Abstract
The number of circulating monocytes increases after stroke. In this study, we assessed the time course and phenotype of monocyte subsets and their relationship with the clinical course and outcome in 46 consecutive stroke patients and 13 age-matched controls. The proportion of the most abundant ‘classical’ CD14highCD16 monocytes did not change after stroke, whereas that of CD14highCD16+ monocytes increased and CD14dimCD16+ monocytes decreased. CD14highCD16 + monocytes had the highest expression of TLR2, HLA-DR and the angiogenic marker, Tie-2; CD14dimCD16+ monocytes had the highest expression of costimulatory CD86 and adhesion molecule CD49d. Platelet-monocyte interactions were highest in CD14highCD16 monocytes and lowest in CD14dimCD16+ monocytes. In adjusted models, 1/CD14highCD16 monocytes were associated with poor outcome (OR: 1.38), higher mortality (OR: 1.40) and early clinical worsening (OR: 1.29); 2/CD14highCD16+ monocytes were inversely related to mortality (OR: 0.32); and 3/CD14dimCD16+ monocytes were inversely related to poor outcome (OR: 0.74) and infarction size (r= 0.45; P = 0.02). These results illustrate that the predominant monocyte subtype conveys harmful effects after stroke, which include stronger interaction with platelets. Alternatively, rarer subpopulations of monocytes are beneficial with a phenotype that could promote tissue repair and angiogenesis. Therefore, monitoring of monocyte subtypes may emerge as a useful tool at the bedside for stroke patients.
Introduction
The role of the immune system in stroke is increasingly recognized, but further studies are necessary to elucidate the multifaceted characteristics of the adaptive and innate immune responses in this condition (Hallenbeck et al, 2006). Monocytes are the focus of attention in stroke, as they are major cells of innate immunity that may also affect adaptive immunity (Gordon, 2003). Monocytes can differentiate into macrophages and dendritic cells and are crucial for the initiation of the immune response to pathogens and of a variety of endogenous molecules released from injured tissues, such as heat shock proteins, RNA, fibronectin or fibrinogen (Kariko et al, 2004).
Monocytes can reach the central nervous system as early as 4 h from the onset of acute brain ischemia, although maximal infiltration occurs after 7 days (Garcia et al, 1994). In experimental ischemia, monocytes can promote inflammation, but they may also contribute to the resolution of inflammation (Manoonkitiwongsa et al, 2001; Yu et al, 2007). In patients with acute stroke, there is a significant increase in the number of circulating monocytes, and this increment is particularly conspicuous in patients who experience a stroke-associated infection (SAI) (Chamorro et al, 2006). Stroke may also induce monocyte deactivation, which is characterized by decreased HLA-DR expression and impaired production of proinflammatory cytokines on stimulation with lipopolysaccharide (Emsley et al, 2007; Haeusler et al, 2008). A higher expression of Toll-like receptor (TLR)4 in monocytes of patients with acute stroke was recently associated with poor prognosis (Urra et al, 2009a; Yang et al, 2008), in agreement with experimental data showing that TLR4-deficient mice have minor infarctions and less inflammatory response after an ischemic insult (Caso et al, 2007).
Monocytes are a heterogeneous population (Grage-Griebenow et al, 2001), with proinflammatory or anti-inflammatory phenotypes depending on their stage of differentiation and on the mechanism by which they are activated (Gordon, 2003). In mice, monocytes are best characterized according to their chemokine receptor expression pattern. The Ly6Chigh subset is actively recruited to inflamed tissues through monocyte chemotactic protein-1 receptor (CCR2), and the Ly6Clow subset is characterized by fractalkine-dependent recruitment to noninflamed tissues (Geissmann et al, 2003). Human monocytes are best characterized according to their expression of lipopolysaccharide receptor (CD14) and Fcγ receptor III (CD16) (Ziegler-Heitbrock, 2000). The majority of peripheral blood monocytes consist of cells strongly CD14+ and CD16- (CD14highCD16-) and are usually regarded as ‘classical monocytes.’ These monocytes share with Ly6Chigh monocytes the high expression of CCR2. The remaining monocytes express CD16 but have different expression levels of CD14. The minor subtype with a lower CD14 expression (CD14dimCD16 +) produces more tumor necrosis factor-± compared with classic monocytes after stimulation with lipopolysaccharide (Beige et al, 2002), but shares the chemokine expression pattern with murine Ly6Clow monocytes. CD14highCD16+ monocytes have been less well characterized. They could be a transitional form between the earlier subtypes and are the main producers of IL-10 in blood (Skrzeczynska-Moncznik et al, 2008).
In this study, we evaluated whether changes in monocyte subsets and phenotypes were associated with the clinical course after acute stroke. Our results stress (1) the differences between monocyte subtypes in their phenotype and their interaction with platelets; and (2) the unequal contribution of different monocyte subsets to major consequences of acute stroke, such as mortality, early clinical worsening, long-term outcome and risk of SAI.
Materials and methods
Subjects
As part of the IRIS (Immune Response In Stroke) study (Urra et al, 2009a, b), we assessed the immune changes induced by ischemic (n = 35) or hemorrhagic (n = 11) stroke. Ischemic and hemorrhagic strokes are frequently individualized in research studies, but growing evidence indicates that both groups share many of the immune responses (Emsley and Tyrrell, 2002; Zhao et al, 2007), a finding which is consistent with the danger model of immunity proposed by Matzinger (2002).
