of ischemia and at 15 minutes of reperfusion (
Mitochondrial Oxygen Consumption
Mitochondrial oxygen consumption was measured using an Oxygraph (Oroboros Instrument, Innsbruck, Austria) at 25°C. Mitochondria (~0.3 mg of proteins) were incubated in 2 mL medium containing 60 mmol/L KCl, 150 mmol/L sucrose, 20 mmol/L Tris-HCl, 5 mmol/L KH2PO4, and glutamate 20 mmol/L at pH 7.4. Respiration state 3 was initiated by the addition of 0.2 mmol/L ADP and state 4 without ADP. The oxygen consumption was expressed in nmol O2/min per mg of protein. The respiratory control rate (RCR) was calculated as the ratio of the state 3/state 4.
Statistical Analysis
Values are expressed as mean ± s.d. or s.e.m. ([
14
C]-CBF values). Mean BFV between left and right ICA, and CBF values taken at five different caudorostral intervals were compared using repeated-measures ANOVA on the different groups of animals. Caudorostral profiles of CBF in reperfusion were likewise compared using repeated-measures ANOVA.
RESULTS
Physiologic data indicated that P7 rat pups with a mean body weight of 17.19 ± 1.82 g had a pH of 7.32 ± 0.02, a PaO2 of 79.7 ± 12.5 mm Hg, and a Pa
Injury Produced by Ischemia–Reperfusion
Electrocoagulation of the left MCA in association with 50 minutes left and right CCAo produced a lesion in the left parietal cortex as shown on ADC (apparent diffusion coefficient) maps as early as 10 minutes after ischemia (Figure 1, upper), on T2-weighted images taken at 2 hours (Figure 1, middle) and on cresyl violet-stained sections at 48 hours (Figure 1, lower).
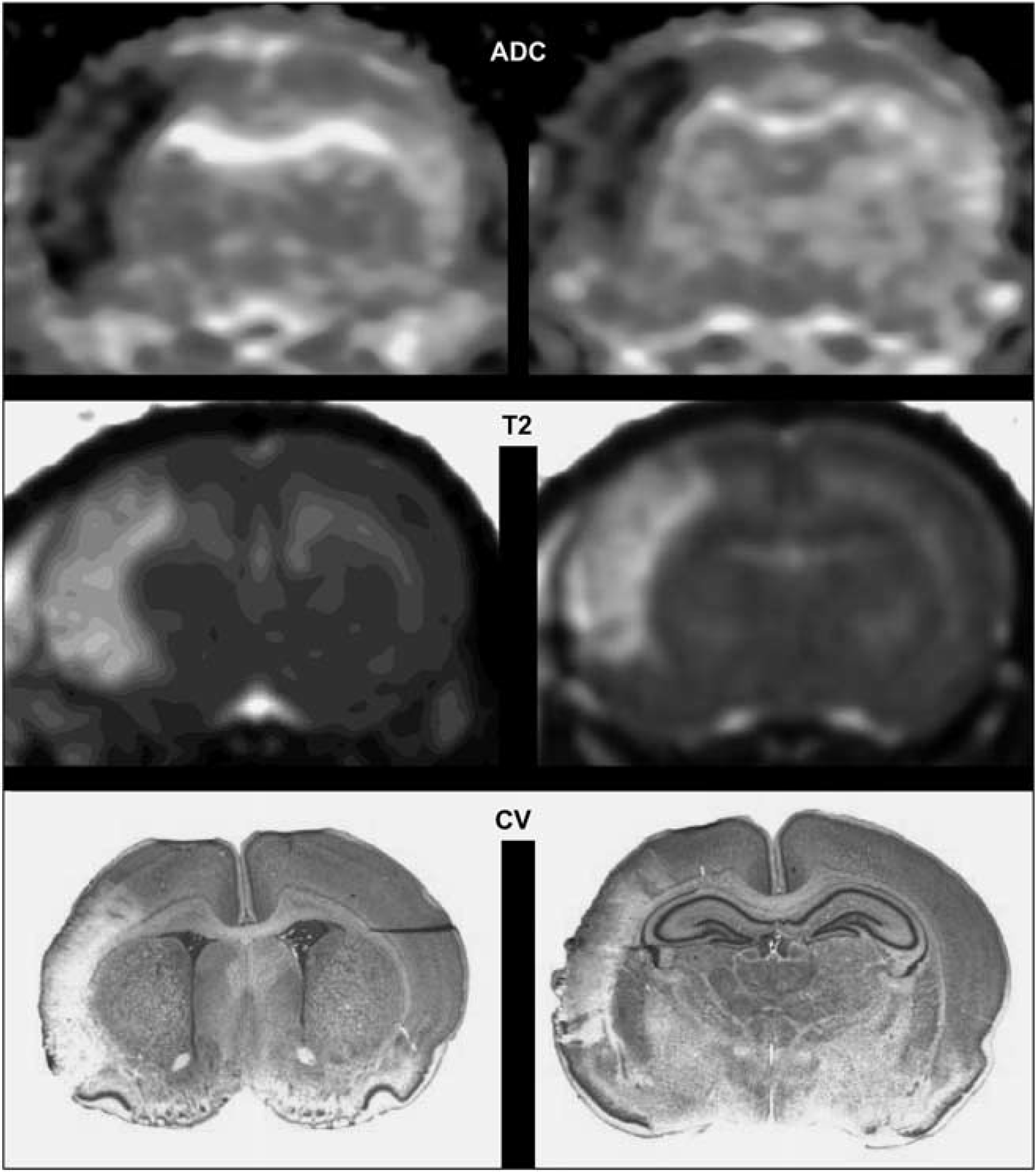
Ischemia–reperfusion induces cortical infarct in P7 rat brain. Upper: Representative apparent diffusion coefficient (ADC) map at 10 minutes after ischemia–reperfusion showing a cortical lesion at Bregma 1.32 and −3.24 mm, respectively. Middle: Representative T2-weighted image at 2 hours after ischemia–reperfusion showing the cortical lesion. Lower: Representative cresyl violet (CV)-stained sections from this animal at 48 hours after ischemia–reperfusion.
