Abstract
Supercompensated brain glycogen may contribute to the development of hypoglycemia unawareness in patients with type 1 diabetes by providing energy for the brain during periods of hypoglycemia. Our goal was to determine if brain glycogen content is elevated in patients with type 1 diabetes and hypoglycemia unawareness. We used
Introduction
Hypoglycemia is the factor that prevents most patients with diabetes from achieving the glycemic targets known to reduce the microvascular complications of the disease (The Diabetes Control and Complications Trial Research Group, 1993; UK Prospective Diabetes Study Group, 1998). Hypoglycemia is common and when recurrent can lead to hypoglycemia unawareness; the serious clinical syndrome where a falling blood sugar is not detected until the development of neuroglycopenia (Cryer, 2004; Dagogo-Jack et al, 1993).
The mechanisms responsible for hypoglycemia unawareness remain uncertain. One possible mechanism is that recurrent hypoglycemia leads to upregulated fuel availability to the brain, either in the form of glucose (free or in glycogen reserves) or in the form of alternative fuels. Prior work suggested glucose uptake may increase (Boyle et al, 1994; Criego et al, 2005; McCall et al, 1986) or alternative fuels such as lactate may support a greater fraction of oxidative metabolism (Jiang et al, 2009; Mason et al, 2006) after recurrent hypoglycemia, both of which may serve to enhance fuel availability during subsequent periods of hypoglycemia. Another potential mechanism to enhance fuel availability during hypoglycemia is to increase brain glycogen content via a phenomenon termed ‘supercompensation’. In the adult brain, glycogen is primarily localized in astrocytes (Wiesinger et al, 1997) and may support brain function either by being used locally in astrocytes, which would spare extracellular glucose for neurons, or by being exported to neurons in the form of lactate (Brown et al, 2005; Dienel et al, 2007; DiNuzzo et al, 2010; Sickmann et al, 2009; Swanson et al, 1992; Wender et al, 2000). Glycogen is mobilized during hypoglycemia both in rodents (Canada et al, 2011; Herzog et al, 2008; Suh et al, 2007) and in humans (Öz et al, 2009). Furthermore, we have found that healthy humans and rats have increased rates of glycogen synthesis after an episode of hypoglycemia, suggesting that such an event may occur in patients with diabetes after hypoglycemia (Choi et al, 2003; Öz et al, 2009). Similarly, Alquier et al (2007) found a twofold increase in hypothalamic glycogen content in rodents exposed to recurrent 2-deoxy-
In the current study, we sought to gain insight into the contribution of glycogen supercompensation to hypoglycemia unawareness by using 13C nuclear magnetic resonance (NMR) spectroscopy to assess brain glycogen content and metabolism in subjects with type 1 diabetes and hypoglycemia unawareness. As a first step of testing the hypothesis that an increase in brain glycogen content after hypoglycemia contributes to cerebral energy demands during subsequent periods of hypoglycemia, we compared brain glycogen levels of subjects with type 1 diabetes and hypoglycemia unawareness as assessed by a standardized questionnaire to those of age-, gender-, and body mass index (BMI)-matched controls.
Materials and methods
Subjects
Five subjects with type 1 diabetes mellitus and hypoglycemia unawareness (one female, four males, age 57±4 years, BMI 25±4 kg/m2, mean±standard deviation) and five gender-, age-, and BMI-matched healthy volunteers (one female, four males, age 57±3 years, BMI 24±2 kg/m2) were studied after giving informed consent using procedures approved by the Institutional Review Board: Human Subjects Committee. Age was matched within 4 years and BMI was matched within 4 kg/m2 between subject pairs. Patients with hypoglycemia unawareness had at least two episodes of hypoglycemia per week for the preceding 2 weeks, had a hemoglobin A1c <7.5%, and had their hypoglycemia unawareness verified by a standardized questionnaire (Clarke et al, 1995).
