Abstract
About 15 million strokes occur each year worldwide. As the number one cause of morbidity and acquired disability, stroke is a major drain on public health-care funding, due to long hospital stays followed by ongoing support in the community or nursing-home care. Although during the last 10 years we have witnessed a remarkable progress in the understanding of the pathophysiology of ischemic stroke, reperfusion induced by recombinant tissue-type plasminogen activator (tPA—Actilyse) remains the only approved acute treatment by the health authorities. The objective of the present review is to provide an overview of our present knowledge about the impact of tPA on the neurovascular unit during acute ischemic stroke.
Keywords
Introduction
Stroke concerns 15 million people every year worldwide and causes 5.7 million deaths, accounting for 9% of all deaths. The majority of ischemic strokes, the most common type of stroke, result from an acute thrombosis. A thrombus consists in blood cells trapped in a matrix of fibrin. Dissolution of this fibrin clot (fibrinolysis) is enzymatically driven by the trypsin-like serine protease, plasmin. The active form plasmin is generated from a precursor (zymogen), known as plasminogen. The conversion of plasminogen into plasmin requires proteolytic cleavage by naturally occurring plasminogen activators, which in mammals can be tissue-type plasminogen activator (tPA) or urokinase-type plasminogen activator (Rijken and Sakharov, 2001). Tissue-type plasminogen activator, urokinase-type plasminogen activator, plasmin (or a genetically modified plasmin named microplasmin), and Desmoteplase (DSPA) (a recombinant plasminogen activator derived from bat salivary glands) are available through recombinant technologies (for review on mechanisms of action of fibrinolytic agents, see Lijnen and Collen, 1995). In addition, other bacterial/microbial thrombolytics exist, including streptokinase and staphylokinase.
The tPA (Figure 1) is a 69-kDa glycoprotein, which consists in a single polypeptide chain of 527 or 530 amino acids. It is synthesized within cells as a polypeptide chain held by 34 disulfides bonds and released as a single chain enzyme (sctPA). Limited proteolysis by plasmin in the extracellular space cleaves the chain between arginine 275 and isoleucine 276, converting it into a two-chain (tctPA) form (chain A or heavy chain and chain B or light chain). Chain A contains four distinct domains: a finger (F) domain composed by residues 6 to 43, homologous to the first domain of the fibronectin. This domain has a high affinity for the fibrin; an epidermal growth factor (EGF) domain, composed by residues 44 to 92, homologous with the human and murine EGF. This domain is involved in the hepatic clearance with the finger domain; two kringle (K) domains homologous to the K domains of plasminogen consisting in residues 93 to 176 and 176 to 275. The K2 and F domains form an interaction site for fibrin. The B chain contains the catalytic protease (P) domain and is composed of the last 230 residues. The active site is constituted of Histidine 322, Asparagine 371, and Serine 478. Due to its high affinity for fibrin, tPA activates clot-bound plasminogen 100-fold more efficiently than circulating plasminogen, although it has a very short half-life in the blood (3 to 5 minutes) (Tanswell et al, 1989). These characteristics have led to successful clinical trials of thrombolytic therapy in acute stroke treatment (NINDS-tPA Study Group, 1995).
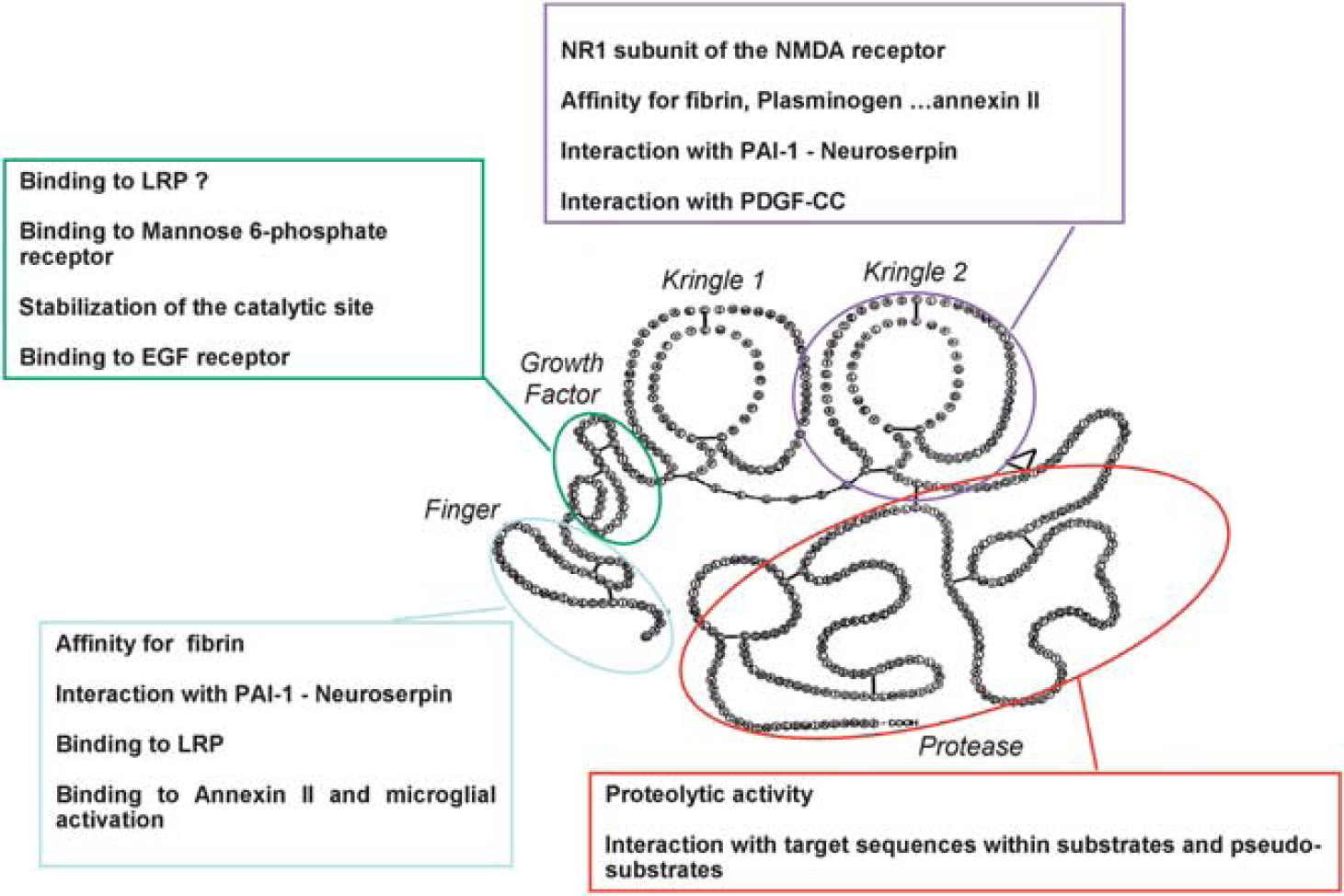
Tissue-type plasminogen activator (tPA) is a ‘JANUS’ protease. tPA is a five domain serine protease including a Finger domain, an epidermal growth factor (EGF)-like domain, two kringle domains, and a catalytic domain. Pleiotropic functions of tPA as related to the specificity of each of these domains. LRP, lipoprotein receptor-related protein; PAI-1, plasminogen activator inhibitor type 1; NMDA receptor,
However, growing body of evidence suggests that, in the context of acute ischemic stroke, tPA affects the integrity of the neurovascular unit, a functional entity involving microvessels, pericytes, astrocytes, neurons, axons, and other supporting cells such as microglia and oligodendrocytes. Indeed, both protective and deleterious effects of tPA on this ‘unit’ have been reported. This paper will review the current knowledge of benefits and risks of tPA in patients with acute ischemic stroke, preclinical evidences that could explain the suboptimal efficacy/safety ratio of tPA, and promising approaches that could enhance the efficiency of thrombolysis.