Dynamic Blood-Flow Monitoring During Ischemia and Early Reperfusion
During ischemia, while the two CCAs were occluded, mBFVs increased to 138 ± 51% (
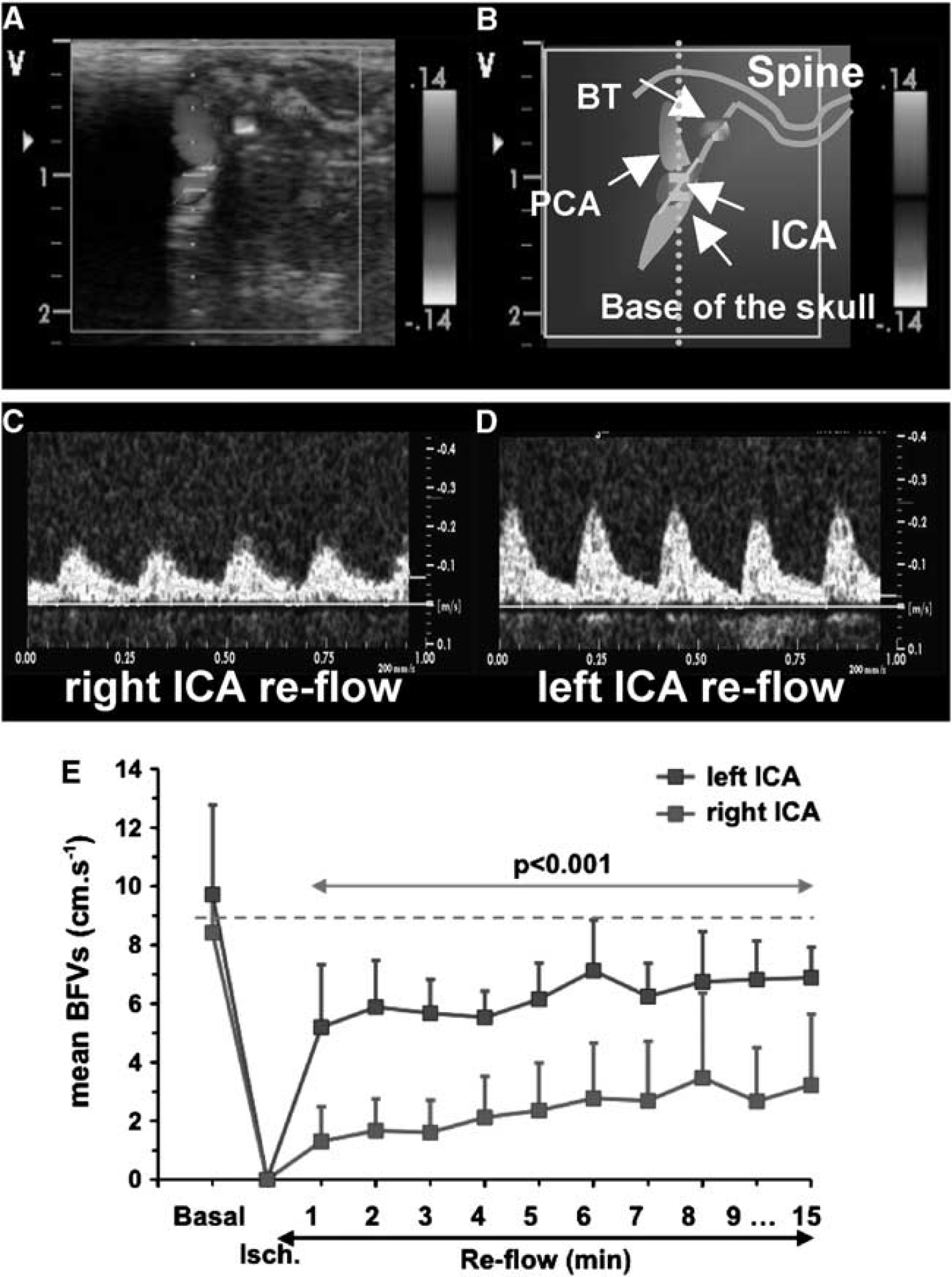
Time course of mean blood-flow velocities (mBFVs) in the left and right internal carotid artery (ICA) using ultrasound (US) imaging during ischemia and reflow (
Representative data illustrating the pattern of blood flow before and after reperfusion using LSC imaging are shown in Figure 3. Immediately after combination of MCA and bilateral CCAo blood flow fell down (Figure 3C) in the MCA territory. After CCAo release and reflow, blood flow through superficial anastomotic arterial connections occurred between the occipital pole (supplied by the BT and posterior cerebral arteries) toward the MCA territory (Figures 3D to 3F, black arrows in Figure 3E). In the right MCA