Abstract
We attempted to assess regional differences in cerebral P-glycoprotein (P-gp) function by performing paired positron emission tomography (PET) scans with the P-gp substrate (
Introduction
The multidrug efflux transporter P-glycoprotein (P-gp, ABCB1), which is highly expressed in the luminal endothelium of the human blood–brain barrier (BBB), limits entry into the brain for a large number of endogenous and exogenous compounds (Löscher and Potschka, 2005). Changes in cerebral P-gp function and expression are thought to be implicated in several neurologic disorders, such as epilepsy, depression, and Parkinson's and Alzheimer's disease (Löscher and Potschka, 2005). Positron emission tomography (PET) with the radiolabeled P-gp substrate (
Verapamil is very effectively transported by P-gp in the BBB, thereby possessing low brain uptake, which affords low counting statistics and hampers the mapping of regional differences in cerebral P-gp function (Langer et al, 2007). Assessment of regional differences in cerebral P-gp function and expression is currently of high interest, as, for instance, regional overexpression of P-gp in epileptic brain tissue is thought to contribute to the phenomenon of drug resistance in epilepsy by impeding access of anti-epileptic drugs to their sites of action (Löscher and Potschka, 2005).
A promising strategy to overcome the limitation of low brain uptake of VPM is to perform VPM PET scans after P-gp modulation with P-gp inhibiting drugs, at doses that do not completely inhibit the P-gp pump. Two P-gp inhibitors have so far been used in human PET studies, cyclosporine A (Sasongko et al, 2005) and tariquidar (Wagner et al, 2009). The third-generation P-gp inhibitor tariquidar (Fox and Bates, 2007) is safer than cyclosporine A for use in human subjects and was shown to lack interactions with metabolism and plasma protein binding of VPM (Wagner et al, 2009). We have previously performed paired VPM PET scans in healthy volunteers, before and after intravenous administration of tariquidar at a dose of 2 mg/kg (Wagner et al, 2009). Tariquidar administration was shown to result in a mean 24% increase in VPM
In this study, we assessed regional differences in tariquidar-induced modulation of VPM brain uptake, which might be indicative of regional differences in cerebral P-gp function and expression.
Materials and methods
This study was performed at the Department of Clinical Pharmacology at the Medical University of Vienna. The study protocol was approved by the local Ethics Committee. All subjects were given a detailed description of the study and their written consent was obtained before enrolment in the study. Five healthy male subjects (mean age ± s.d., 32 ± 8) were included into the study. The study was conducted as a pilot study, so no
Each study participant underwent two 40-min dynamic VPM PET scans (mean injected dose: 384 ± 13 MBq) at an interval of 200 mins, and serial arterial blood sampling
Arterial plasma samples were analyzed for polar [11C]metabolites of VPM using an earlier described assay (Abrahim et al, 2008). An arterial input function was constructed by correcting total activity counts in arterial plasma for polar [11C]metabolites of VPM as described earlier (Langer et al, 2007).
For data analysis, both a voxel-based parametric imaging approach and a region of interest (ROI)-based approach were used. Parametric
Imaging data for ROI analysis were processed following previously published procedures (Langer et al, 2007). A maximum probability atlas based on 30 subjects and containing 83 regions (Hammers et al, 2003) was used to define 41 different brain ROIs, whereby regional data from the left and right hemispheres were averaged. In addition, the choroid plexus ROI was manually defined. A 1-tissue-2-rate-constant compartment model was used to estimate the influx and efflux rate constants of activity across the BBB. For estimation of
Individual regional
Limitations
In theory, regional brain uptake of VPM could become dependent on regional cerebral blood flow under conditions of P-gp modulation (Liow et al, 2009). These differences could bias regional differences in
Another limitation of the methodology used with regard to a possible application in epilepsy patients is the fact that the hippocampus, an important brain structure in epilepsy, could not be analyzed due to spill in of activity from the adjacent choroid plexus. However, this problem might at least partly be overcome by using higher-resolution PET cameras than in this study.