Tissue-Type Plasminogen Activator-Mediated Thrombolysis in Ischemic Stroke: Bed Side
Two placebo-controlled randomized trials have shown substantial benefit of early treatment with intravenous recombinant tPA (rtPA) in patients with acute ischemic stroke (NINDS Study Group, 1995; Hacke et al, 2008), and several meta-analyses of randomized trials support the use of rtPA within 4.5 hours after ischemic stroke onset (Lees et al, 2010; Wardlaw et al, 2009; Lansberg et al, 2009). The National Institute of Neurological Disorders and Stroke (NINDS) trial was the first to test rtPA within 3 hours of stroke onset at the dosing regimen of 0.9 mg/kg (maximum 90 mg), with 10% given as an initial bolus and the remainder infused over 1 hour. In this study, the absolute benefit of tPA treatment ranged from 11% to 13% depending on the stroke outcome scale. For every 100 patients treated, as compared with placebo, ∼32 benefited in a clinically important manner (Saver et al, 2010). Following these results, rtPA has been approved in several countries and recommended in most guidelines for the management of patients with acute stroke (Adams et al, 2007). However, widespread adoption of rtPA therapy proceeded slowly. Among the different potential obstacles restricting the use of rtPA, the time window has been the most important. Many patients do not arrive at the hospital within 3 hours after stroke onset, and even then, the time required to thoroughly evaluate the patient and make the treatment decision may jeopardize the chances of initiating thrombolysis.
Expansion of the Time Window up to 4.5 Hours: European Cooperative Acute Stroke Study III
To explore a longer time window, a pooled analysis of six trials (2,775 patients) that had differing time windows (up to 6 hours) for enrollment (NINDS Parts 1 and 2, European Cooperative Acute Stroke Study (ECASS) I and II, and ATLANTIS Parts A and B) was performed (Hacke et al, 2004). This pooled analysis showed that the benefit of rtPA diminished as time elapsed and that a favorable outcome was still observed if treatment was given between 3 and 4.5 hours, with an odds ratio for favorable outcome of 1.4 with rtPA as compared with placebo.
In 2002, the European Medicines Agency was cautious and granted license for the use of rtPA in ischemic stroke within 3 hours of symptoms onset on condition of the completion of a prospective registry of (1) patients treated with rtPA within the 3-hour time window (Safe Implementation of Thrombolysis in Stroke-Monitoring Study (SITS-MOST) study, see below) and (2) a randomized, placebo-controlled trial of rtPA between 3 and 4.5 hours after stroke onset. Thus, the ECASS III multicenter, prospective, randomized trial was conducted in Europe and tested rtPA 3 to 4.5 hours after stroke onset in 821 patients (Hacke et al, 2008). The dosing regimen was the same as in NINDS trial. The proportion of patients who had a favorable outcome (modified Rankin Scale score<2) at 90 days was significantly greater with rtPA than with placebo (52.4% vs. 45.2%; odds ratio=1.34, 95% confidence interval (95% CI)=1.02 to 1.76; risk ratio=1.16, 95% CI=1.01 to 1.34), corresponding to number needed to treat to benefit of 14 (absolute improvement of 7.2%). Additional outcomes and subgroup analyses published in a separate paper showed that rtPA was effective across a broad range of subgroups of patients, including those aged 65 years or more and those with a severe stroke at baseline (Bluhmki et al, 2009).
The ECASS III trial has represented an important step forward in the treatment of acute stroke, allowing an increase in the number of eligible patients. However, the ECASS III trial results do not mean that we have more time to initiate the treatment (Saver, 2006). From the moment a patient arrives at the door, the focus must remain on the door-to-needle time (Lyden, 2008). The updated pooled analysis of ECASS, ATLANTIS, NINDS, and EPITHET trials (3,670 patients) have demonstrated that the benefit of rtPA decreased substantially over time from stroke onset. In this analysis, no benefit was seen after around 4.5 hours (Lees et al, 2010). On the basis of ECASS III and SITS-MOST studies results, the European Stroke Organization and subsequently the American Heart Association and American Stroke Association changed their guidelines (Del Zoppo et al, 2009; Ringleb et al, 2008).