Thirteen age-adjusted relatives of the patients, all free of symptomatic central nervous system disorders but not of stroke risk factors, served as controls. Patients had a prestroke modified Rankin Scale (mRS) score ≥ 2 and a National Institutes of Health Stroke Scale (NIHSS) score > 3 on admission, when first evaluated at a median time delay of 180 mins (IQR 120 to 350) after stroke onset. Exclusion criteria included the use of antibiotics, immunosuppressants or steroids within the preceding 3 months. A recent (3 months) history of infection was also excluded after a structured questionnaire. This study was approved by the local ethics committee and all participants or legal representatives signed a written informed consent. Neurologic impairment was assessed daily using the NIHSS score until day 7, and again at day 90. Early clinical worsening was defined as an increase in the NIHSS score of 4 or more points during the first week. Stroke-associated infection was defined as body temperature > 37.8°C, in patients with suggestive symptoms (i.e., cough, dyspnea, pleuritic pain, urinary tract symptoms), or white blood cell count >11,000/mL or <4000/mL, pulmonary infiltrate on chest X-rays, or cultures positive for a pathogen during the first week (Chamorro et al, 2005). Global functional outcome and neurologic impairment were assessed with the mRS and NIHSS scores at day 90. Favorable outcome was defined as mRS of 0 to 2 and NIHSS of 0 or 1.
Neuroimaging
Patients had a brain computed tomography scan or magnetic resonance imaging (MRI) on admission and a second MRI using DWI (diffusion-weighted imaging) sequences within 31 h (SD: 8.8) from stroke onset. In patients with ischemic stroke, the lesion volume was calculated in the second DWI-MRI using MRIcro software (Chris Roden; University of Nottingham, Nottingham, UK) by investigators blinded to clinical and immunological data.
Monocyte Subsets and Phenotype
The phenotype of monocytes was analyzed immediately after blood extraction by blinded investigators, in samples harvested before the start of any medications, and between 8:30 and 9:00 hours on days 2, 7 and 90 after admission. The following monoclonal antibodies were used: TLR2, CD16 (FcγRIII) and IgG1 isotype control conjugated to fluorescein isothiocyanate (FITC); TLR4, CD49d (±4 subunit of VLA-4), CD86 (B7-2), HLA-DR (MHC class II receptor), CD16, Tie-2 (angiopoietin receptor) and IgG1 isotype control conjugated to phycoeritrin (PE); CD45 (leukocyte common antigen) conjugated