Abstract
Manganese (Mn2+)-enhanced magnetic resonance imaging studies of the neuronal pathways of the hypothalamus showed that information about the regulation of food intake and energy balance circulate through specific hypothalamic nuclei. The dehydration-induced anorexia (DIA) model demonstrated to be appropriate for studying the hypothalamus with Mn2+-enhanced magnetic resonance imaging. Manganese is involved in the normal functioning of a variety of physiological processes and is associated with enzymes contributing to neurotransmitter synthesis and metabolism. It also induces psychiatric and motor disturbances. The molecular mechanisms by which Mn2+ produces alterations of the hypothalamic physiological processes are not well understood. 1H-magnetic resonance spectroscopy measurements of the rodent hypothalamus are challenging due to the distant location of the hypothalamus resulting in limited measurement sensitivity. The present study proposed to investigate the effects of Mn2+ on the neurochemical profile of the hypothalamus in normal, DIA, and overnight fasted female rats at 14.1 T. Results provide evidence that γ-aminobutyric acid has an essential role in the maintenance of energy homeostasis in the hypothalamus but is not condition specific. On the contrary, glutamine, glutamate, and taurine appear to respond more accurately to Mn2+ exposure. An increase in glutamine levels could also be a characteristic response of the hypothalamus to DIA.
Introduction
Manganese (Mn2+) is an essential metal necessary for the normal functioning of a variety of physiological processes: it has a role in immune system functioning, regulation of cellular energy, and is cofactor for a variety of enzymes involved in neurotransmitter synthesis and metabolism (Aschner et al, 2007; Benedetto et al, 2009). However, it is also a neurotoxicant and induces psychiatric and motor disturbances as well as cognitive deficits such as memory and information processing impairments, reduced learning capacity, cognitive slowing, and decreased mental flexibility (Aschner et al, 2007; Benedetto et al, 2009). The molecular mechanisms by which Mn2+ produces alterations of the hypothalamic physiological processes are not well understood.
Recently, Mn2+-enhanced magnetic resonance imaging techniques demonstrated a growing potential for structural imaging (Silva et al, 2008) and the identification of neuronal pathways through tract tracing studies (Pautler, 2004). Manganese has the potential to enter excitable cells through voltage-gated calcium channels and acts as a marker of brain activation (Aoki et al, 2004). Previous Mn2+-enhanced magnetic resonance imaging studies established that several hypothalamic nuclei (the paraventricular nuclei of the hypothalamus (PVN), the lateral hypothalamus (LH), the ventromedial nucleus of the hypothalamus, and the arcuate nucleus) of rats and mice show specific enhancement patterns following systemic administration of MnCl2 associated with food-related challenges (Parkinson et al, 2009; Chaudhri et al, 2006). These enhancements were related to neuronal activity in the aforementioned hypothalamic nuclei.
In relation to these novel and promising techniques, dosage and administration routes of Mn2+ have been investigated (Silva and Bock, 2008), since acute exposure to high doses of Mn2+ can lead to hepatic failure and cardiac toxicity (Silva and Bock, 2008; Silva et al, 2004). Only a few NMR (nuclear magnetic resonance) spectroscopic studies were conducted to investigate the influence of Mn2+ on brain metabolism (Zwingmann et al, 2003, 2004, 2007). In rat models, Mn2+ affected both astrocytic and neuronal integrity (Zwingmann et al, 2003, 2004, 2007). Moreover, Mn2+ neurotoxicity has been associated with changes in brain energy metabolism and stimulation of oxidative glucose metabolism as well as protective astrocytic response (Zwingmann et al, 2004). Manganese in brain is preferentially accumulated in astrocytes and is known to stimulate the production of reactive oxygen and nitrogen species resulting in oxidative/nitrosative stress (Zwingmann et al, 2004, 2007). Additionally, Mn2+ was shown to induce cell swelling (Rama Rao et al, 2010). On the contrary, a model of dehydration-induced anorexia (DIA) was shown to induce cell shrinkage due to hyperosmolar challenge in rats (Jaimes-Hoy et al, 2008; Salter-Venzon and Watts, 2008, 2009).
Another benefit of using experimental models of Mn2+ exposure could therefore be the possibility to investigate neuronal and astrocytic mechanisms of regulation of the metabolism.
In the present work, our aim was to investigate the effects of Mn2+ on the proton neurochemical profile of the hypothalamus under disrupted energy balance conditions and following homeostatic changes. The following questions were addressed:
How does Mn2+ affect hypothalamic metabolism? What are the energetic effects of acute Mn2+ exposure? Can Mn2+ modify the metabolic response to a specific challenge? Can this modification be characterized?
The neurochemical profiles of the hypothalamus of healthy, dehydrated-induced anorexic (Jaimes-Hoy et al, 2008; Watts, 1999), and overnight food suppressed (OFS) rats were measured and examined with and without the influence of Mn2+.
Materials and methods
Animal Preparation
All studies were performed following the approval of Service de la consommation et des affaires vétérinaires du canton de Vaud (Switzerland). Thirty female Wistar rats (225 to 250 g body weight on day 0 (D0)) were divided into three groups: the control group (CTL, n=10), the OFS group (n=10), and the DIA group (n=10). Magnetic resonance (MR) experiments were conducted on one rat per group per session. Animals were acclimatized in an animal facility under humidity and temperature-controlled conditions on a 12-hour light/dark cycle during 1 week before the beginning of the experiments. Animals were allowed ad libitum access to standard chow and drinking water during this period.
