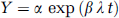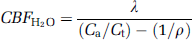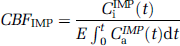Abstract
An 15O-labeled water (H215O) steady-state method for quantitative measurement of cerebral blood flow (CBF), which is less stressful to small animals with a few point blood sampling, was developed. After a simulation using a dose meter to achieve stable H215O radioactivity in the blood with a multiprogramming syringe pump programmed for slowly increasing injection volume, 10 rats were studied with the injection method. Arterial blood was sampled every minute during 6-minute positron emission tomography (PET) scans. After the PET scan, N-isopropyl-p-[125I]-iodoamphetamine (125I-IMP) was injected into the same rat to measure CBF using the autoradiography method based on a microsphere model. Regions of interest were placed on the whole brain in H215O-PET and 125I-IMP-autoradiography images, and CBF values calculated from both methods were compared. Radioactivity in the dose meter achieved equilibrium ~1 minute after starting the H215O injection. In rat studies, radioactivity in the blood and brain rapidly achieved equilibrium at 2 minutes after administration. The correlation of CBF values of H215O PET (49.2 ± 5.4 mL per 100 g per minute) and those of 125I-IMP autoradiography (49.1 ± 5.2 mL per 100 g per minute) was excellent (y = 1.01 x −0.37, r2 = 0.97). The H215O steady-state method with a continuously increasing injection is useful for CBF measurement in small animal studies, especially when multiple scans are required in the same animal.
Introduction
Small animal models for cerebrovascular diseases have recently been studied using 15O positron emission tomography (PET) (Temma et al, 2006). 15O-labeled water (H215O) and 15O-gas PET for measurement of cerebral blood flow (CBF) and oxygen metabolism usually requires multipoint arterial blood sampling (Mintun et al, 1984). However, the number of blood samplings is limited due to the small blood volume in animals, especially when multiple scans are required in the same animal. To avoid the stress caused by blood sampling, several techniques have been proposed to obtain an input function without arterial blood sampling in small animal PET studies using 18F-FDG (18F-fluorodeoxyglucose) or 11C-acetate (Laforest et al, 2005; Wu et al, 1996; Kim et al, 2006). However, the region of interest-based method with dynamic PET data was less accurate because of a partial volume effect and other errors and estimates (Laforest et al, 2005). Although a factor analysis technique successfully extracted the time-activity curve of blood from dynamic PET images in mice and rats (Wu et al, 1996; Kim et al, 2006), these techniques depended on the spatial resolution of PET scanners to obtain clear cardiac ventricle images. Because of high radioactivity in the lung, it is difficult to apply these methods to 15O-gas PET studies.
The steady-state method with 15O-labeled carbon dioxide (C15O2) and 15 O-labeled oxygen (15O2) was developed to calculate the cerebral hemodynamic state with a few arterial blood samplings as a simple and practical method in clinical studies (Frackowiak et al, 1980; Lammertsma et al, 1983). Jones et al (1985) applied the steady-state method to H215O PET with continuous intravenous tracer infusion to measure CBF in a clinical PET study because arterial radioactivity during C15O2 inhalation is unstable due to the condition of subject's breathing. However, the steady-state H215O method has not been applied to animal studies because the total volume of tracer injected is substantial even though the injection speed is very slow.
The purpose of this study was to develop a new steady-state method with a slowly increasing H215O injection using a multiprogramming syringe pump to measure CBF in rats. The CBF values obtained from the H215O steady-state method (CBFH2O) were compared with CBF measured by autoradiography using the N-isopropyl-p-[125I]-iodoamphetamine (125I-IMP) microsphere method (CBFIMP) to evaluate the accuracy of the PET measurement of CBF in rats.
Materials and methods
Preparation of H215O
15O2 gas was produced by a 15N (p, n) 15O nuclear reaction with a 2.5% O2/N2 gas target at an proton current accelerated for ~10 minutes using an in-house cyclotron Eclipse HP/RD (Siemens, Knoxville, TN, USA). The H215O was synthesized by reduction of 15O2 with H2 gas. The synthesized H215O gas was trapped in a saline solution and then passed though a 0.22-μm millipore filter before experimental use.
Evaluation of the Program for the H215O Injection
The program for a multiprogramming syringe pump (FP-2000, Melquest, Toyama, Japan) was designed to combine the bolus and slowly increasing (B/SI) the H215O injection by using a PE-50 tube (i.d. 0.5 mm, o.d. 0.9 mm, length 60 cm) as a venous line. In this method, the injection speed was controlled rapidly for the first 5 seconds to fill the dead volume of the venous line with a small amount of overshoot, the rate was changed moderately for 40 seconds to increase blood radioactivity, followed by a slow but gradually increased administration rate to compensate for the decay of blood 15O radioactivity (Figure 1A). The rapid injection rates in the initial two phases were programmed to accelerate equilibrium of radioactivity in the rat's body. To compensate for the decay of blood radioactivity in the slowly increasing injection velocity, the injection rate was continuously changed under the assumption of an inverse decay function as expressed by the following equation:
where Y (μL/min) is the injection velocity for the syringe pump, Λ (/min) is the decay constant of 15O, and t (minute) is time after H215O injection. α (μL/min) and β (dimensionless) are the constants for this injection method. The velocity gradually increases according to reciprocal function of the decay of 15O in the body, and the initial velocity of α is estimated from the radioactivity concentration at the end of phase 2. To determine the two constants in Eq. (1) for the steady-state condition, radioactivity in the rat's body was simulated by collecting the tracer into an empty vial, which was connected to the injection system with prefilled H215O. The vial was put in a dose meter (Capintec Inc., Ramsy, NJ, USA) and total radioactivity without decay correction was measured as a function of time (Figure 1B).
