Abstract
Substantial experimental data and recent clinical evidence suggesting that tissue reperfusion is a better predictor of outcome after thrombolysis than recanalization necessitate that patency of microcirculation after recanalization should be reevaluated. If indeed microcirculatory blood flow cannot be sufficiently reinstituted despite complete recanalization as commonly observed in coronary circulation, it may be one of the factors contributing to low efficacy of thrombolysis in stroke. Although microvascular no-reflow is considered an irreversible process that prevents tissue recovery from injury, emerging evidence suggests that it might be reversed with pharmacological agents administered early during recanalization. Therefore, therapeutic approaches aiming at reducing microvascular obstructions may improve success rate of recanalization therapies. Importantly, promoting oxygen delivery to the tissue, where entrapped erythrocytes cannot circulate in capillaries, with ongoing serum flow may improve survival of the underreperfused tissue. Altogether, these developments bring about the exciting possibility that benefit of reperfusion therapies can be further improved by restoring microcirculatory function because survival in the penumbra critically depends on adequate blood supply. Here, we review the available evidence suggesting presence of an ‘incomplete microcirculatory reperfusion’ (IMR) after focal cerebral ischemia and discuss potential means that may help investigate IMR in stroke patients after recanalization therapies despite technical limitations.
The best conceivable treatment for acute ischemic stroke is restoration of blood flow to the ischemic region, which is currently provided by thrombolysis. Unfortunately, a short time window limits the use of recanalization therapies, and not infrequently, thrombolysis is complicated by hemorrhage and edema because of increased vascular permeability.1–3 There is also a not well-recognized potential drawback associated with recanalization therapies, which is characterized by incomplete restoration of the microcirculatory flow in some parts of the ischemic tissue after reopening of the occluded blood vessel; the so-called ‘no-reflow phenomenon’.4–7 This phenomenon enjoyed considerable scientific interest after its first description in the 1970s;8–10 later, it lost its popularity because of the claims that it might be an experimental artifact,11–14 in addition to the concerns that it might be an epiphenomenon of the established tissue injury.9,10 However, substantial experimental data accumulating since the past decades15–20 and the recent clinical evidence suggesting that tissue reperfusion (restoration of microcirculatory blood flow) is a better predictor of outcome after thrombolysis than recanalization (reopening of the occluded artery)21,22 necessitate that this phenomenon should be reevaluated. If indeed the microcirculatory blood flow cannot be sufficiently reinstituted after brief focal cerebral ischemia despite complete recanalization as commonly observed in the coronary circulation,5,6 it might be one of the factors decreasing the efficacy of cerebral thrombolysis done within the time window when penumbral cells are still viable but microcirculatory obstructions have emerged. In addition to its potential use as a negative prognostic factor, understanding the conditions of reversibility of microvascular occlusions may create the possibility of improving recovery after recanalization therapies.
Here, we review the available evidence and discuss the potential means that may help investigate the presence and significance of incomplete microcirculatory reperfusion in thrombolysis patients. We also suggest to reserve the term ‘no-reflow’ for the impaired perfusion of the microvessels seen after long-lasting global ischemia and after prolonged or severe focal ischemia when microvessel occlusions are irreversible and prevent recovery from injury. The term ‘incomplete microcirculatory reperfusion’ (IMR) can be used to describe the potentially reversible ‘no-reflow’ observed after brief focal ischemia when there is still salvageable penumbral tissue. This formulation is based on the assumption that the microvascular responses are heterogeneous within the mini-cores and mini-penumbras at the territory-at-risk after focal ischemia as proposed by del Zoppo and colleagues. 23 Although the evidence is yet insufficient and the ischemic thresholds (severity, duration, etc) determining the reversibility of microvascular injury remain to be clarified, this distinction may be of clinical significance because the IMR can reduce the success of recanalization therapies as will be discussed below. The aim of this review is to bring this potentially important subject to the attention of stroke researchers with the hope of finding the imaging correlates of IMR having satisfactory resolution, and of developing therapeutic interventions.