to peridinin-chlorophyll-protein; and CD14 (lipopolysaccharide core-ceptor) and IgG1 isotype control conjugated to allophyco-cyanin (all from Pharmingen (San Jose, CA, USA), except for TLR2 and TLR4 (Serotec, Oxford, UK) and Tie-2 (R&D Systems, Minneapolis, MN, USA)). The monoclonal antibodies were mixed with the cell suspension and incubated for 15 mins at room temperature in the dark. After erythrocyte lysis and two washes, acquisition was carried out on a FACSCalibur flow cytometer (BD Biosciences, San Diego, CA, USA). A total of 60,000 events were acquired for monocyte subtype quantification; To have enough numbers of the minor subtypes, 20,000 monocytes were acquired for phenotyping. CellQuest® software (BD Biosciences) was used for analysis. Monocyte subsets were analyzed as shown in Figure 1. Surface molecule expression was quantified by converting median fluorescent intensity values into molecules of equivalent soluble fluorochrome (MESF) units using standardized fluorescent beads (Quantum™ FITC and Quantum™ PE Medium Level, BangsLabs, Fishers, IN, USA). As different monocyte subsets differed in their staining with isotype control antibodies, MESF units were calculated after subtraction of the isotype control MESF units.
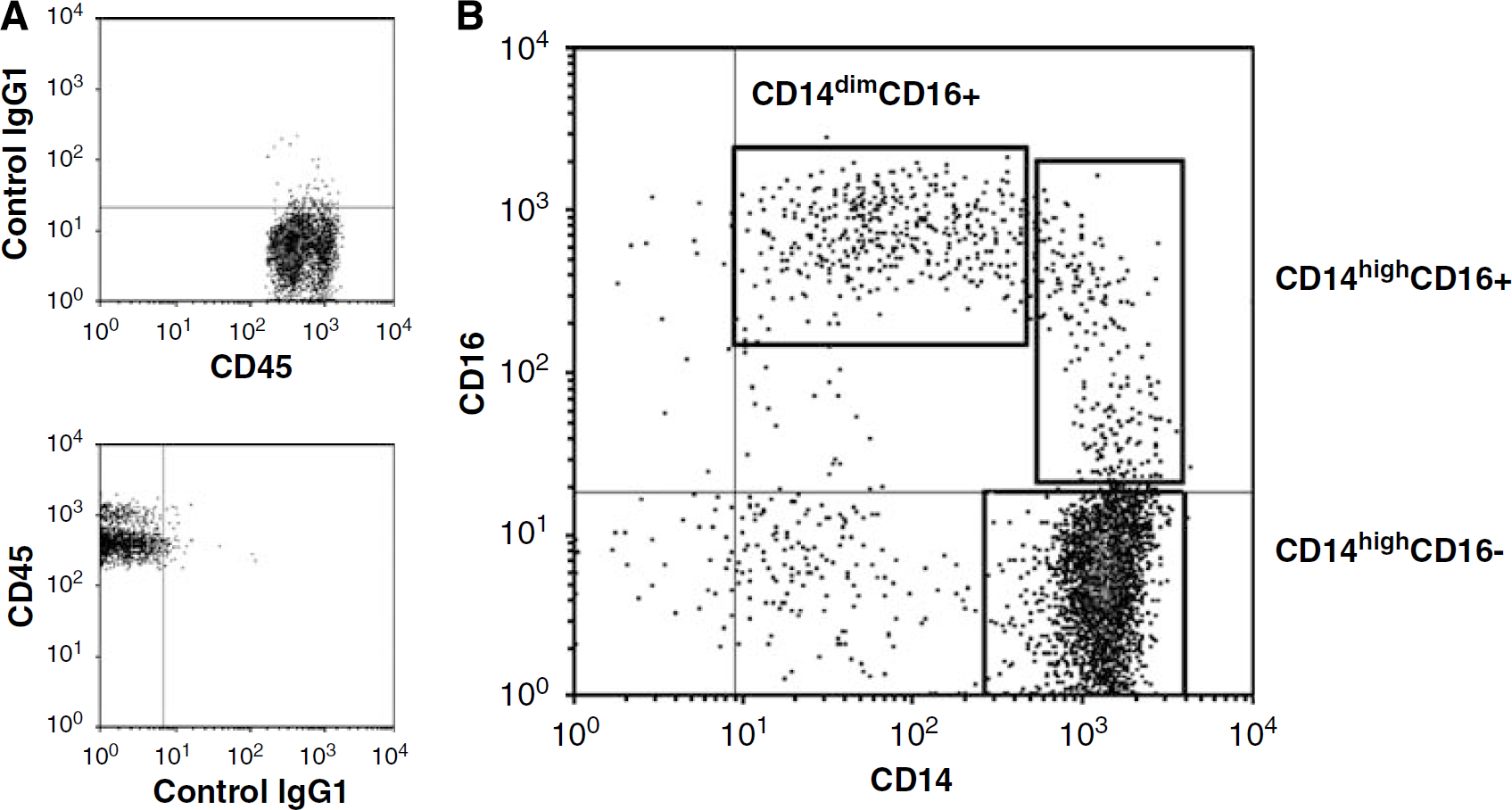
Quantification of monocyte subsets. Positivity for CD14 and CD16 staining was determined according to the staining with isotype controls (
Platelet-Monocyte Complexes
After incubating with anti-CD45, anti-CD14, anti-CD16 and anti-CD42b or with isotype control antibodies, the samples were lysed and washed once with PBS. After gating on each of the monocyte subsets, platelet-monocyte complexes were identified as events expressing the platelet marker, CD42b. Positivity for each monocyte subset was determined according to the expression of the isotype control in the same subset. We chose the present protocol because, unlike a no-lyse no-wash method that was used earlier in our laboratory, it does not require DNA staining. Before the start of this study, we compared both methods and found no significant differences (unpublished observations).
Neurohormonal Response
Between 08:30 and 09:00 hours of day 1, serum levels of Cortisol and plasma levels of unconjugated metanephrine were measured by enzyme immunoassays, as reported earlier (Chamorro et al, 2007).
Statistical Analysis