Recanalization Rates on Tissue-Type Plasminogen Activator
Data on recanalization rate after intravenous rtPA are limited to observational studies, as none of the major randomized trials has assessed the baseline occlusion status or recanalization after treatment. Yet, it is clearly established that early recanalization (assessed until 24 hours from symptoms onset) is the strongest predictor of good clinical outcome. Depending on timing when recanalization status was assessed and on technique used, the recanalization rates after intravenous rtPA varied widely, with an overall estimate of ∼46% (Rha and Saver, 2007). The speed of clot dissolution by thrombolysis appears to affect the evolution of the ischemic lesion and clinical outcome (Delgado-Mederos et al, 2007). Vessel recanalization rates also vary depending on thrombus location with lower rates in proximal vs. distal arteries, suggesting that the burden of thrombus is important (Rha and Saver, 2007; Del Zoppo et al, 1992; Saqqur et al, 2007). Patients with extracranial large vessel occlusion have also very low rates of recanalization (Saqqur et al, 2007; Bhatia et al, 2010). Data on recanalization according to stroke etiologic subtype are limited. It has been suggested that patients with cardioembolic ischemic stroke are more likely to have recanalization, which is moreover faster and more complete, than patients with noncardioembolic stroke (Molina et al, 2004). Indeed considering the rtPA pharmacology, the drug is more likely to be active on fibrin-rich clots than on platelet-rich clots (see below).
Innovative Strategies to Enhance Recombinant Tissue-Type Plasminogen Activator's Effects
Thus, although the overall recanalization rate with intravenous rtPA is about twice the spontaneous rate, it is still <50%. This low recanalization rate is in agreement with the fact that only about one third of patients benefit of rtPA and has encouraged novel techniques to increase this relatively modest effect of intravenous fibrinolytic therapy. Development of catheters suitable for navigation into brain vasculature has opened the possibility for endovascular therapies including intraarterial fibrinolysis. As intraarterial approach allows a direct access to the clot, the technique should theoretically permit the use of lower doses of fibrinolytic, be combined with mechanical thrombus disruption devices, and therefore offer the potential of restoring blood flow more efficiently than intravenous therapy. Although intraarterial fibrinolysis has been used for decades in acute stroke patients, this therapy is not approved by the US Food and Drug Administration or by the European Medicines Agency. No single randomized clinical trial has demonstrated a statistically significant beneficial effect (defined by a modified Rankin Scale 1 or 2) of intraarterial fibrinolysis compared with control groups and not all studies used rtPA (Del Zoppo et al, 1998; Furlan et al, 1999; Keris et al, 2001; Macleod et al, 2005; Ogawa et al, 2007). However, despite differences in fibrinolytic drug used and in associated interventions (intravenous thrombolysis or mechanical disruption), the meta-analysis of these trials shows that intraarterial fibrinolysis is associated with high recanalization rates and better outcomes (Lee et al, 2010). Symptomatic intracerebral hemorrhage (ICH) is significantly more frequent in intraarterial fibrinolysis compared with control groups (8.9% vs. 2.3%), but mortality is similar in both groups (20.5% vs. 24.0%). Partial or complete recanalization was obtained in 64.6% of patients treated with intraarterial fibrinolysis, but complete recanalization was obtained in only 19.0% in mean, showing that intraarterial fibrinolytic therapy alone may not be a definitive solution to rapidly and fully recanalize occluded cerebral arteries.
The main limitations of intraarterial thrombolysis are the need for availability of an interventionalist and the additional time required to begin thrombolysis. Thus, a combined intravenous and intraarterial approach was explored in pilot studies (Lewandowski et al, 1999; Ernst et al, 2000; Hill et al, 2002; Suarez et al, 2002). The Interventional Management of Stoke (IMS) program began in 2001 aiming at further investigate the feasibility and safety of a combined intravenous–intraarterial approach within 3 hours of stroke onset in patients with moderate-to-large ischemic stroke (NIHSS10). IMS I and II studies (IMS Study Investigators, 2004; IMS II Trial Investigators, 2007) used a similar protocol with initial intravenous rtPA (0.6 mg/kg, 60 mg maximum over 30 minutes) followed by additional rtPA at the site of the thrombus (up to 22 mg) in case of visible arterial clot on early angiography for up to 7 hours from stroke onset. Unlike IMS I, IMS II used the EKOS microinfusion catheter consisting in end-hole infusion lumen with a 1.7-MHz radially emitting ultrasound transducer in place of classical microcatheter contrast injection leading to increased recanalization rates (Tomsick et al, 2008). These 2 nonrandomized studies enrolled 80 patients each. In comparison with historical control subjects from the NINDS trial, the results suggested that the combined approach to recanalization might be more effective than standard intravenous rtPA alone. The RECANALISE study has evaluated the combined approach using a before–after design (Mazighi et al, 2009). In comparison with 107 patients treated with intravenous rtPA only in the first period, the 53 patients treated with intravenous–intraarterial approach had higher recanalization rates (87% vs. 52%; risk ratio=1.49, 95% CI=1.21 to 1.84) and were more likely to have an early neurologic improvement defined as National Institute of Health Stroke Scale score of 0 or 1 or improvement of 4 points at 24 hours (60% vs. 39%; risk ratio=1.36, 95% CI=0.97 to 1.91). However, there was no significant difference in the 90-day favorable outcomes defined as a Rankin Scale of 0 to 2 (57% vs. 44%,
Ultrasound-enhanced thrombolysis is a promising approach including improvement in drug delivery, increase in binding of rtPA to fibrin, and reversible alteration of the fibrin structure (Daffertshofer and Hennerici, 2003). So far, three different ultrasound technologies have been used: transcranial Doppler, transcranial color-coded duplex, and low-frequency ultrasound. A recent meta-analysis of small randomized trials and nonrandomized studies has shown that sonothrombolysis with high-frequency ultrasounds appears to be safe, leads to higher recanalization rates (odds ratio=2.99; 95% CI=1.70 to 5.25) and higher likelihood of independence when compared with intravenous thrombolysis alone (Tsivgoulis et al, 2010), as it was suggested by the first results of the CLOTBUST trial (Alexandrov et al, 2004). However, the single trial using low-frequency ultrasounds was stopped because ultrasounds were associated with a significant increase in risk of symptomatic ICH (including atypical subarachnoid hemorrhages; Daffertshofer et al, 2005). The potential benefit of high-frequency ultrasounds results needs to be confirmed in new and larger randomized-controlled studies.