territory, LSC images (Figures 3G to 3J) showed a decrease in CBF during right CCAo (Figure 3H), and its gradual recover during early reflow (Figures 3I to 3J). The LSC images also identified venous and arterial blood flow (Figures 3K to 3N). Blood-flow recovery was gradual and incomplete during reflow in the MCA territory (Figures 3M and 3N) but observed in the terminal segment of the MCA (Figure 3N). Measures of rCBF indicated that after MCA occlusion, CBF declined to 55 ± 8%, and further declined to 18 ± 5% of baseline after bilateral CCAo (Figure 3O) in the ipsilateral cortex. After bilateral CCAo release, rCBF gradually increased from 30 ± 11% at 1 minute to 44 ± 9% at 20 minutes (Figure 3O,
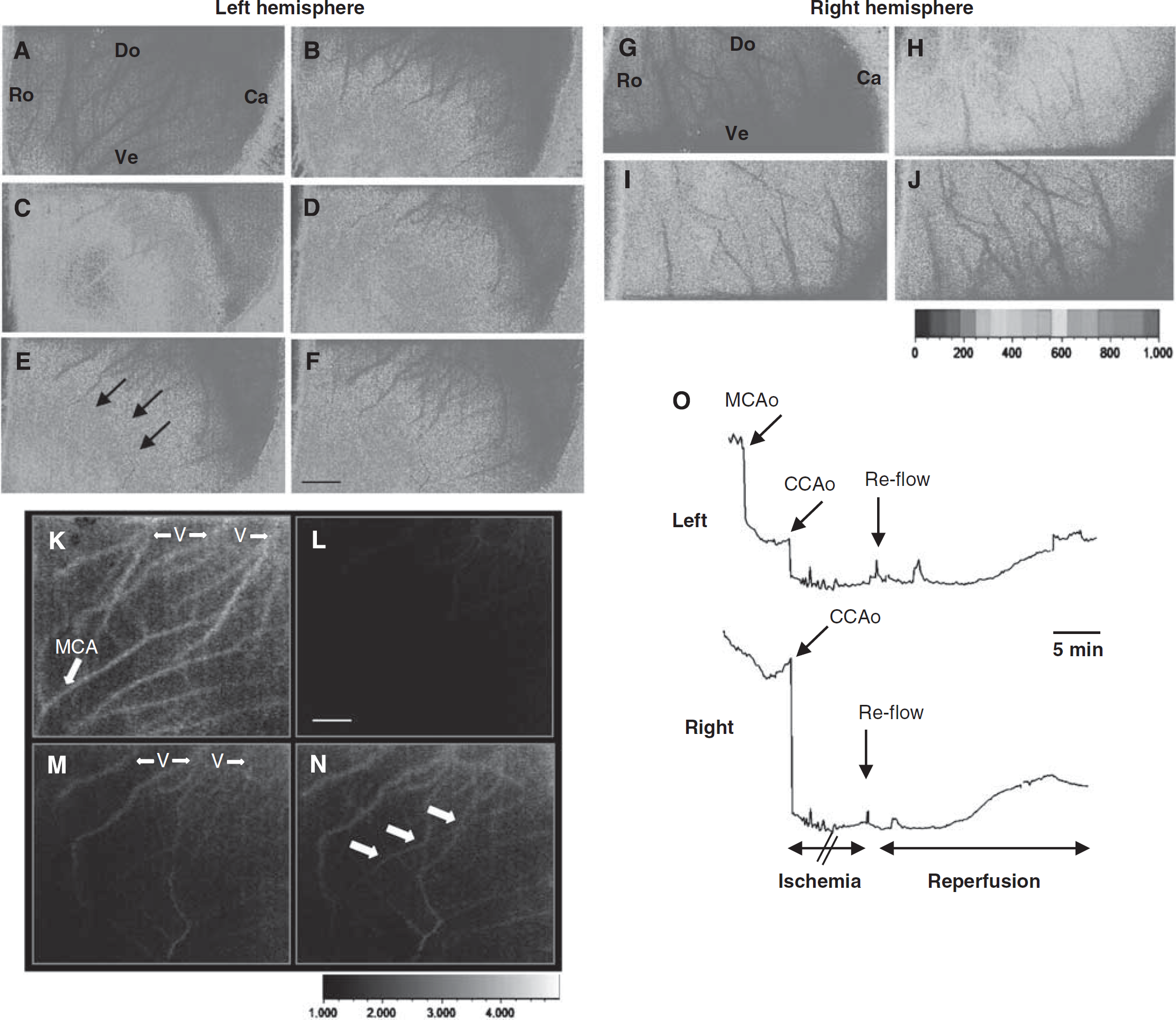
Typical example of spatio-temporal evolution of blood changes in the cerebral cortex in response to neonatal ischemia and reperfusion. (