On day 0 (D0), animals were placed in individual cages. Animals from the CTL group and the OFS group received water and food ad libitum; the dehydrated group (DIA) received 2.5% NaCl (Sigma, Zofingen, Switzerland) as drinking solution (Jaimes-Hoy et al, 2008; Watts, 1999). On D3 at 1800 hours, the OFS rats had their chow removed until the MR scan took place, 24 hours later. The amounts of food and drink were measured at 1800 hours on D0, D1, D3, D7, D10, and D14. The rats’ body weight was also measured. On D3, rats from each group were anesthetized with 2% isoflurane in a mixture of O2 and N2O via a facemask. The tail vein was catheterized for intravenous infusion of MnCl2. Each rat received an intravenous infusion of 100 mg/kg at 100 mmol/L MnCl2 at a rate of 0.5 mL to 1 mL/h via the tail vein to avoid cardiac arrest. Animals were all the time gently manipulated to avoid stress and suffering. During infusion and until animals were completely recovered from anesthesia, their temperature was carefully monitored and kept at 37°C±1°C.
Five female Wistar rats per group did not undergo MnCl2 infusions and were scanned on D4 after the start of anorexic protocol (D0) for the DIA group of rats.
During the MR experiments, rats were anesthetized with 2% isoflurane in a mixture of O2 and N2O. The rats were positioned in a dedicated holder with head fixation. Their body temperature was maintained at 37.5°C±0.5°C using a temperature-controlled water circulation and a rectal probe. Their respiration rate was monitored throughout the experiment. Magnetic resonance imaging and magnetic resonance spectroscopy (MRS) were performed on D4 24 hours after MnCl2 infusion.
Proton Magnetic Resonance Spectroscopy in the Hypothalamus and Hippocampus
Localized proton spectroscopy was performed using Spin-Echo full intensity Acquired Localized (SPECIAL) (Mlynárik et al, 2006), using echo time/repetition time (TE/TR)=2.8/4,000 milliseconds in combination with outer volume suppression and vapor water suppression (Tkác et al, 1999). A voxel of interest of 50 μL was localized in the hippocampus and the PVN–LH region for all the rats using the previously acquired 3D-gradient echo (GRE) images. All first- and second-order shims were readjusted using Fast Automatic Shimming by Mapping along Projections (FASTMAP) (Gruetter and Tkác, 2000) in the voxel of interest, which resulted in water linewidth of 23±5 Hz. To obtain adequate signal-to-noise ratio, 640 scans were acquired. For absolute quantification, water signal was acquired using identical parameters, except without water suppression and averaging eight scans.
Quantification and Statistics
The in vivo1H-MR spectra were processed using linear combination (LC) model (Provencher, 1993). In this study, 18 metabolite concentrations were quantified using databases of simulated spectra of metabolites (Mlynárik et al, 2006). Only metabolites with Cramer–Rao lower bounds under 20% were kept for further analysis.
All data are presented as mean±standard error of the mean (s.e.m.) unless otherwise stated. Two-tailed unpaired Student's t-tests were applied to compare data acquired in the three groups of rats. A P value<0.05 is considered significant.
Results
3D-gradient echo T1-weighted images obtained at 14.1 T were used to position carefully the voxels of interest in the hippocampus and the hypothalamus of female rats. 3D images provided an increased spatial resolution in the rat brain and voxels of interest were positioned with reference to the Paxinos atlas (Paxinos and Watson, 1998). Despite challenges due to the depth of the hypothalamus in the rat, good quality spectra were obtained using a surface radiofrequency (RF) coil at 14.1 T and relative to the hippocampus (see Supplementary file). Eleven metabolites were quantified reliably in the hypothalamus with Cramer–Rao lower bounds under 20%. The comparison of 1H-MRS spectra of the hypothalamus with spectra of the hippocampus in normal female rats demonstrated important differences such as lower Glu, Tau, N-acetyl-aspartate (NAA), PCr, PCr+Cr (tCr) concentrations in agreement with the previous findings in mice (Lei et al, 2010). A lower γ-aminobutyric acid (GABA) peak was also found in female rats. A comparison of 1H-MRS spectra of the hypothalamus with spectra of the hippocampus in female rats subjected to dehydration by drinking a hypertonic saline solution demonstrated higher concentrations of most metabolites in both the hippocampus and the hypothalamus (relative differences in hypothalamus: ΔTau=+106%, ΔtCr=+40%, Δmyo-Ins=+92%, ΔGABA=+160%; in hippocampus: ΔTau=+30%, ΔtCr=+50%, Δmyo-Ins=+72%). Interestingly, GABA was lower in the hippocampus of DIA rats (ΔGABA=−50%).
1H-MRS spectra are presented in Figure 1 for each condition CTL, DIA, and OFS with and without MnCl2. Twenty-four hours after infusion of MnCl2 in the rat tail vein, CTL rats demonstrated a significant increase in GABA and Lac concentrations (t-test, P=0.01) compared with CTL rats with no MnCl2 infusion (Figure 2A). MnCl2-infused DIA rats demonstrated no significant difference with non-MnCl2-infused DIA rats except for Tau (t-test, P=0.01) (Figure 2B). Under DIA condition, all metabolite concentrations increased relative to the CTL condition. However, no significant differences were detected between CTL and DIA rats in the presence of MnCl2. In particular, the relative difference for GABA between CTL and DIA was only 3.3%. To verify that changes seen between MnCl2 and non-MnCl2-infused DIA rats were solely related to osmotic changes due to hypertonic saline in the rat hypothalamus, a third group of rats was introduced. These rats had their food suppressed overnight (OFS). Five of them received an infusion of MnCl2. Their hypothalamic neurochemical profiles 24 hours post-MnCl2 infusion showed lower metabolite concentrations compared with OFS rats without MnCl2 (ΔGlu=−28%, t-test, P=0.03; ΔTau=−55%, t-test, P=0.04; ΔNAA, ΔCr∼−40% but P>0.05) (Figure 2C). Glc was undetectable and GABA and myo-Ins levels increased, although not significantly, relative to OFS rats without MnCl2. In MnCl2-infused OFS rats, metabolite concentrations had a tendency to decrease relative to their CTL (ΔGABA=−6%, ΔTau=−17%, ΔGlu=−18%, ΔLac=−40%), whereas most metabolite concentrations increased significantly in OFS rats without MnCl2 relative to their CTL. Figure 3 compares metabolite concentrations for CTL, DIA, and OFS rats (Figure 3A) and for CTL (+Mn2+), DIA (+Mn2+), and OFS (+Mn2+) (Figure 3B). While significant differences were found between the neurochemical profile of CTL and the neurochemical profiles of DIA and OFS, these differences appear to be masked in the presence of Mn2+.