Another way to enhance fibrinolysis and prevent the risk of reocclusion is the use of antiplatelet agents in combination with rtPA. The most widely used antiplatelet agent is aspirin, but there is currently no randomized trial that has assessed the effect of aspirin in combination with rtPA and it is even recommended that aspirin should not be started within the 24 hours after thrombolysis (Adams et al, 2007). However, ∼30% of stroke patients treated by intravenous rtPA receive antiplatelet agents at the time of their stroke. In the Safe Implementation of Treatments in Stroke (SITS) International Stroke Thrombolysis Register (SITS-ISTR), patients who received aspirin, clopidogrel, aspirin and clopidogrel, or aspirin and dipyridamole at the time of their stroke were no more likely to have a good prognosis than those who did not receive antiplatelet agents after adjustment for baseline characteristics (Diedler et al, 2010). The results were in agreement with those of previous studies (Cucchiara et al, 2009). The issue of whether intravenous rtPA in combination to aspirin is superior to intravenous rtPA alone is currently addressed in the randomized ARTIS trial (Zinkstok et al, 2010). Combining glycoprotein IIb/IIIa inhibitors with intravenous or intraarterial tPA has been shown to be feasible and apparently safe in small observational studies, but no large randomized studies are available (Deshmukh et al, 2005; Mangiafico et al, 2005; Qureshi et al, 2006). Considering that the use of abciximab within 5 hours of stroke onset is associated with an increased risk of symptomatic or fatal ICH (Adams et al, 2008), the approach combining abciximab and intravenous rtPA is likely to be illusory. The randomized CLEAR study assessed the safety of the eptifibatide in combination with intravenous rtPA within 3 hours of acute stroke (Pancioli et al, 2008). The combination was apparently safe and, although there was no trend toward efficacy, a new trial is currently ongoing (http://www.strokecenter.org/trials/trialDetail.aspx?tid=995).
Based on experimental data and early human experience, hypothermia appears a promising neuroprotective therapy for acute ischemic stroke. Hypothermia could improve the efficacy and potentially extends the time window for tPA-induced reperfusion. The recent ICTuS-L trial assessed the feasibility and safety of induced hypothermia after ischemic stroke and thrombolysis. Fifty-nine patients were randomized to receive either hypothermia (33°C) immediately after rtPA-induced thrombolysis for 24 hours, followed by 12 hours of controlled rewarming or normothermia (Hemmen et al, 2010). Target temperature was reached in 20/28 patients. At 3 months, 18% of patients treated with hypothermia had good outcome compared with 24% in the normothermia group. A higher rate of pneumonia was reported in the hypothermia group (50% vs. 10%,
Finally, another very promising approach is to improve the selection of patients who are most likely to benefit from thrombolysis, i.e., those who still have salvageable cerebral tissue (ischemic penumbra). EPITHET has shown that mismatch on magnetic resonance imaging treatment by tPA was associated with attenuation of infarct volumes and nonsignificant better functional recovery at 3 months (Davis et al, 2008). This tissue-based approach could also be applied in patients with unknown-onset or wake-up stroke (Mackey et al, 2011; Barreto et al, 2009). A few thrombolysis trials with penumbra assessment have been carried out, most showing encouraging results, and new trials are currently ongoing (see Donnan et al, 2009 for review).
Safety of Recombinant Tissue-Type Plasminogen Activator-Induced Thrombolysis
The most serious complication of rtPA treatment is the occurrence of a large ICH, most often in the core of the cerebral infarction. When this complication occurs, there is a fatality rate of ∼50%. There have been several definitions of symptomatic ICH after rtPA treatment in trials and safety-monitoring studies, all based on computerized tomography scan imaging. Most studies defined symptomatic ICH as any hemorrhage associated with neurologic deterioration, as indicated by an increase in the NIHSS score of 4 or more points (Hacke et al, 1995; Wahlgren et al, 2007). However, clinical deterioration in patients with ICH could also be due to an increase in cerebral infarction or cerebral edema, coinciding with hemorrhagic transformation. Thus, in ECASS III, the ICH had to have been identified as the predominant cause of the neurologic deterioration to be considered symptomatic (Hacke et al, 2008).
After the publication of the NINDS and other trials, there have been concerns over the applicability of rtPA in daily practice, especially regarding the risks of ICH. In NINDS trial, the risk of symptomatic ICH was 6.4% in the rtPA group compared with 0.6% in the placebo group (
Several clinical, biological, and imaging predictors of ICH have been identified (Derex and Nighoghossian, 2008). Although longer stroke onset-to-treatment time may theoretically be associated with a higher risk of ICH, most studies and the pooled analysis of randomized trials failed to show an association between timing of rtPA administration and symptomatic ICH. There is very few evidence on the benefit/risk profile of rtPA in patients over 80 years from randomized trials. Yet, ∼25% of stroke patients in developed countries are aged over 80 years, and this proportion will increase over the next decades. The NINDS included only 42 patients over 80 and other trials did not enroll those patients. Thus, thrombolytic therapy is not currently licensed for patients >80 years in the European Union. By contrast, most safety-monitoring studies have included patients without an age limit and most stroke centers use rtPA in elderly patients. In published safety-monitoring studies, old age is associated with poor clinical outcome and with an increased risk of symptomatic ICH (Wahlgren et al, 2008). Of note, a small uncontrolled retrospective study showed that patients aged over 90 years (
A few studies have shown that the use of biomarkers, such as matrix metalloproteinases (MMPs, especially MMP-9), may help identify patients at high risk of symptomatic ICH (Derex and Nighoghossian, 2008). These proteinases, which are released at the acute stage of ischemia, increase the blood–brain barrier (BBB) permeability as they damage some components of the vessel and the basal lamina. High plasmatic levels of vascular adhesion protein 1 and cellular fibronectin have also been associated with symptomatic ICH after thrombolytic treatment (Hernandez-Guillamon et al, 2010; Castellanos et al, 2004, 2007). This approach seems promising, but more data are needed before biomarkers can be used to select the best candidates for thrombolysis.