Distribution of Local Cerebral Blood Flow During Ischemia and Early Reperfusion
In sham-operated rats (
CBF values in the hemibrain contralateral (CL) and ipsilateral (IL) to the MCAo side in the three groups of P7 rats
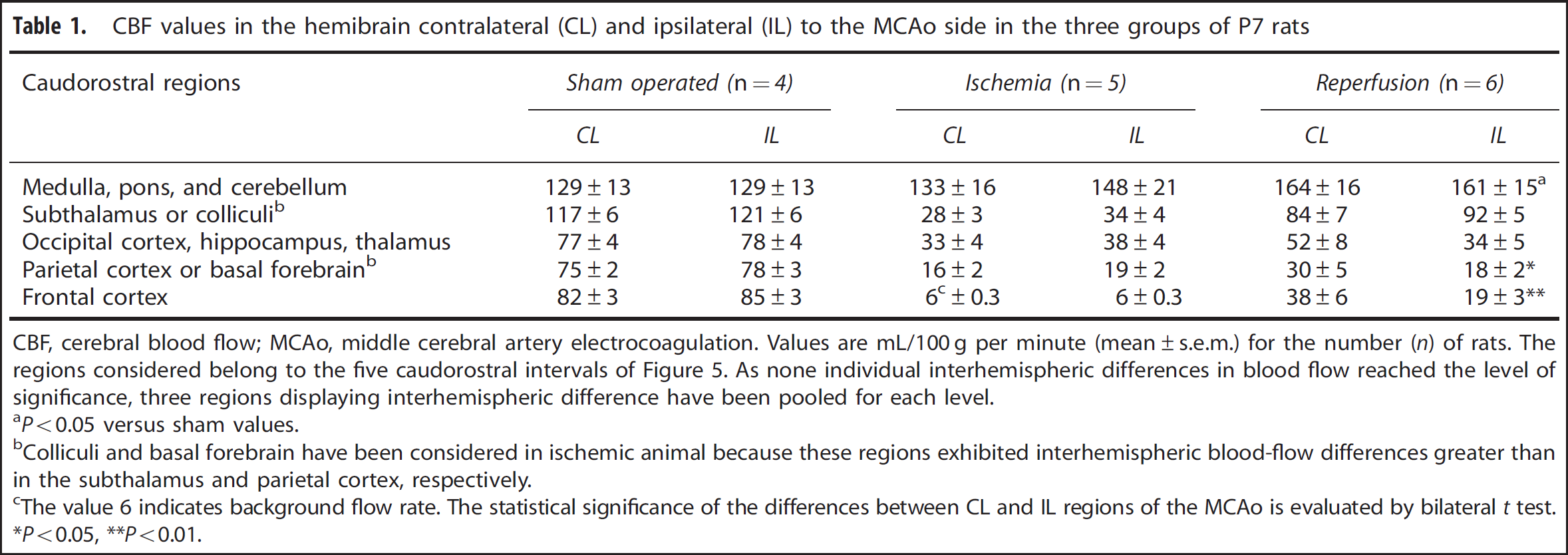
CBF, cerebral blood flow; MCAo, middle cerebral artery electrocoagulation.
Values are mL/100 g per minute (mean±s.e.m.) for the number (
The regions considered belong to the five caudorostral intervals of Figure 5. As none individual interhemispheric differences in blood flow reached the level of significance, three regions displaying interhemispheric difference have been pooled for each level.
Colliculi and basal forebrain have been considered in ischemic animal because these regions exhibited interhemispheric blood-flow differences greater than in the subthalamus and parietal cortex, respectively.