Experimental Design
The night before the experiment, subjects with type 1 diabetes were admitted to the General Clinical Research Center. To eliminate the impact of subcutaneously injected insulin to the insulinemia experimentally controlled during the study the next day, the at home insulin regimen was discontinued upon admission. To manage their diabetes, an intravenous infusion of insulin was administered to maintain their blood glucose between 8.3 and 13.9 mmol/L overnight. This infusion was stopped at the beginning of the study on the next day. Healthy matched controls presented to the General Clinical Research Center in the fasting state on the morning of the study. An intravenous catheter placed antegrade in a forearm was used for infusion and a catheter placed retrograde in the other arm was used for blood sampling. At 1000 h, the protocol created for this study (Figure 1) was begun. Insulin was started at a rate of 0.5 mU/kg/min and a bolus injection of 20 g of [1-13C]glucose (Cambridge Isotope Laboratories, Andover, MA, USA, prepared as 20% weight/volume
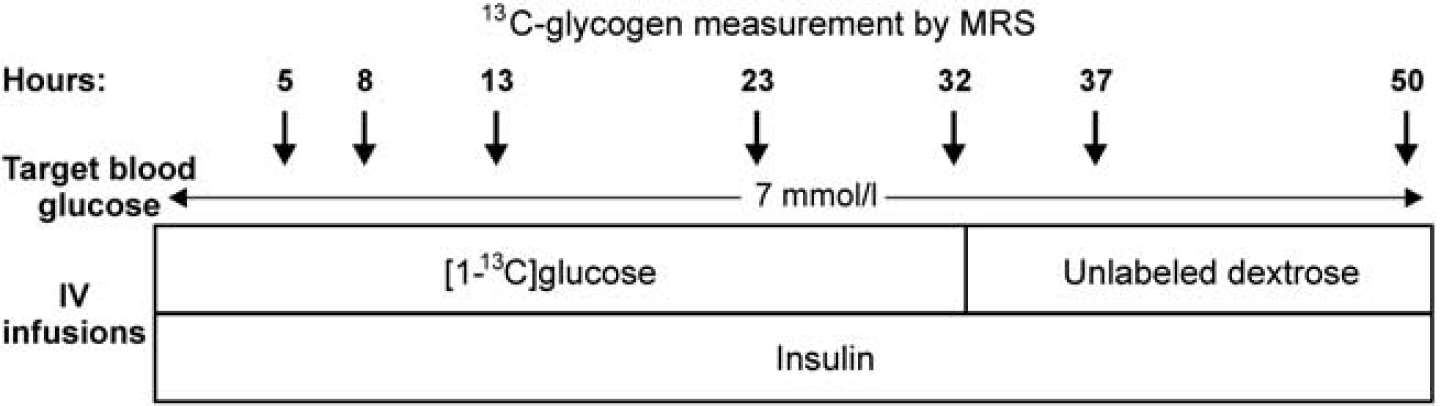
Experimental protocol. Timeline of [1-13C]glucose, unlabeled dextrose, and insulin infusions and the approximate times of MR scans are shown.
Two control subjects were studied twice with different blood glucose target levels several months apart.