IMPAIRED MICROVASCULAR PATENCY AFTER ISCHEMIA
An impaired reflow because of loss of microvascular patency (no-reflow) was first noted by Ames
CAN INCOMPLETE MICROCIRCULATORY REPERFUSION UNFAVORABLY AFFECT TISSUE SURVIVAL AFTER RECANALIZATION?
The incomplete microcirculatory reperfusion after prolonged focal ischemia could be an epiphenomenon of the established tissue injury; therefore, its reversal may not be of functional significance. In contrast, incomplete restoration of microcirculatory blood flow may negatively affect tissue recovery if reopening of the occluded artery is achieved early when there is still salvageable penumbral tissue. Considering that microvascular obstructions are already present 1 hour after MCA occlusion,37,38 and that a salvageable penumbra may exist at least 4.5 hours in some stroke patients, 1 improving microcirculatory reperfusion seems to be a promising strategy after recanalization therapies despite the lack of direct evidence connecting the microvasculature obstructions to the evolution of tissue injury as well as the findings in animal models to patients.
Parallel to observations in other tissues,
39
the idea that incomplete microcirculatory reperfusion could compromise tissue recovery after focal cerebral ischemia was primarily put forward by del Zoppo
DO PERICYTES CONTRIBUTE TO INCOMPLETE MICROCIRCULATORY REPERFUSION?
Endothelial swelling as well as compression caused by swollen astrocyte endfeet were postulated as the mechanism of narrowing of capillary lumina that, together with mechanisms that involve endothelial cell adhesion, promotes aggregation of blood cells and fibrin in capillaries.15,16,27 Recently, it has been shown that pericytes on microvessels contract during ischemia and remain contracted despite reopening of the occluded artery in a mouse model of focal ischemia
50
(Figure 1). This mechanism may significantly contribute to microvascular clogging because the active contraction of pericytes is likely to increase the microcirculatory resistance far greater than compression of the lumen by swollen endfeet because large volume changes at the periphery of a cylindrical structure causes relatively smaller changes in diameter. Moreover, segmental constrictions giving a sausage-like appearance to ischemic capillaries (see also Morris