The value 6 indicates background flow rate. The statistical significance of the differences between CL and IL regions of the MCAo is evaluated by bilateral
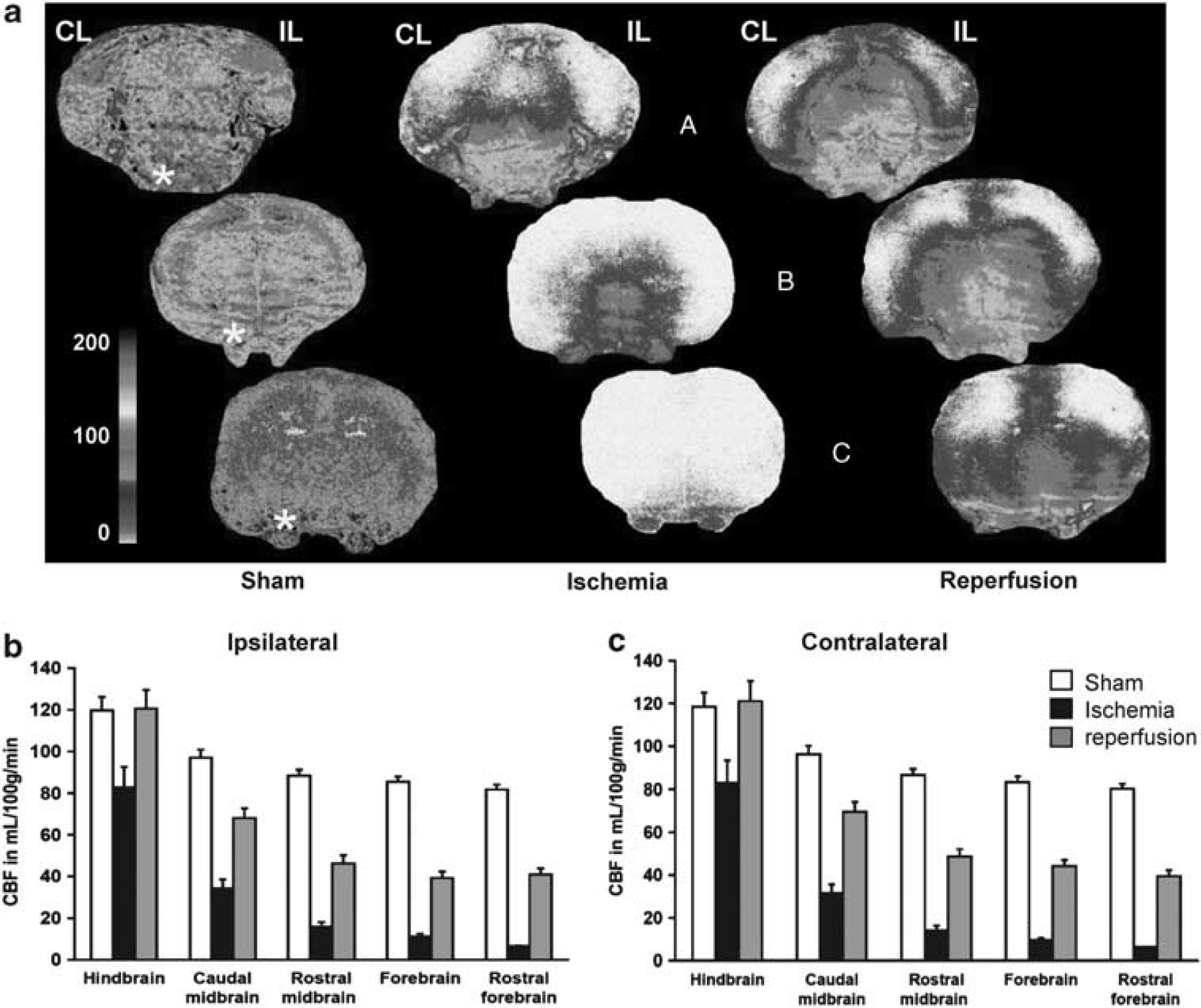
Cerebral blood flow (CBF) distribution in sham-operated, ischemic, and reperfused P7 rat pups. (
Rostrocaudal Blood-Flow Profiles During Early Reperfusion
The changes in local CBF with respect to sham-operated rats at different caudorostral levels are illustrated in Figure 5. A caudorostral gradient is still present during ischemia (not shown) and reperfusion, but perfusion in ventral and medial regions remained less affected. This observation was substantiated in distinguishing blood flow in ventrodorsal and mediolateral regions. A detailed analysis of the results provided evidence that blood-flow decline is steeper in dorsal and lateral regions, comparatively to sham-operated rats (Figure 5). Thus, a ventrodorsal and a mediolateral blood-flow gradient are superimposed on the main caudorostral gradient.