In intravenous–intraarterial thrombolysis studies, the risk of symptomatic ICH was up to 10% according to ECASS-II definition, which is higher than that observed in patients treated with intravenous tPA alone (IMS Study Investigators, 2004; IMS II Trial Investigators, 2007; Mazighi et al, 2009). This higher rate of symptomatic ICH can be related to the severity of stroke and to the technique itself. Indeed, intraarterial procedure requires the use of heparin and microcatheter contrast injections increase the risk of ICH (Khatri et al, 2008). In IMS, internal carotid occlusion and atrial fibrillation were also independent risk factor for symptomatic ICH (Khatri et al, 2008).
Tissue-Type Plasminogen Activator-Mediated Thrombolysis in Ischemic Stroke: Bench Side
Clinical trials and daily use of thrombolysis have highlighted that rtPA has several drawbacks in acute stroke setting. Most of the effects of rtPA can be replicated in ischemic stroke animal models. For instance, volumes of ischemic lesions are significantly smaller in tPA-deficient mice compared with nondeficient mice (Wang et al, 1998; Nagai et al, 1999). Similarly, inhibition of tPA in a murine stroke model results in a decrease in ischemic lesion volumes (Yepes et al, 2000). Also, we have shown that the use of rtPA in a thromboembolic stroke murine model was associated with deleterious effects when infused at a late stage after stroke onset (4 hours) and that these deleterious effects could not be only explained by the increase in the risk of hemorrhage (Orset et al, 2007; Macrez et al, 2010; García-Yébenes et al, 2011). Consequently, tPA is a double-sided molecule, with beneficial effects due to its fibrinolytic activity and deleterious effects increasing with time after stroke onset that counteract the global benefits of reperfusion (Benchenane et al, 2004).
Deleterious Effects of Tissue-Type Plasminogen Activator in Experimental Stroke Include Both Actions in the Blood and in the Brain Parenchyma
In the circulation, main substrate for endogenous or exogenous tPA is plasminogen, which promotes thrombolysis (Figures 2 and 3). By contrast, in the brain tissue, tPA substrates extend above tPA/plasmin(ogen)-driven extracellular matrix degradation (Figure 3). Indeed, tPA has been reported to interact with the low density lipoprotein receptor-related protein (LRP), the
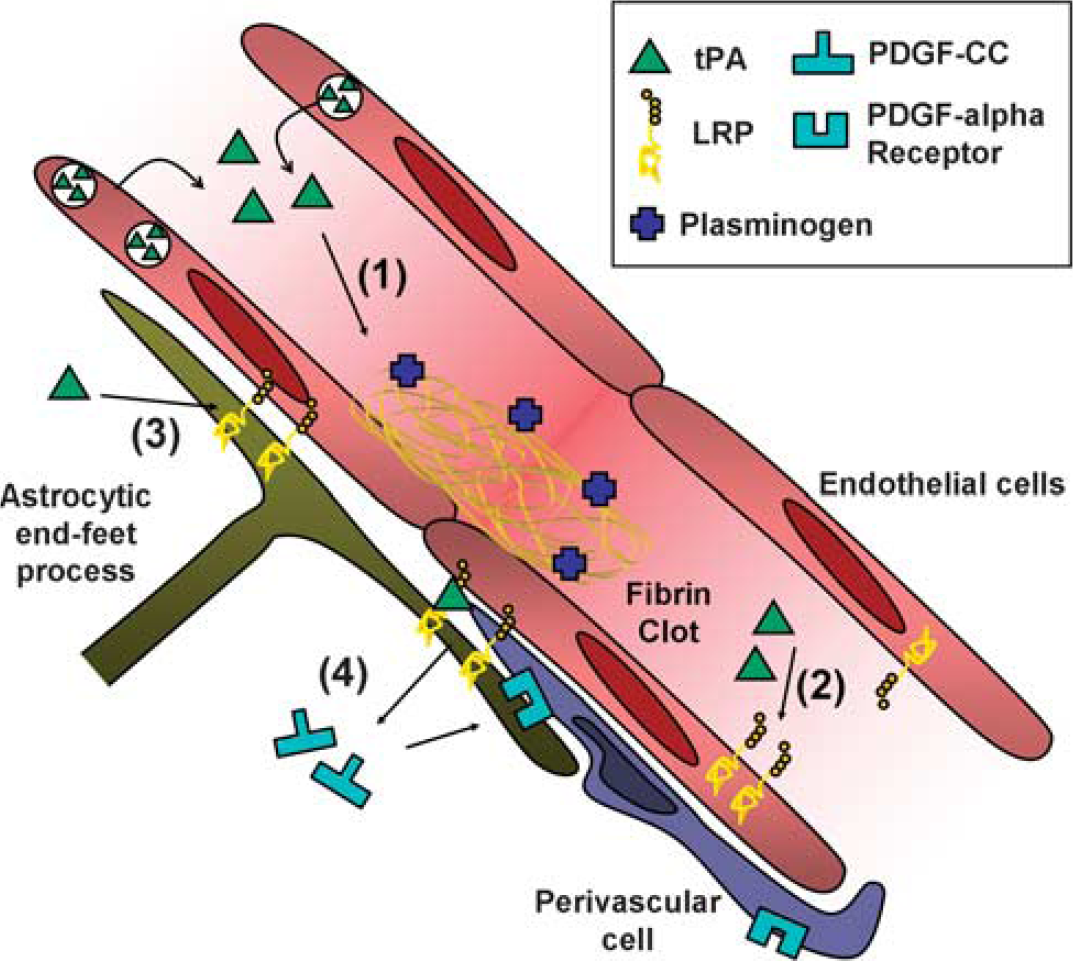
Effect of exogenous and endogenous tPA at the blood–brain barrier (BBB). (1) Tissue-type plasminogen activator (tPA)-mediated plasminogen activation promotes clot lysis and subsequent blood flow restoration. (2) tPA interacts with endothelial LRPs, leading to NF
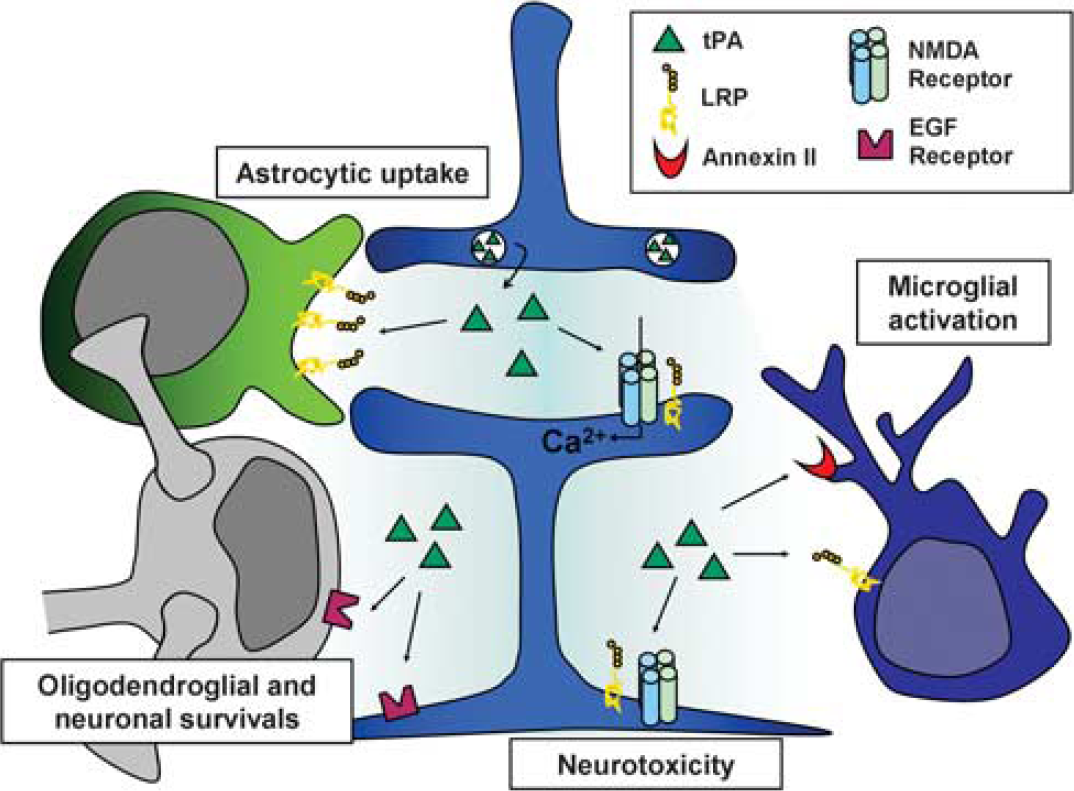
