Abstract
Intra-arterial (IA) injection represents an experimental avenue for minimally invasive delivery of stem cells to the injured brain. It has however been reported that IA injection of stem cells carries the risk of reduction in cerebral blood flow (CBF) and microstrokes. Here we evaluate the safety of IA neural progenitor cell (NPC) delivery to the brain. Cerebral blood flow of rats was monitored during IA injection of single cell suspensions of NPCs after stroke. Animals received 1×106 NPCs either injected via a microneedle (microneedle group) into the patent common carotid artery (CCA) or via a catheter into the proximally ligated CCA (catheter group). Controls included saline-only injections and cell injections into non-stroked sham animals. Cerebral blood flow in the microneedle group remained at baseline, whereas in the catheter group a persistent (15 minutes) decrease to 78% of baseline occurred (P < 0.001). In non-stroked controls, NPCs injected via the catheter method resulted in higher levels of Iba-1-positive inflammatory cells (P = 0.003), higher numbers of degenerating neurons as seen in Fluoro-Jade C staining (P < 0.0001) and ischemic changes on diffusion weighted imaging. With an appropriate technique, reduction in CBF and microstrokes do not occur with IA transplantation of NPCs.
Introduction
Stroke is the third leading cause of death in the United States behind heart disease and cancer (Dickerson et al, 2007; Rosamond et al, 2007). Each year 700,000 people experience stroke, resulting in ~270,000 deaths. Despite its enormous impact on death and disability rates, there are few treatment options available for the majority of patients (Reeves et al, 2005). However, recent advances in the field of experimental stem cell research have created promising new avenues for stroke therapies. Although the growing body of evidence demonstrating the beneficial effects of stem cell-based therapies in animal stroke models has prompted various clinical trials (Roitberg, 2004; Savitz et al, 2004), the optimal route of stem cell delivery remains unresolved.
Intravascular transplantation is a minimally invasive approach that may circumvent the need for stereotactic transplantation of cells. The most obvious intravascular transplant option is intravenous infusion based on ease and clinical precedent. However, several groups have shown that intravenous infusion results in transplanted cells becoming trapped in filtering organs, including the liver, spleen, and lungs (Fischer et al, 2009; Hauger et al, 2006; Kraitchman et al, 2005; Li et al, 2010; Pendharkar et al, 2010), as well as minimal engraftment of cells in the brain. In contrast, intra-arterial (IA) injection may represent a more effective transplant paradigm providing a more direct route to the lesion with a higher level of cellular survival and engraftment (Bliss et al, 2007; Fischer et al, 2009; Guzman et al, 2008; Harting et al, 2009; Li et al, 2010).
It has been reported that IA transplantation carries the risk of reduction in cerebral blood flow (CBF) associated with microstrokes (Walczak et al, 2008). Although Brenneman et al (2010) and Li et al (2010) have suggested that this can be circumvented by using mononuclear cells, it remains unclear whether this observation is related to the injection technique or if it is inherent to the IA cell transplantation approach. As researchers further explore the advantages of a targeted IA stem cell delivery technique to the injured brain, the safety of an IA stem cell transplantation paradigm must be evaluated. The aim of this study was to analyze the effect of IA stem cell injection on CBF and the risk of microstrokes in two technically different cell injection approaches. We show that delivering neural stem cells into the carotid artery using a microneedle injection technique with preserved flow within the artery does not lead to alterations in CBF and does not cause microstrokes.
Materials and methods
Cell Culture and Labeling
Murine neural progenitor cells (NPCs; C17.2 cell line) were cultured in Dulbecco's modified Eagle's medium (Gibco, Grand Island, NY, USA) supplemented with 10% fetal bovine serum (Gibco), 5% horse serum (Gibco), and 1%
Experimental Stroke Model
All animal procedures were approved by the Stanford University Administrative Panel on Laboratory Animal Care. Middle cerebral artery (MCA) occlusion was induced in 240 to 290 g female Wistar rats, as previously described (Longa et al, 1989). Briefly, a silicon-coated 4-0 filament (Doccol Corporation, Redlands, CA, USA) was advanced up the internal carotid artery until it occluded the middle cerebral artery. The occlusion time was 2 hours, followed by reperfusion. Sham or non-stroked animals received the same initial procedure, but their common carotid artery (CCA) was merely exposed and middle cerebral arteries were left untouched.
