Abstract
Although platelet-derived growth factors (PDGFs) and receptors (PDGFRs) are abundantly expressed in the central nervous system, their functions largely remain elusive. We investigated the role of PDGFR-β in tissue responses and functional recovery after photothrombolic middle cerebral artery occlusion (MCAO). In the normal adult mouse brain, PDGFR-β was mainly localized in neurons and in pericyte/vascular smooth muscle cells (PC/vSMCs). From 3 to 28 days after MCAO, postnatally induced systemic
Keywords
Introduction
Stroke currently ranks first in adult disability worldwide and second in mortality in most regions (Strong et al, 2007). For the prevention and care of the disease, numbers of therapeutic strategies including neuroprotective agents have been developed (Warburton et al, 2011). In addition, owing to the discovery of adult neurogenesis in the mammalian central nervous system (CNS), regeneration-based treatments for CNS injury are expected as promising therapeutic strategies in which interactions among neurons, glial cells, and the vascular system should be elucidated and controlled appropriately (Okano et al, 2007).
Platelet-derived growth factor (PDGF) family members include PDGF-A, PDGF-B, PDGF-C, and PDGF-D, which are assembled as disulfide-linked homodimers or heterodimers. Two types of PDGF receptors, namely PDGFR-α and PDGFR-β, undergo overlapping, but distinctive, signal transduction in various cell types (Tallquist and Kazlauskas, 2004). Both PDGFs and PDGFRs are widely expressed in the CNS (Sasahara et al, 1991) and are upregulated in neurons, reactive astroglial cells, and pericyte/vascular smooth muscle cells (PC/vSMCs) after cerebral stroke in human and animal models (Iihara et al, 1996; Krupinski et al, 1997; Renner et al, 2003).
Both PDGF-B and PDGF-C protect neurons in several different animal models of neuronal injury including ischemia (Iihara et al, 1997; Tang et al, 2010). Using inducible knockout (KO) mutant, we have shown that PDGFR-β expressed in neurons, mediates neuroprotective signals against various insults both
The null mutants of PDGF-B or PDGFR-β cause neonatal lethality owing to severe vascular abnormalities including kidney glomerulus (Lindahl et al, 1997, 1998). In contrast, postnatally induced systemic PDGFR-β KO in mouse does not show apparent adverse effects both on physiologic parameters and on the morphology of the kidney for a long term, but disturbs the adaptive responses of the stressed kidney glomerulus in our previous report (Nakagawa et al, 2011). These findings indicate that the role of PDGFR-β could be different between organogenesis and postnatal life, and raise the possibility that PDGFR-β may also have a role in the repair process after cerebral insult in the adult mouse brain. Accordingly, using two kinds of PDGFR-β KO mice (postnatally induced systemic KO and neuroepithelium-derived cells KO), we examined the role of PDGFR-β signal in CNS tissue responses to focal cerebral ischemia induced by photothrombotic middle cerebral artery occlusion (MCAO).
Materials and methods
All experimental animal procedures were conducted according to ‘the Institutional Animal Care and Use Committee at the University of Toyama’ (University of Toyama, Sugitani, Toyama City, Japan). All of our study protocols were approved by the Ethics Committee of the University of Toyama before the study began, and we were given permission to carry out the study.
Conditional Knockout Mice
Mutant mice, in which exons 4 to 7 of
In another line of PDGFR-β conditional KO mice,
Genomic Polymerace Chain Reaction
The recombinant allele of
Quantitative Real-Time Polymerace Chain Reaction
The efficiency of
Focal Cerebral Ischemia
Mice were subjected to permanent MCAO according to a modified method (Sugimori et al, 2004). Five- to six-month-old male mice were anesthetized with halothane (2% for induction, 1% for maintenance with a face mask; Takeda Pharmaceutical, Osaka, Japan) carried by a mixture of 70% nitrous oxide and 30% oxygen. Rectal temperature was maintained throughout the surgical procedure at 37°C±0.5°C using a homeothermic blanket (Harvard Apparatus, Holliston, MA, USA). The left middle cerebral artery (MCA) was exposed using a stereoscopic surgical microscope (Conan Medical, Hyogo, Japan). A 6-mW krypton laser with 568 nm (Melles Griot, Tokyo, Japan) was used to irradiate the distal MCA for 4 minutes, after intravenous administration of a photosensitizing rose Bengal dye solution (Wako, Osaka, Japan) at a dose of 20 mg/kg body weight over 90 seconds. After that, a secondary 4-minute laser irradiation was administrated to the proximal MCA. The left common carotid artery was then tightly ligated to induce a reproducible neocortical infarct (Brint et al, 1988). The temporalis muscle and skin were subsequently reconstructed; mice were subsequently maintained at a warm temperature and returned to the home cage until resuscitation. Sham controls were treated identically, excluding laser exposure and common carotid artery ligation.
Mouse Behavior
Following a previously described method (Orset et al, 2007), an open-field test was performed before and after MCAO. Each mouse was gently placed at the center of a cubic chamber (48 × 48 × 30 cm3). The mouse could move freely in the chamber for 15 minutes. The spontaneous locomotor activity (by the total distance covered) and exploration behavior (by the number of entries and time spent in the center zone) were assessed using an automated open-field system equipped with Scanet MV-10 computer software (Melquest, Toyama, Japan).
Infarction Volume Measurement
Under deep anesthesia with sodium pentobarbital (intraperitoneal injection, 50 mg/kg body weight; Dainippon Sumitomo Pharma, Osaka, Japan), mice were transcardially perfused with phosphate-buffered saline, followed by perfusion and immersion in 4% paraformaldehyde before being embedded in paraffin. Coronal sections (10-μm thick) were prepared with 200-μm intervals so that the entire ischemic lesion was covered. Hematoxylin—eosin staining was performed for measurement of areas of interest. Morphologic and morphometrical analyses were performed using a microscopy system (BX 50; Olympus, Tokyo, Japan) connected to a digital camera (DP70; Olympus). Measurements of areas of interest were conducted using MetaMorph software (Molecular Devices, Osaka, Japan). Lesion volumes were estimated using an indirect method to avoid effects of tissue swelling or shrinkage: 100% × (contralateral hemisphere volume−noninfarct ipsilateral hemisphere volume)/contralateral hemisphere volume (Swanson et al, 1990).