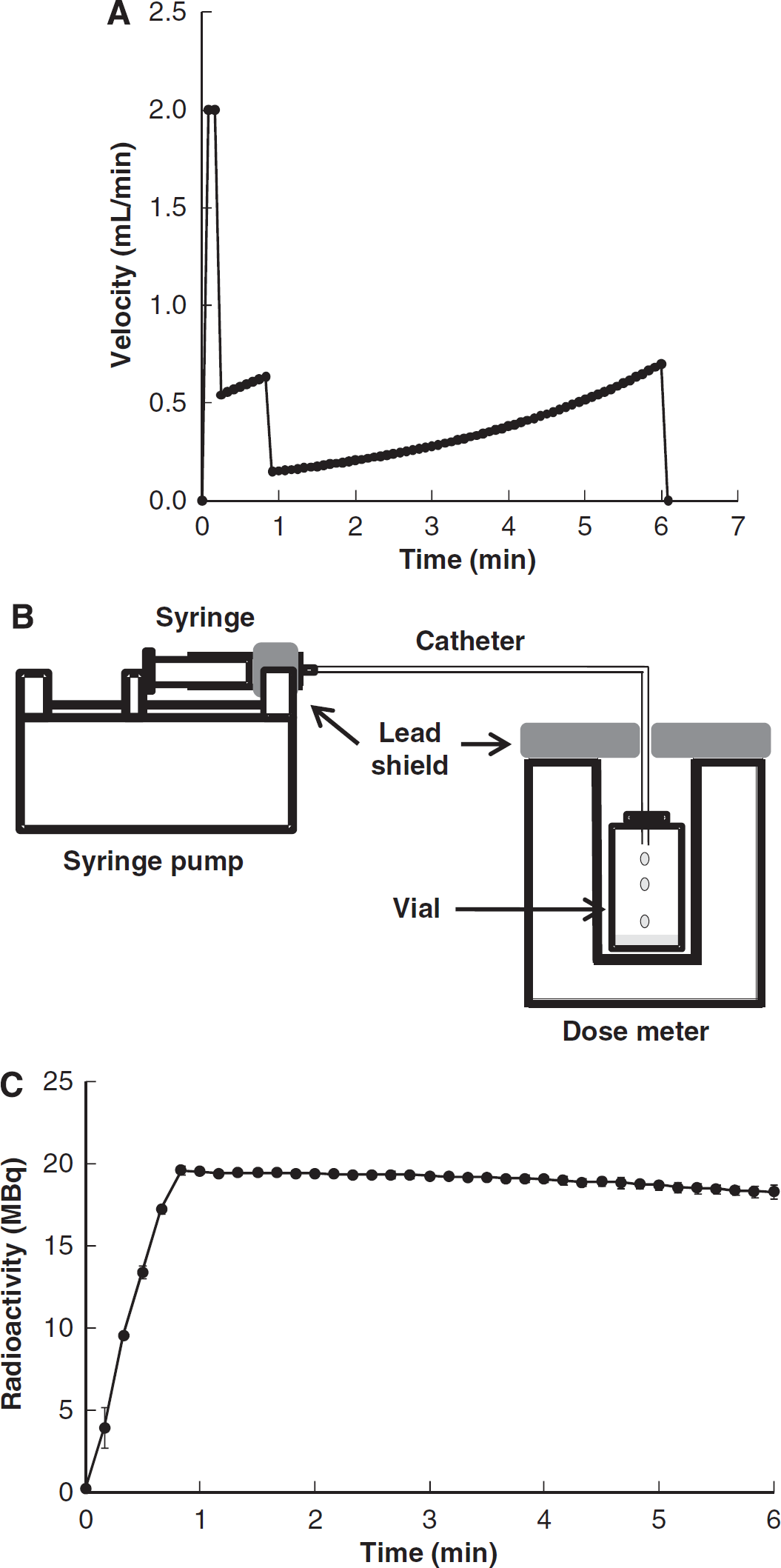
Program of the bolus and slowly increasing (B/SI) injection method and 15O-labeled water (H215O) radioactivity measured with a dose meter. (
Animal Positron Emission Tomography and Autoradiography
Animal studies were approved by the Animal Care Committee at the University of Fukui and conducted in accordance with the international standards for animal welfare and institutional guidelines. Adequate measures were taken to minimize pain or discomfort. Male Sprague–Dawley rats from Japan SLC Inc. (Hamamatsu, Japan) were housed for 1 week under a 12-hour light/12-hour dark cycle with free access to food and water. The rats were fasted with no food overnight with water supplied ad libitum.