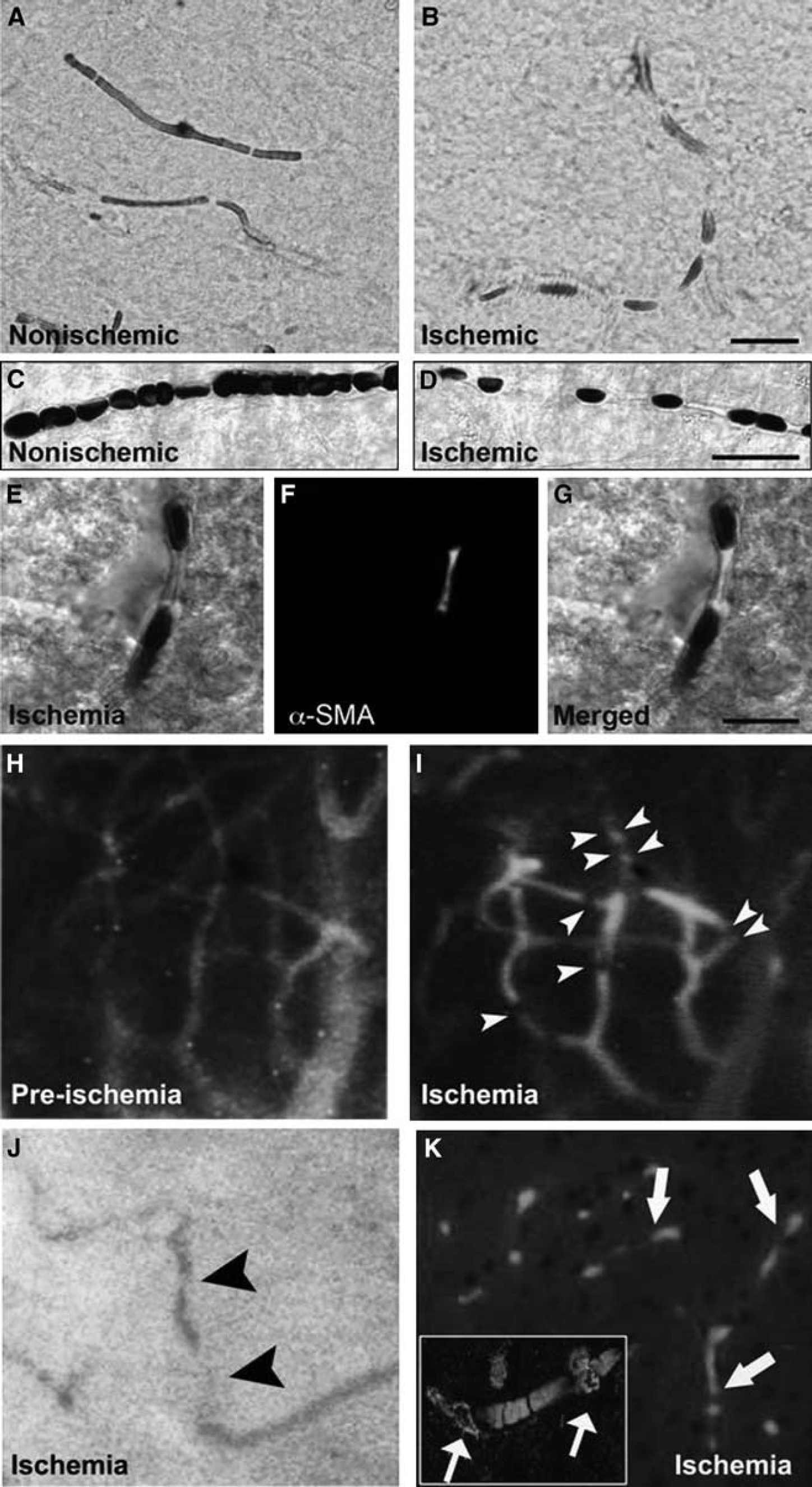
Cerebral ischemia induces intermittently spaced capillary constrictions, among which erythrocytes are entrapped. Brain sections in upper panels illustrate the cortical capillaries filled with horseradish peroxidase (HRP) injected before killing the mouse. In contrast to uninterruptedly filled nonischemic microvessels (
CAN INCOMPLETE MICROCIRCULATORY REPERFUSION HAVE CLINICAL SIGNIFICANCE?
Although the above reviewed evidence supports a role for IMR in stroke pathophysiology, the direct evidence is still missing because of the technical limitations, both experimentally and in clinical imaging. Technical insufficiencies also preclude the investigation of microcirculatory dynamics and associated metabolic changes in required resolution, especially in patients. Recent developments in multiphoton microscopy,43,68 optical coherence tomography, and phosphorescence quenching,69,70 as well as magnetic resonance imaging (MRI), may provide further insight and help to assess the potential impact of this interesting phenomenon on the outcome of recanalization therapies. Contrary views reported in the past were mostly based on examination of capillary patency with Evans blue,11,13 which may have filled the periphery of microvessel lumen with circulating serum and created the illusion of a patent lumen despite the presence of entrapped blood cells.
19
Indeed, capillary patency was found to be significantly compromised 1 hour after MCA occlusion when a tracer with different characteristics (e.g. fluorescein isothiocyanate–dextran 2,000 kDa) was used.37,38 Similarly, detection of regional cerebral blood flow with iodoantipyrine or laser Doppler flowmetry showed varying results ranging from clear local reperfusion deficits to near-normal recovery after MCA recanalization.13,71–74 Relatively free circulation of the radioactive tracer with serum may have caused the iodoantipyrine technique to underestimate the microcirculatory deficit caused by capillary cellular aggregates. Doppler shifts created by erythrocytes bypassing through patent capillaries and arteriolo-veneous anastomoses or thoroughfare channels may have led to pseudonormalized signal after recanalization.