Immunohistochemistry and Immunofluorescence Staining in Paraffin-Embedded Tissue Sections
Paraffin-embedded, coronal tissue sections (10-μm thick) through the injured cortex were prepared according to the AMex method (Suzuki et al, 2002). In brief, transcardial perfusion with ice-cold phosphate-buffered saline was followed by perfusion and immersion in periodate-lysine-paraformaldehyde fixative containing 4% paraformaldehyde. After dehydrated in acetone at 4°C overnight, tissues were cleared in methyl benzoate for 1 hour and in xylene for 1 hour, respectively. These tissues were then embedded in paraffin. Deparaffinized coronal tissue sections were processed for antigen retrieval using target retrieval solution (pH 9.0; Dako, Cupertino, CA, USA) at 120°C for 4 minutes. Nonspecific immunoreactions were blocked at room temperature for 30 minutes using the Protein-Block kit (Dako). For immunostainings with mouse primary antibodies, sections were treated with Histofine Mouse Stain kit (Nichirei, Tokyo, Japan) for 1 hour before the Protein-Block kit. Sections were incubated at 4°C overnight with the following primary antibodies: goat polyclonal anti-PDGFR-β (1:100; R&D Systems, Minneapolis, MN, USA), mouse monoclonal anti-alfa smooth muscle actin (α-SMA; 1:100; Dako), rabbit polyclonal anti-laminin (1:1,000; Dako), and rabbit polyclonal anti-glial fibrillary acidic protein (GFAP; 1:1,000; Dako). Positive reactions were detected using the appropriate Histofine Simple Stain Mouse System (Nichirei) and were visualized following 3,3′-diaminobenzidine tetrahydrochloride (Dako) reaction. Nuclear counter stainings were conducted with hematoxylin. Images were captured using a light microscope (Olympus) and processed using Photoshop software (version 7.0, Adobe, San Jose, CA, USA).
To test the efficiency of
To specifically identify PC/vSMC coverage and proliferation, double-immunofluorescence staining for α-SMA/laminin and Ki67 (a cellular proliferation marker)/α-SMA were used. Deparaffinized sections were processed for antigen retrieval, blocking, and incubated at 4°C overnight with the following primary antibodies: mouse monoclonal anti-α-SMA (1:100; Dako), rabbit polyclonal anti-laminin (1:1,000; Dako), and rat polyclonal anti-Ki67 (1:100; Dako). Stainings were visualized by incubating with donkey anti-mouse IgG conjugated with Alexa Fluor 488/594 (1:500; Molecular Probes), donkey anti-rabbit IgG conjugated with Alexa Fluor 594 (1:500; Molecular Probes), and donkey anti-rat IgG conjugated with Alexa Fluor 488 (1:500; Molecular Probes). All sections were mounted with the Vectashield mounting medium with 4′,6-diamidino-2-phenylindole (Vector Laboratories, Burlingame, CA, USA) and imaged using a confocal microscope (TCS-SP5, Leica, Heidelberg, Germany). Montages were created using Photoshop software.
Western Blot
For western blot analysis, 2-mm thick, coronal tissue sections were harvested from the middle of the ischemic lesions. The cortical ischemic lesion was then dissected along the outer margin of the lesion and designated as the ipsilateral cortex. The cerebral cortex of the similar anatomic site was obtained from another side of the tissue section and designated as the contralateral cortex. All brain tissues were homogenized in RIPA buffer (50 mmol/L Tris-HCl, 150 mmol/L NaCl, 0.5% sodium deoxycholate, 1% NP-40, 0.1% SDS, 2 mmol/L Na3VO4, 500 mmol/L NaF, 58 μg/mL aprotinin, 10 μg/mL leupeptin, and 2 mmol/L PMSF) using the Multi-Beads Shocker system (Yasui-Kikai, Osaka, Japan) at 4°C. Protein (15 μg) samples were separated by SDS-PAGE using a gradient gel (5% to 20%; ATTO, Tokyo, Japan), followed by electroblotting onto polyvinylidene difluoride membranes (ATTO) and incubation in a blocking buffer of 5% nonfat milk at room temperature for 1 hour. Membranes were incubated with the following primary antibodies: rabbit polyclonal anti-PDGFR-β (1:500, Upstate Biotechnology, Charlottesville, VA, USA), rabbit polyclonal anti-PDGFR-α (1:1,000, Santa Cruz Biotechnology, Santa Cruz, CA, USA), rabbit polyclonal anti-phospho-Tyr857 PDGFR-β (1:1,000, Santa Cruz Biotechnology), rabbit polyclonal anti-phospho-Tyr742 PDGFR-α (1:1,000, Novus Biologicals, Littleton, CO, USA), rabbit polyclonal anti-α-SMA (1:250, Abcam, Cambridge, MA, USA), rabbit polyclonal anti-laminin (1:250, Santa Cruz Biotechnology), goat polyclonal anti-GFAP (1:250, Santa Cruz Biotechnology), and mouse monoclonal anti-glyceraldehyde-3-phosphate dehydrogenase (1:5,000, Chemicon, Temecula, CA, USA) at 4°C overnight. Immunoreactive bands were visualized by incubating with appropriate horseradish peroxidase-linked secondary antibodies, followed by enhanced chemiluminescence detection using enhanced chemiluminescence plus western blotting detection reagents (Amersham Biosciences, Little Chalfont, UK). The immunoreactive bands of targeted proteins were quantified using the VH Analyzer software (VH-H1A5, Keyence, Osaka, Japan), normalized with the GAPDH protein band, and then relative to sham values.
Fluorescein Isothiocyanate-Labeled Albumin Leakage and Immunofluorescence Staining in Frozen Tissue Sections
To assess BBB permeability after cerebral ischemia, fluorescein isothiocyanate (FITC)-labeled albumin (Sigma-Aldrich; 5 mg/mL in Ringer HEPES buffer: 147 mmol/L NaCl, 4 mmol/L KCl, 3 mmol/L CaCl2, 1.2 mmol/L MgCl2, 15 mmol/L Hepes, 5 mmol/L glucose, 1% bovine serum albumin; pH 7.4) was transcardially infused (Cavaglia et al, 2001). In brief, at 1, 3, 6, and 14 days after MCAO, mice were anesthetized with sodium pentobarbital. Heparin (100 Units/kg in 0.1 mol/L phosphate-buffered saline buffer) was intravenously injected (right jugular vein), followed by transcardial (left ventricle) perfusion with FITC-labeled albumin solution at a rate of 1 mL/min for 5 minutes. To avoid the effects of systemic blood pressure, the same amount of blood was simultaneously withdrawn through the right jugular vein. The brains were rapidly removed, fixed in 4% paraformaldehyde for 3 days, and immersed in 30% sucrose for 2 days. The brains were then embedded in Tissue-Tek O.C.T. compound (Sakura Finetek, Torrance, CA, USA). Five coronal tissue sections (30-μm thick) with 100-μm intervals were collected from the ischemic center of each brain using a cryostat (CM1900, Leica). Fluorescein isothiocyanate-albumin-infused brain sections were further immunostained. The floating sections were permeabilized in 0.3% Triton X-100 solution at room temperature for 1 hour, and then incubated with primary antibodies against α-SMA (1:100, mouse monoclonal antibody, Dako) and PDGFR-β (1:100, goat polyclonal antibody, Neuromics) in a blocking buffer of 10% normal donkey serum at 4°C for 2 days. Secondary antibodies were raised in appropriate hosts and conjugated with Alexa Fluor 594 and Alexa Fluor 633 (1:500, Molecular Probes). To determine glial scar formation, rabbit polyclonal anti-GFAP antibody (1:1,000; Dako) or rabbit polyclonal anti-vimentin antibody (1:100; Abcam) were incubated with floating sections without FITC-labeled albumin perfusion, followed by secondary antibody conjugated with Alexa Fluor 488 (1:500, Molecular Probes) or Alexa Fluor 594 (1:500, Molecular Probes), respectively. All sections were mounted on glass slides with the Vectashield mounting medium with 4′,6-diamidino-2-phenylindole and imaged using a confocal microscope (Leica). Montages were created using the Photoshop software.