Laser Doppler Flow Measurements and Cell Transplantation
Twenty-four hours after stroke, a laser Doppler probe was installed using dental acrylic on the ipsilateral cranium, on the thinned skull above the MCA region under the temporal muscle to measure CBF in the MCA region. Owing to variability in signal from bone thickness and vasculature and the possibility of affecting our findings, the probe was positioned at the location with the highest sustained signal and the values were recorded as a percentage of that baseline. Before cell transplantation, the CCA was exposed and the external carotid artery and pterygopalatine artery were ligated. In the group injected using the catheter method, the CCA was ligated, followed by placement of a catheter (25G) distal to the ligation (Figure 1A). This allowed us to compare with similar techniques using PE 20 (Walczak et al, 2008) and PE 50 (Li et al, 2001) tubing to catheterize the vessels leading to the MCA. Cells (1 × 106) in 1 mL saline were injected directly into the CCA over the course of 40 seconds. In the group transplanted with the microneedle method (5 μL Hamilton syringe and custom made 33G microneedle), cells (1 × 106) in 5 μL saline were injected into the CCA with preserved flow in the CCA and internal carotid artery (ICA) (Figure 1B), and bleeding was controlled by the application of bioabsorbable Gelfoam (Pfizer, New York, NY, USA) at the injection site for 5 minutes. Pressure was maintained at the site until bleeding ceases. The stroke controls underwent MCA occlusion stroke surgery and received IA saline injection of 1 mL using the catheter method or 5 μL using the microneedle method. The cell doses, flow, and volumes in this study were based on previously published methods (Guzman et al, 2008; Li et al, 2001; Walczak et al, 2008) to compare their effects. The non-stroke controls received sham surgeries but were administered their respective cell infusions via catheter or microneedle injection technique (see Table 1 for details). Laser Doppler measurements for all groups were recorded before transplantation after preparation of vessels. For the catheter group, the baseline was recorded after unilateral CCA occlusion, taking the 10% to 20% CBF reduction caused by CCA occlusion out of the equation. Initial animals were measured for up to 25 minutes without any changes noticed past 15 minutes (data not shown). Subsequent measurements for all groups were recorded for 15 minutes.
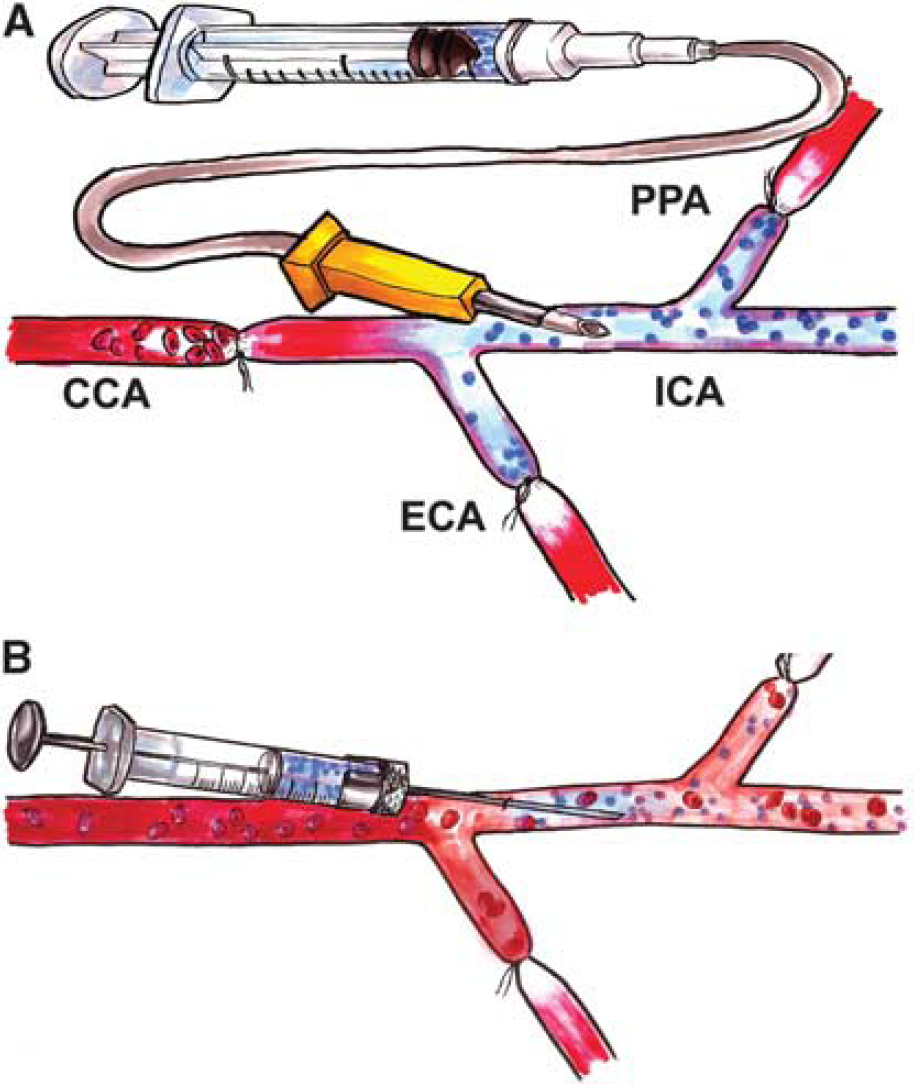
Schematic of the catheter cell injection method. (
Table demonstrating the various groups and their animal numbers used in the study

Both methods include controls that were either unstroked or were given saline instead of cells.