Nuclear Magnetic Resonance Spectroscopy
13C-glycogen levels in the brain were measured using methods described before (Öz et al, 2009). Briefly, measurements were performed on a 4-T magnet (Oxford Magnet Technology Inc., Witney, UK) interfaced to a Varian UNITY
Modeling Glycogen Turnover
A model of glycogen metabolism (Öz et al, 2007) was fitted to the time courses of 13C-glycogen using the software SAAM II (The SAAM Institute, Seattle, WA, USA). The plasma glucose IE time courses of each subject were used as the input function. Glycogen synthase (
Comparison of glucose, insulin, glucose isotopic enrichment, and glycogen measurements between subject groups (mean±standard deviation between subjects,
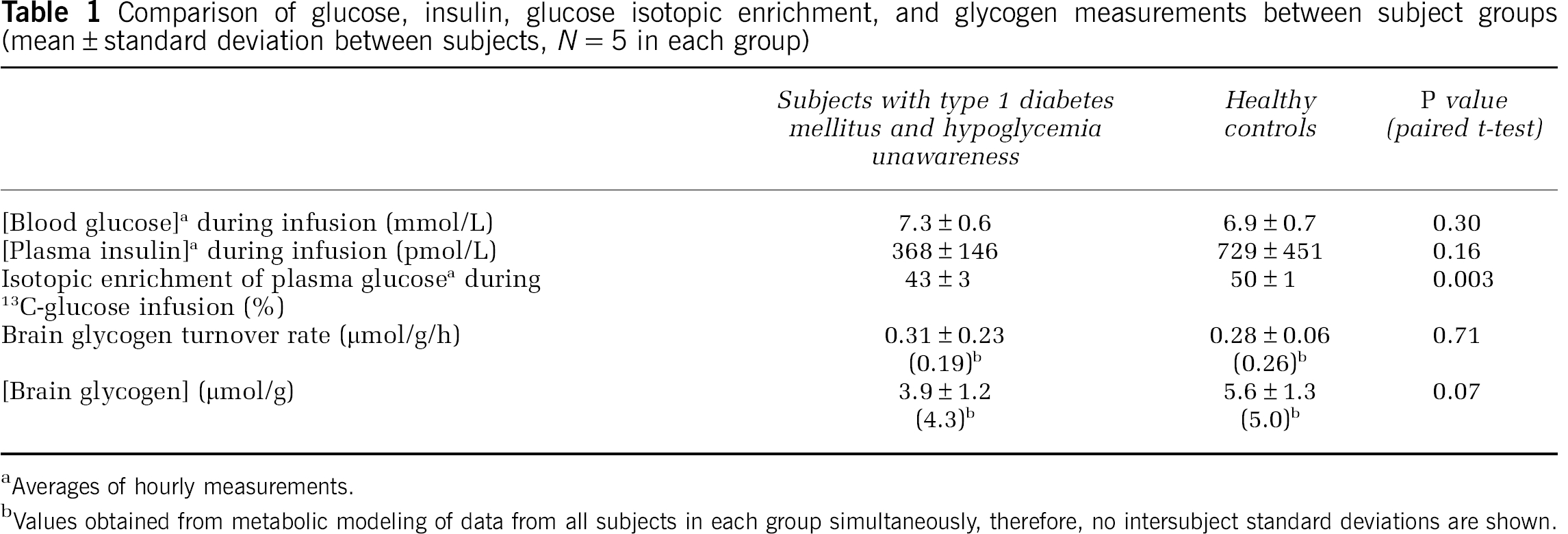
Averages of hourly measurements.
Values obtained from metabolic modeling of data from all subjects in each group simultaneously, therefore, no intersubject standard deviations are shown.
Statistical Analysis
Summary statistics and paired
Results
Effect of Blood Glucose Concentrations on Glycogen Labeling by [1-13C]glucose
To compare glycogen metabolism in subjects with type 1 diabetes and hypoglycemia unawareness to that in matched healthy controls, we infused [1-13C]glucose intravenously to both subject groups over ∼32 hours and monitored 13C label incorporation into and wash-out from glycogen by 13C NMR in the occipital lobe (Figure 1). While the target blood glucose level in these studies (7 mmol/L) was easily reached and exceeded on average in the patient group (Table 1), it was not reached in a subset of the controls. Therefore, the study was repeated in two control subjects both to determine the effect of blood glucose concentration on glycogen labeling kinetics and to better match average blood glucose levels of patients and controls. The newly synthesized glycogen levels were higher at higher average blood glucose concentrations in both of these volunteers (Figure 2), demonstrating the need to match blood glucose levels well between patients and their controls. Because the repeat studies of these subjects matched the average blood glucose levels of their matched patients better, the data from these repeat studies (Study 2 in Figures 2A and 2B) were used for the patient—control comparisons from here on. Note that the experimental conditions in these repeat studies were identical to the patient studies except for the glucose infusion rate, which was higher in all control studies compared with patient studies because patients required less infusion of labeled glucose to maintain target blood glucose. Therefore, no bias was introduced by using the repeat studies of these two control volunteers, who agreed and were able to participate twice in the study, in the comparisons with patient data.