Image Analysis and Quantification
Areas of PDGFR-β, α-SMA-positive stainings and areas of laminin-positive blood vessels were determined within randomly selected three squares of 0.16 mm2 per section in the ischemic border and core, respectively, using a Biorevo BZ-9000 microscope (Keyence) and BZ-II Analyzer software (Keyence). Three nonadjacent coronal sections per mouse were randomly selected and examined.
To determine the PC/vSMC coverage of blood vessels, the colocalization rates between α-SMA and laminin signals from microvessels were analyzed using LAS AF software (Leica). In each animal, four randomly selected squares of 0.0441 mm2 per section in the ischemic border of the cerebral cortex were analyzed in three nonadjacent coronal sections.
The percentage of Ki67-positive cells within α-SMA-positive cells was determined within three randomly selected 0.16 mm2 squares per section in the ischemic border of the cerebral cortex. Three nonadjacent coronal sections per mouse were randomly selected and examined.
To quantify FITC-albumin leakage, five frozen coronal tissue sections were prepared from the ischemic center of each mouse, and the lower-magnified entire view of the ischemic lesion was acquired throughout the entire depth of each section, i.e., eight images were acquired at every 3.75-μm depth using a confocal microscope (Leica; Supplementary Figure 1). On each acquired image, the fluorescence mean intensity was automatically read within three randomly selected 0.04 mm2 squares in the ischemic core, 8 to 10 fields covering the entire ischemic border, and 9 randomly selected cortical fields in the contralateral hemisphere using LAS AF software (Leica).
The astrocyte density in the glial scar at 14 days after MCAO was quantified using paraffin-embedded tissue sections stained for GFAP immunohistochemistry. The number of cell nucleus with cytoplasmic GFAP staining was counted in three consecutive 0.09 mm2 squares in the vicinity of the lesion per section, the squares of which are illustrated in Figure 6C, using a light microscope (Olympus) and analysis software (Olympus). In each animal, three nonadjacent sections were examined.
Statistics
Quantitative data were expressed as mean±s.e.m. Comparisons between two experimental groups were made using unpaired Student's
Results
In this study, MCAO was induced in 188 mice (Floxed,
Platelet-Derived Growth Factor Receptor-β is Depleted in Pericyte/Vascular Smooth Muscle Cells and Neurons of Esr-KO Mice
We confirmed the efficiency of
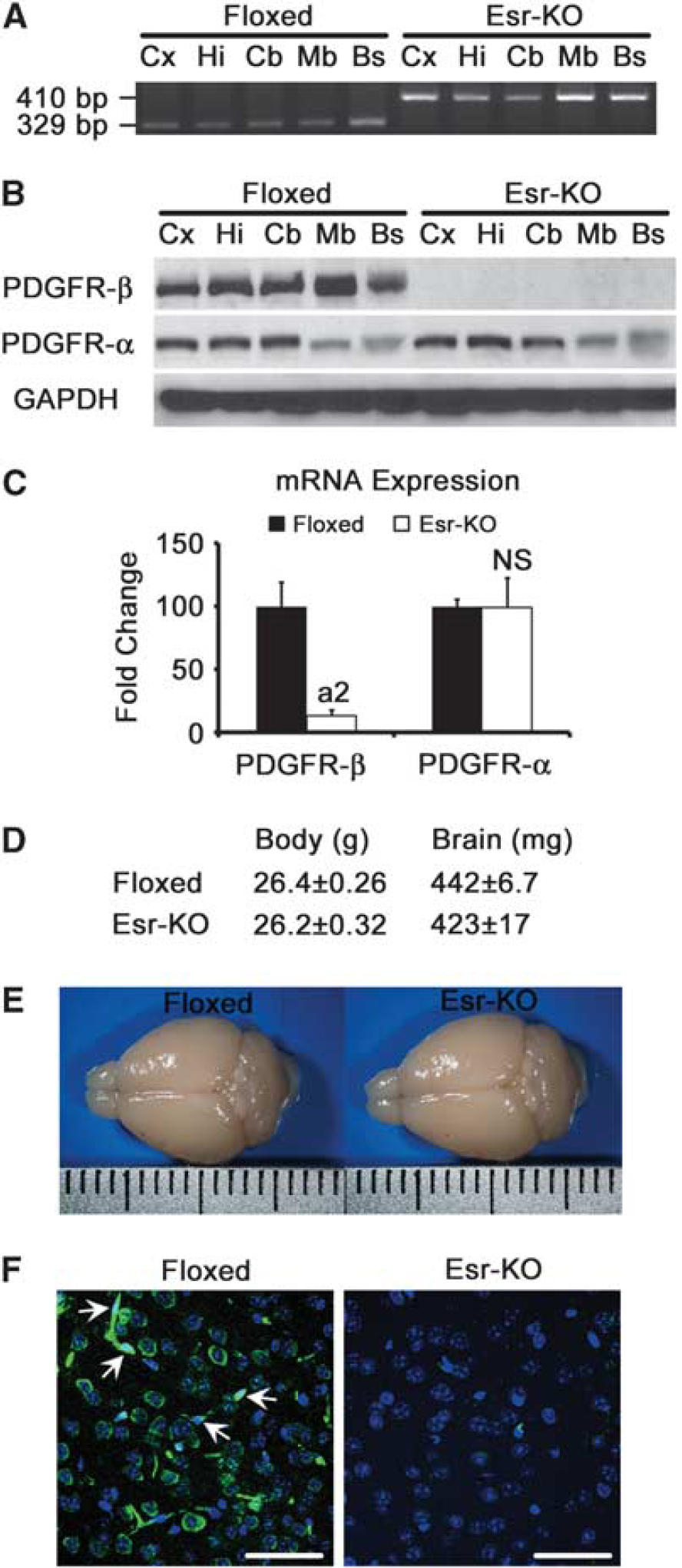
Specific
In western blot, PDGFR-β was consistently detected in all brain tissues obtained from Floxed mice, but was decreased to undetectable levels in those tissues from Esr-KO mice (Figure 1B). Platelet-derived growth factor receptor-α was detected at similar levels between two groups in all tissues examined (Figure 1B).
Real-time PCR analysis showed that PDGFR-β mRNA expression was decreased by ∼85% in the cerebral cortices of adult Esr-KO compared with Floxed mice (Figure 1C,
Body weight, brain weight, and macroscopic appearance of brains from Esr-KO mice were indistinguishable from those of Floxed mice before MCAO (Figures 1D and 1E).
To further characterize PDGFR-β depletion in the cerebral cortex, immunostaining of PDGFR-β was conducted. Immunofluorescence clearly showed that PDGFR-β was expressed in neurons and in PC/vSMCs of Floxed mice as reported previously (Ishii et al, 2006; Virgintino et al, 2007), and mostly disappeared in both cell types of adult Esr-KO mice (Figure 1F). Throughout the experiments, our immunostaining did not show PDGFR-β in astrocytes in the adult mouse brain, although cultured astrocytes isolated from the neonatal mouse brain showed abundant PDGFR-β expression that was phosphorylated after PDGF-BB stimulation (Supplementary Figure 2).