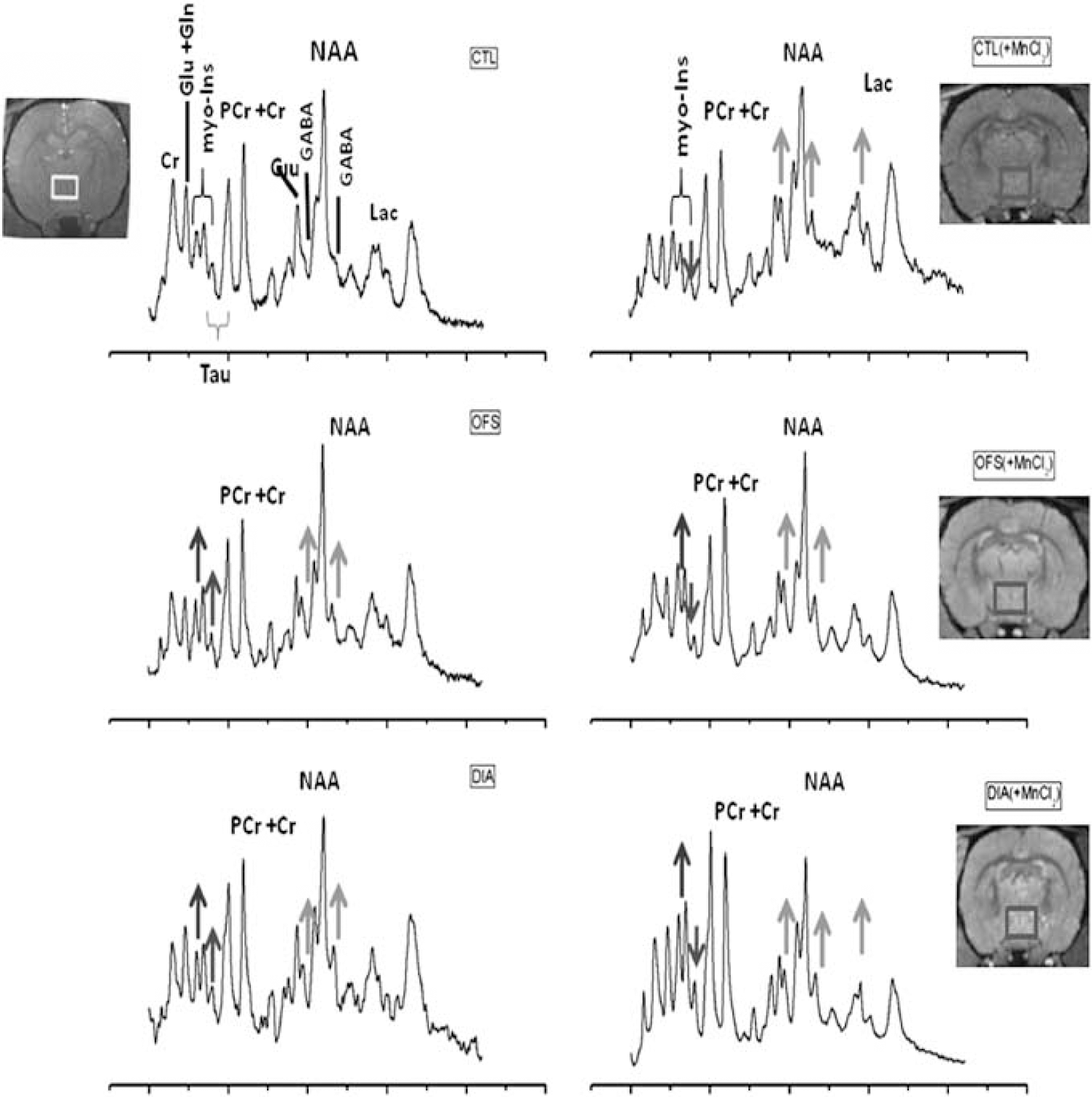
1H-nuclear magnetic resonance (NMR) spectra obtained at 14.1 T of the rat hypothalamus in control (CTL), dehydrated-induced anorexic (DIA), and overnight food-restricted (OFS) rats with metabolite assignments. Peak assignments: Cr, creatine; GABA, γ-aminobutyric acid: Gln, glutamine; Glu, glutamate; Lac, lactate; myo-Ins, myo-Inositol; NAA, N-acetyl-aspartate; Tau, taurine. The voxels of interest are overlaid on 3D-T1-weighted gradient echo images (TR/TE=20/5 milliseconds, flip angle=70°, field of view=25 × 25 × 25 mm3, matrix size=256 × 256 × 128, coronal slices, BW=25 KHz, five averages). Spectra were also obtained 24 hours after an intravenous injection of 100 mg/kg MnCl2 in CTL (MnCl2), DIA (MnCl2), and OFS (MnCl2) rats. Colored arrows indicate increases or decreases of the metabolites of interest relative to the CTL spectrum: red for GABA and lactate; green for myo-Ins; and violet for Tau. The color reproduction of this figure is available at the Journal of Cerebral Blood Flow and Metabolism journal online. BW, bandwidth; TE, echo time; TR, repetition time.