75
Therefore, labeling erythrocytes might be a more reasonable approach to study the microcirculatory occlusions
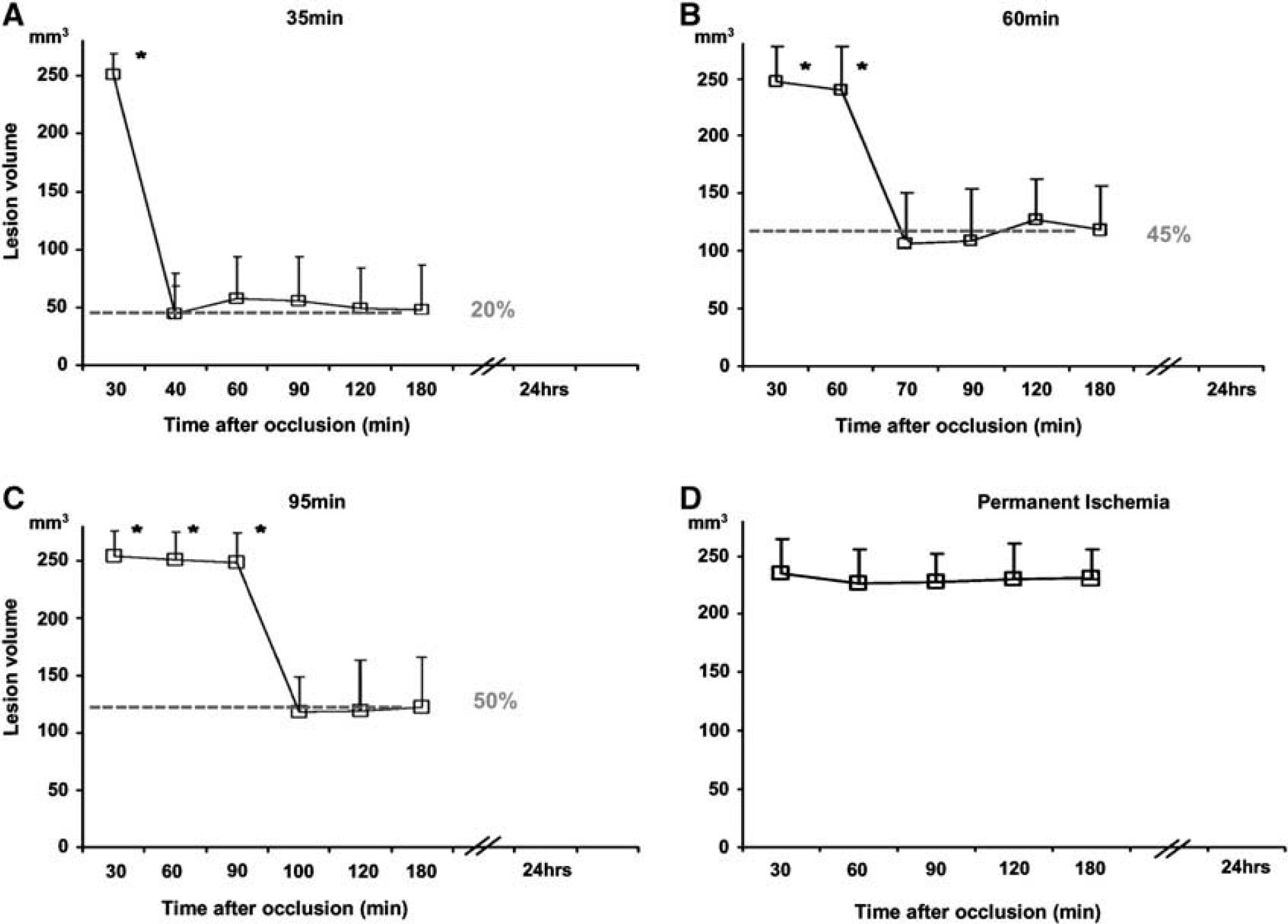
Temporal evolution of cerebral blood flow changes on magnetic resonance image-derived lesion volumes after (
CAN WE IMPROVE THE STROKE OUTCOME AFTER THROMBOLYSIS?