Platelet-Derived Growth Factor Receptor-β Deletion Increases Infarction Volume and Impairs Functional Recovery After Cerebral Ischemia
Middle cerebral artery occlusion induced reproducible and well-demarcated ischemic lesions in the cerebral cortices of Esr-KO and Floxed mice (Figure 2A).
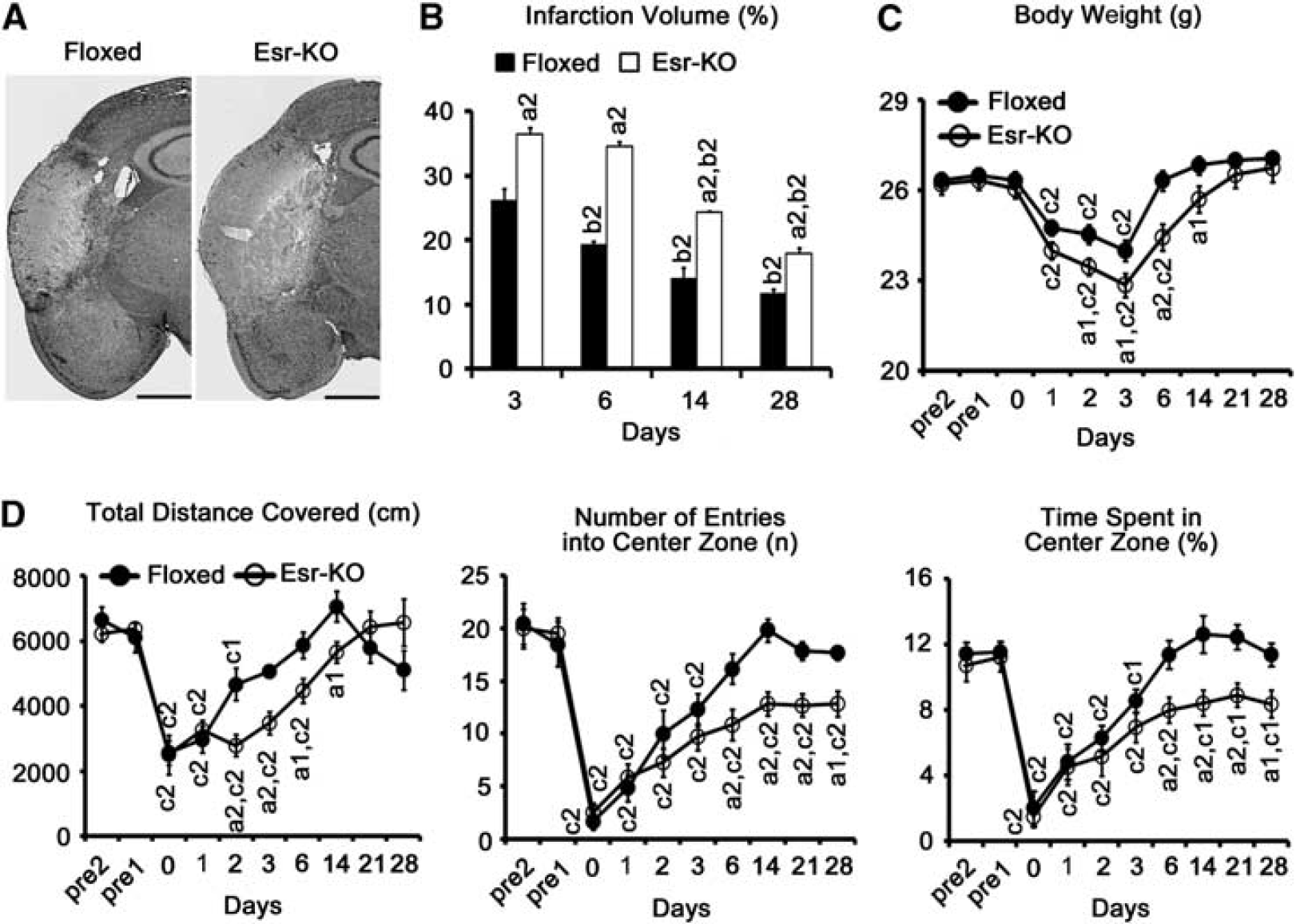
Infarction volume was significantly larger in Esr-KO than in Floxed mice at 3, 6, 14, and 28 days after MCAO (Figure 2B,
The body weight of Esr-KO mice was significantly lost at 1 day and recovered to pre-MCAO level at 14 days after MCAO, which showed obviously delayed recovery compared with Floxed mice that recovered to pre-MCAO level at 6 days (Figure 2C,
To test functional recovery after cerebral ischemia, the open-field test was performed before and after MCAO. Before MCAO, three parameters of the open-field test: the total distance covered (indicating the spontaneous locomotor activity), the number of entries into the center zone, and the time spent in the center zone (indicating the exploration behavior) were at comparable levels in Floxed and Esr-KO mice (Figure 2D). After MCAO, recovery of the total distance covered was significantly delayed in Esr-KO than in Floxed mice (
Platelet-Derived Growth Factor Receptor-β Deletion Decreases Pericyte/Vascular Smooth Muscle Cell Coverage and Proliferation in Lesions After Cerebral Ischemia
According to western blot, PDGFR-β protein levels decreased at 12 hours in Floxed lesions compared with the sham-operated group, subsequently increased from 1 to 6 days, and then sustained high levels until 28 days. In Esr-KO lesions, PDGFR-β levels were almost undetectable (Figure 3A). Levels of α-SMA were distinctly increased in Floxed lesions from 3 to 28 days compared with the sham-operated group, and were higher than those in Esr-KO lesions during this period (Figure 3A). After transient decreases, laminin levels were similarly restored in the 14-day lesions in the two groups (Figure 3A). In the contralateral side, there were no evident differences of α-SMA and laminin levels between the two groups, except apparently decreased PDGFR-β levels in Esr-KO mice.