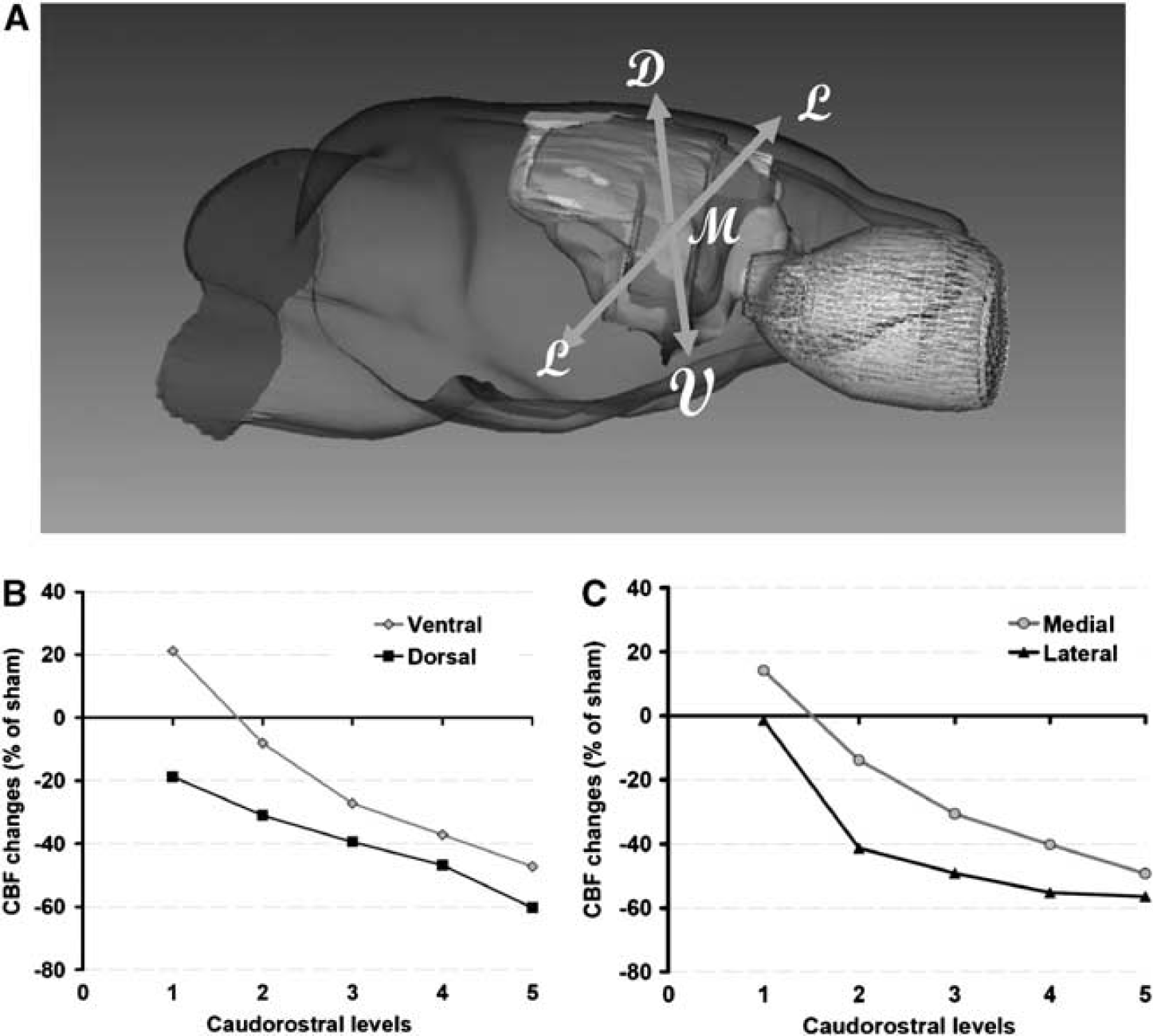
Regional cerebral blood flow (CBF) profiles during reperfusion. (
Effects of Unilateral Occlusion of the Middle Cerebral Artery on Hemispheric Blood-Flow Distribution
The effects of unilateral occlusion of the MCA were evaluated by side-to-side differences in blood flow (Table 1). None of the interhemispheric differences observed in sham-operated and ischemic rats were significant. However, a trend to increase was observed in the ipsilateral midbrain and hindbrain of ischemic animals (Table 1). In contrast, significantly lower blood flows were found in midbrain and forebrain regions ipsilateral to the occlusion in reperfused animals. Table 1 also showed that reperfusion in ipsilateral forebrain cortical areas remained very low, with at most 20 mL/100 g per minute.