Ischemic awnd hemorrhagic strokes were compared with controls as one single clinical group in the main analysis, although exploratory analyses were also performed by stroke subtype. Differences in numerical values were calculated with the Student t-test, Mann-Withney U-test, the paired t-test or analysis of variance, as appropriate. Correlations were calculated with parametric and nonparametric coefficients. Early clinical worsening, SAI, outcome, mortality and infarct volume at follow up were dependent variables assessed in logistic or linear regression models, assessing the effect of monocyte subtypes at admission after adjusting for age and baseline stroke severity, which are strong, accurate and validated independent outcome predictors (Weimar et al, 2004). All tests were performed using the SPSS software version 14.0 (SPSS Inc., Chicago, IL, USA). P-values <0.05 were considered statistically significant.
Results
Demographics and Clinical Course of Patients
The main clinical and demographic characteristics of the study population are summarized in Table 1.
Demographics, risk factors and clinical evolution of the study population
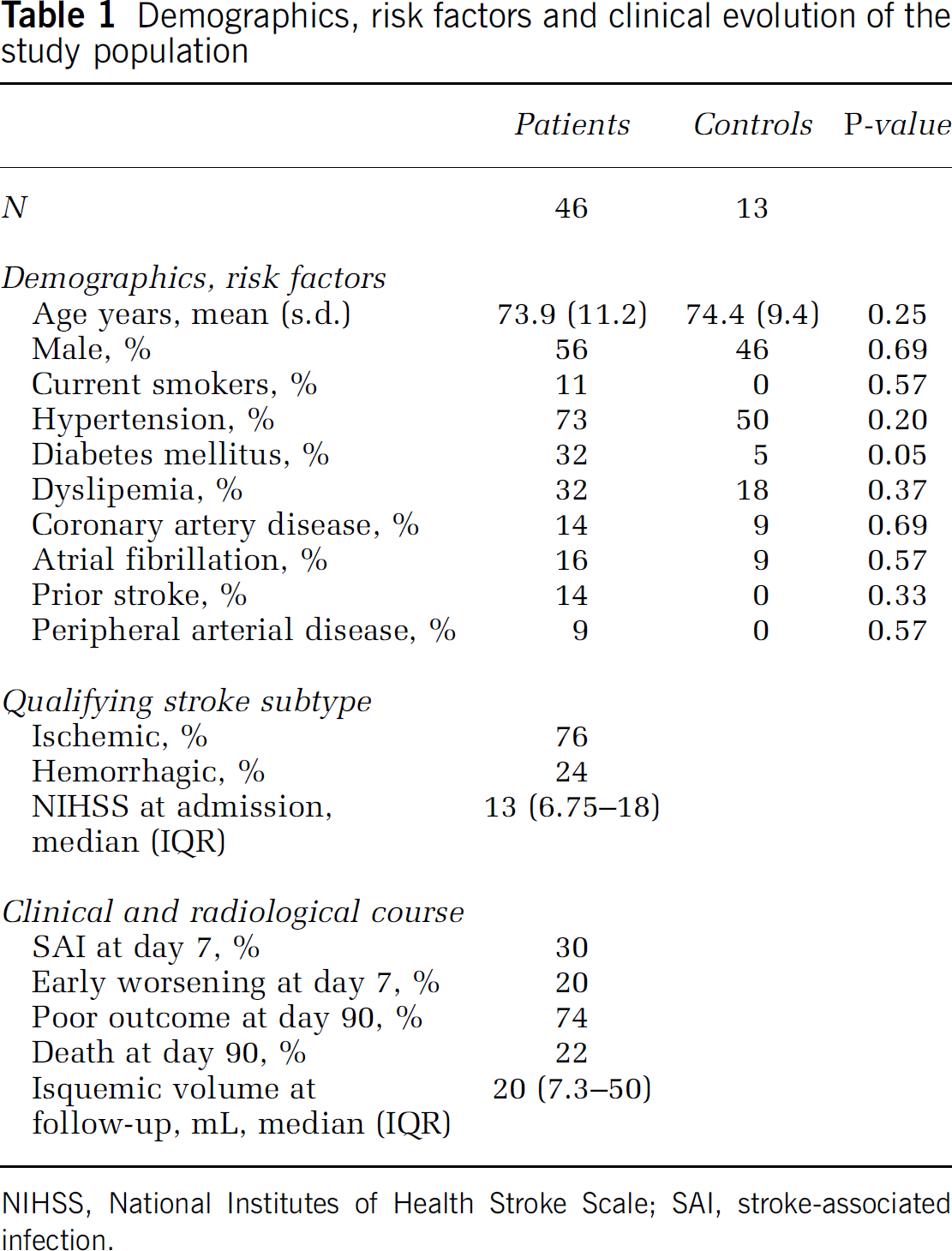
NIHSS, National Institutes of Health Stroke Scale; SAI, stroke-associated infection.
Monocyte Subtypes and Phenotype
Compared with controls, the total number of monocytes was similar in patients on admission and at day 90, but higher at day 2 and at day 7 (Figure 2A), as reported earlier (Urra et al, 2009a). Furthermore, the proportion of CD14highCD16- monocytes did not change significantly after stroke, but there was a 38% relative increase in CD14highCD16+ monocytes and a 41% relative decrease in CD14dimCD16+ monocytes. There were no differences in the proportion of monocyte subtypes at day 90 (Figures 2B to 2D). Ischemic and hemorrhagic strokes had similar longitudinal changes in the proportion of monocyte subsets (see Supplementary Table).
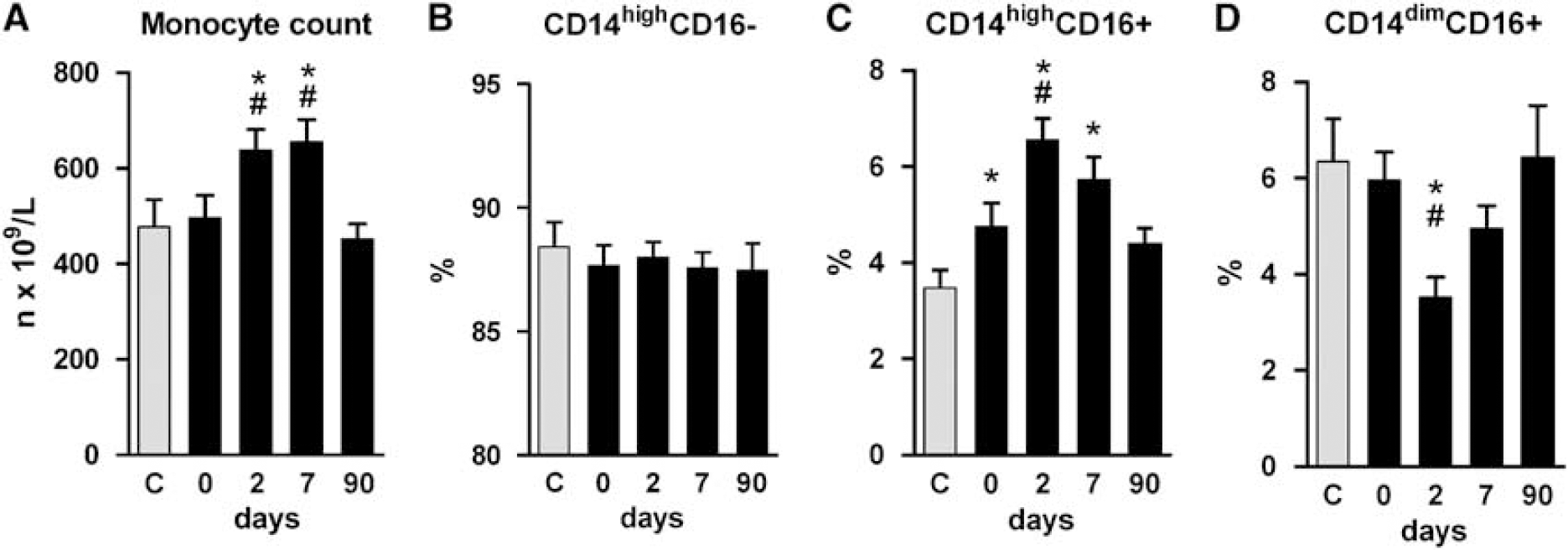
Time course of monocyte subtypes in acute stroke. The number of circulating monocytes increased after stroke (
The phenotype of monocyte subtypes disclosed significant differences (Figure 3). CD14highCD16-monocytes showed the lowest expression of HLA-DR, CD49d, CD86 and Tie-2; CD14highCD16 + monocytes showed the highest expression of TLR2, HLA-DR and Tie-2, and intermediate levels of CD49d and CD86; and CD14dimCD16+ monocytes showed the highest expression of CD49d and CD86. The expression of TLR4 disclosed no significant differences between monocyte subtypes. There were no major differences in phenotypes among the monocyte subtypes of patients and controls (data not shown).