Bioluminescence and Magnetic Resonance Imaging
Bioluminescence imaging was conducted on the IVIS Spectrum system (Xenogen Corporation, Alameda, CA, USA). Rats were administered an intraperitoneal injection of 100 μL
Magnetic resonance imaging (MRI) was performed on a 7T microSigna scanner (Varian/GE Healthcare, Palo Alto, CA, USA). Serial diffusion weighted imaging (DWI) was performed using repeated sequences of six or eight levels of diffusion gradient amplitudes corresponding to b-values varying from 0 to 1,500 second/mm2. Diffusion-sensitizing gradients were applied along the Superior—Inferior direction (long axis of the brain). Apparent diffusion coefficient (ADC) maps were generated from the diffusion-weighted images using a voxel-by-voxel fitting routine as described earlier (Yenari et al, 1996). Relative ADC decreases were measured by copying the ROI delineated from the DWI scans to the fitted ADC map. The ADC was measured within this ROI. The ROI was then reflected onto the opposite hemisphere and ADC was measured in this comparable non-ischemic region. High-resolution MRI was obtained from paraformaldehyde-fixed brains immersed in fomblin (Specialty Fluids Co, Valencia, CA, USA). Brains were placed in a custom-built solenoidal radiofrequency coil and imaged using a three-dimensional FIESTA (Heyn et al, 2006) sequence (repetition time/echo time = 7.8/3.9 ms; resolution = 100 μm isotropic voxels; number of excitations = 1). For co-registration of both modalities we used Image J, whereas the DWI was set at 50% opacity and based on anatomical landmarks was superimposed onto the corresponding FIESTA image.
Histology
Twenty-four hours after IA delivery, animals were transcardially perfused with PBS followed by 4% paraformaldehyde before being prepared for immunochemistry. Standard immunohistochemical staining techniques were used on free-floating sections. Sections or cells were incubated in blocking solution (PBS containing 3% serum) for 1 hour at room temperature. Sections were incubated overnight at 4°C in primary antibodies diluted in PBS containing 1% serum. The following antibody was used: Iba-1 (Wako, Osaka, Japan). After several washes in PBS/1% serum, sections were incubated with secondary antibodies overnight. Either a biotinylated secondary antibody coupled with streptavidin-horseradish peroxidase and reacted with 3,3'-diaminobenzidine (DAB) or a fluorescence-labeled secondary antibody was used.
For the Fluoro-Jade C staining, 40-μm-section slides were immersed in 1% sodium hydroxide and 80% ethanol for 5 minutes and were rinsed for 2 minutes in 70% ethanol, 2 minutes in distilled water and then incubated in 0.06% potassium permanganate solution for 20 minutes. Slides were then transferred into a 0.0001% solution of Fluoro-Jade C (Histo-Chem Inc., Jefferson, AR, USA) dissolved in 0.1% acetic acid. This was followed by three 1-minute rinses of distilled water. Dried slides were cleared in xylene for 1 minute and coverslipped with a xylene-based mounting media (Richard Allan Scientific, Kalamazoo, MI, USA). Sections were analyzed on light microscopy or on a laser scanning confocal microscope (LSM5; Zeiss, Thornwood, NY, USA).
2,3,5-Triphenyl Tetrazolium Chloride Staining
Immediately after killing animals, 2 mm slices of the brain were immersed in 2% 2,3,5-triphenyl tetrazolium chloride solution for 15 minutes at room temperature. The slices were then photographed and the stroke area measured using image J. The total stroke volume was expressed in percent of the volume of the intact hemisphere.
Cell Quantification
All quantification was performed in non-stroked controls (sham controls, see Table 1) to analyze the effect of potential microvascular obstruction isolated from changes observed in stroked animals. All cell counts were performed using Image J (NIH) image analysis software (Bethesda, MD, USA). The degenerating neurons were quantified on Fluoro-Jade C-stained sections on three consecutive sections per animal for all groups (n = 3 for each group). Sections at the level of the striatum were chosen for quantification. The number of Fluoro-Jade-positive cells in the hemisphere ipsilateral to the cell injection was counted on slides blinded for the treatment group. Fluoro-Jade C-positive cells and Iba-1-positive cells were counted in predefined ROI from the dorsal, lateral, and ventral cortex using Image J.
Statistical Analyses
GraphPad Prism 5 software (San Diego, CA, USA) was used for statistical analyses. For comparisons between two or more groups, analysis of variance followed by Bonferroni's test for multiple comparisons between treatment groups was conducted. The data passed the normality test using Kolmogorof—Smirnoff test. For comparisons between two groups, Student's t-test was performed. Fisher's exact test was used for categorical variables.
Results
Cerebral Blood Flow During Intra-Arterial Cell Transplantation
Laser Doppler measurements showed that CBF remained at baseline during microneedle IA cell injection (Figure 2A). The same was true for the microneedle control groups (no stroke + cells and stroke + saline, Figure 2C). In contrast, injecting cells with the catheter method resulted in a 65% decrease in CBF during the first minute of cell injection and partially recovered to 60% to 70% of baseline after 15 minutes (Figure 2A). Both the initial drop and the overall CBF values in the catheter method were significantly decreased compared with the microneedle method (initial drop, P < 0.05; overall difference P < 0.0001; Figure 2A). The mortality in the catheter method was 35% at 24 hours, whereas no death was recorded using the microneedle technique. To determine whether the cells or the injection method caused the reduction in CBF, non-stroked animals were injected with cells (sham group) and stroked animals were injected with saline (control group). Only the CBF in saline-infused animals recovered to baseline (Figure 2B, P < 0.0001). Bioluminescence imaging confirmed that both the microneedle and catheter methods successfully delivered NPCs to the brain (Figure 3A). Photon flux was observed from cell-transplanted animals, but not saline controls, indicating luciferase reporter gene activity and thus the presence of NPCs.