Oxidative Phosphorylation During Ischemia and Early Reperfusion
During ischemia, mitochondria isolated from ipsilateral and contralateral cortex showed a similar and significantly decreased (~50% of the mean sham value) state 3, complex-1-dependent phosphorylating, respiration rate and uncoupled respiration along with poorer RCR (
Oxygen consumption during phosphorylating respiration (state 3) and respiratory control ratio (RCR) in mitochondria isolated from the ipsilateral (IL) and contralateral (CL) cortex of ischemic-rat pups
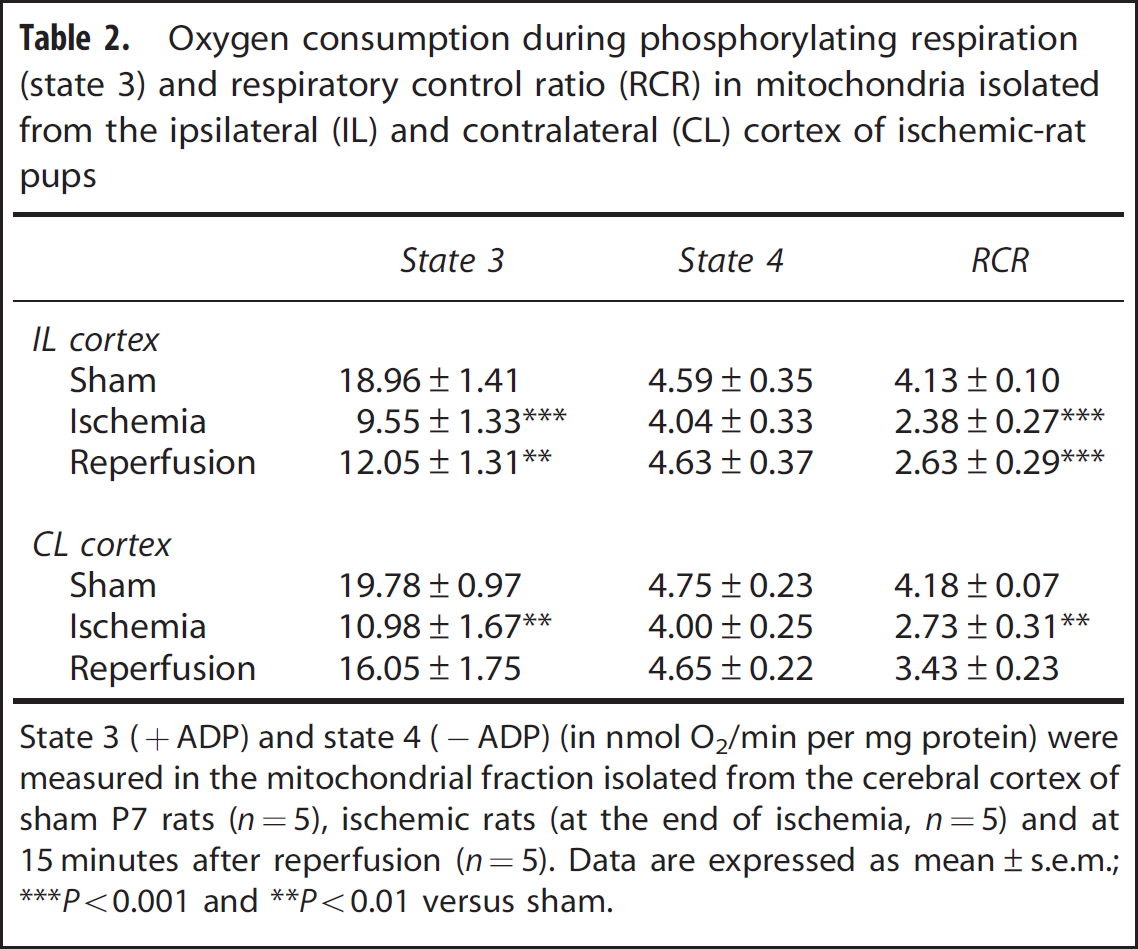
State 3 (+ADP) and state 4 (−ADP) (in nmol O2/min per mg protein) were measured in the mitochondrial fraction isolated from the cerebral cortex of sham P7 rats (
DISCUSSION
To our knowledge, this is the first study that defines arterial and multiregional CBF after cerebral stroke in the immature rat. A progressive incomplete reperfusion is depicted in the cortex during early reflow in both carotids. The gradual ipsilateral reperfusion is correlated with reduced local CBF, and reduced cortical mitochondrial respiratory function. In the contralateral hemisphere, reperfusion is also gradual with a recovery in cortical mitochondrial respiratory function. Collectively, these data indicate that the magnitude of reflow and cerebral energy metabolism after ischemia–reperfusion did not return to basal values early after reperfusion in the P7 rat, and highlight an efficient collateral network during ischemia and reperfusion.
Monitoring BFVs by using ultrasound imaging indicates no significant hyperemia over basal values in the three pre Willis large arteries. In both ICAs, reflow was potent but not maximum, and recovered to 60% to 70% within 15 minutes. Recovery became complete at 24 hours as previously reported.23–25 The early difference in mBFVs between the left and right ICA observed at early reflow may be explained by the higher level of vasodilation reached during ischemia in the left compared with the right hemisphere, because of the upstream permanent left MCA occlusion. Conceivably, at the time of CCA's reflow, blood could reflow toward both hemispheres with the same arterial pressure, but as the vascular network is more dilated in the left side (with MCAo), blood-flow rates and BFVs reached higher values than in the right side.