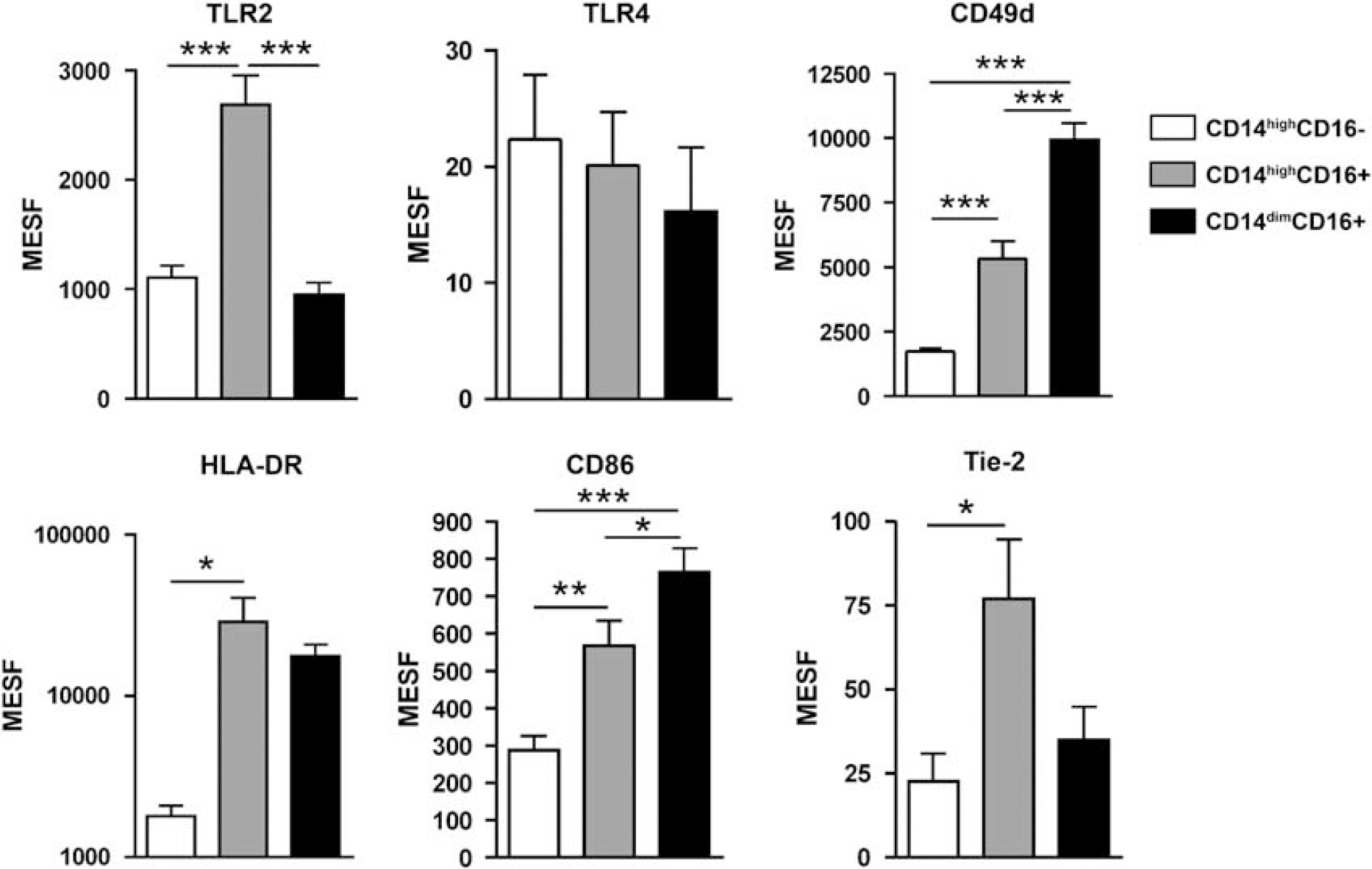
Phenotype of monocyte subtypes. Surface expression was quantified in MESF units. Values are mean ± s.e.m. *P < 0.05, **P <0.01 and ***P < 0.001, ANOVA. n = 16 patients.
Platelet-Monocyte Complexes
The ability to interact with platelets differed significantly between monocyte subsets. Thus, CD14highCD16- monocytes showed the highest and CD14highCD16+ monocytes the lowest proportion of platelet-monocyte complexes (Figure 4).