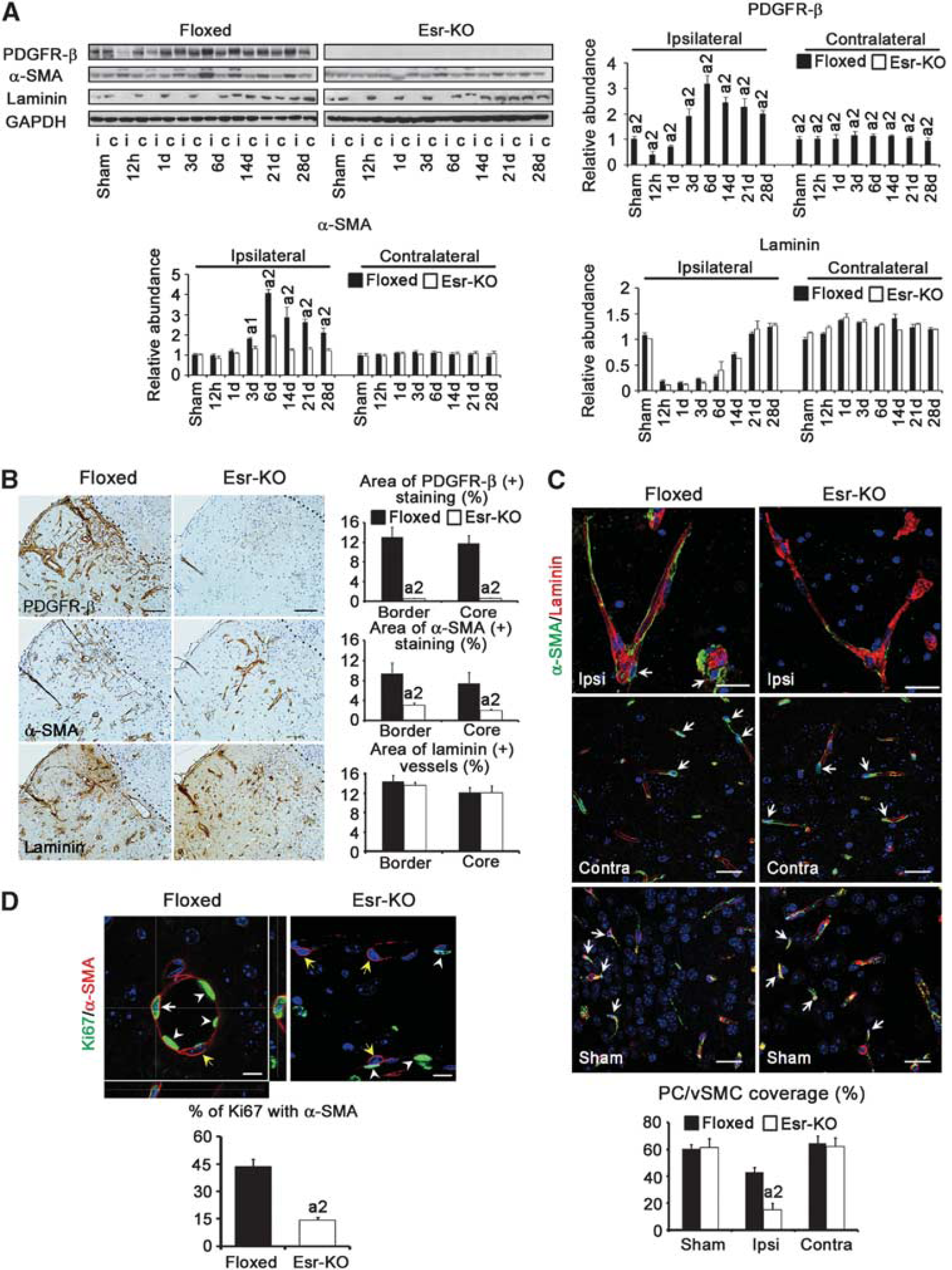
In the immunohistochemistry of 6-day ischemic lesions, many blood vessels were positively stained for PDGFR-β and α-SMA in Floxed mice compared with those in Esr-KO mice, and these increases were further quantitatively approved by morphometrical measurement (Figure 3B). In contrast, the features and morphometry of laminin staining, a marker of basement membrane of vascular endothelial cells, were similar between two groups of mice, indicating that angiogenesis occurred at similar extents between Esr-KO and Floxed mice. This notion was further confirmed by PCR-array study showing that the sets of genes related to angiogenesis were not significantly alternated in the lesions of Esr-KO mice compared with Floxed mice (Supplementary Table). In the contralateral side, α-SMA and laminin were similarly stained in the blood vessels, and these stainings were at comparable levels in morphometry between two groups, although PDGFR-β immunoreactivities were mostly undetectable in the contralateral cerebral cortices of Esr-KO mice (data not shown).
Pericyte/vascular smooth muscle cell coverage in the blood vessels in the ischemic lesion, which was measured by the percentage association between α-SMA and laminin stainings, was significantly lower in Esr-KO (14.9%±4.8%) than in Floxed mice (43.1%±3.4%) at 6 days after MCAO (Figure 3C). Pericyte/vascular smooth muscle cell coverage of Esr-KO was similar to that of Floxed mice in the contralateral cerebral cortices, and in the sham-operated ipsilateral cerebral cortices, respectively (Figure 3C).
The proliferation of PC/vSMCs was measured by the immunofluorescence of Ki67. In 6-day lesions, the percentage of Ki67-positive cells within α-SMA-positive PC/vSMCs was significantly lower in Esr-KO (14.2%±1.7%) than in Floxed mice (43.7%±3.9%) (Figure 3D).
Platelet-Derived Growth Factor Receptor-β Deletion Increases Vascular Permeability Correlating With the Loss of Pericyte/Vascular Smooth Muscle Cell Coverage After Cerebral Ischemia
The leakage of FITC-albumin tracer appeared in the ischemic border at 1 day after MCAO, and extended towards the ischemic core until 6 days in Floxed and Esr-KO mice (Figure 4A). When compared, the leakage was more intense in Esr-KO than in Floxed mice (Figure 4A), and this increase was found to be significant in the ischemic border of 3- and 6-day lesions, and in the ischemic core of the 6-day lesion by quantitative analyses (Figure 4D). In 14-day lesions, leakage was decreased and fluorescent tracer was primarily localized within a fine meshwork of blood vessels in Esr-KO and Floxed mice (Figure 4A). The sham-operated and contralateral cerebral cortices from two groups did not show abnormally increased leakage of FITC-albumin, respectively (Figures 4B and 4D). Leakage was consistently detected in the hypothalamus of each mouse where the BBB is physiologically absent, and this was used as an indicator to proof appropriate measurement of BBB leakage as described previously (Cavaglia et al, 2001).