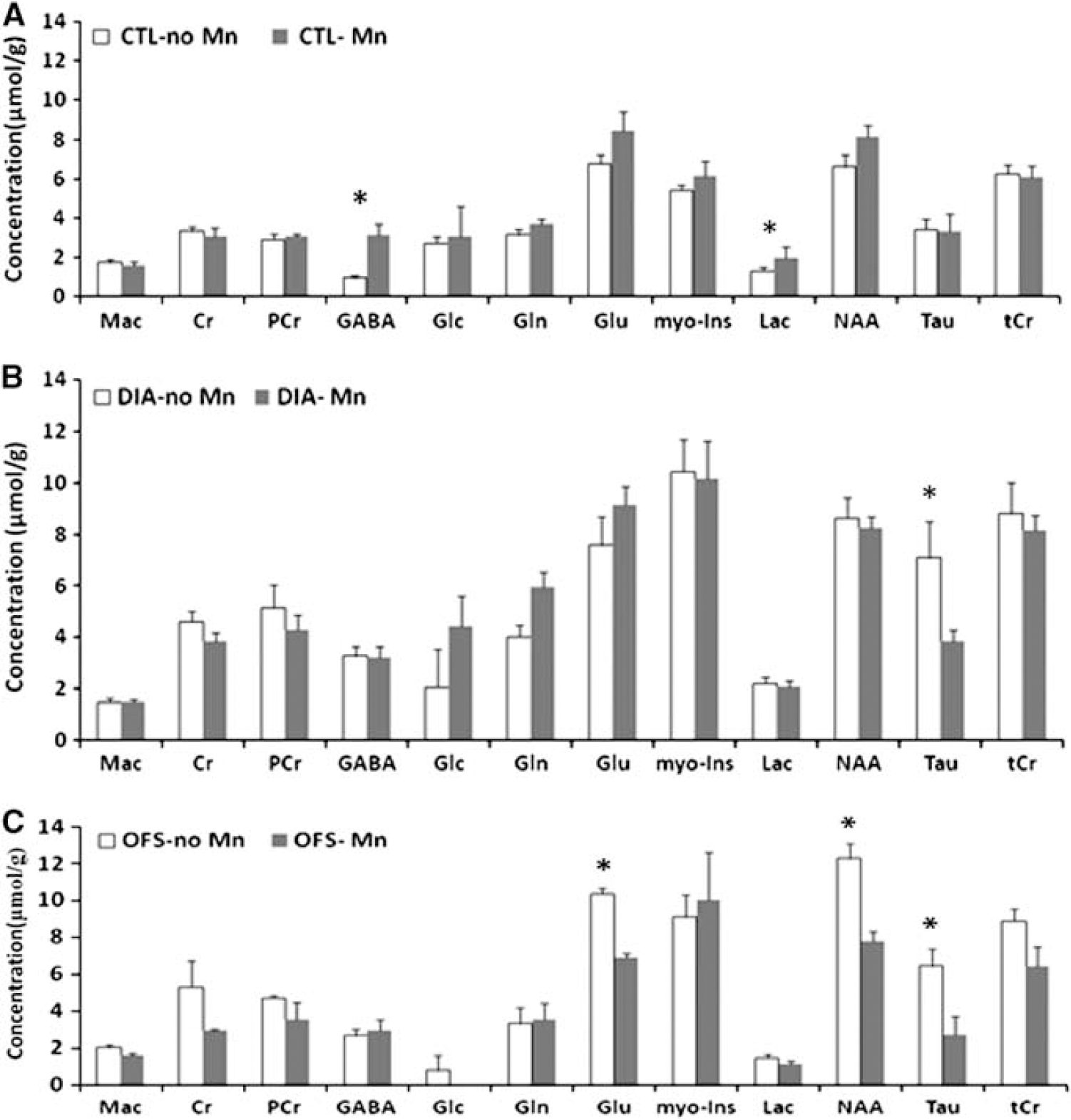
Effect of manganese (Mn2+) on the neurochemical profile of control (CTL), dehydration-induced anorexia (DIA), and overnight food suppressed (OFS) rats. (
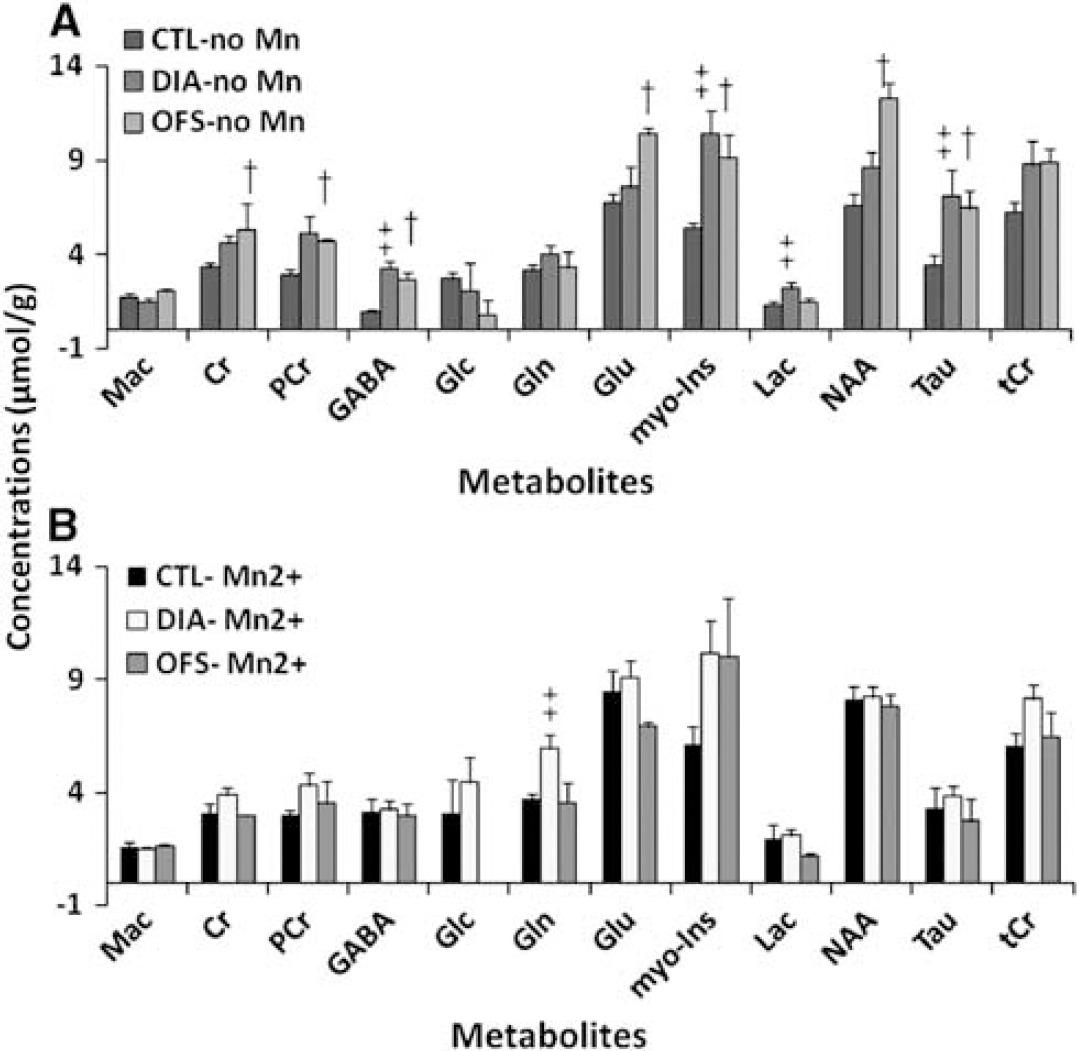
Comparison between control (CTL), dehydration-induced anorexia (DIA), and overnight food suppressed (OFS) rats. (
The relative concentration changes (percentage of CTL) are presented in Figure 4. Significant differences were found between DIA and OFS (two-tailed t-test; Gln, Glu, GABA: P<0.0001; Lac: P=0.0048; NAA: P=0.015; Tau: P<0.05). Significant differences also exist between CTL (+Mn2+), OFS (Mn2+), and DIA (+Mn2+) relative to the CTL group (two-tailed t-test between CTL (+Mn2+) and DIA (+Mn2+): PCr, Gln: P<0.0001; between OFS (Mn2+) and DIA (+Mn2+): Gln, Glu, Lac: P<0.0001). Statistically significant differences between groups of rats exposed to Mn2+ and rats nonexposed are also depicted in Figure 4. Although averaging groups of rats challenged by different conditions