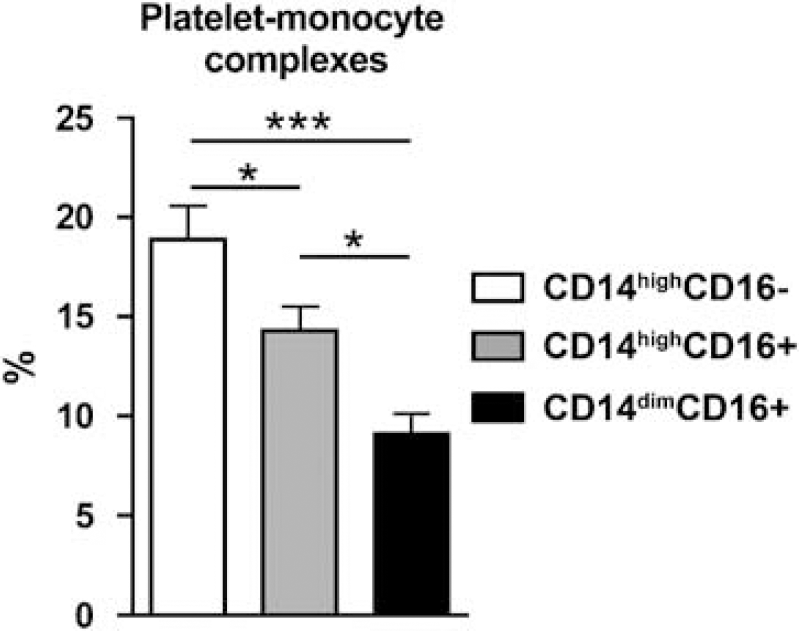
Platelet-monocyte interactions in different monocyte subtypes. The proportion of platelet-monocyte complexes was measured in each monocyte subtype. Values are mean ± s.e.m. *P <0.05, ***P < 0.001, ANOVA. n = 16 patients.
Monocyte Subtypes and Neurohormonal Response
Cortisol and metanephrine levels did not show any significant correlations with monocyte subtypes on admission. In exploratory analyses, Cortisol levels were inversely correlated with the proportion of CD14dimCD16+ monocytes at day 2 (ρ = −0.42; P = 0.01) and at day 7 (r =−0.36; P = 0.03), but not with other monocyte subtypes.
Association between Monocyte Subtypes on Admission, Infarction Volume and Clinical Events
Infarction volume on MRI at day 1 was positively correlated with the proportion of CD14highCD16-monocytes (0.54; P = 0.004), and inversely correlated with CD14highCD16 + (−0.43; P = 0.02) and CD14dim CD16+ monocytes (−0.45; P = 0.02) at admission. In models adjusted for age and admission NIHSS, CD14highCD16- monocytes were predictors of larger infarctions at day 1 (B: 9.08, P = 0.01), whereas CD14dimCD16+ monocytes (B: −10.44, P = 0.04) were predictors of smaller infarctions.
In logistic regression models adjusted for age and stroke severity, an increased proportion of CD14highCD16- monocytes on admission was associated with early clinical worsening, poor outcome and mortality at day 90 (Table 2, Figure 5). Furthermore, a decreased proportion of CD14highCD16 + monocytes on admission predicted mortality at day 90, and a decreased proportion of CD14dimCD16+ monocytes predicted poorer outcome at 3 months (Table 2). The proportion of monocyte subtypes at stroke admission did not differ between patients with or without SAI (Table 2). In exploratory analyses, SAI was associated with an increased proportion of CD14highCD16+ monocytes at day 2 (OR: 1.63, 95% CI: 1.09 to 2.45, P = 0.02) (Figure 5).