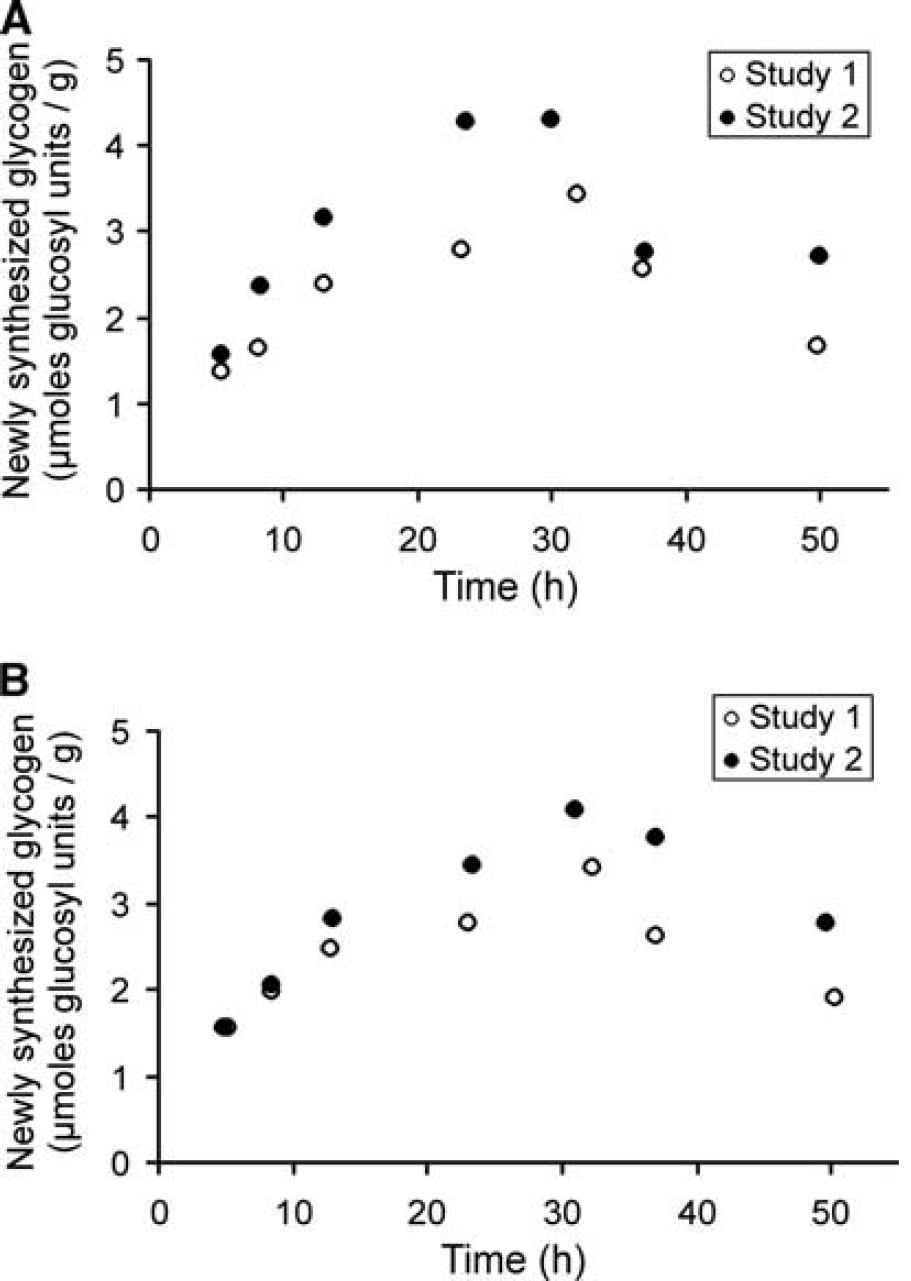
Newly synthesized glycogen levels in two healthy volunteers (shown in
Blood Glucose, Insulin, Isotopic Enrichment, and Brain Glycogen in Patients with Type 1 Diabetes Versus Controls
The average blood glucose levels matched well between patients and their controls (Table 1; Figure 3A). The plasma insulin levels tended to be higher in controls (Figure 3B), but not significantly on average (Table 1). The 13C IE of plasma glucose was higher in controls (Figure 3C; Table 1), and therefore it was critical to correct for this enrichment when determining newly synthesized glycogen levels (see Materials and