Unlike the situation in most stroke patients, the arterial occlusion was not produced with a clot, and recanalization was not attained with thrombolytic agents in most of the animal studies reviewed above. This may be of clinical significance because protracted recanalization as typically seen during thrombolysis may differently affect the microcirculation from prompt recanalization induced by the release of mechanical occlusion. However, a few studies in which MCA was embolized with autologous clots and tissue plasminogen activator (tPA) was used for recanalization reported that tissue reperfusion was only partly restored despite complete recanalization.79–81 This finding conforms to the idea of an impaired microcirculation despite recanalization and also raises the possibility that microemboli originating from the disintegrated thrombus could contribute to the clogging of microvessels. In stroke patients, clots occluding the arteries fragment, either spontaneously or by thrombolytics, may move downstream to obstruct distal arterial branches and sometimes arterioles (> 50
CLINICAL EVIDENCE SUGGESTING A ROLE FOR INCOMPLETE MICROCIRCULATORY REPERFUSION IN RECANALIZATION THERAPIES
Although recanalization is vital for the treatment of stroke, a subset of patients do not show any clinical improvement despite recanalization. 89 Among many other factors, incomplete reperfusion at the microcirculatory level might be one of the reasons for this discrepancy. Despite the well-established nature of IMR in experimental models of cerebral ischemia, the incidence and relevance of this phenomenon in the clinical setting has not been analyzed systematically. One major reason for the paucity of human data in this regard was the absence of clinical tools that provided temporal information regarding the