appear arbitrary, the ‘gross’ effect of Mn2+ on the neurochemical profile of the hypothalamus is clearly shown for Gln in DIA, Glu, and NAA in OFS and Tau for all conditions.
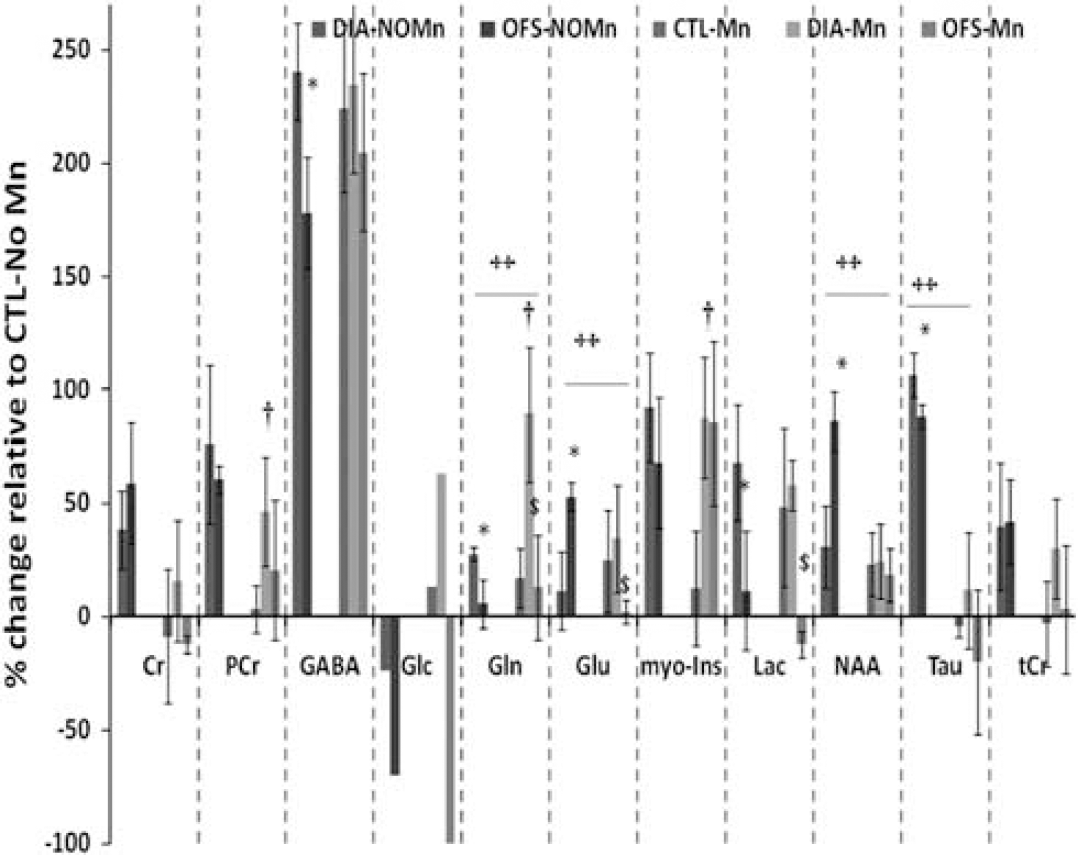
Effect of manganese (Mn2+) on metabolites of the hypothalamus under normal (control, CTL), dehydration-induced anorexia (DIA), and overnight fasted (OFS) conditions. The calculated mean percentages of the CTL without manganese (±s.d.) are represented. Significant differences were found between DIA and OFS (∗two-tailed t-test; Gln, Glu, γ-aminobutyric acid (GABA): P<0.0001; Lac: P=0.0048; NAA: P=0.015; Tau: P<0.05). Significant differences also exist between CTL (+Mn2+), OFS (Mn2+), and DIA (+Mn2+) relative to the CTL group (two-tailed t-test ‡between CTL (+Mn2+) and DIA (+Mn2+): PCr, Gln: P<0.0001; $; between OFS (Mn2+) and DIA (+Mn2+): Gln, Glu, Lac: P<0.0001). Statistically significant differences between groups of rats exposed to Mn2+ and rats nonexposed are also depicted(++P<0.05).