methods). Newly synthesized cerebral glycogen levels were higher in controls than in patients on average across all time points (
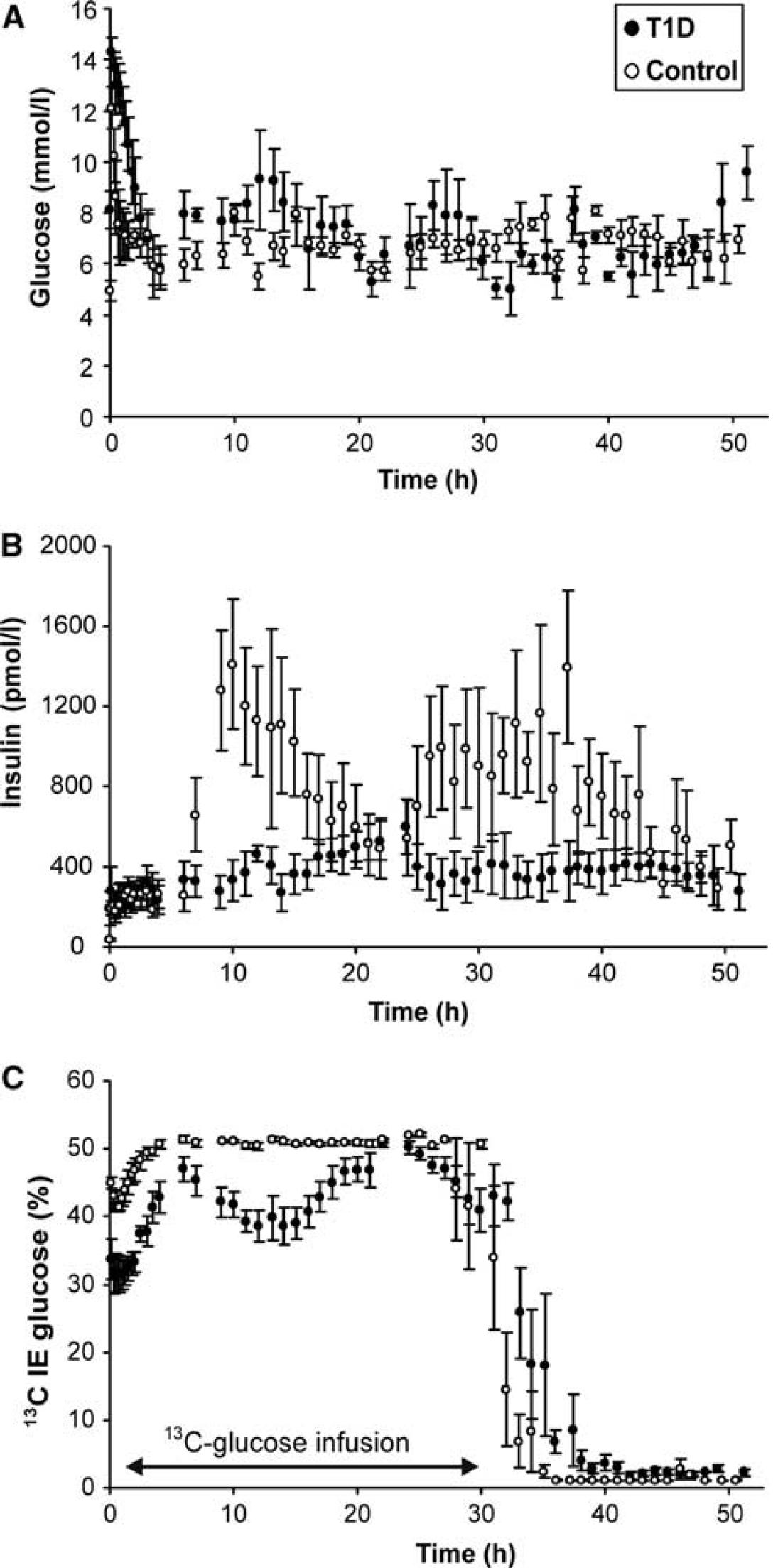
Time courses of average (±standard error of the mean) blood glucose (
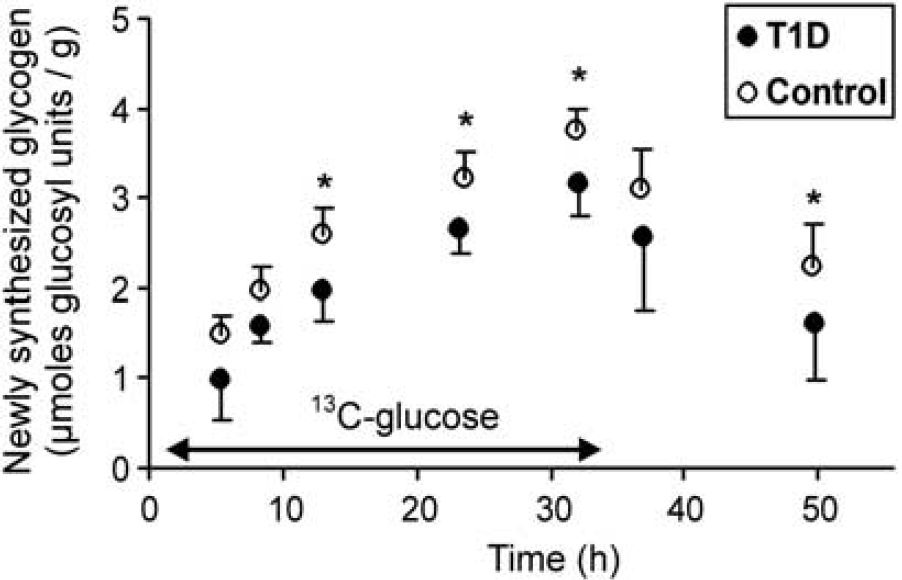
Newly synthesized glycogen concentrations (average±standard deviation) in patients with type 1 diabetes (T1D) and age-, gender-, and body mass index (BMI)-matched controls during the 13C-glucose+insulin infusion (shown with arrow) followed by 12C-glucose+insulin infusion. Newly synthesized glycogen levels were significantly higher in controls than in patients on average across all time points (
To distinguish these two possibilities, estimates for brain glycogen content and turnover rates were obtained for each subject by fitting a model of glycogen metabolism to their 13C-glycogen time courses using their individual IE time courses. This analysis indicated similar glycogen turnover rates in the two groups, but higher glycogen levels in controls than in patients (Table 1). While higher glycogen content in controls was also found when fitting the model to the data from all five subjects in each group simultaneously, this analysis also indicated a higher glycogen turnover rate in controls than in patients (Table 1).