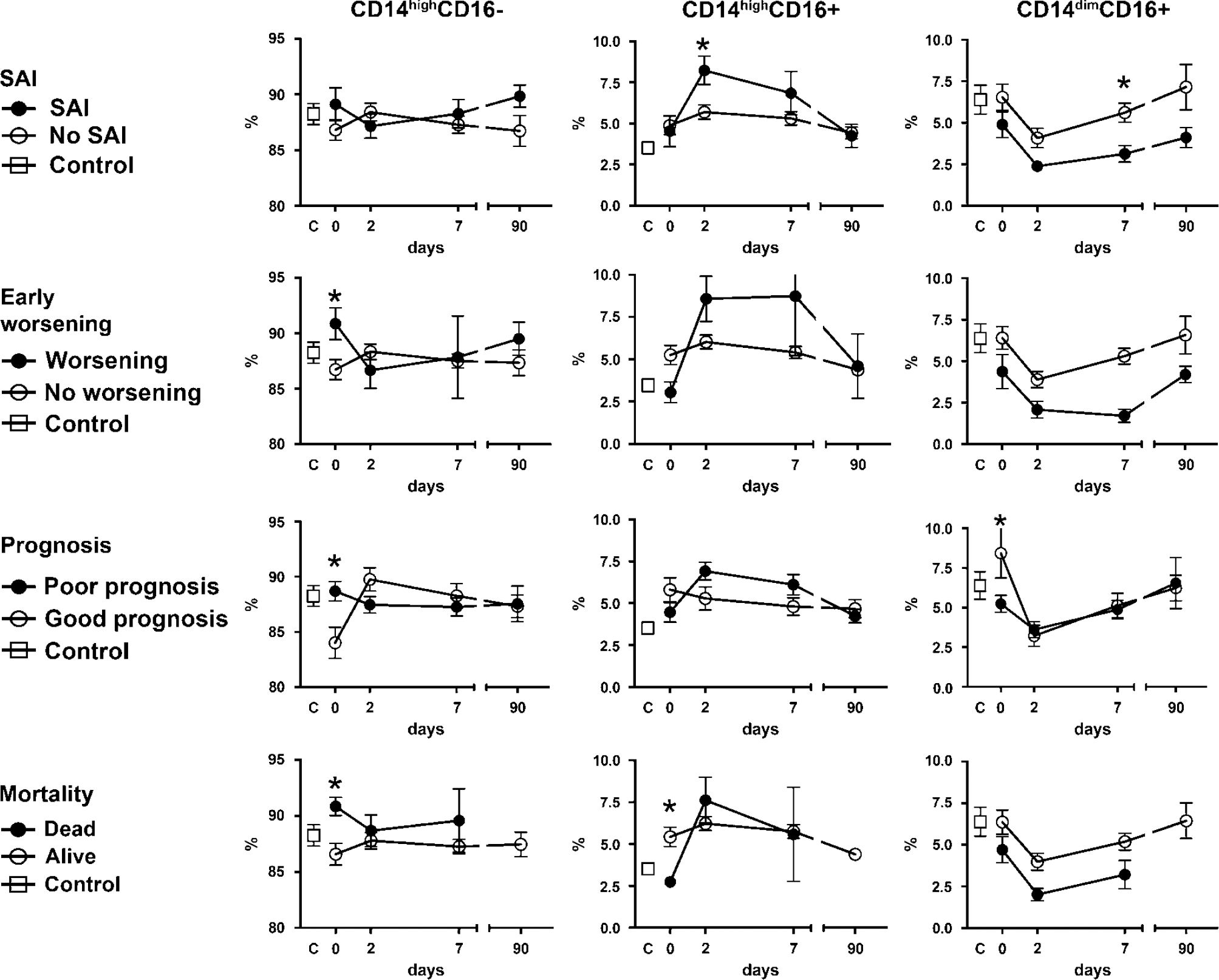
Monocyte subtypes and clinical end points. Course of monocyte subtypes according to the presence or absence of clinical end points. Values are mean ± s.e.m. *Significant differences (P < 0.05) in adjusted models, n (controls) = 12; SAI: n (day 0) = 13, n (day 2) = 13, n (day 7) = 11, n (day 90) = 8; No SAI: n (day 0) = 23, n (day 2) = 26, n (day 7) = 30, n (day 90) = 26. Early worsening: n (day 0) = 8, n (day 2) = 8, n (day 7) = 4, n (day 90) = 2; no worsening: n (day 0) = 28, n (day 2) = 31, n (day 7) = 37, n (day 90) = 32. Poor prognosis: n (day 0) = 28, n (day 2) = 30, n (day 7) = 29, n (day 90) = 22; good prognosis: n (day 0) = 8, n (day 2) = 9, n (day 7) = 12, n (day 90) = 12. Dead: n (day 0) = 9, n (day 2) = 9, n (day 7) = 5, n (day 90) = 0; alive: n (day 0) = 27, n (day 2) = 30, n (day 7) = 36, n (day 90) = 34.
Independent predictors of clinical end points
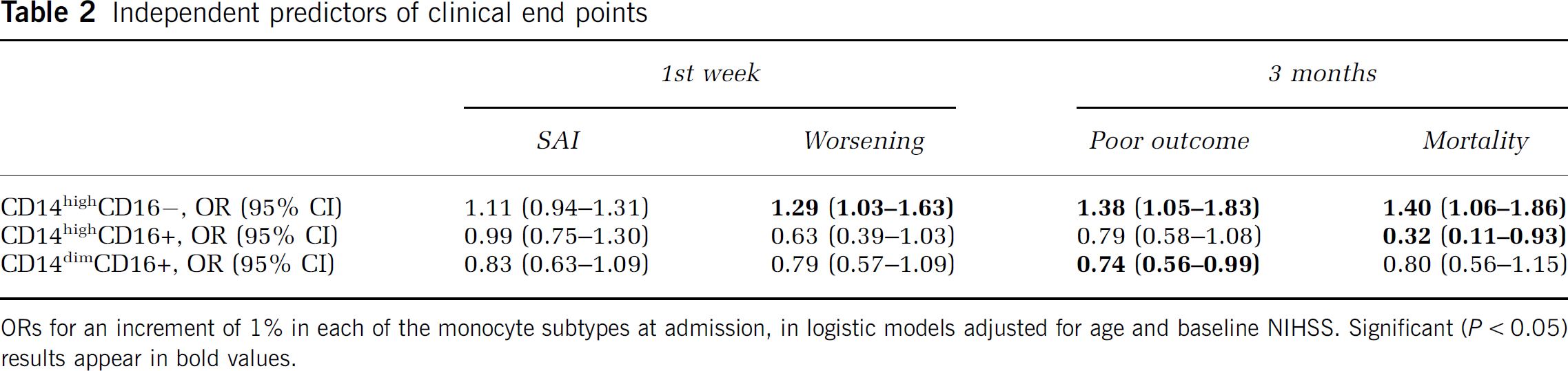
ORs for an increment of 1% in each of the monocyte subtypes at admission, in logistic models adjusted for age and baseline NIHSS. Significant (P < 0.05) results appear in bold values.
Discussion
Recent studies have established that monocytes increase early after stroke onset and that phenotypic variations in the surface of monocytes are associated with the clinical course of stroke (Haeusler et al, 2008; Urra et al, 2009a). In this study, (1) we define and quantify the monocyte subsets that follow acute stroke; and (2) we recognize the main traits of monocytes responsible for increased stroke mortality, poorer functional outcome, early clinical worsening, infarction growth and risk of SAI.