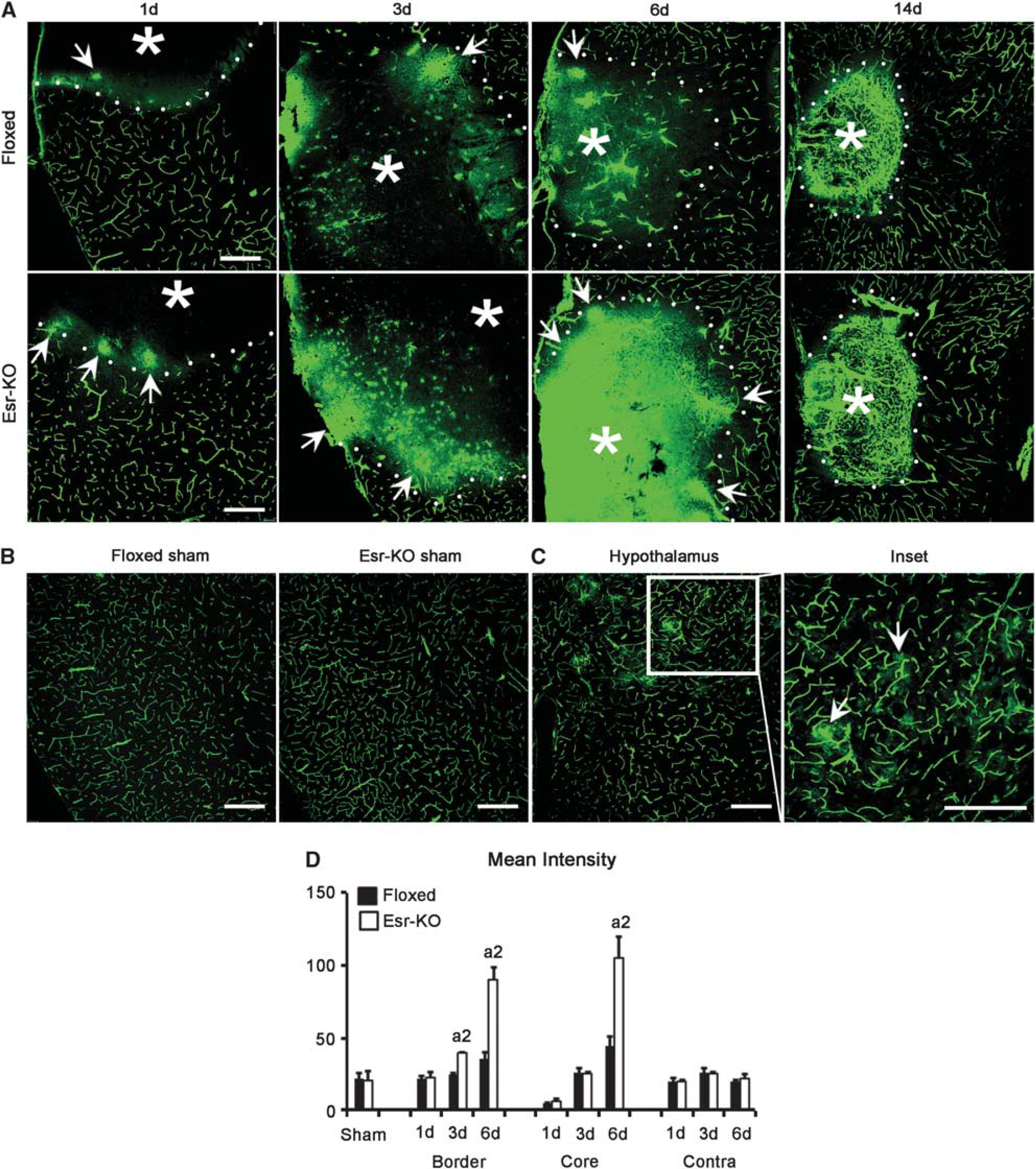
We further tested the hypothesis that PDGFR-β knockdown exacerbates BBB permeability owing to the loss of PC/vSMC coverage after MCAO. In 6-day lesions of Floxed mice, BBB leakage of fluorescent tracer was slight, and FITC-albumin containing blood vessels was consistently associated with PC/vSMCs, as determined by double-immunofluorescence staining of α-SMA and PDGFR-β in frozen-tissue sections from FITC-albumin-infused brains (Figure 5). In contrast, in 6-day lesions of Esr-KO mice, BBB leakage was extensive, and FITC-albumin containing blood vessels were frequently unassociated with α-SMA- and PDGFR-β-positive PC/vSMCs (Figure 5). Thus, the loss of PC/vSMCs and increased BBB leakage were closely localized, and the causal relationship was suggested between the two phenomena.
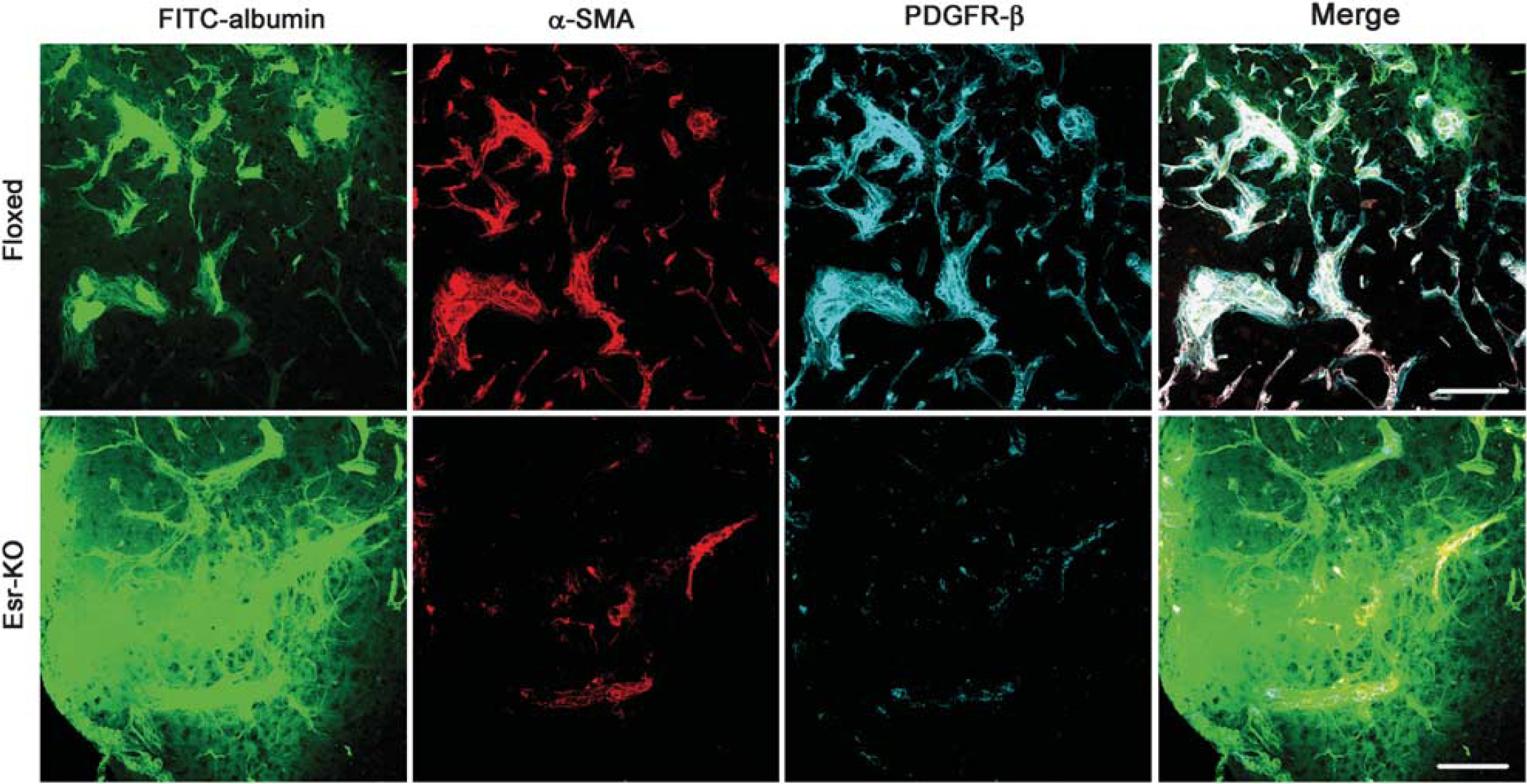
Increased vascular permeability correlates with the loss of PC/vSMCs owing to
Platelet-Derived Growth Factor Receptor-β Deletion Disturbs Glial Response to Ischemic Injury
According to western blot analysis, GFAP levels in the ipsilateral cerebral cortices were increased from 14 days and after in 2 groups and were significantly lower in Esr-KO than in Floxed mice (Figures 6A and 6B). Glial fibrillary acidic protein levels were comparable in the contralateral cortices between the two groups.