Tissue-type plasminogen activator (tPA) in the brain parenchyma. In the brain parenchyma, tPA, released by neurons, was reported to potentiate NMDA receptors signaling to display either neurotrophic or neurotoxic effects, to reduce apoptotic neuronal and oligodendroglial deaths, to activate microglia after binding to annexin II and LRP and to be cleared from the extracellular space by astrocytes through an LRP-dependent mechanism. LRP, lipoprotein receptor-related protein; NMDA,
Tissue-Type Plasminogen Activator at the Blood–Brain Barrier
A growing body of evidence indicates that tPA mediates increase in the permeability/leakage of the BBB (Lo et al, 2002). The pathways described are multiple and involve several components of the neurovascular unit (Figure 2). Indeed, in an embolic stroke animal model, treatment with rtPA induces the extravasation of Evans blue dye and overactivation of MMP-3 and MMP-9, two markers of BBB permeability. Studies in knockout mice argue that both MMP-9 and MMP-3 are responsible for tPA-induced parenchymal hemorrhages after stroke. Additionally, injection of rtPA into the cerebrospinal fluid induces a dose-dependent increase in permeability of the BBB, and this effect is inhibited by treatment with either the receptor-associated protein (an antagonist of the LRP) or by anti-LRP antibodies (Yepes et al, 2003). Since endothelial expression of LRP gradually increases during oxygen glucose deprivation (Suzuki et al, 2009), this could explain the observed time-dependent increase in hemorrhage rate after thrombolysis. In addition, it has been recently demonstrated in experimental models that expression of LRP also increased in perivascular astrocytes early after the onset of the ischemic insult onset and that its interaction with both endogenous and exogenous tPA-induced shedding of its ectodomain into the vascular basement membrane. Altogether, these events were associated with detachment of astrocytic end-feet from the basal lamina and subsequent increased BBB permeability (Polavarapu et al, 2007). In addition, it has been reported that rtPA could mediate the local activation of latent platelet-derived growth factor-CC (PDGF-CC) and subsequent activation of PDGF-α receptors on pericytes after cerebral ischemia (Macrez et al, 2011a, 2011b), an effect leading to hemorrhagic transformation (Su et al, 2008). Indeed, intraventricular injection of rtPA or active PDGF-CC, in the absence of ischemia, led to significant increase in BBB permeability. In contrast, coinjection of neutralizing antibodies to PDGF-CC with tPA blocked this increased permeability, indicating that PDGF-CC is a downstream substrate of tPA within the BBB. Since ischemia–reperfusion alone induces time-dependent changes in BBB integrity (Hamann et al, 1996), blood-derived tPA can reach the brain parenchyma and interact with all the components of the neurovascular unit.