In this regard, the study found an increase in the proportion of CD14highCD16+ monocytes that peaked at 48 h after stroke. Alternatively, the most abundant classical subtype, CD14highCD16- monocytes, did not change significantly after stroke, whereas the CD14dimCD16 + subtype decreased. These changes occurred to a similar extent in patients with ischemic and hemorrhagic stroke. In adjusted models, poor outcome, increased mortality and early clinical worsening after stroke were associated with an increased proportion of CD14highCD16- monocytes. However, other subtypes of monocytes could be associated with clinical benefits, as mortality was inversely related to CD14highCD16 + monocytes, and poor outcome and infarction size were inversely related to CD14dimCD16+ monocytes. Although it may seem surprising how minor subsets of monocytes have such an effect on infarct size and outcome, their unique phenotype and function may contribute towards explaining these results. Overall, these results illustrate the complexities of the monocytic response and attest the diverse susceptibility of different monocyte subtypes to be produced and released into the systemic circulation after acute stroke.
Earlier studies have shown that poor outcome after stroke is associated with innate responses signaled through TLR4 in monocytes (Urra et al, 2009a; Yang et al, 2008). In this study, the expression of TLR4 disclosed no significant differences between monocyte subtypes, suggesting that other differences in monocyte subtypes affect stroke outcome. The harmful clinical effects of CD14highCD16- monocytes agree with recent experimental data in mice, showing that, after myocardial infarction, the corresponding Ly6Chigh monocytes accumulate through CCR2 to mediate early proteolysis, inflammation and phagocytosis (Nahrendorf et al, 2007). In experimental brain ischemia, CCR2 deficiency is associated with reduced infarct size, decreased blood-brain barrier permeability, lesser infiltration of monocytes and neutrophils, and reduced expression of proinflammatory cytokines during reperfusion (Dimitrijevic et al, 2007). Thus, the deleterious effects of CD14highCD16- monocytes could be at least partially explained by an increased inflammatory drive at ischemic areas.
Platelet activation and increased platelet-monocyte interaction have been described in patients with stroke (Htun et al, 2006; Marquardt et al, 2002), and they have been related to worse outcome (Zeller et al, 2005). Platelet-monocyte interaction occurs through P-selectin and P-selectin glycoprotein ligand-1 (Yang et al, 1999). The higher proportion of platelet-monocyte complexes found in CD14highCD16- monocytes in this study could result from a higher expression of P-selectin glycoprotein ligand-1 in human CD14highCD16- monocytes and in murine Ly6Chigh monocytes (An et al, 2008).
This study highlighted, for the first time, a beneficial role of CD14highCD16+ monocytes in patients with stroke. This monocyte subtype is the main producer of IL-10 (Skrzeczynska-Moncznik et al, 2008), an anti-inflammatory cytokine with beneficial effects in patients with stroke (Vila et al, 2003). Therefore, the increased number of CD14highCD16+ monocytes observed in the study identifies a new trait of the immune syndrome that follows acute stroke to minimize the inflammatory consequences of acute brain damage. Yet, CD14highCD16+ monocytes could also be beneficial through its richer expression of proangiogenic Tie-2 (Murdoch et al, 2007) and their strong phagocytic activity (Skrzeczynska-Moncznik et al, 2008). Together, angiogenesis and phagocytosis could facilitate the clearance of injured brain tissue after stroke (Manoonkitiwongsa et al, 2001; Yu et al, 2007). The meaning of the high expression of TLR2 in this population remains to be further investigated.
In contrast to other subtypes, CD14dimCD16 + monocytes decreased after stroke. The depletion of CD14dimCD16+ monocytes could be the result of hypercortisolism, because glucocorticoids selectively induce apoptosis of CD14dimCD16+ monocytes (Dayyani et al, 2003). Indeed, we found higher levels of Cortisol in the patients and their inverse correlation with the proportion of circulating CD14dimCD16+ monocytes. Nonetheless, we cannot exclude the possibility that CD14dimCD16+ monocytes migrated to the injured brain as CD14dimCD16 + monocytes had the highest expression of CD49d, which mediates monocyte diapedesis.
In this study, the increased proportion of CD14dimCD16+ monocytes was associated with better outcome. Contrarily, in other diseases such as sepsis and coronary heart disease (Ziegler-Heitbrock, 2007), harmful effects have been attributed to these monocytes in relation to a proinflammatory cytokine production pattern (Beige et al, 2002). However, experimental data in mice indicate that Ly6Clow monocytes, comparable with human CD14dimCD16+ monocytes, acquire a less-inflammatory phenotype after migrating to tissues, where they upregulate the expression of genes involved in tissue repair (Auffray et al, 2007; Nahrendorf et al, 2007).
Patients who developed SAI were characterized by the most pronounced increase in CD14highCD16 + monocytes, indicating that SAI may be the consequence of an increased anti-inflammatory drive rather than an independent negative prognostic factor (Vargas et al, 2006). In addition, SAI was also associated with the deepest decrease of CD14dimCD16 + monocytes, that is, the subtype with the highest expression of costimulatory and adhesion molecules that are needed to respond adequately to pathogens.
Taken together, the results of this study indicate that each colony of monocytes has a different time course in patients with acute stroke and is associated with a distinct clinical pattern. Overall, classic CD14highCD16- monocytes were detrimental, whereas the minor subsets of CD16-expressing monocytes were associated with a favorable clinical course. Therefore, monitoring of monocyte subtypes could help to identify the individual risk of impending complications early after stroke onset. Further studies will nevertheless be required to unravel the molecular mechanisms linking circulating monocytes with brain tissue fate and the therapeutic implications that might derive.
Footnotes
Acknowledgements
We thank Francisca Ruiz for technical assistance, and Dr Álvaro Cervera for the help in the evaluation of the patients. Patients and their relatives are acknowledged for their consent.
The authors state no conflict of interest.