Ten rats (303.0 ± 17.0 g) were anesthetized with intraperitoneal injection of chloral hydrate (0.4 mg/g body weight, intraperitoneally). A PE-50 catheter was inserted into the femoral artery for blood sampling and the femoral vein for H215O administration. The PET studies were performed using a small animal PET scanner (SHR-41000, Hamamatsu Photonics, Hamamatsu, Japan) (Yamada et al, 2008). The scanner acquires 213 slices covering an axial length of 160 mm, with a three-dimension mode and achieving a resolution of ~2.0 mm full width at half maximum in the transaxial direction and 2.8 mm full width at half maximum in the axial direction. The rats were placed in a supine position on the scanner bed, and the limbs were fixed using surgical tape. The orientation of the cranial position was determined using a laser beam on the scanner. Before emission scans, a transmission scan was performed for attenuation correction using a 68Ge/68Ga external source. A measure of 9 mL of ~555 MBq H215O was filled in a 10-mL syringe and set up on the syringe pump.
A 6-minute list-mode PET scan was started with intravenous administration of H215O using a syringe pump with a B/SI injection program. During the PET scan, rats were heated using an electric lamp to maintain the body temperature. Approximately 50 μL of arterial blood was sampled every 1 minute, and radioactivity in the blood samples was immediately measured with a well scintillation counter (ARC380, Aloka, Tokyo, Japan), and radioactivity concentration of each sampled blood was corrected for the decay from the sampling time. Fifteen minutes after the H215O-PET scan, 125I-IMP (1.11 MBq) was injected into the same rat and arterial blood was withdrawn for 10 minutes after tracer administration with a constant rate of 100 μL/min using the same syringe pump. To obtain the area under the curve of the true input function, collected blood was put into 5 mL of octanole and a lipophilic fraction was extracted (Kuhl et al, 1982). Rats were killed using a guillotine at 10 minutes after the tracer injection. The brains were immediately removed, frozen in isopentane, and sliced into 30 μm thickness slices in a cryostat at approximately −20°C. The brain slices were mounted on glass slides and exposed to an imaging plate with 125I standards for 24 hours to obtain autoradiography. Exposed imaging plates were scanned by a fluoro-image analyzer (FLA-7000, Fuji Film, Tokyo, Japan). Body temperature was monitored during the experiment using animal body temperature control (ATC-1000, World Precision Instruments, Sarasota, FL, USA). Arterial blood gas and other physiological parameters were measured before the H215O and 125I-IMP injection using a blood gas analyzer (ABL555, Radiometer, Copenhagen, Denmark) and a hemooximeter (OSM3, Radiometer, Copenhagen, Denmark).
Cerebral Blood Flow Calculation
The H215O-PET images were reconstructed using the Fourier rebinning-filtered back projection method with attenuation correction, and calibrated by the cross-calibration factor. The CBFH2O using the steady-state method was calculated from PET data and arterial blood radioactivity concentration using the following equation:
where Λ (/min) is the decay constant of 15O, Ca (Bq/mL) is the mean arterial H215O concentration, Ct (Bq/mL) is the average brain radioactivity concentration for 2 minutes calculated from 3 to 5 minutes dynamic data without decay correction. ρ is a brain–blood partition coefficient for water, which was fixed as 0.91 mL/g (Herscovitch and Raichle, 1985).
In 125I-IMP-autoradiography studies, CBFIMP using the microsphere method was calculated according to the following equation:
where CiIMP(t) (Bq/mL) is the total 125I-IMP radioactivity concentration in the brain on autoradiography images and E is the first-pass extraction fraction. CaIMP (t) (Bq/mL) is the input function obtained from the arterial radioactivity concentration of 125I-IMP and its area under the curve was obtained using the radioactivity concentration of the octanol extraction as mentioned above. The counts for CiIMP (t) were calibrated by using agar standard including measured 125I radioactivity, which was exposed simultaneously on the imaging plate. For E of 125I-IMP, 0.92 was used from a previous report (Di-Rocco et al, 1993).
Data Analysis
Magnetic resonance imaging (MRI) scans were taken under chloral hydrate anesthesia (0.4 mg/g body weight, intraperitoneally) using a 3.0-Tesla MR scanner (Signa Horizon, GE Medical Systems, Milwaukee, WI, USA) before the PET scans. The rats were placed and fixed between a pair of surface coils during the MR scan. T2-weighted images of the rat's brain were acquired using a fast spin echo method (repetition time 5050 milliseconds, echo time 85 milliseconds, 256 × 256 matrix, slice thickness 1.0 mm, field of view 6.0 cm, phase-field of view 4.8 cm, number of excitations 3). The MRI and H215O images were coregistered using the Dr View software (AJS, Tokyo, Japan). The MRI slices were used to draw region of interests. Three coronal slices of 125I-IMP-autoradiography images were used for drawing region of interests on the autoradiography at the following four slice levels of corresponding MRI and H215O-PET images, referring to the rat brain atlas: frontal cortex, sensorimotor cortex, visual cortex, striatum, and thalamus (see Figure 5). The region of interests were applied to H215O and 125I-IMP images to compare global and regional CBF values.