status of recanalization and reperfusion in patients with acute ischemic stroke. Moreover, the current imaging techniques are far from to match the spatial resolution obtained by neuropathologic examination of microvascular bed in experimental animals. However, with the advent and widespread use of modern neuroimaging tools like computed tomography (CT) angiography, CT perfusion, MRI angiography, and MRI perfusion, together with more common application of intravenous and intraarterial thrombolysis in the acute stroke setting, we have started to get a better picture of the relationship between recanalization and reperfusion in humans (Table 1). The development and standardization of new imaging modalities that enable repetitive monitoring of tissue perfusion status, like arterial spin labeling MRI perfusion or transcranial Doppler ultrasound perfusion, will hopefully help us to better understand the nature of these complex pathophysiological events in humans in the near future.
Studies serially analyzing recanalization and reperfusion in ischemic stroke patients
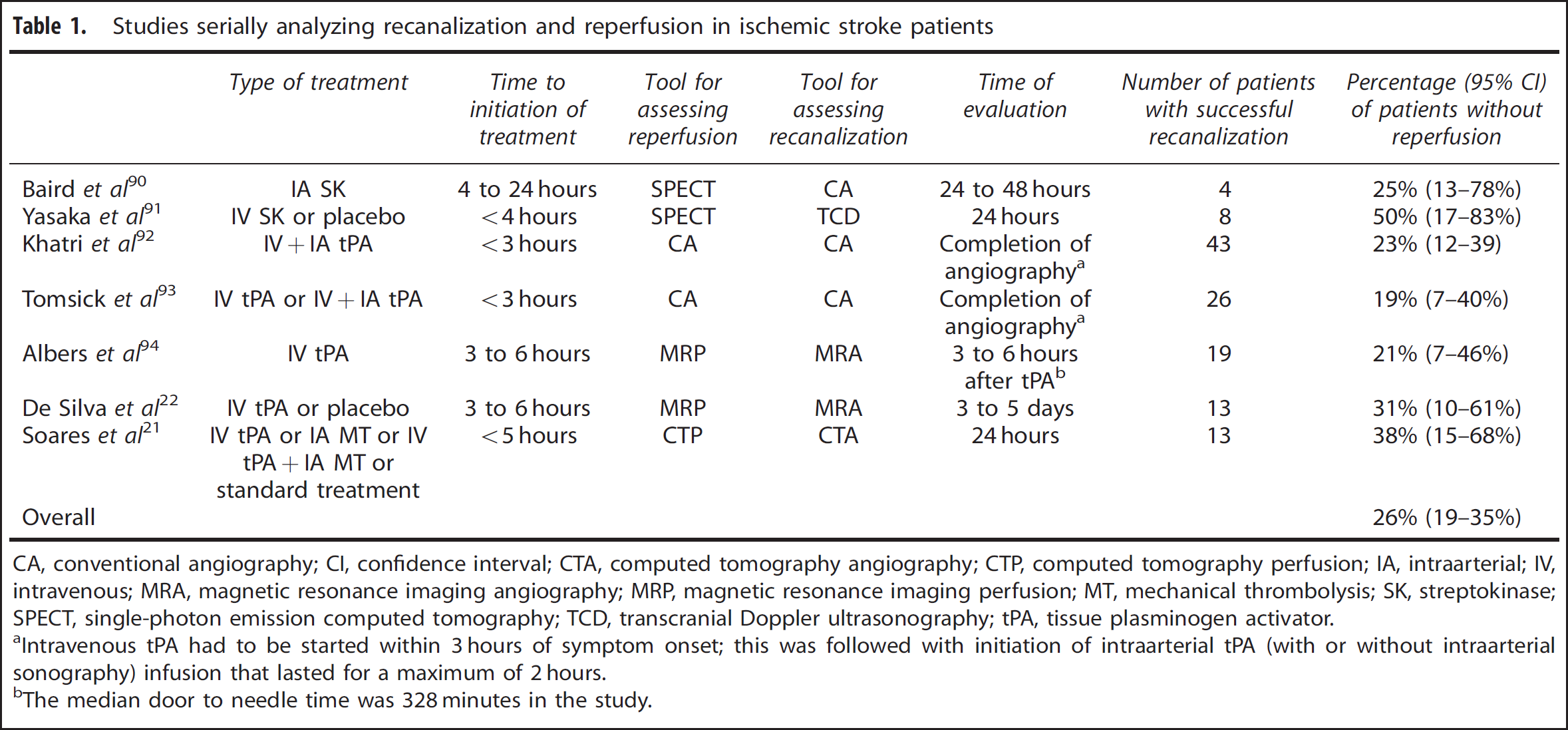
CA, conventional angiography; CI, confidence interval; CTA, computed tomography angiography; CTP, computed tomography perfusion; IA, intraarterial; IV, intravenous; MRA, magnetic resonance imaging angiography; MRP, magnetic resonance imaging perfusion; MT, mechanical thrombolysis; SK, streptokinase; SPECT, single-photon emission computed tomography; TCD, transcranial Doppler ultrasonography; tPA, tissue plasminogen activator.
Intravenous tPA had to be started within 3 hours of symptom onset; this was followed with initiation of intraarterial tPA (with or without intraarterial sonography) infusion that lasted for a maximum of 2 hours.
The median door to needle time was 328 minutes in the study.