Results and discussion
Up to now only little is known about the regional distribution pattern of P-gp in the human brain. Most animal studies conducted to date have focused on describing expression levels of P-gp in epileptic brain tissue relative to nonepileptic brain tissue without comparing P-gp expression levels across different brain regions (Löscher and Potschka, 2005). In one recent study, regional brain uptake of the P-gp substrate radiotracer 11C-
In our study, we used a novel human brain imaging protocol that included the performance of paired brain PET scans with the validated P-gp probe VPM, before and after administration of a moderate dose (i.e., one that does not completely inhibit P-gp) of the third-generation P-gp inhibitor tariquidar (2 mg/kg body weight) (Wagner et al, 2009). The 2 mg/kg dose was chosen because this has been the maximum dose given to human subjects in earlier clinical studies (Fox and Bates, 2007). An earlier study had shown excellent (about 4% for
In rats, the half-maximum effect dose of tariquidar for increasing VPM brain
Statistical parametric mapping analysis revealed clusters that showed higher response to tariquidar treatment in terms of VPM
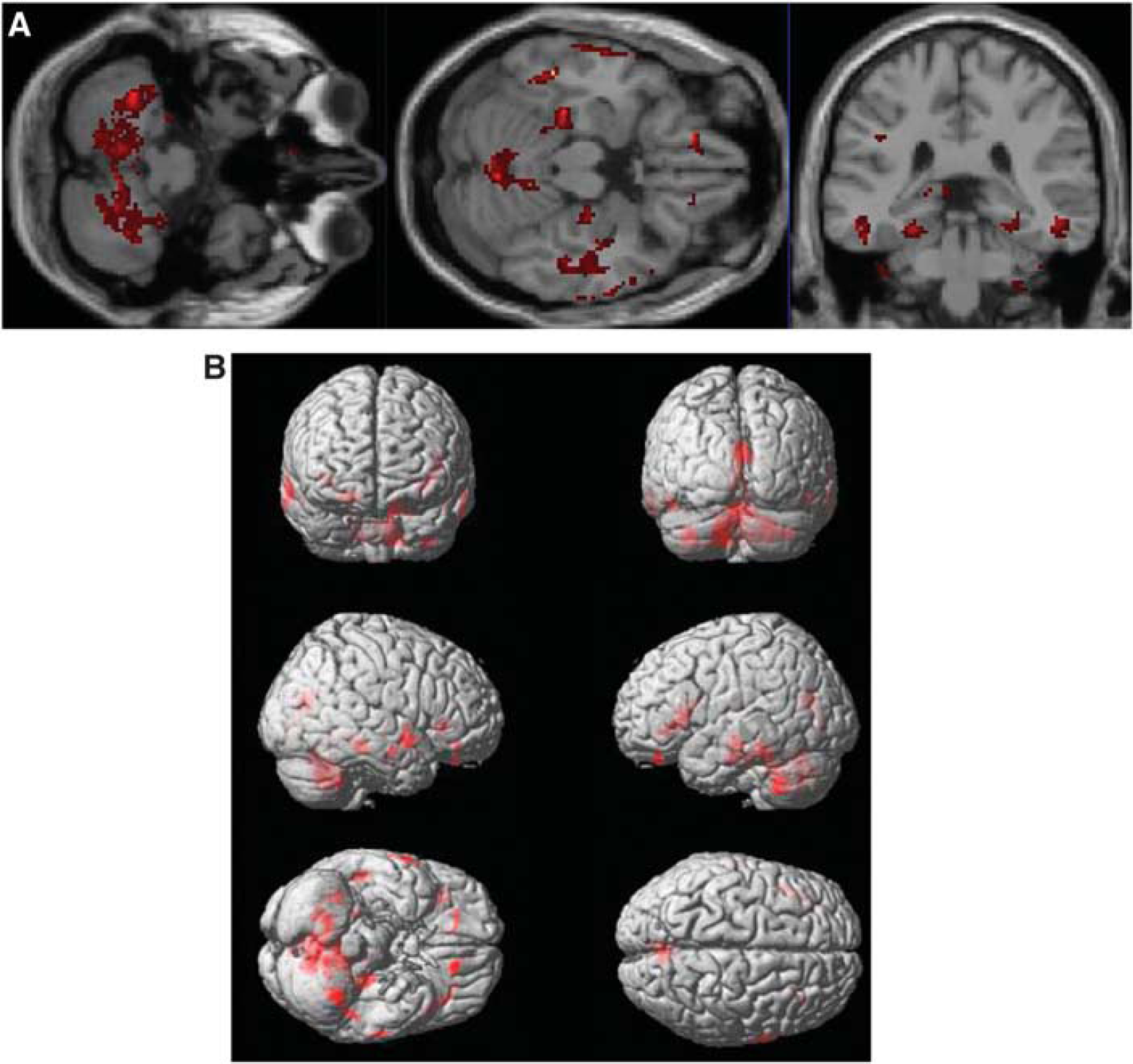
Axial and coronal slices (
We hypothesize that an increased response to tariquidar treatment reflects lower functionality and density of P-gp. In baseline experiments (i.e., before administration of tariquidar, scan 1),
By using the ROI-based analysis approach with a standardized maximum probability brain atlas, time-activity curves of 42 ROIs were generated.