Discussion
Manganese is an essential element for normal rodent and human development. It is required for the appropriate functioning of a variety of physiological processes (Aschner et al, 2007; Benedetto et al, 2009). Recently, magnetic resonance imaging techniques have taken advantage of the paramagnetic properties of this compound identified as a potential indirect noninvasive neuronal marker (Duong et al, 2000) and allowing the identification of various neuronal pathways within the rodent brain in tract tracing studies (Pautler et al, 1998, 2004). However, excessive levels of Mn2+ are toxic for the central nervous system (Stanwood et al, 2009). Silva et al (2004) have reported behavioral disorders such as somnolence, convulsions, tremors, and respiratory effects in both rats and mice at high Mn2+ doses (>90 mg/kg) independent of the route of administration. Symptoms similar to Parkinson's disease have also been reported (Benedetto et al, 2009; Olanow, 2004; Pal et al, 1999). Moreover, Mn2+ was also reported to disrupt the astrocytes glutamine transporter expression and function in astrocytic cell cultures (Sidoryk-Wegrzynowicz et al, 2009) and to alter dopaminergic and GABAergic neurons within the basal ganglia (Stanwood et al, 2009). Although the mechanisms underlying Mn2+ neurotoxicity remain unclear and chronic exposure to Mn2+ is a topic of interest since neuropathologic changes in humans lead to neuronal loss, gliosis and Alzheimer disease, the present study examined the effects of acute exposure to Mn2+ in relation to previous Mn2+-enhanced magnetic resonance imaging studies in the hypothalamus.
The hypothalamus is responsible for metabolic processes of the autonomic nervous system. This extremely complex brain region coordinates many behavioral and physiological mechanisms as well as complex homeostatic processes. It must respond to many different signals internally and externally generated. Moreover, the hypothalamus is richly connected with the central nervous system. For an appropriate understanding of the function of hypothalamus, we challenged it with dehydration and fasting and investigated its metabolic response to Mn2+ exposure.
Comparison of the Neurochemical Profiles of the Hypothalamus and the Hippocampus of the Rat
The neurochemical profile of the normal rat hypothalamus was characterized by low PCr, Glu, Tau, and NAA concentrations relative to those of the hippocampus in agreement with a previous study in mice at 14.1 T (Lei et al, 2010). However, GABA and myo-Ins concentrations were also lower relative to the rat hippocampus, whereas these metabolites had significantly higher levels in the mouse hypothalamus relative to the hippocampus (Lei et al, 2010). γ-Aminobutyric acid is known to have an important inhibitory role in the regulation of the hypothalamic–pituitary–adrenocortical axis (Cullinan et al, 2008) and especially at the level of the paraventricular nucleus (Cullinan et al, 2008). No GABAergic neurons are located within the PVN itself (Cullinan et al, 2008), which may explain the lower GABA levels measured compared with the hippocampus in healthy rats.
In response to changes in brain homeostasis, metabolite concentrations in the hippocampus and the hypothalamus increased. A significant increase of GABA in DIA rats relative to CTL rats but also relative to the hippocampus was noticed.
In response to changes in osmolality, a cell shrinks or swells and finally exhibits a regulatory volume increase or decrease. On hypernatremic challenge, the cell loses water until intracellular and extracellular osmolality are equal. The recovery of water is mediated through accumulation of inorganic (Na+, K+, Cl−) and organic (glutamate, glutamine, taurine, myo-Inositol) osmolytes. Gln, Glu, Tau, GABA, myo-Ins rise in the brain of chronic hypernatremic rats (Sterns and Silver, 2007) and return to their baseline levels as soon as normal osmolar conditions are recovered.
A reduction in brain water due to dehydration would be expected to decrease brain metabolite concentrations, 4 days after initiation of DIA. Dehydration-induced anorexia rats lost ∼15% of their initial weight (Just and Gruetter, 2011). If it is assumed that the entire weight loss can be attributed to dehydration then a 15% total body water loss can be assumed and metabolite concentrations can be downscaled to 15%, which might be overestimated in the brain, since cellular dehydration is compensated by the continuous ingestion of water loaded with sodium and volume regulation occurs.