The first hints regarding the presence of impaired reperfusion despite successful recanalization in humans came from studies that carried out serial single-photon emission CT studies combined with transcranial Doppler ultrasound or digital subtraction angiography in patients receiving streptokinase as part of clinical trials.90,91 In one study, out of five patients with an initial perfusion defect on serial single photon emission CT together with an intracranial artery occlusion, one patient had no cerebral reperfusion despite partial recanalization of MCA with intraarterial streptokinase. 90 Recanalization and reperfusion status were fairly correlated in the other three patients, and one additional patient had undergone partial reperfusion despite unsuccessful recanalization. 90 In another study, which compared the efficacy of intravenous streptokinase against placebo, out of 10 patients with initial vessel occlusion, 4 had successful recanalizaton, yet impaired reperfusion. 91 Reperfusion and recanalization were correlated in the remaining eight patients; six attained successful recanalization and reperfusion, whereas the remaining two reached neither end point. 91 Both studies showed that perfusion status, rather than successful or unsuccessful recanalization, had a significant impact on clinical outcome at follow-up.90,91
These studies were followed by analyses carried out in cohorts of patients receiving intravenous or intraarterial tPA in the acute ischemic stroke setting. In the International Management of Stroke-I (IMS-I) trial, which tested the efficacy and safety of intravenous and intraarterial tPA combination therapy, digital subtraction angiography was used for assessing both recanalization and reperfusion. 92 The analyses have indicated that despite a partial or complete recanalization of the proximal occlusion (graded as arterial occlusive lesion (AOL) score II or III), there was no evidence of distal branch filling (graded as Thrombolysis in Myocardial Infarction (TIMI) score of 1) in 10 out of 43 patients. 92 In the following IMS-II trial, the same treatment approach was combined with low-energy sonography applied by a micro-catheter. Similar to IMS-I, an AOL score of II or III was observed in 26 patients, but optimal reperfusion was achieved (graded as Thrombolysis in Cerebral Infarction (TICI) score of 2 or 3) in only 21 patients. 93 Both studies showed, once again, that clinical outcome was better correlated with reperfusion status rather than recanalization.92,93 Although angiography by itself cannot be considered as an appropriate tool for assessing tissue perfusion, these results highlight that there is a discordance between proximal and distal vascular beds in terms of vascular patency in a subset of stroke patients after recanalization.