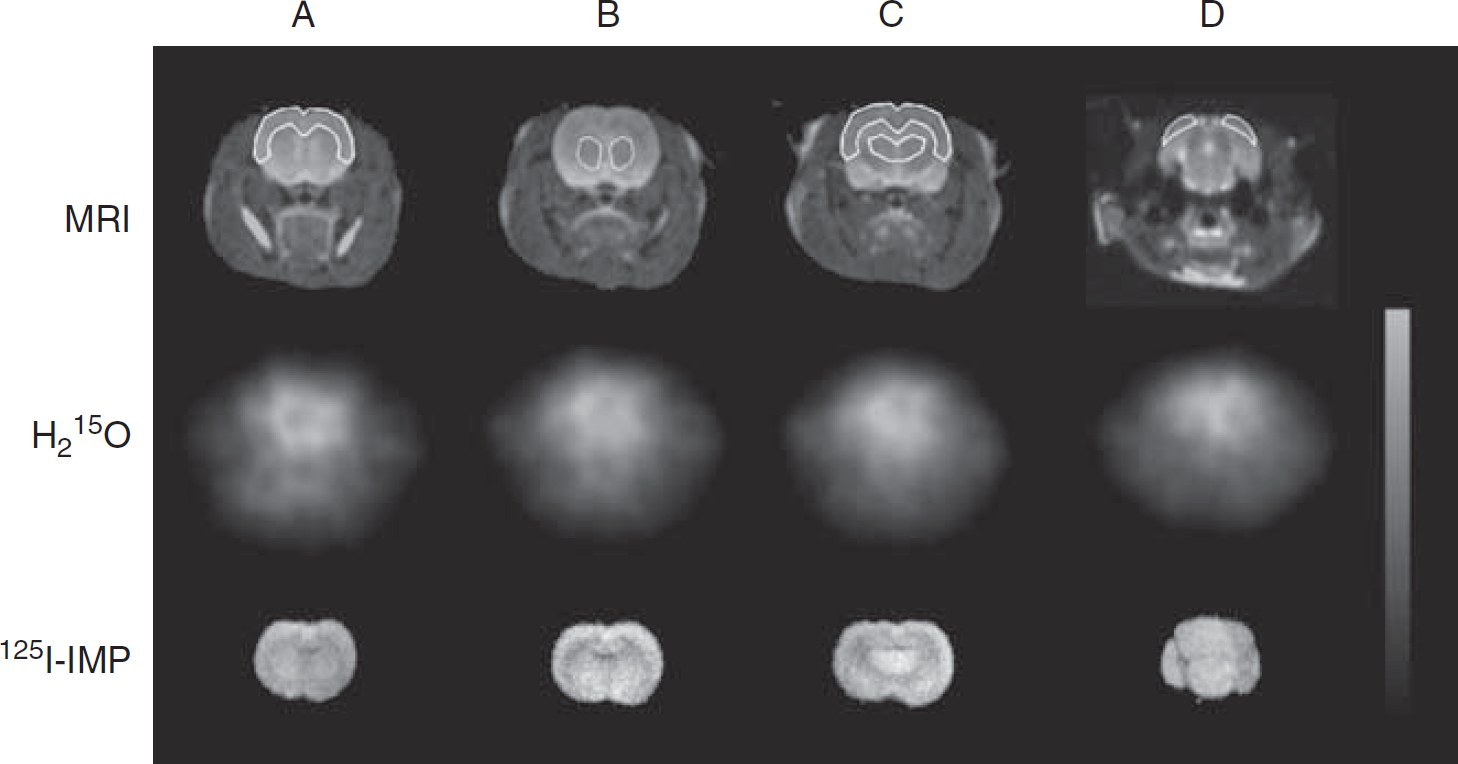
Magnetic resonance imaging (MRI), 15O-labeled water (H215O), and N-isopropyl-p-[125I]-iodoamphetamine (125I-IMP) images at the same slices. Region of interests were placed on the whole brain, the regions of frontal cortex (A), striatum (B), sensorimotor cortex (C), thalamus (C), and visual cortex (D) using MRI. The same region of interests was applied on the H215O and 125I-IMP images at the same slice levels.
The CBFH2O and CBFIMP values were compared using the Student's t-test. Blood gas data in the rats were compared for the two experiments using a paired t-test. A P value of < 0.05 was considered to be statistically significant.
Results
Radioactivity measured by a dose calibrator using the B/SI injection method is given in Figure 1C. The increasing injection velocity was controlled to achieve a steady state and the constants of α and β in Eq. (1) were determined to be α = 117 (μL/min) and β = 1.0 × 10−2 in our setting of the syringe and catheter. Total radioactivity reached a plateau ~1 minute after starting the injection, which was kept within a 5% difference. In application of the B/SI injection method, the H215O radioactivity concentration rapidly achieved equilibrium in the whole blood and brain ~2 minutes after H215O administration (Figure 2). The radioactivity after equilibrium in the rat body was ~20 MBq, which was below the saturation count of our small animal PET scanner.