Discussion
Here, we investigated cerebral glycogen metabolism in patients with type 1 diabetes and hypoglycemia unawareness for the first time. By labeling glycogen with 13C-glucose, we observed lower concentrations of newly synthesized brain glycogen in patients than age-, gender-, and BMI-matched controls, which was likely due to lower brain glycogen content based on metabolic modeling. In addition, we demonstrated that higher blood glucose levels lead to higher newly synthesized glycogen in the brain, confirming direct control of brain glycogen content by glucose concentrations.
There are methodological considerations to be made in analyzing our findings. Regarding patient selection, we have not performed a hypoglycemic clamp to confirm blunted counterregulatory response that is a critical component of the hypoglycemia-associated autonomic failure (Cryer, 2004). However, the presence of blunted counterregulation has been repeatedly demonstrated in subjects with type 1 diabetes and hypoglycemia unawareness (Criego et al, 2005), as assessed by the standardized questionnaire we have used here; therefore, the patients enrolled in our study can be assumed to have hypoglycemia-associated autonomic failure. Our studies use 13C NMR, which is the only
There are also metabolic factors to consider in assessing our findings. Glucose and insulin levels can affect glycogen synthesis and labeling (Choi et al, 2003; Morgenthaler et al, 2006). High blood glucose levels can induce glycogen synthesis, as shown by the repeat studies we performed in two control subjects. Therefore, matching blood glucose levels was critical for a reliable comparison of glycogen labeling kinetics between the groups. Although blood glucose levels were comparable between groups when using the repeat studies in controls, plasma insulin levels tended to be higher in controls (Figure 3B). The higher newly synthesized glycogen levels in controls could therefore be attributed to relatively higher insulin levels, since insulin has been shown to lead to glycogen deposition in rat brain (Choi et al, 2003; Morgenthaler et al, 2006). This difference in insulin levels may not explain the group difference in newly synthesized glycogen levels at the first measurement point (at 5 hours after the start of the experiments; Figure 4), because insulin levels started to differ between groups only after ∼7 hours after the start of the experiments (Figure 3B). Of note, the 7-h time point occurred after the first meal subjects were given following the first scan. Thus, the higher insulin levels after this point were likely due to the expected postprandial insulin response in controls. It remains possible that the diabetic subjects displayed some insulin resistance by virtue of being chronically hyperglycemic, which may have contributed to the early difference in newly synthesized glycogen levels in the absence of different serum insulin levels.
While higher insulin levels/sensitivity in controls may explain the higher glycogen levels in controls, our study clearly showed that the diabetic subjects did not have higher glycogen levels than controls under the controlled blood glucose conditions of our experiment, implying that glycogen supercompensation does not contribute to the development of hypoglycemia unawareness in humans with type 1 diabetes. In reaching this conclusion, our sample size was small due to the difficulties involved in completing these studies, namely the long duration of each experiment during which stable plasma glucose levels and enrichment need to be maintained, the sophisticated NMR methodology and the expense associated with large amounts of 13C-glucose infused (Tesfaye et al, 2011). However, the good intersubject and intrasubject reproducibility of our methodology (Öz et al, 2007) has allowed us to detect statistically significant group differences with similar subject numbers in prior investigations by virtue of measuring time courses in individual subjects (Öz et al, 2009). Our conclusion regarding the involvement of brain glycogen supercompensation in hypoglycemia unawareness is in agreement with recent work in animal models exposed to recurrent hypoglycemia. Using a rat model, Herzog et al (2008) found that brain glycogen concentrations were not increased above baseline 6 and 24 hours after acute or recurrent hypoglycemia, despite finding blunted counterregulatory response in rats exposed to recurrent hypoglycemia. In mice, Canada et al (2011), observed brain glycogen supercompensation 6 hours after acute and recurrent hypoglycemia, but glycogen content had returned to normal by 27 