Strikingly, levels of NAA also increased in DIA (P>0.05) and OFS (P<0.05) rats. N-acetyl-aspartate is believed to be a marker of neuronal density, integrity, and function. N-acetyl-aspartate reductions have been interpreted as reflecting neurotoxicity, neurodegeneration, or impaired mitochondrial energy metabolism. Consistent with this hypothesis, levels of NAA are reduced in a range of pathologic states with dramatic reductions in patients with tumor or stroke (Zimmerman and Wang, 1997). However, NAA has been identified as an osmolyte (Baslow, 2003). N-acetyl-aspartate is liberated into extracellular space in response to changing osmotic conditions but NAA was also hypothesized to be a molecular pump in which translocation creates an osmotic environment that passively removes water from the intracellular milieu of neurons (Baslow, 2003; Steen and Ogg, 2005).
Studies of psychiatric disorders have also shown that NAA levels can increase, suggesting a restoration or repair of neuronal viability (Moore et al, 2000; Mickael et al, 2003). The administration of lithium chloride to humans increased whole brain NAA levels, suggesting an increase of neuronal viability. However, lithium chloride also induces anorectic effects (Parkinson et al, 2009; Ishii et al, 2004). There were no significant difference in Mn2+ uptakes in hypothalamic regions involved in the control of food intake (arcuate nucleus, PVN, and supraoptic nucleus) between overnight fasted mice and mice receiving lithium chloride (Parkinson et al, 2009). The upregulation of NAA in OFS rats could be attributed to a transient neuroprotective effect of NAA in neurons in response to a short fasting period. This hypothesis is consistent with studies, demonstrating that fasting induces neuroprotective effects (Davis et al, 2008; Chiba et al, 2007).
γ-Aminobutyric Acid Responses to Hypertonicity and Manganese
Without MnCl2, the hypothalamic inhibitory neurotransmitter GABA increased significantly in both DIA and OFS rats relative to CTL rats. γ-Aminobutyric acid is known to suppress feeding in rats and impact on body weight loss (Turenius et al, 2009). The majority of stress-responsive corticotropin-releasing hormone (CRH) neuronal inputs to the PVN contain the inhibitory neurotransmitter GABA, highlighting the importance of GABAergic neurotransmission in the regulation of the neuroendocrine stress response (Cullinan et al, 2008). Neurons of the PVN synthesize thyrotropin-releasing hormone and CRH hormones having an important role in energy homeostasis (Jaimes-Hoy et al, 2008). Under restricted food regime, rats show decreased thyrotropin-releasing hormone expression in the PVN while anorexic animals show increased thyrotropin-releasing hormone expression in the PVN. However, a reduction of CRH expression compared with CTL animals (Jaimes-Hoy et al, 2008) in the PVN was found in both food-restricted and DIA animals but to a greater extent in DIA rats. This finding parallels the increase of GABA seen in DIA and OFS rats but to a greater extent in DIA rats. Yet, CRH-R2, one of the receptors of CRH signal involved in anorexigenic effects (de Gortari et al, 2009) was downregulated in the PVN of DIA rats but not in food-restricted animals (de Gortari et al, 2009). GABAergic projections from regions surrounding the PVN (anterior hypothalamic area, dorsomedial hypothalamic nucleus, LH, medial preoptic area) convey inhibitory signals to CRH neurons in response to a stressor such as DIA and food deficiency and limit hypothalamic activity by an important braking mechanism that includes increase of GABA release at the level of the PVN (Cullinan et al, 2008). It was suggested (Watts, 1999) that CRH neurons of the LH are specifically activated by the dehydration-induced stimulus and project to neurons in the PVN so that food inhibition due to low energy intake is overridden by dehydration mechanisms.
It is known that infertility can result when food intake fails to compensate for increased energy demands such as in anorexia nervosa. MnCl2 stimulates the release of luteinizing hormone releasing hormone from the hypothalamus but simultaneously increases GABA release that in turn inhibits luteinizing hormone releasing hormone release (Prestifilippo et al, 2008). These processes also involve the activity of nitric oxide synthase (Prestifilippo et al, 2008) and may ultimately contribute to suppressed reproductive function in both male and female rats (Prestifilippo et al, 2008). As a consequence, further increases of GABA levels would have been expected in rats under both anorexic and Mn2+ condition. Nonetheless, GABA levels were similar in CTL (+Mn2+), DIA (+Mn2+), and OFS (+Mn2+) rats. Moreover, GABA concentrations were not significantly increased in DIA (+Mn2+) and OFS (+Mn2+) rats relative to their corresponding DIA and OFS rats. This suggests that the effects of dehydration and food restriction and MnCl2 are not summed. MnCl2 appears to blur the effects of anorexia and food restriction on GABA. Dehydration-induced anorexia animals demonstrated similar metabolite levels with and without MnCl2 except for Tau. As pointed out earlier, both challenges should induce GABA release in the PVN but GABA levels were similar in DIA and DIA (+Mn2+) rats, suggesting that the effects of one condition may override the effects of the other. Salt loading and Mn2+ induce opposing effects at the cellular level. When exposed to hypertonicity, a cell shrinks until intracellular and extracellular osmolalities are equal. In contrast, Mn2+ induces cell swelling (Rama Rao et al, 2010). Cell swelling and cell shrinkage have opposing effects on Ca2+ concentration and hormonal secretion (Ben-Tabou De-Leon et al, 2003, 2006). Due to dehydration, morphological plasticity of astrocytes in the PVN occurred. Thus, synaptic and extrasynaptic transmission in magnocellular neurons of the PVN were modified with increased synaptic release of glutamate and GABA but reduced ability of Tau to bind to glycinergic receptors and modulate neuronal excitability (Oliet and Bonfardin, 2010).