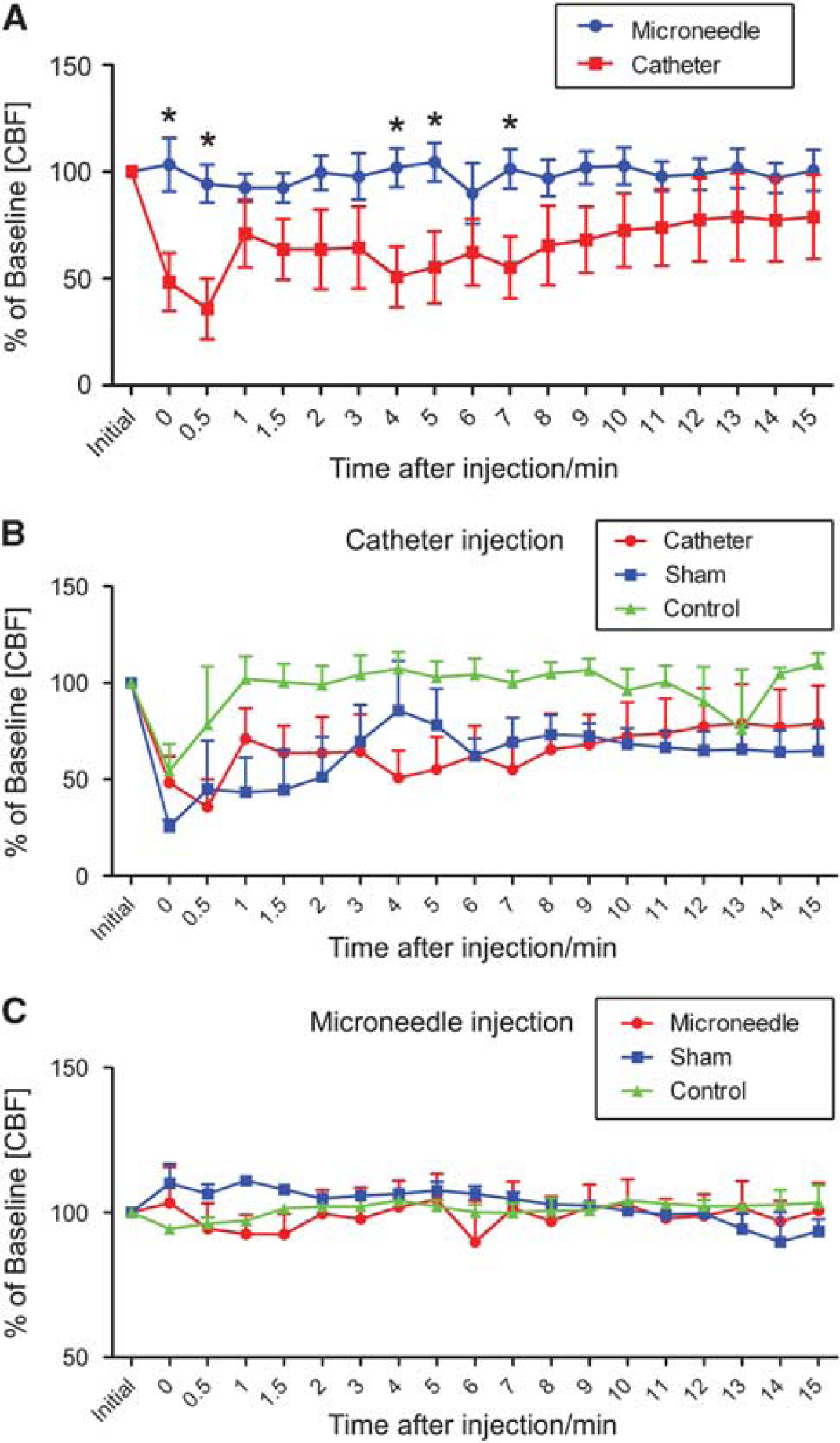
Laser flow Doppler analysis of cerebral blood flow (CBF) after intra-arterial stem cell injections. (
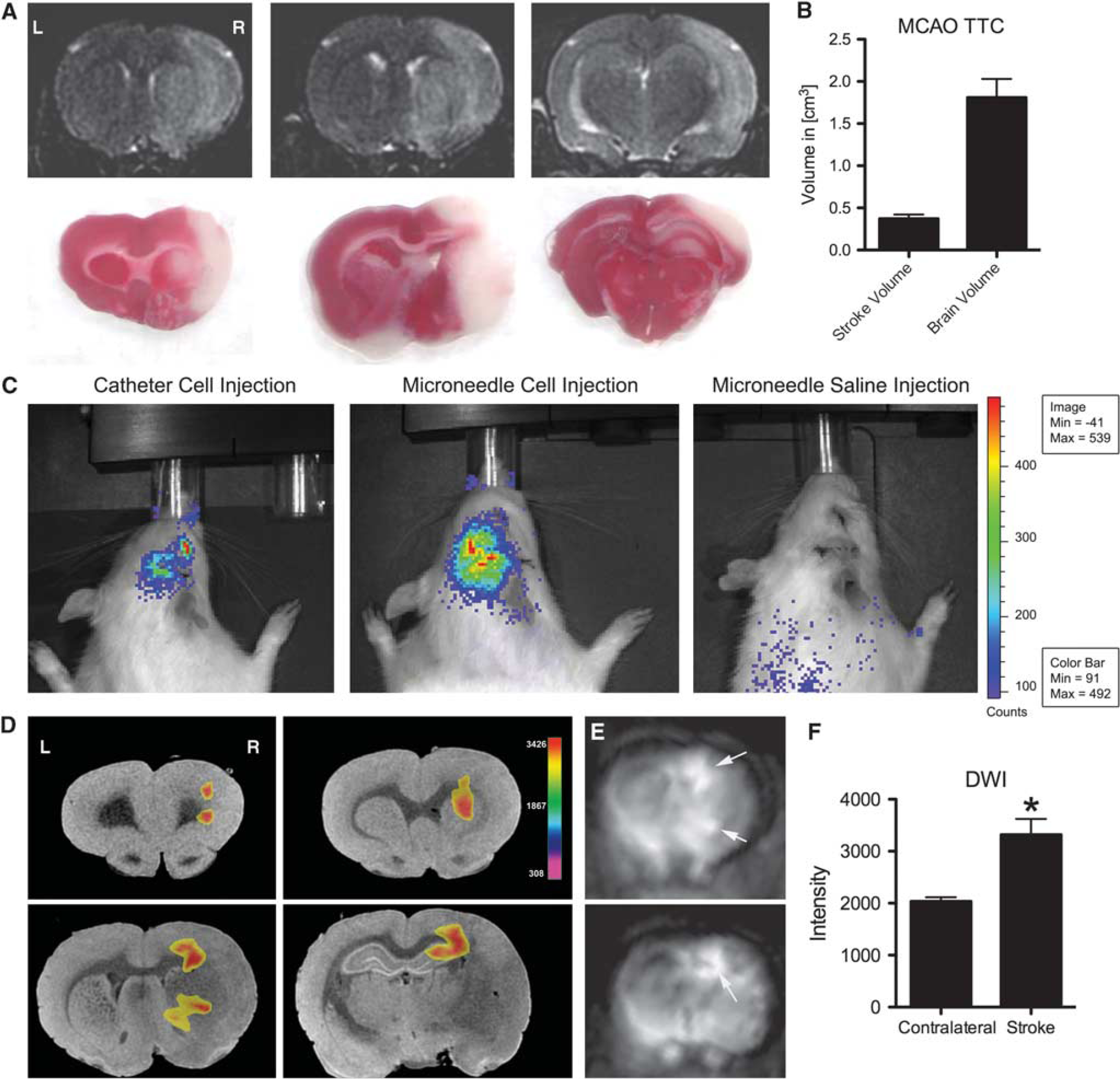
Magnetic resonance and bioluminescence imaging (BLI). (
Diffusion Weighted Imaging After Intra-Arterial Transplantation
We used DWI to further explore whether the decreased CBF gave rise to small ischemic events in experimental animals. Non-stroked cell-transplanted animals were used to isolate the injection method as the cause of microstrokes. Microneedle-injected groups did not show post-transplant diffusion-weighted changes in the ipsilateral cortex; however, catheter-injected animals showed DWI-positive regions in the ipsilateral cortex with an average percent reduction of ADC compared with contralateral normal brain area of 29%, indicating microstrokes (Figures 3D—F).
Neuronal Degeneration
To further explore the extent of damage resulting from decreased flow, we studied neuronal degeneration. Fluoro-Jade C staining was used to label degenerating neurons in cell-transplanted animals. Again, non-stroked animals were used to exclude neuronal damage from the experimental stroke model. The microneedle technique did not result in any appreciable staining of degenerating neurons above background (Figure 4B). In contrast, the catheter method gave rise to distinct pockets of dying neurons within the ipsilateral cortex (Figures 4C and 4D). There were significantly more Fluoro-Jade C-positive cells in the catheter group as compared with the microneedle group (P < 0.0001; Figure 4E).