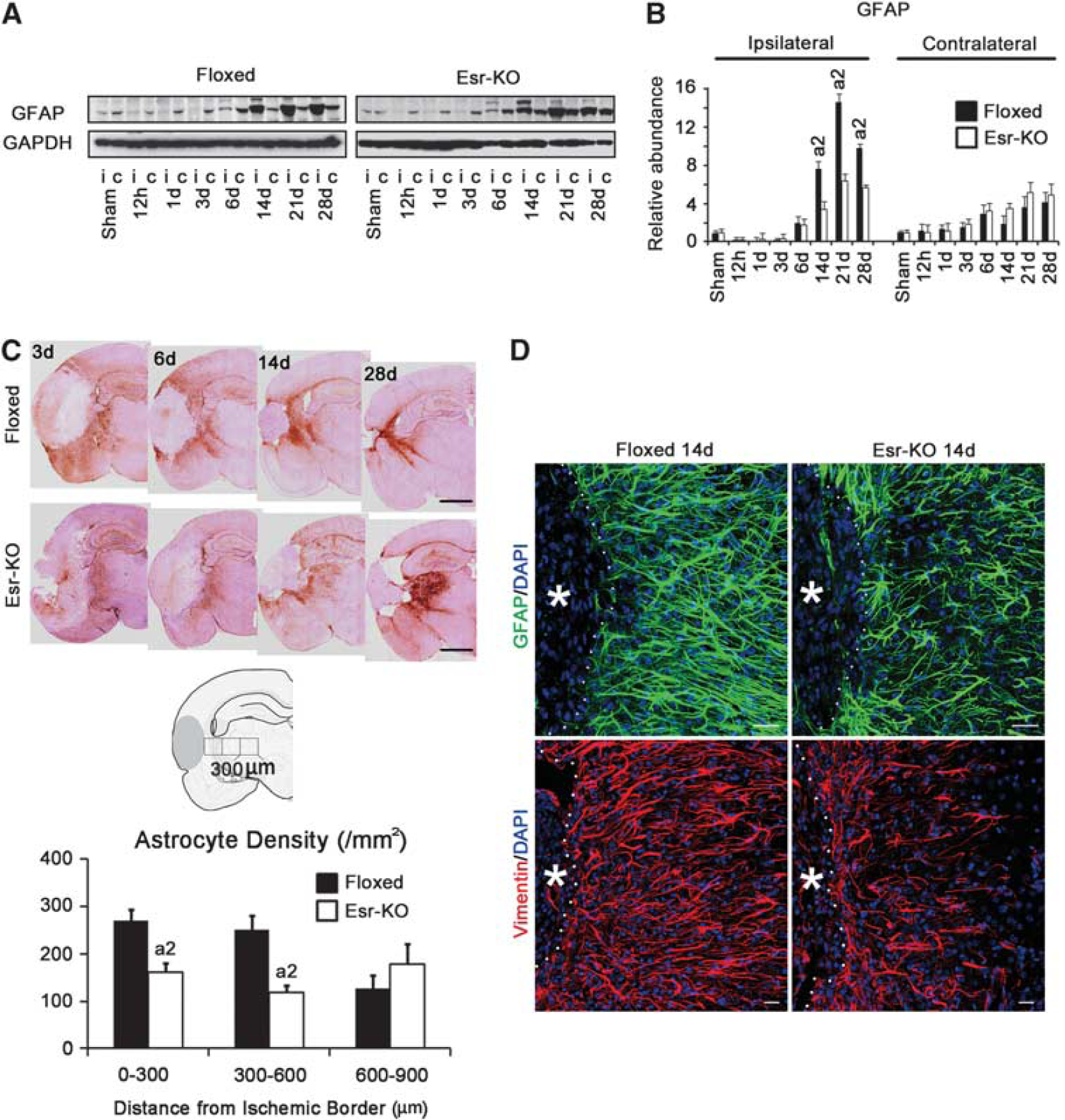
In immunohistochemistry, many GFAP-positive astrocytes accumulated in the ischemic border of Floxed mice at 3, 6, 14, and 28 days after MCAO, but these responses of astrocytes were much less in Esr-KO mice (Figure 6C, upper). Quantitative analysis of astrocyte density in the ischemic border was significantly lower in Esr-KO than in Floxed mice at 14 days after MCAO (Figure 6C, lower). In immunofluorescence staining, the density of GFAP- or vimentin-positive astrocytes in the ischemic border was less in Esr-KO than in Floxed mice at 14 days (Figure 6D).
Platelet-Derived Growth Factor Receptor-β Expression in Pericyte/Vascular Smooth Muscle Cells Increases Vascular Maturation and Tissue Responses After Cerebral Ischemia
Our previous study clearly showed that PDGFR-β was mostly depleted in neurons, but was well preserved in PC/vSMCs in Nestin-KO mice (Ishii et al, 2006), although nestin is reportedly expressed in both cell types (Alliot et al, 1999). Accordingly, we induced MCAO in Nestin-KO and tried to further examine the role of PDGFR-β expressed in different cell types.
As expected, PDGFR-β-positive blood vessels were frequently seen in 6-day ischemic lesions in Nestin-KO and Floxed mice, but were few in Esr-KO mice (Supplementary Figure 3). Blood vessels, as detected by laminin, were distributed to similar extents in all mouse lines.