Tissue-Type Plasminogen Activator Within the Brain Parenchyma
In the brain parenchyma, tPA had initially been shown to promote kainate-induced neurotoxicity (Tsirka et al, 1995) and then to potentiate NMDA receptor signaling, with several potential mechanisms: (1) an upregulation of NR2B-containing NMDA receptors (Pawlak et al, 2005); (2) a catalytic-independent activation of an ERK1/2-GSK3 signal transduction pathway (Medina et al, 2005); (3) the plasmin-independent proteolytic cleavage of the amino terminal domain of the NMDA receptor NR1 subunit (Nicole et al, 2001; Fernández-Monreal et al, 2004; Benchenane et al, 2007); (4) an overactivation of extrasynaptic NR2D-containing NMDA receptors (Baron et al, 2010). Although the fine mechanism remains debated, tPA should be now considered as a neuromodulator of glutamatergic signaling mediated by NMDA receptors (Benchenane et al, 2007; Samson and Medcalf, 2006) leading to an increased calcium influx, overactivation of MAP kinase ERK1/2 signaling and finally to an increased neurotoxicity. It is also interesting to note that NMDA-dependent nitric oxide production has been recently demonstrated to be dependent on tPA (Parathath et al, 2006), unveiling a possible role of tPA in the control of local cerebral perfusion. In contrast to its well-admitted exacerbating effect on neuronal excitotoxic necrosis (Nolin et al, 2008), tPA modulates apoptotic neuronal (Liot et al, 2006) and oligodendrocyte death independently of its proteolytic activity (Correa et al, 2011). Together with the previous observations that nonproteolytically active tPA can activate microglia after binding to annexin II (Siao and Tsirka, 2002), these antiapoptotic mechanisms of tPA strongly support the notion of a ‘cytokine-like’ or even ‘neurotrophic-like’ function of this molecule. However, these data are in contradiction with other reports (Pawlak et al, 2005; Liu et al, 2004), suggesting that tPA promoted NMDA-induced apoptosis. The protection of oligodendrocytes from apoptosis provided by exogenous tPA (Correa et al, 2011) raises the intriguing possibility that thrombolysis could improve stroke outcome even when arterial reperfusion fails. Since interaction of tPA with oligodendrocytes is mediated by its EGF domain, it is also interesting to note that other plasminogen activators lacking the EGF domain (such as reteplase) should be devoid of these protective effects.
Tissue-Type Plasminogen Activator and Cerebrovascular Inflammation
Interaction of tPA with glial cells raises questions about the influence of thrombolysis on poststroke inflammation. Indeed, cerebral ischemia/reperfusion rapidly leads to the release of pro-inflammatory cytokines (including TNF and IL-1β) (Zhang et al, 1999; Stoll et al, 1998) from injured brain tissues (especially from activated microglia). Subsequently, brain microvascular endothelial cells are activated and express adhesion molecules (ICAM-1, VCAM-1, E-Selectin) allowing transendothelial migration of circulating cells (Frijns and Kappelle, 2002). In the subacute stage, resident and infiltrating cells produce MMP (Gidday et al, 2005) and inflammatory cytokines. Altogether, these phenomena increase BBB permeability and further amplify inflammatory response, eventually leading to hemorrhagic transformation and neuronal death (for review of the role of MMPs in neuroinflammation, see Rosenberg, 2002). Interestingly, microglial activation was demonstrated to be significantly decreased in tPA-deficient mice (Rogove et al, 1999). Mice lacking LRP on microglia have reduced ischemic edema and reduced MMP-9 elevation after stroke (Zhang et al, 2009), suggesting that tPA interaction with microglial LRP contributes to the development of inflammation, BBB leakage, and cerebral edema. Notably, delayed tPA treatment in rats increases endothelial expression of ICAM-1 and neutrophil accumulation. Accordingly, strategies blocking leukocyte adhesion or activation increase therapeutic time window for thrombolysis in preclinical studies (Zhang et al, 1999; Bowes et al, 1995). It has been demonstrated that direct interaction of endothelial LRP receptors with tPA induces NF
Tissue-Type Plasminogen Activator and Platelet-Derived Serpins
After arterial recanalization, platelets (among other blood components) accumulate on the injured vasculature and thus, could impair reperfusion. Since thrombolytic effects of tPA are mediated by plasmin, which degrades fibrin, tPA-induced thrombolysis is significantly more efficient on fibrin-rich clots than on platelet-rich clots (Levi et al, 1992; Zhu et al, 1999). But other factors may contribute to the limited efficiency of tPA on platelet-rich clots. Indeed, on activation, platelets release large amounts of plasminogen activator inhibitor type 1 (a serine protease inhibitor or serpin), which inhibits tPA proteolytic activity and blocks plasminogen activation. Accordingly, plasminogen activator inhibitor type 1 blockade promotes tPA-mediated thrombolysis in experimental models. Recently, it has also been shown that protease nexin 1, which accumulates in thrombus because of its presence in platelets, also impairs tPA-mediated thrombolysis by direct inhibition of fibrin bound plasmin (Boulaftali et al, 2011). Altogether, these mechanisms decrease thrombolysis efficiency on platelet-rich clots and could contribute to the low recanalization rate of ischemic stroke patient treated with tPA. Whether platelet-derived serpins could alleviate tPA-induced BBB injuries remains, however, to be investigated.
Tissue-Type Plasminogen Activator and Ageing
Whereas most of the preclinical studies are conducted in young and healthy animals, ischemic stroke mainly occur in elderly people with increased vascular risk factors and comorbidities (Jaramillo et al, 2006). Interestingly, it has been demonstrated that ageing was associated with a decrease in expression and activity of tPA in the brain parenchyma of mice, and that this was associated with a decrease ischemic lesion volumes and with protection of the barrier function of the neurovascular unit during cerebral ischemia (Roussel et al, 2009). Thus, although controlling the endogenous availability of tPA could be of therapeutic interest, its transcriptional regulation in nerve cells remains poorly investigated (Shin et al, 2004; Lee et al, 2008). Interestingly, it has been recently proposed that the transcription of the neuronal tPA may be influenced by epigenetic regulation (Obiang et al, 2011).