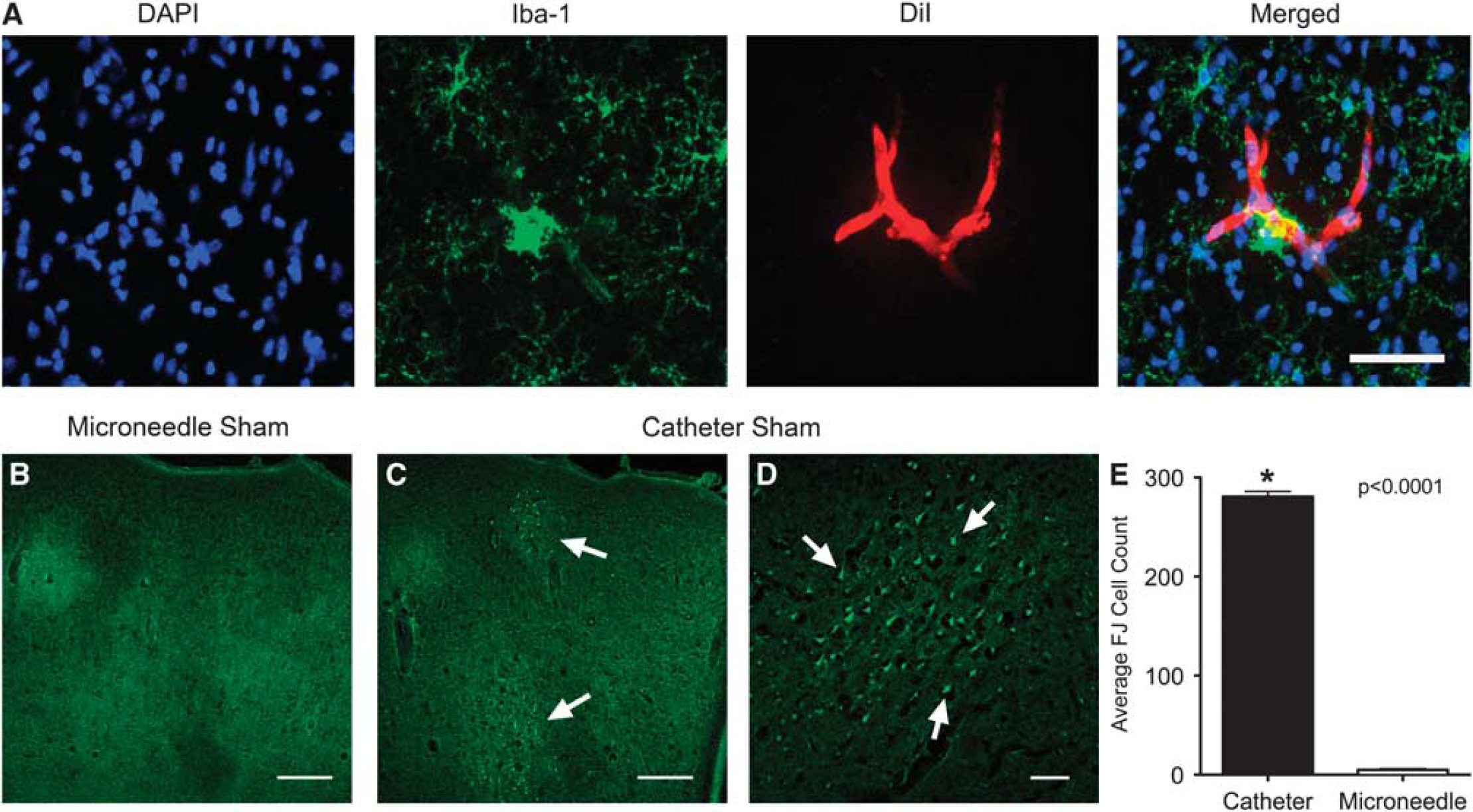
Histological analysis for neuronal degeneration in non-stroked (Sham) animals. (
Inflammation Caused by Microstrokes
To explore differences in post-transplant inflammation as an additional metric of ischemia between the microneedle and catheter groups, sections from non-stroked cell-transplanted animals were stained for Iba-1 immunoreactive microglia (Figures 5A—F). Animals injected using the catheter method had a significantly higher density of Iba-1-positive cells as compared with animals injected using the microneedle method (P < 0.05; Figure 5G).