Infarction volume significantly increased in Nestin-KO mice at 3 and 6 days after MCAO compared with Floxed mice without tamoxifen (Floxed (−) TM, Figure 7A), which might suggest the neuroprotective role of PDGFR-β. However, when compared with Esr-KO mice, infarction volume in Nestin-KO mice was similar at 3 days (Figures 2B and 7A, 36.4%±1.09% in Esr-KO; 33.4%±1.29% in Nestin-KO;
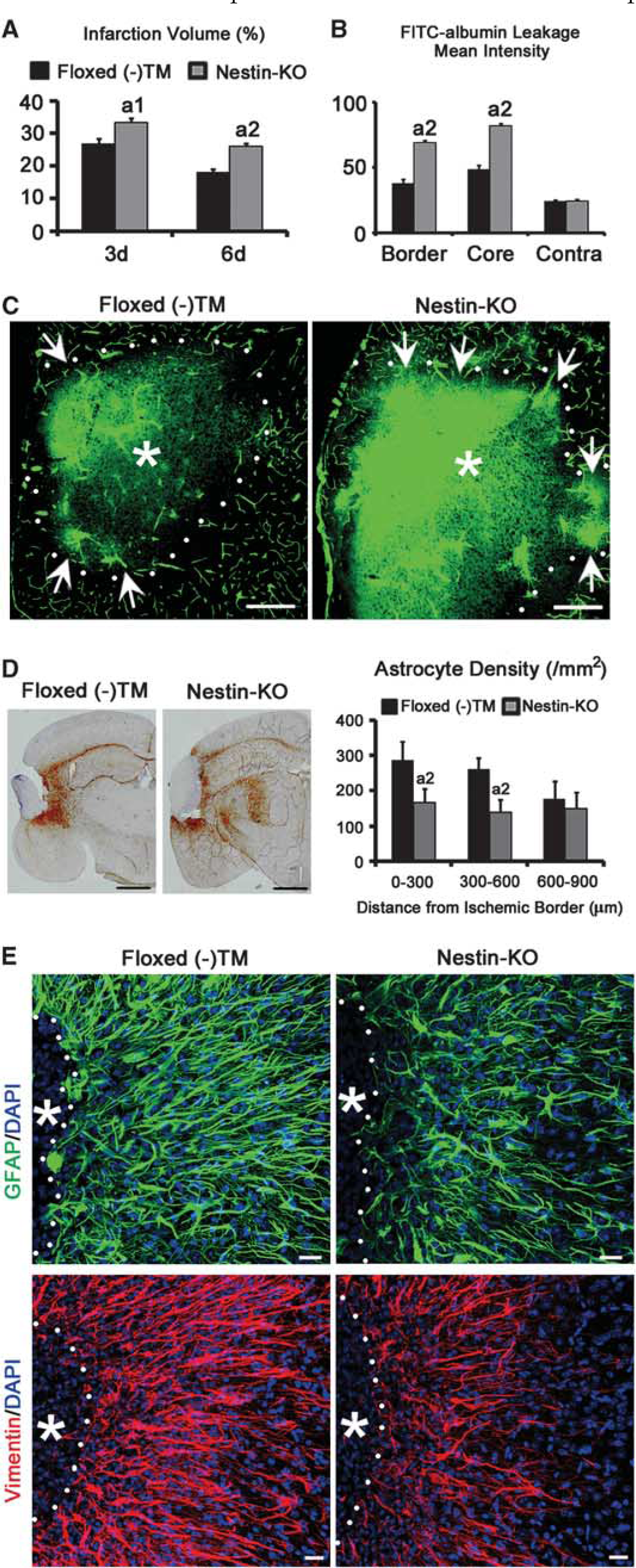
Infarction volume and BBB leakage are worse, and glial scar formation is disrupted in Nestin-KO mice after MCAO. (
In the ischemic border and core of 6-day lesions, FITC-labeled albumin leakage was significantly extensive in Nestin-KO compared with Floxed (−) TM (Figures 7B and 7C, ischemic border: 69.1±1.9 in Nestin-KO, 36.5±4.5 in Floxed (−) TM,
Immunohistochemistry of GFAP showed that astrocyte density in the ischemic border of Nestin-KO (Figure 7D, 0 to 300 μm: 166±41; 300 to 600 μm: 138±36) was significantly lower than Floxed (−) TM (Figure 7D, 0 to 300 μm: 287±51,
Discussion
We induced systemic PDGFR-β deletion, and conducted comprehensive analyses on the postnatal role of PDGFR-β in the acute phase of neuroprotection and tissue remodeling after focal cerebral ischemia. To the best of our knowledge, this study first reported that after MCAO, PDGFR-β conditional KO mice showed increased infarction volume and impaired functional recovery compared with control mice throughout the experiments. In these KO mice, disturbed coordinated responses of the different cell types, including neurons, glial cells, and blood vessel-associated cells were observed.
In non-ischemic Esr-KO mice, PDGFR-β expression was substantially decreased in western blot and real-time PCR analysis compared with Floxed mice. In accordance with these, the positive immunoreaction products detected in neurons and PC/vSMCs in Floxed mice mostly disappeared in Esr-KO mice in immunofluorescence staining. Platelet-derived growth factor receptor-β has been repeatedly shown to be expressed in neurons and PC/vSMCs (Smits et al, 1991; Virgintino et al, 2007). Furthermore, PDGFR-β is widely expressed in cerebral cortical neurons, and is involved in the dopamine receptor-mediated regulation of
In sham-operated or nonischemic cerebral cortices of Esr-KO mice, PC/vSMCs were well preserved in immunostaining and western blot for α-SMA, even though these cells were negative for PDGFR-β staining. Furthermore, vascular structures were preserved, and abnormal FITC-albumin leakage was not detected in these regions. In contrast, congenital disturbance of the PDGF-B/PDGFR-β signal axis results in the decreased numbers of PC/vSMCs and abnormal BBB leakage (Armulik et al, 2010; Bell et al, 2010). These findings are compatible with a notion that the PDGF-B/PDGFR-β signal axis is essential for the development/organogenesis, but not for the maintenance of PC/vSMC populations and BBB function in the normal adult brain. Along this line, the congenital deficits of PDGF-B/PDGFR-β signal severely disturb the organogenesis of kidney glomerulus (Lindahl et al, 1998); however, postnatal deletion of
At 3 days after MCAO, the ischemic lesions were larger in Esr-KO than in Floxed mice. Nestin-KO mice showed large 3-day lesions to the similar extent as did Esr-KO mice. These data from two different KO mice, in which PDGFR-β was commonly depleted in neurons, collaboratively indicated the neuroprotective effects of PDGFR-β that was expressed in neurons in acute cerebral ischemia. In this line, PDGF-BB and PDGF-CC pretreatments suppress lesion formation after ischemic insults (Iihara et al, 1997; Tang et al, 2010). It is a possibility that PDGFR-β protects neurons from ischemia by suppressing glutamate-induced excitotoxicity and oxidative stress, because neuron death induced by these stresses is augmented after
In our study,
In this study, infarction volume was well correlated with the degree of BBB leakage as shown by the following. In Esr-KO mice, 6-day lesions exhibited extensive BBB leakage and remained as large as 3-day lesions. The lesion volume in Nestin-KO mice was similar to that in Esr-KO mice at 3 days when BBB leakage was low in both strains, and was smaller than that in Esr-KO mice at 6 days when BBB leakage was less in Nestin-KO mice than in Esr-KO mice. Compared with Floxed mice, lesion volume and BBB leakage were exacerbated in Nestin-KO mice at 6 days. Blood—brain barrier dysfunction results in brain edema and detrimental clinical outcomes after stroke (Sandoval and Witt, 2008). Accordingly, PDGFR-β signal is believed to contribute to better clinical outcome by restoring BBB functions in early angiogenesis within the lesions after cerebral ischemia.
Pericyte/vascular smooth muscle cells induce expression of occludin, a tight junction protein that regulates BBB permeability, in cultured cerebral vascular endothelial cells (Hori et al, 2004). In addition, BBB tight junction proteins, such as ZO-1, occludin, and claudin-5, are reduced in pericyte-deficient mice (Bell et al, 2010). These results suggest that altered BBB-specific gene expression could underlie increased BBB leakage in Esr-KO mice, in which there were few PC/vSMCs in newly formed blood vessels.
In addition to PC/vSMCs, astrocytes are the primary constituents of BBB structure, and are involved in BBB repair after injury (Bush et al, 1999). Astroglial scar formation was disturbed in both Nestin-KO and Esr-KO, and these facts indicated that
In addition to the neuroprotective effects, this study showed that the PDGFR-β signal is involved in the functional recovery and local tissue responses after focal cerebral ischemia. Therefore, our data gave novel perspective that enhancement of PDGFR-β signal may potentially minimize CNS injury, improve clinical outcomes, and may be a novel therapeutic candidate for ischemic stroke, by targeting neurons, PC/vSMCs and astrocytes.
Footnotes
Acknowledgements
The authors thank Yoichi Kurashige for technological assistance, and Masako Tonami for preparing the manuscript (University of Toyama, Japan).
The authors declare no conflict of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