Can We Improve the Efficacy/Safety Ratio of Tissue-Type Plasminogen Activator?
The risk of hemorrhagic transformation carried by the use of tPA has initiated an era of investigation for alternative thrombolytic agents. With advances in DNA technology, chemistry and proteomics, several second and third generation of tPA derivatives, genetically engineered molecules targeting the blood clot and even ‘exotic’ plasminogen activators have progressively reached clinical trials, including Streptokinase, DSPA, Tenecteplase (TNK-tPA), Microplasmin, Reteplase, Ancrod, and Pro-urokinase. However, none has demonstrated benefits so far. Conceptually, the ideal thrombolytic agent should be an effective fibrinolytic agent, devoid of harmful effects on brain cells and BBB. It has been recently demonstrated that the minimal structural requirements for tPA-mediated pro-excitotoxic activity is reteplase, i.e., K2 and protease domains (Lopez-Atalaya et al, 2008). Using an
Relevant adjunctive therapy to tPA reported in the last 5 years
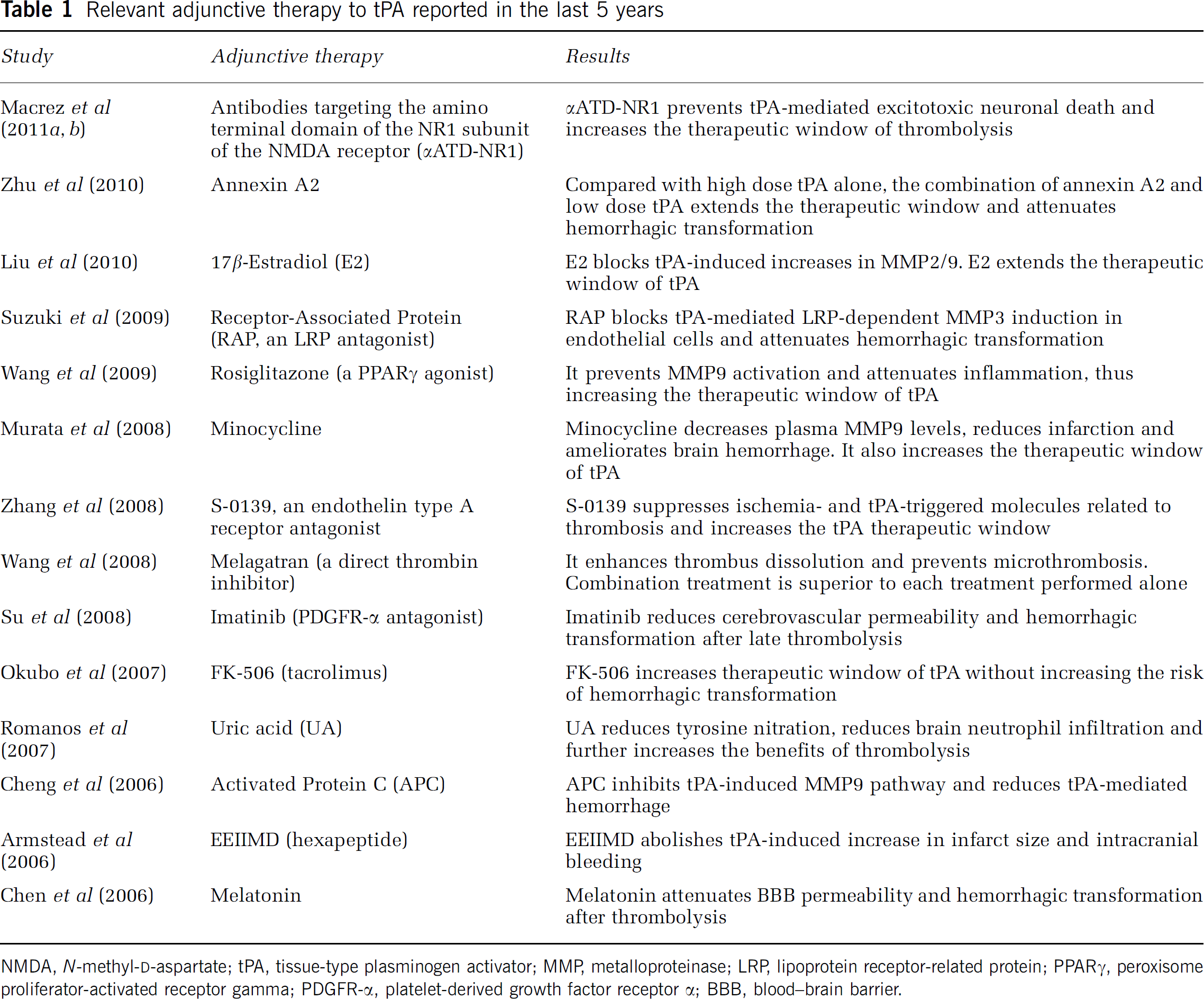
NMDA,
Conclusion
Thrombolytic therapy using recombinant human tPA is a widely accepted therapy for acute stroke. However, despite an important advance in the management of stroke patients, more research is needed to increase the benefit and safety of tPA. First, a number of efforts remain to be done to ensure that patients are promptly identified and efficiently treated. Second, combined strategies to increase the rate of recanalization and prevent early reocclusion are warranted. Third, the selection of the best candidates for thrombolysis using imaging of the ischemic penumbra is very promising. Finally, there seems to be a potential for targeted strategies to prevent the induction of brain toxicity and hemorrhagic complications by tPA. Elaboration of such brain cocktails requires strong collaboration between researchers and clinicians.
Footnotes
The authors declared no conflict of interest. DV is involved in the development of pharmaceuticals that target reperfusion (Desmoteplase) and tPA toxicity (Macrez et al, 2011a, ![]() ).
).