Previous investigations also suggest that signaling of astrocytes to neurons is performed through Ca2+-dependent release of glutamate. We suggest that under hypertonic conditions, the Mn2+ enhancement seen in the PVN (and corresponding to neuronal activation) is mediated through astrocytic signaling. This hypothesis appears consistent with in vitro findings, showing that astrocytes increased their oxidative and anaerobic glucose metabolism to protect against energy failure and oxidative stress induced by Mn2+. Simultaneously, Mn2+ induced neuronal energy failure (Zwingmann et al, 2003). For the maintenance of neuronal energy and neurotransmitter metabolism, the hypothesis of an astrocyte–neuron ‘lactate shuttle’ (Pellerin et al, 2007; Zwingmann et al, 2003) might hold.
γ-Aminobutyric acid levels increased significantly for all the conditions explored in the present study including with Mn2+. Hence, GABA appears to be a marker of homeostatic changes within the rat hypothalamus but it cannot be considered as a specific marker.
Spectroscopic Analysis of the Hypothalamus Energy Metabolism in Manganese Neurotoxicity
The characteristic and significant GABA increases in fasted animals (Violante et al, 2009) and MnCl2-infused animals (Zwingmann et al, 2004, 2007) relative to normal animals were already described following the measurements of brain extracts using Carbon-13 (13C)-NMR spectroscopy. These metabolic changes in the glutamine–glutamate–GABA cycle were attributed to impaired cerebral energy metabolism. This hypothesis was further confirmed since both fasted and MnCl2-infused animals demonstrated significantly increased lactate levels measured ex vivo relative to normal animals. Lactate synthesis is known to counteract decreased mitochondrial energy production (Zwingmann et al, 2004, 2007). Lactate also increased in our in vivo study for CTL (+Mn2+) rats but not for OFS (+Mn2+) rats. Violante et al (2009) attributed the increased GABA levels of fasted mice to a reduction in the GABA degradation pathway since no significant increases in Glu and Gln were detected. Without MnCl2, Glu levels increased significantly in our OFS rats relative to CTL. This would point toward increased synthesis of GABA through Glu and Gln precursors while in OFS (+Mn2+) rats, no significant difference was detected relative to CTL, suggesting a decreased degradation of GABA (Violante et al, 2009). However, as pointed out earlier, increasing levels of GABA did not appear to be condition specific including in the presence of MnCl2 (Figure 4). On the contrary, Mn2+ clearly influenced Tau, NAA, Cr, and PCr as well Glc levels relative to the CTL group of rats. Moreover, Gln and Glu levels appeared more specifically affected by Mn2+ for DIA (+Mn2+) and OFS (+Mn2+) rats. Zwingmann et al (2004, 2007) showed that Mn2+ disturbs glucose metabolism, which is supported by the results of the present study. Additionally, increases in glutamine levels following exposure of cells to Mn2+ were attributed to an impaired efflux of Gln from astrocytes or an impaired uptake of Gln from neurons (Zwingmann et al, 2003). These results were consistent with a increased glutamine synthetase activity in Mn2+-intoxicated rats (Zheng et al, 1999) and match as well with findings that simultaneous increases in Gln levels with decreases in Tau levels are consistent with alterations in astrocyte morphology (Oliet and Bonfardin, 2010). In DIA (+Mn2+), Gln increases were further exacerbated compared with all the other groups of rats and relative to CTL, but we also noted that a significant difference between Gln levels in DIA and OFS rats remains with and without Mn2+. In future studies, the role of glutamine in hypothalamic astrocytes and neurons of anorexic rats should be further investigated.
Significantly lower Tau levels were noticed for both OFS (+Mn2+) and DIA (+Mn2+) rats relative to non-MnCl2-infused rats. This change was not noticed in CTL rats. Relative to CTL rats, Tau levels did not change significantly for the OFS (+Mn2+) and DIA (+Mn2+) conditions, whereas higher Tau levels were measured in OFS and DIA rats without MnCl2.
Taurine is an amino acid that has neuroprotective properties and is involved as an osmoregulator (Song and Hatton, 2003). Taurine uptake was increased in astrocyte cultures exposed to a hyperosmotic condition (Aschner et al, 2001), while Tau variations have already been observed in short-term food-deprived animals (Benuck et al, 1995). As in the present study, Tau concentrations in the globus pallidus of rats measured in vivo by NMR spectroscopy (Zwingmann et al, 2007) after MnCl2 infusion demonstrated significant decreases compared with control rats. Taurine is also an important neurotransmitter known to modulate GABA neurochemistry (Namima et al, 1982, 1983) and share GABA transporters in the brain. Here, due to Mn2+ exposure taurine responded inversely to GABA. Previous data suggested that Mn2+ increases GABA by inhibition of GABA transporter function. In turn, the osmoregulatory and neuromodulatory functions of taurine are altered and perturb neural function (Fordahl et al, 2010).
Myo-Inositol concentrations were also increased with MnCl2 in the globus pallidus compared with controls without MnCl2 whereas NAA levels were decreased (Zwingmann et al, 2007). Moreover, these changes appear to be regional specific (Zwingmann et al, 2007).
Conclusion
In the present work, the metabolism of the hypothalamus in rats was characterized both under normal conditions and following changes in brain energy homeostasis using a model of DIA and overnight fasting in rats. The data presented in this study show that it is necessary to perform region-specific 1H-MRS for an accurate characterization of the neurochemical profile of the hypothalamus. In addition, compared with previous studies (Lei et al, 2010), it shows that an improved characterization is performed when challenges such as dehydration, fasting, and Mn2+ infusion are applied: the data of the present study provide evidence that GABA has an essential role in the maintenance of energy homeostasis in the hypothalamus. However, results also reveal that GABA is a nonspecific marker of Mn2+ intoxication in the hypothalamus. On the contrary, glutamine, glutamate, and taurine appear to respond more accurately to Mn2+ exposure. An increase in glutamine levels could be a characteristic response of the hypothalamus to DIA.
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