Monitoring cerebral perfusion in the penumbra by laser flowmetry indicates that reperfusion in the penumbra is gradual during the first 20 minutes of reflow and reaches the level obtained with permanent MCA occlusion in some animals at 30 minutes. Studies in adult rats have shown that there is a hyperemia at the early stage of reperfusion after focal or global ischemia. 26 This hyperemia could be related to the intense vasodilation of the local vascular network, because of the delayed collateral recruitment efficient from 1 hour (or more). 27 In contrast, we do not provide evidence for such hyperemia during early reflow in rat pups. Absence of hyperemia could reflect the absence of an important vasodilation consecutive to the very early potent blood-flow redistribution though the collateral network during ischemia (efficient from a few minutes). 16 The LSC imaging has become a useful tool to map spatial and temporal dynamic changes in tissue CBF, 28 and to identify extensive anastomotic connections between cerebral arteries.29,30 Here, we show the recruitment of anastomotic connections between distal segments of the occipital arteries (BT and PCA) and MCA at early reflow. Interestingly, recruitment of anastomotic connections between distal segments of the ACA and MCA provides immediate and persistent collateral blood flow to ischemic territories during ischemia in adult rat, but these connections disappear (anastomoses no more perfused) after spontaneous reperfusion. 29 Indeed, before reflow, flow in anastomoses follows the arterial blood pressure gradient from the vascular territory normally perfused with a good blood pressure, toward the vascular territory downstream the occluded feeding artery where the blood pressure is weak. At reflow, normalization of the blood pressure in the artery supplying the ischemic territory annuls the blood pressure gradient between the two extremities of the anastomoses, thus blood flows through itself.
Measurement of local tissue perfusion in the brain of neonates requires overcoming difficulties related to how to administer the diffusible radiotracer and to reliably evaluate tracer concentration irrigating the brain. For ischemia studies, the tracer is usually administered by intraperitoneal or subcutaneous infusion, which requires approximate estimation of the timing of the beginning and peak of the systemic tracer concentration.9,10 The peak of the curve is measured on blood collected from the severed neck at the end of tracer infusion, and the duration of measurement is long (1 to 2 minutes). As our experimental paradigm involves a general anesthesia during CBF measurement, we circumvent the inconvenience related to approximation on the time course of tracer concentration, by infusing the tracer into the jugular vein, and measuring the peak of the arterial concentration by intracardiac puncture exactly at the end of infusion. We found that the tracer concentration measured in the cardiac blood sample was at least twice that in the severed neck blood sample. Further accuracy on the arterial tracer concentration curve was provided by the use of programmed infusion, which reproducibly produced a ramp arterial input function.18,31 Compared with other studies, our CBF values are substantially higher than those reported in unanesthetized neonate rats.10,31,32 Our methodological CBF procedure can hardly account for the differences, since it is very close to that of Nehlig
Mitochondrial dysfunction is the most fundamental mechanism of cell damage in cerebral HI in the developing brain. 34 However, differences are observed between HI and stroke in P7 rat brain. Indeed, at 30 minutes of reoxygenation/reperfusion the activity of the respiratory chain in mitochondria isolated from HI mice is restored, exhibiting near-normal complex-I-dependent phosphorylating respiration rates, 35 whereas such recovery is not established in our neonatal stroke rat model at 15 minutes (this study) and 30 to 40 minutes 21 after reperfusion. In contrast, respiration rates recovered in the contralateral side, although CBF recovery was still incomplete. The later data are in accordance with the particular metabolism that mediates a better tolerance to hypoxia in neonatal than in adult brain. 36 After several hours of reperfusion after HI mitochondria again exhibit a profound secondary decline in their ability to generate ATP,37,38 known as a secondary energy failure. Previous results showed that adult rats subjected to 2 hours of focal ischemia, followed by recirculation, show an initial, partial, 39 or complete 40 recovery of mitochondrial O2 consumption, to be followed by a gradual decrease in RCR. 39 Therefore, our data suggest that cerebral blood-flow redistribution and mitochondrial O2 consumption differ between neonatal and adult rat during blood recirculation, and between HI and stroke (this study) in the immature brain.
In conclusion, the use of three separate techniques to evaluate blood flow in the brain during and after reperfusion in a neonatal stroke model provides useful information as each of them complements the others. Laser Speckle Flow and Laser Doppler Flow reflect red blood-cell velocity within microvessels as a measure of tissue perfusion in the brain surface, whereas color-coded pulsed Doppler ultrasound monitors red blood-cell vascular flow in the pre Willis arteries, reflecting downstream hemodynamic activity to cerebral hemispheres and more probably to deeper cerebral tissues. Blood flow autoradiography, which gives CBF in all cerebral regions, reflects tissue perfusion by blood plasma, as [ 14 C]-iodoantipyrine is a diffusible tracer. Altogether, our study highlights the strong similarity of results given by the three techniques, indicating an absence of hyperemia during early reflow according to respiration rate, and a prominent role for blood flow providing from the hindbrain and caudal midbrain supplied by the BT in an acute stroke model in the P7 rat. Gradual reperfusion found in the ipsilateral hemisphere also appeared to be fairly well correlated with impaired RCR.
DISCLOSURE/CONFLICT OF INTEREST
The authors declare no conflict of interest.