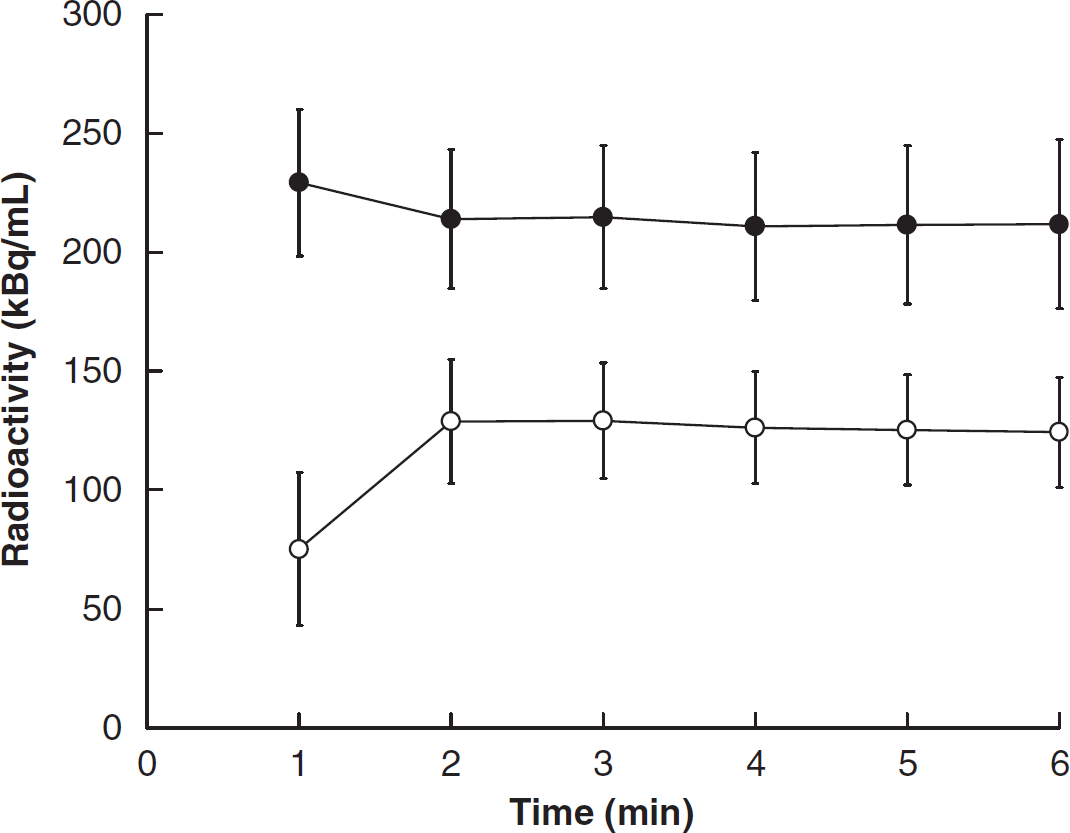
Time-activity curve of the whole blood and whole brain using the 15O-labeled water (H215O)-SS method with bolus and slowly increasing (B/SI) injection. Radioactivity of H215O achieved equilibrium in the whole blood (●) rapidly and the whole brain (○) at ~2 minutes after H215O injection.
Figure 3 shows representative H215O-PET images. The H215O accumulation in the brain at 2 to 4 minutes was little different from that at 3 to 5 minutes. The body temperature of all rats was kept between 32°C and 38°C, and the mean CBFH2O and CBFIMP values in the whole brain were 49.2 ± 5.4 and 49.1 ± 5.2 (mL per 100 g per minute), respectively. The CBFH2O values were well correlated with CBFIMP as given in Figure 4A (y = 1.01x–0.37, r2 = 0.97). Figure 4B shows changes in CBFH2O as a function of body temperature. The CBFH2O values were closely associated with body temperatures in rats. Table 1 shows mean values of arterial blood gas and other physiologic data in all rats measured before H215O and 125I-IMP injection. None of these data were significantly different between two phases (P > 0.05), and they were not associated with rat body temperature. Figure 5 shows the corresponding coronal slice images of MRI, H215O PET, and autoradiography of 125I-IMP. The CBFH2O in the regional cerebral areas is given in Table 2. The CBFH2O values were corrected into cCBFH2O using the regression line in Figure 4B to adjust CBFH2O from 32°C to 38°C to 37°C in terms of the rat's body temperature. Each cCBFH2O in the regional cerebral areas showed a small s.d. compared with CBFH2O.