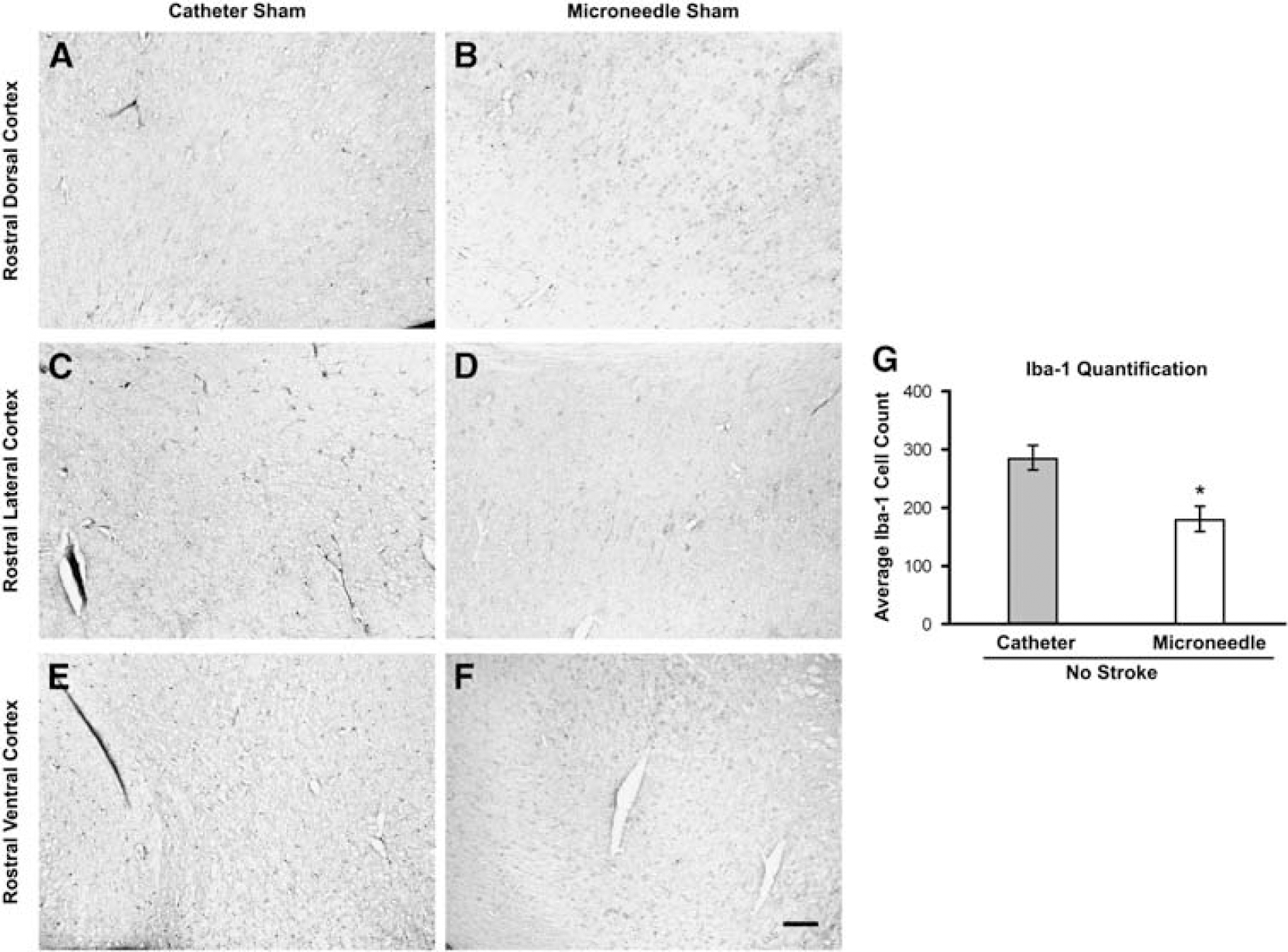
(
Discussion
Intra-arterial transplantation is a promising method for targeted delivery of stem cells to the ischemic brain. The intravascular paradigm relies on the ability of stem cells to migrate toward an ischemic lesion in a targeted manner, mediated by endogenous chemoattractant and adhesion molecules (Guzman et al, 2008; Guzman, 2009). Unlike intravenous infusion, IA transplantation results in less entrapment of cells in filtering organs and has the capacity for higher engraftment of cells in the brain. While several groups have showed the efficiency of IA delivery (Chung et al, 2009; Harting et al, 2009; Li et al, 2010; Pendharkar et al, 2010; Walczak et al, 2008), recent concerns have been raised based on CBF measurements that IA delivery may result in the development of microstrokes from the physical obstruction of capillaries by the cells (Walczak et al, 2008). We hypothesized that these observations were due to a transplantation method where the CCA flow is interrupted during cell injection and that this would favor microvascular obstruction by the injected cells (Figure 1A). Hence, in this study, we examined IA transplantation using an alternative method using a microneedle with preserved flow within the carotid artery during cell injection (Figure 1B). We found that using a microneedle injection technique with preserved flow in the carotid artery did not result in any significant decrease in CBF throughout the transplantation process (Figures 2A and 2C), whereas the catheter-based method yielded an initial decrease in CBF without recovery to baseline after cell injection (Figures 2A and 2B). This finding, for the catheter method, is similar to another study, which reported cellular occlusion of the microvasculature, decrease in CBF and subsequent microstrokes (Walczak et al, 2008). We found that saline-only controls using the catheter method produced a significant initial decrease, but recovered to the original baseline (Figure 2B). While groups injected with cells by the catheter method had the same initial CBF decrease, they only recovered to 60% to 70% of baseline (Figure 2B). This suggests that the initial decrease in CBF was an artifact of the large volume of injected saline. The failure of CBF to recover to baseline, however, was the result of cellular occlusion of the microvasculature during transplantation, which caused an impedance to flow. Cerebral blood flow is measured using laser Doppler, which relies on beam reflection from moving blood cells (O'Doherty et al, 2009). As a large volume of saline is infused, the dilution of blood cells in the vasculature may falsely present as a reduction in CBF.