More recent studies, using noninvasive tools like CT or MRI to assess vessel status and tissue reperfusion, have also identified the presence of patients with discordant recanalization and reperfusion. In the Diffusion and Perfusion Imaging Evaluation for Understanding Stroke Evolution (DEFUSE) trial, there were 4 patients out of 19 who did not show evidence for reperfusion on MRI after 3 to 6 hours of intravenous tPA despite recanalization. 94 The correlation between recanalization and reperfusion was significant, but moderate with a κ value of 0.58. 94 In the Echoplanar Imaging Thrombolytic Evaluation Trial (EPITHET), among 18 patients receiving intravenous tPA and with technically adequate serial angiography and MRI perfusion studies, recanalization was attained in 13 cases at days 3 to 5, 4 of whom with no success in reperfusion. 22 In multivariate analyses, reperfusion but not recanalization was significantly associated with good neurologic and functional outcome. 22 Consistently, another study has shown that reperfusion was a significant predictor of both clinical and tissue outcomes, whereas none of these outcome parameters were related to recanalization. 21 Out of 13 patients in this study, 5 had impaired reperfusion despite recanalization 21 (Figure 3). Although the supporting data continue to accumulate, we still do not know the exact mechanism of these observations in humans. Multiple mechanisms, ranging from fragmentation and migration of thrombotic particles into distal arterial beds to disturbed flow within the capillaries secondary to IMR, might be related to impaired perfusion on CT or MRI studies, despite restored patency in proximal arteries (hence, recanalization). 95 One promising aspect regarding the potential role of IMR in humans is that both the no-reflow phenomenon and most of the phenomena that are now considered critical in acute ischemic stroke, like the penumbra concept, have been shown to be present in non-human primates.7,96
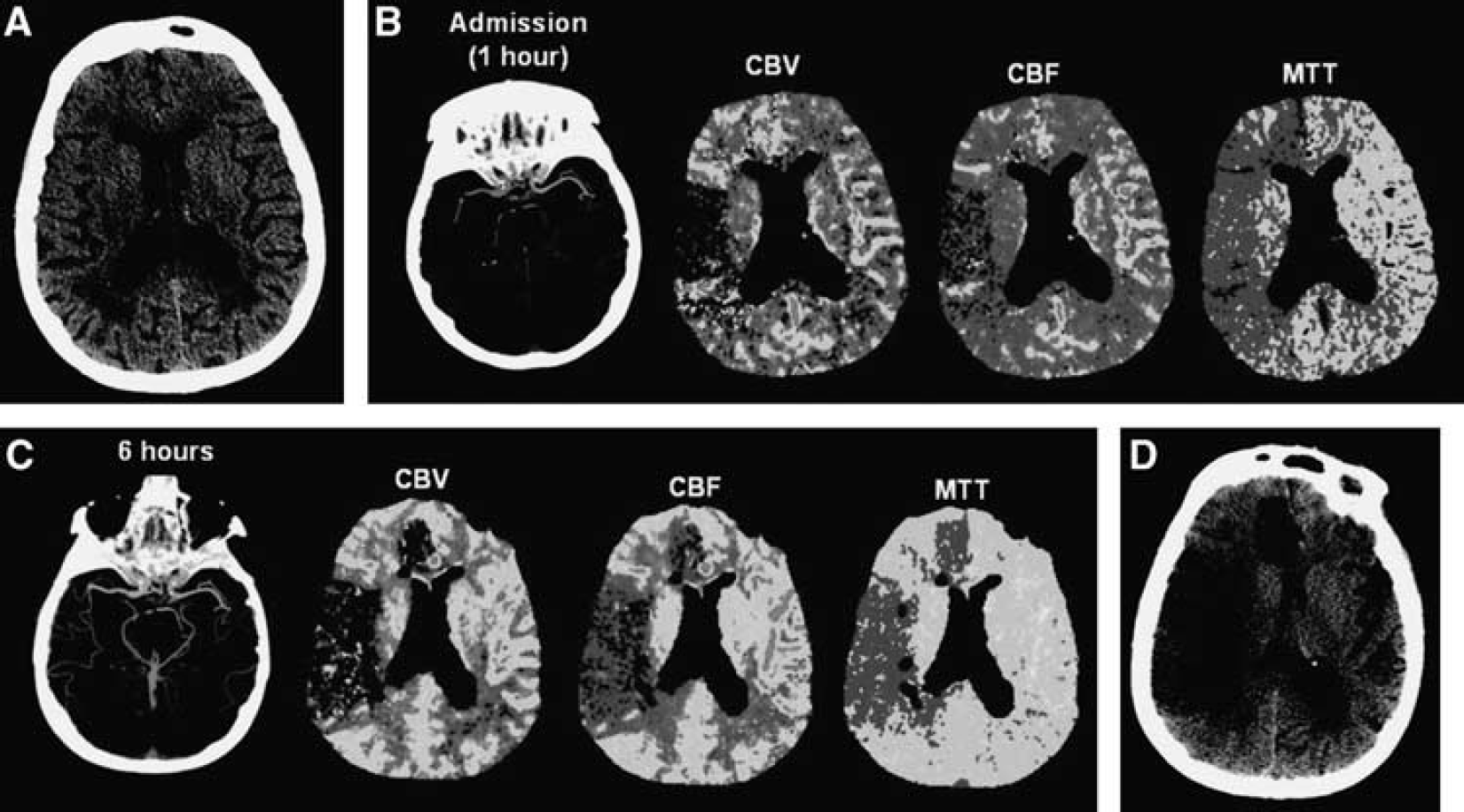
Absence of reperfusion, even in the setting of complete recanalization, may result in a large follow-up infarct volume. (
In conclusion, clinical studies suggest that a state of incomplete reperfusion is observed in approximately one quarter of patients with successful recanalization. The understanding of potential mechanisms that contribute to this microcirculatory insufficiency and development of therapeutic strategies to restore microcirculation should become one of the priorities of stroke research in the near future as both tissue and clinical outcomes heavily depend on successful reperfusion.
DISCLOSURE/CONFLICT OF INTEREST
The authors declare no conflict of interest.