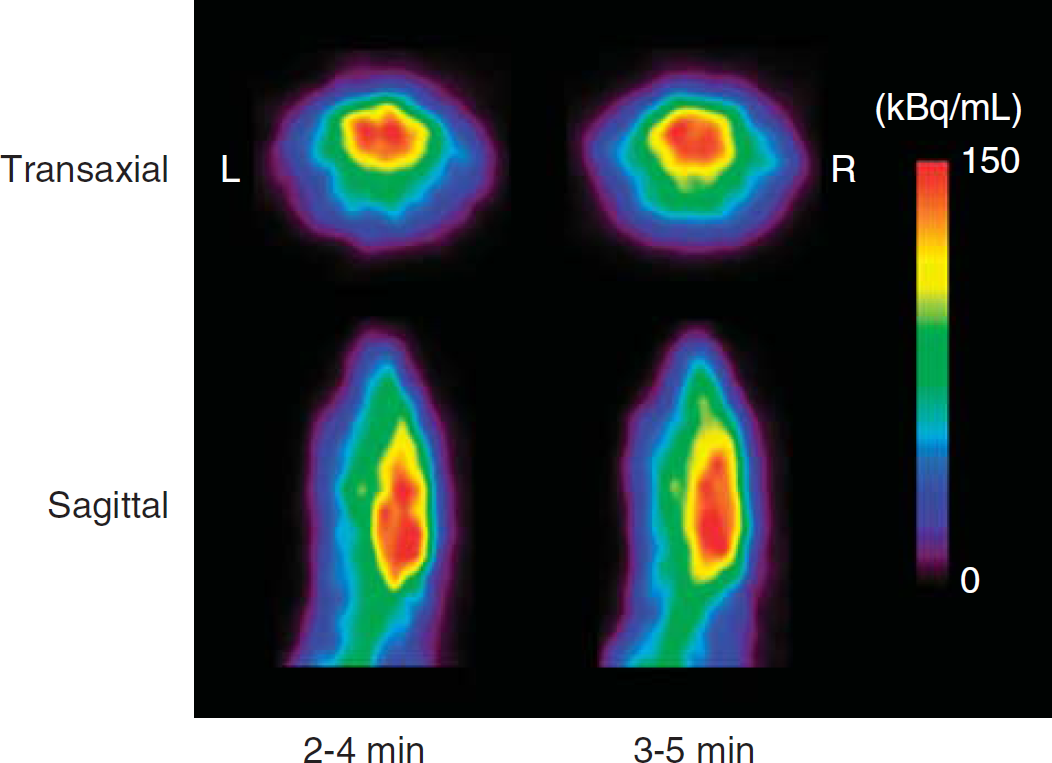
Comparison of 15O-labeled water (H215O)-positron emission tomography (PET) images in the transaxial and coronal direction at 2 to 3 minutes and 3 to 5 minutes. Cerebral accumulation in the H215O images at 2 to 3 minutes was little different from that at 5 to 6 minutes.
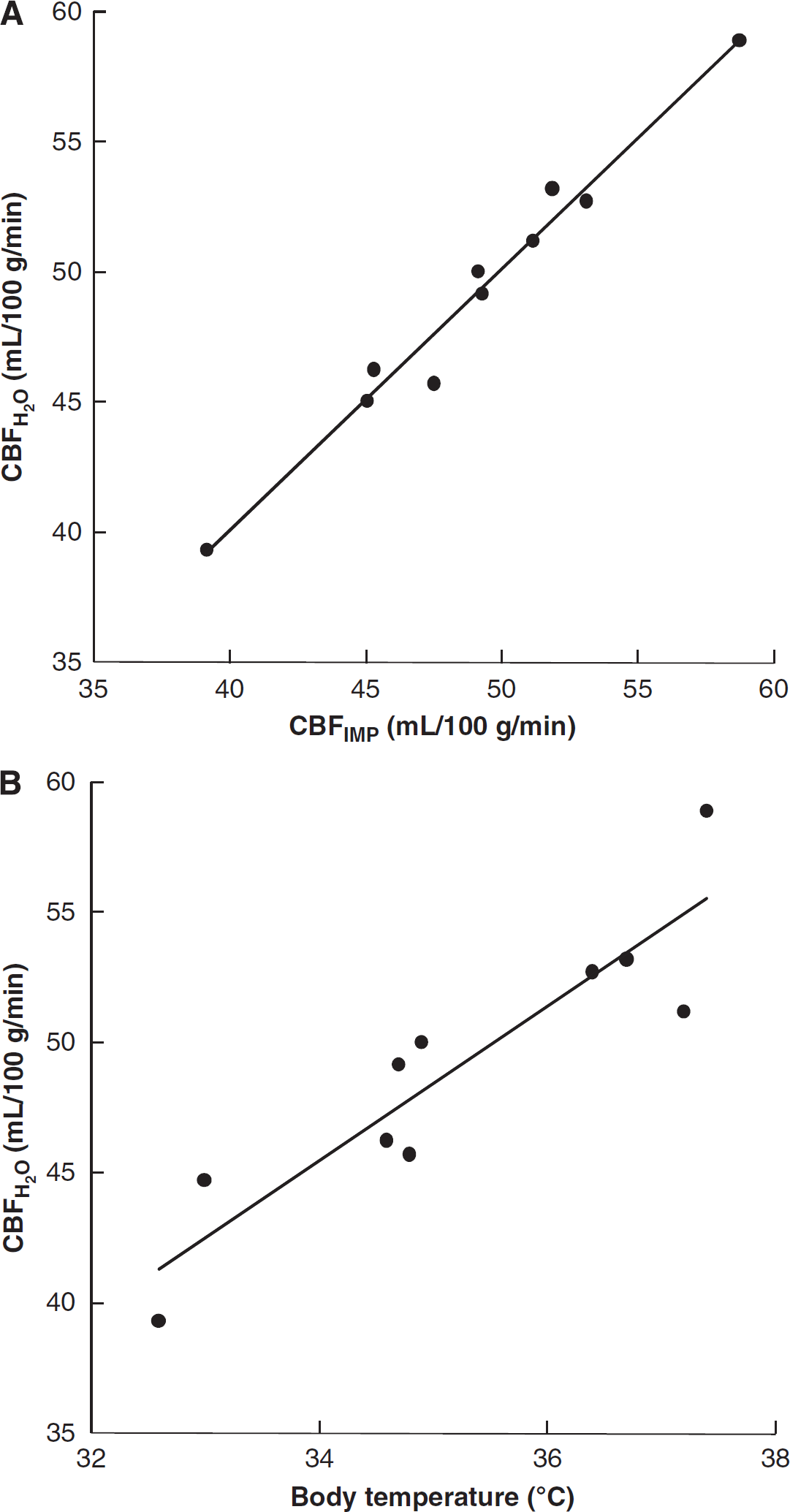
Correlation between the cerebral blood flow (CBF)H2O and CBFIMP values and relationship between CBFH2O and rat body temperature. (
Arterial blood gas data at H215O and 125I-IMP injection

Hb, hemoglobin; Hct, hematocrit; H215O, 15O-labeled water; O2 Sat, arterial oxygen saturation; PCO2, arterial carbon dioxide tension; PO2, arterial oxygen tension; 125I-IMP N-isopropyl-p-[125I]-iodoamphetamine.
NS, not significantly different between the two conditions.
CBFH2O and cCBFH2O (mL per 100 g per minute) in the regional cerebral areas
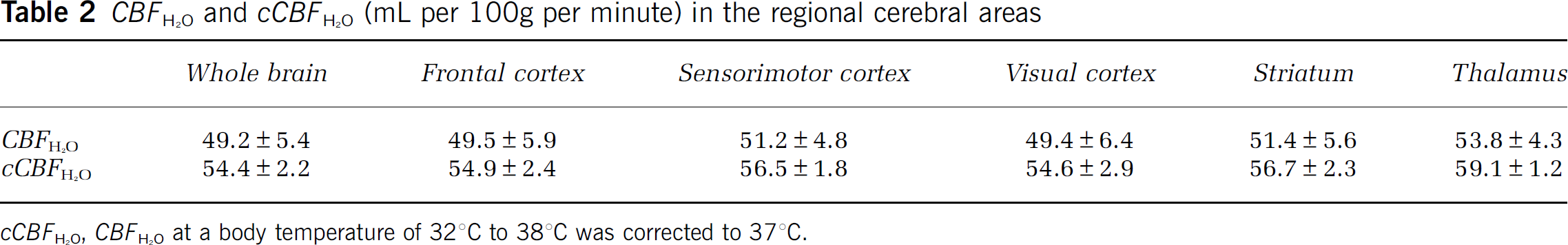
cCBFH2O, CBFH2O at a body temperature of 32°C to 38°C was corrected to 37°C.
Discussion
In this study, the H215O steady-state method was used to measure CBF in the rat brain and the quantitative values were evaluated by the consecutive CBF measurements using the 125I-IMP-autoradiographic method. The steady-state method is ideal for a small animal study because it requires only a few sampling points with a small sampling blood volume after radioactivity has achieved equilibrium, which is less invasive to animals. If C15O2 gas is applied as a tracer for CBF measurement, nasal radioactivity from the tracer gas affects the brain radioactivity count in small animal PET studies, and the brain radioactivity is also affected by respiratory conditions. We applied the B/SI injection of H215O for the steady-state method, and achieved rapid radioactive equilibrium in the brain and blood at 2 minutes after the H215O injection (Figure 2). The small injection volume of H215O of only a 1.5-mL net increase by subtracting the sampling volume from the injection volume during a 6-minute scan is not stressful for rats (Morton et al, 1997), and only slightly affects physical parameters.