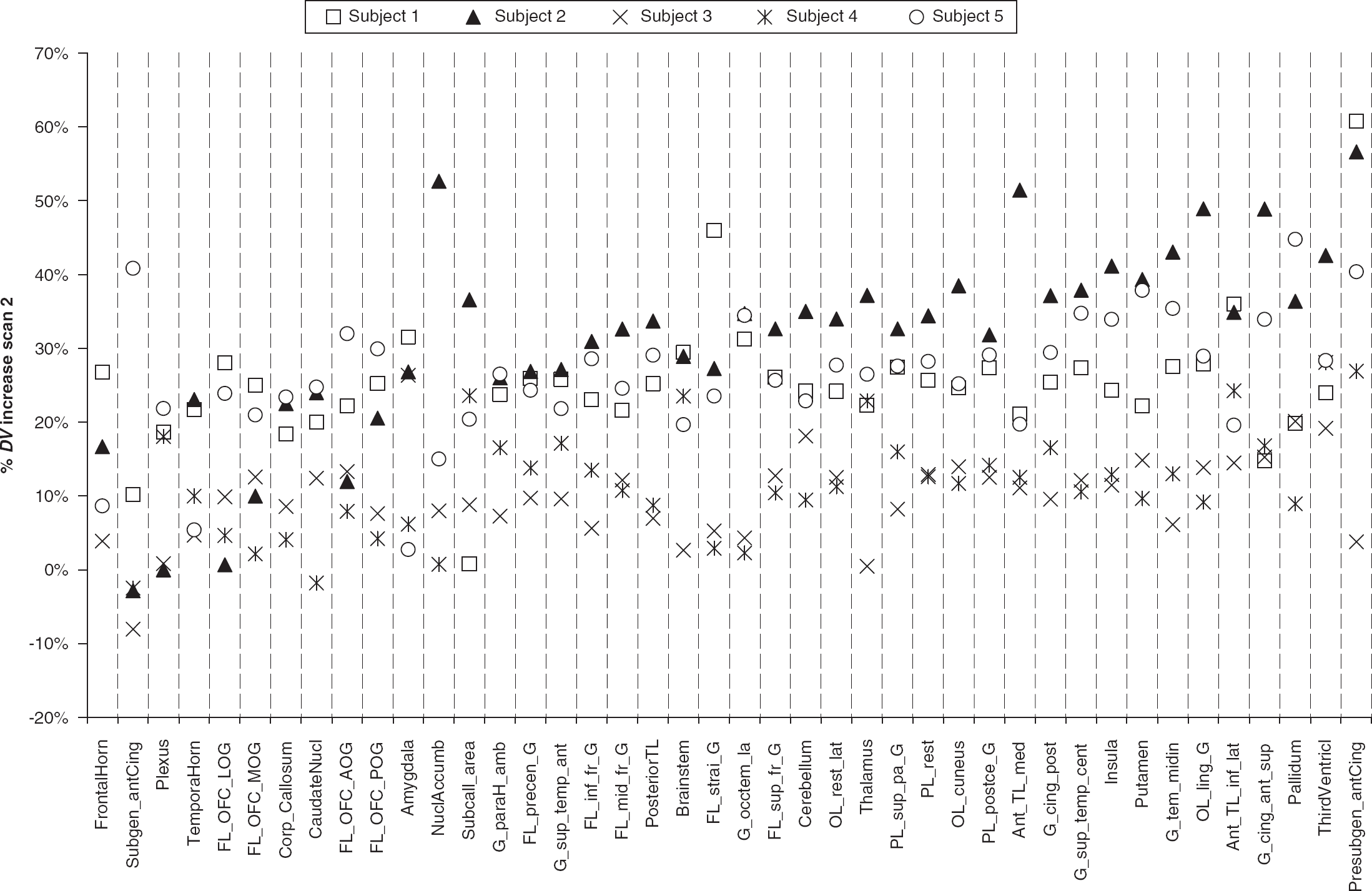
Plot of percent increases in VPM
The different results of ROI analysis and SPM analysis might be related to the fact that (1) only parts of individual brain ROIs displayed moderately higher
Overall, our data point to only moderate, if any, regional differences in P-gp function and expression in the healthy human brain, which might be related to a lack of upregulation of the ABCB1 gene in response to cellular stress triggered by drugs, toxins, or environmental factors (Löscher and Potschka, 2005). In patients, on the other hand, regional transporter expression may be markedly increased, which has been shown for patients with medically intractable epilepsy (Löscher and Potschka, 2005). The setting used in this study might be better suited than VPM baseline scans alone to studying regional differences in cerebral P-gp function and expression in such patients.
Footnotes
Acknowledgements
The research leading to these results has received funding from the European Community's Seventh Framework Programme (FP7/2007-2013) under grant agreement number 201380 (Euripides) and from the Austrian Science Fund (FWF) project ‘Transmembrane Transporters in Health and Disease’ (SFB F35). This study would not have been possible without the excellent support of Rainer Bartosch (Department of Nuclear Medicine), Edith Lackner, Johann Stanek, and Richard Schwameis (Department of Clinical Pharmacology), and Thomas Foki (Department of Neurology). The authors thank Alexander Hammers from the MRC Clinical Sciences Centre (London, UK), and Marie-Claude Asselin and Rainer Hinz from the Wolfson Molecular Imaging Centre (Manchester, UK) for providing help in establishing the brain atlas analysis approach. Divya Maheshwari from AzaTrius Pharmaceuticals Pvt Ltd (London, UK) is gratefully acknowledged for providing vials of tariquidar for i.v. infusion.
The authors declare no conflict of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