Using our luciferase expressing NPCs and BLI, we ruled out that a difference in cell delivery efficiency between the two methods caused the difference in CBF changes observed during and after injection. We observed luciferase activity, and thus transplanted cells in animals in both the microneedle and the catheter group (Figure 3C). This finding suggests that both IA methods resulted in successful cell transplant, but only the catheter method caused significant CBF changes during injection.
We then explored if the reduction of CBF was sufficient to result in microstrokes. To control for ischemic effects from the initial stroke, we performed the IA procedure on animals, which did not undergo MCA occlusion to elucidate effects from cell-induced microstrokes. Using DWI, we observed DWI bright and ADC low regions in the ipsilateral hemispheres in animals injected using the catheter method (Figures 3D—F). These ischemic changes were not seen in the microneedle-injected animals, and suggest that the catheter-based cell injection method causes CBF changes significant enough to result in microstrokes. A similar finding was observed in a clinical study using intraarterial mesenchymal stem cell transplantation for multisystem atrophy (Lee et al, 2008). The authors observed clinically silent DWI-positive lesions in 7 out of 11 patients transplanted with 4 × 107 cells. It is however not excluded that some of those changes could have been related to the angiography itself.
Using Fluoro-Jade C staining, a marker of degenerating neurons (Schmued et al, 2005), we further examined non-stroked cell-injected animals for regions of damage. Fluoro-Jade C immunoreactive degenerating neurons were observed in distinct pockets throughout the ipsilateral cortex of animals injected using the catheter method, but not those injected using the microneedle method (Figures 4B—E). This regional clustering of Fluoro-Jade C-positive cells was consistent with the hypothesis that transplanted NPC leads to capillary obstruction and subsequent local ischemic changes. This was corroborated with a significantly stronger inflammatory reaction (number of Iba-1 immunoreactive monocytic cells) in the ipsilateral cortex in the catheter method as compared with the microneedle method.
Our results indicate that IA delivery of cells, depending on the method, does not decrease CBF and cause microstrokes. We find that decreased CBF and microstrokes are not caused by the intraarterial cell injection per se but are caused by a combination of flow arrest during the catheter insertion followed by microvascular obstruction by injected cells. In contrast, the microneedle method uses the physiologic high pressure CCA flow to carry injected cells to the ischemic region and allow engraftment of cells into the parenchyma without significant blockage of vasculature. An additional explanation is the significant dilution of the single-cell suspension, as it is introduced slowly to the high rate of blood flow in the CCA. Two previously published studies have investigated the safety of IA cell delivery to the ischemic brain (Li et al, 2010; Walczak et al, 2008). Both studies used a catheter-based injection method, one obstructing the CCA (Walczak et al, 2008), which directly compares with our catheter method, and the other reducing flow at the level of the middle cerebral artery (Li et al, 2010; Li et al, 2001). These studies reported a procedural mortality of 41% (Li et al, 2001) and 67% (Walczak et al, 2008), while the mortality in our catheter group was 35%. Two factors could account for these differences. First, the timing of injection was 30 minutes after stroke in the study by Walczak et al (2008), whereas ours and Li et al (2001) used a 24 hours time point for injection. The longer recovery time after stroke could lead to a reduction in procedural mortality. Second, both studies used a 2 hours MCA occlusion time resulting in a larger stroke size (30% to 50% of hemisphere at 4 days after stroke; Li et al, 2001), as compared with our 1.5 hour occlusion time with an average stroke size of 20.4% of the hemisphere. This could also account for the slightly decreased mortality rate found in our catheter group. In contrast, we found no procedural mortality in our group injected using the microneedle technique. Hence, we hypothesize that reduction of CBF caused by the obstructing indwelling catheter in the CCA together with cell-induced microembolic strokes explains the reported increased procedural mortality. In the hypothetical clinical application, microcatheters do not obstruct intracranial vessels and it should be possible to safely inject cells that would reach the perfused penumbral area surrounding the core of the stroke. In situations with poor collateralization, partial, or no reperfusion, our hypothesis would be that cells would reach areas that are still perfused and then migrate into the penumbral area. If blood flow is carrying the stem cells to the border zone of a stroke, no cell-induced reduction in CBF should result and no cell-induced microstroke would be expected.
Conclusion
Intra-arterial delivery is a promising clinically translatable and minimally invasive transplant paradigm for cell-based stroke therapies. Although previously described to carry the risk of inducing further damage caused by decreased CBF and inflammation, our data suggest that IA cell delivery with maintained blood flow can be used to successfully deliver cells into the cerebral vasculature without causing ischemic changes, thereby allowing transplanted cells to undergo targeted migration to the damaged brain. Our results apply to the IA injection of NPCs, and similar studies using mesenchymal stem cells, which are larger (Lee et al, 2008) and potentially more prone to form cell aggregates, will be needed. Further studies on long-term safety, effect on the host microenvironment, and functional recovery after experimental IA stem cell delivery need to be conducted.
Footnotes
Acknowledgements
The authors thank Evan Snyder, The Burnham Institute, for the donation of the C17.2 neural stem cell line and Elizabeth Hoyte for preparation of the illustrations.
The authors declare no conflict of interest.