The steady-state method for CBF measurement was developed in clinical 15O-gas studies with continuous administration of a tracer (Frackowiak et al, 1980; Lammertsma et al, 1983). Jones et al (1985) applied continuous H215O injection for the steady-state method. An approach with a multiprogramming syringe pump has been implemented in 11C or 18F-PET studies by using a bolus infusion followed by constant infusion to achieve a steady-state concentration curve in tissue and plasma (Carson et al, 1993, 1997, 2000). Since the half-life of O-15 is too short to achieve steady-state radioactivity in the brain with a small injection volume, these approaches have not been applied to small animal 15O-PET studies. In the B/SI injection method, the injection velocity of the first phase was set at 32.0 μL/sec to fill the dead volume (~118 μL) of the venous line tube (Figure 1A), followed by the moderate velocity phase to increase the rat's total body radioactivity to be 20 MBq. In the final phase, the velocity was gradually increased to maintain equilibrium by compensating the decay of blood 15O radioactivity with a reciprocal function of the exponential decay curve. The constants of a and b were obtained from a dose calibrator simulation (Figure 1B), because total radioactivity in the body, as well as blood radioactivity concentration, should be constant in the steady-state method. Achieving overall equilibrium of radioactivity in the rat's body is important to keep arterial input at a constant value. After the simulation, equilibrium of radioactivity in the rat's body was successfully achieved at ~2 minutes after the start of injection with a small injection volume.
The CBFH2O values from H215O PET were closely correlated with the CBFIMP values obtained by the autoradiographic method, which is considered to be the gold standard for CBF measurement because of fewer errors such as partial volume effects or other estimates. Several previous studies using different methods showed similar mean CBF values to our CBFH2O mean (Magata et al, 1995, 2003; Temma et al, 2006; Tiwari et al, 2010). The CBF values are well known to be affected by anesthesia, but the rat's body temperature also closely associated with CBFH2O values (Rosomoff and Holaday, 1954). Our results were also varying according to body temperature between 32°C and 38°C as shown in Figure 4B. Although the PCO2 is reported to be correlated with the body temperature (Hägerdal et al, 1975), PCO2 values in the present study were not significantly correlated, and thus, the correlation between CBFH2O and the body temperature was not caused by changes in PCO2. Lower body temperature may reduce brain function, which induces decrease in CBF. The s.d. of cCBFH2O, in which CBFH2O was corrected to be 37°C by using linear regression of body temperature, was smaller than that in previous studies (Table 2). This correction for body temperature would be a good method for precise CBF evaluation using model animals.
Another advantage of the H215O steady-state method in our study is a better image quality obtained from stable radioactivity in the brain averaged for 2 to 3 minutes from a 6-minute scan, as well as fewer blood samples showing fewer errors caused by handling and counting many sequential samples. Furthermore, handling of an injectable radiotracer is easier than radioactive gas. As the B/SI injection method increases the injection dose as a function of time, the largest volume is injected in the last minute. If the PET scanning time is minimized to 5 minutes from the start of H215O injection to use a 2-minute average image from a 3 to 5-minute dynamic scan, the method can reduce the injection volume, and the stress to rats could be minimized, allowing repeated CBF measurements in different conditions.
The bolus injection method usually requires multipoint blood sampling to obtain time–activity curves for the input function. Frequent multipoint blood sampling is also stressful for small animals, and radioactivity in the blood may include substantial errors because of busy handling of blood samples. However, the steady-state method requires fewer arterial samples of 3 to 4 points during the scan, which is less invasive for rats. As the total volume of arterial blood sampling must be limited to ~10% of the total blood volume (Scipioni et al, 1997), it should be < 2.0 mL for 300 g rats.
However, the steady-state method requires high-specific radioactivity of H215O (>62 MBq/mL), as well as a high-performance and wide-range multiprogramming syringe pump because decay of 15O in the blood should be compensated using a slowly increasing injection rate. The mechanical performance of the syringe pump used in this study achieved accuracy of ± 0.1% and a variable velocity of 0.001 to 150 mm/min. The program using high-specific radioactivity resulted in high reproducibility and accuracy. Eriksson et al (2008) developed a regulation system for the target tissue concentration of 11C-flumazenil using a computer-controlled infusion pump. However, the pump could not rapidly and continuously maintain a steady-state level radioactivity concentration in the whole brain. As a stable injection protocol is needed for stable measurement of CBF, H215O radioactivity at the initial setting of the syringe pump should be constant to avoid correction of the injection program for every experiment.
The steady-state method may induce systemic underestimation because of tissue heterogeneity between gray and white matter as compared with the bolus injection method (Herscovitch and Raichle, 1983; Herscovitch et al, 1983). This underestimation is usually observed in the regions of relatively greater CBF, especially under the conditions of hypercapnia or acetazolamide administration. However, our results showed regional values corresponding to CBFIMP in the normal stable condition, as well as those of previous studies, indicating that this is a reliable method for animal studies with model animals.
In conclusion, an H215O steady-state method for small animal PET was established using the B/SI injection method, which can provide precise and stable CBFH2O values. This method is useful for repeated measurement of CBF in small animals with lower stress because of the smaller injection volume with only a few blood samples. The CBFH2O values calculated from this method were well correlated with CBFIMP values from autoradiography. The s.d. of the cCBFH2O was also smaller than that in previous studies.
Footnotes
Acknowledgements
The authors thank the staff of the Biological Imaging Research Center, University of Fukui for their technical help.
The authors declare no conflict of interest.