hours, at a time when others had observed impaired counterregulation in mice subjected to recurrent hypoglycemia (Jacobson et al, 2006). Taken together, these findings suggest that any effect recurrent hypoglycemia might have on the subsequent detection of and response to acute hypoglycemia is not mediated by glycogen supercompensation. However, it remains possible that subjects with type 1 diabetes and poor blood glucose control have even lower brain glycogen levels, and also that hypoglycemia in the type 1 diabetic population may increase brain glycogen relative to their own baseline. Thus, it is possible that diabetes itself alters brain glycogen content/metabolism in such a way as to confound the response to recurrent hypoglycemia. Consistently, 13C labeling of brain metabolites from [1-13C]glucose was reduced in rodent models of both type 1 (Garcia-Espinosa et al, 2003) and type 2 diabetes (Sickmann et al, 2010). Furthermore, a trend for lower cortical glycogen levels was observed in Zucker diabetic fatty rats relative to controls despite elevated blood glucose levels (Sickmann et al, 2010). Future studies in which hypoglycemia-associated autonomic failure is induced in healthy volunteers will be needed to further investigate the impact of recurrent hypoglycemia on brain glycogen metabolism without the confounding effects of diabetes. In addition, studies of the hypoglycemia response of glycogen in patients with type 1 diabetes, both during and after induced hypoglycemia similar to our prior studies in healthy volunteers (Öz et al, 2009), may provide additional insight and more clinically relevant physiological conditions. Also note that the current results were obtained in a localized brain region in the occipital lobe and we cannot exclude the possibility that glycogen supercompensation in the hypothalamus, similar to what was seen with recurrent neuroglucopenia in rats (Alquier et al, 2007), may contribute to the development of hypoglycemia-associated autonomic failure. Unfortunately, the current
The current findings regarding glycogen content/metabolism in type 1 diabetes also need to be considered within the context of fuel transport into the brain. Some studies suggested that recurrent hypoglycemia leads to an upregulation of glucose transport (Boyle et al, 1994; Criego et al, 2005; McCall et al, 1986), while others did not (Segel et al, 2001). Other studies suggested upregulation of alternative fuel transport/utilization after recurrent hypoglycemia (Jiang et al, 2009; Mason et al, 2006). If a fuel deficiency present during hypoglycemia is compensated for by increased uptake of glucose or an alternative fuel like lactate, brain glycogen may not be mobilized during hypoglycemia. If glycogen mobilization is the signal responsible for supercompensation during hypoglycemia recovery, this may explain why diabetic subjects with hypoglycemia unawareness do not have higher brain glycogen content than controls. Future experiments will be necessary to address this hypothesis. Other reasons for lower brain glycogen content in patients than in controls are unclear at this stage. This observation cannot be due to overall structural tissue damage in patients because all 13C-glycogen concentrations were corrected for the cerebrospinal fluid content of the occipital volume-of-interest. Namely, 11% to 17% of the NMR voxel consisted of cerebrospinal fluid in all subjects, with no difference between the subject groups (data not shown).
In conclusion, our studies have revealed that—under conditions of matched average blood glucose and insulin levels—patients with type 1 diabetes and hypoglycemia unawareness do not have higher brain glycogen levels than healthy controls. We infer from these findings that brain glycogen levels are not supercompensated in patients with type 1 diabetes and hypoglycemia unawareness, and therefore that supercompensation of brain glycogen levels does not appear to contribute to the development of hypoglycemia unawareness in the setting of type 1 diabetes. However, further studies will be necessary to investigate the effects of recurrent hypoglycemia in humans without the potential confounding effects of diabetes.
Footnotes
Acknowledgements
The authors thank the nurses and medical assistants of the General Clinical Research Center for their enthusiastic support of the glucose infusion studies and the staff of the Center for MR Research for maintaining and supporting the NMR system, in particular Dr Gregor Adriany for invaluable help with the NMR coil.
The authors declare no conflict of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
