Abstract
Brain dysfunction is frequently observed in sepsis as a consequence of changes in cerebral structure and metabolism, resulting in worse outcome and reduced life-quality of surviving patients. However, the mechanisms of sepsis-associated encephalopathy development and a better characterization of this syndrome
Introduction
Although it is well accepted that the brain may be affected during sepsis, it was only recently shown that the presence of brain dysfunction impacts directly as an independent determinant of poor prognosis in patients with sepsis (Sprung et al, 1990; Ely et al, 2004; Lin et al, 2008). Growing evidence supports that an early central autonomic dysfunction contributes to the hemodynamic changes found in septic shock patients (Sharshar et al, 2003) and long-term cognitive impairment can be present in sepsis survivors (Siami et al, 2008). Sepsis-associated encephalopathy, or sepsis-associated delirium, is a diffuse cerebral dysfunction characterized by an acute onset of an impairment of cognitive function, which can vary from inattention, disorientation, agitation, stupor and coma. The animal model of sepsis using cecal ligation and puncture (the CLP model) is able to mimic clinical sepsis-associated encephalopathy as it induces autonomic dysfunction (Kadoi and Goto, 2004; Pancoto et al, 2008) and long-term cognitive impairment (Barichello et al, 2005).
Magnetic resonance imaging (MRI) techniques offer unique capabilities in observing morphologic and metabolic characteristics of the brain
In addition, MRI offers the unmatched possibility of acquiring quantitative data on a subset of metabolites
Here, we used MRI and spectroscopy techniques to monitor the consequences of a septic challenge to the mouse brain. Measurements in septic mice brains consisted of
Materials and methods
Experimental Design
Mice were first subjected to an MRI session during which baseline values were acquired. Animals were then subjected to CLP surgery or sham operation. A complete MRI data set was acquired during the development of sepsis at 6 h after surgery. After the MRI session, animals were kept for at least 18 h. For animals that were still alive 24 h after surgery, we acquired a second MRI data set at that time point.
Cecal Ligation and Puncture Model
The CLP protocol was approved by the Institutional Animal Care and Use Committee and performed in accordance to the guidelines set out by the National Institutes of Health. Cecal ligation and puncture was performed as previously described (Gomes et al, 2006). Sixteen 8- to 10-week-old male C57BL/6 mice were anesthetized with isoflurane (2% to 3%) under aseptic conditions. A laparotomy was performed with a 2 cm midline incision through the linea alba. The cecum was exposed and ligated with sterile 3-0 silk below the ileocecal junction. The cecum was punctured once with an 18-gauge needle and squeezed to empty its content through the puncture. The cecum was returned to the peritoneal cavity, and the abdominal muscle and skin incisions were closed. After the surgery, 0.5 mL of sterile saline was administered subcutaneously to the animals. Mice in the sham operation group were subjected to identical procedures, except that ligation and puncture of the cecum were omitted. The lethality of our model was approximately 40% in the first 24 h and 60% in 144 h.
Magnetic Resonance Imaging
Mice were anesthetized using 2% isoflurane and 800 mL/min oxygen, and maintained with a constant body temperature of 37°C under general anesthesia using 1% isoflurane during the imaging session. Their respiratory rate was monitored. All MRIs were acquired on a 7 T, 30 cm horizontal bore magnet (Bruker Biospin, Ettlingen, Germany), using a G060 gradient set and a whole-body multi-rung 38 mm resonator.
Morphologic Images
Morphologic images were acquired using a rapid acquisition, relaxation-enhanced pulse sequence with
Diffusion-Weighted Imaging
Diffusion-weighted images were acquired using a single-shot echo-planar encoded imaging sequence with a repetition time of 1,325 ms, an echo time of 63 ms, and a flip angle of 90°. Geometric distortions inherent to this encoding scheme applied at an operating proton frequency of 300 MHz were greatly diminished by careful shimming of the volume of interest using the ‘fast automatic shimming technique by mapping along projections' method (Gruetter, 1993). The diffusion-sensitizing scheme of Stejskal and Tanner (1965) was used. Diffusion-sensitizing gradients (7 ms) were applied along the rostro-caudal axis, separated by 14 ms and located at symmetric positions on either side of the refocusing pulse. Five images were obtained with different gradient gains, resulting in
In Vivo Proton Spectroscopy
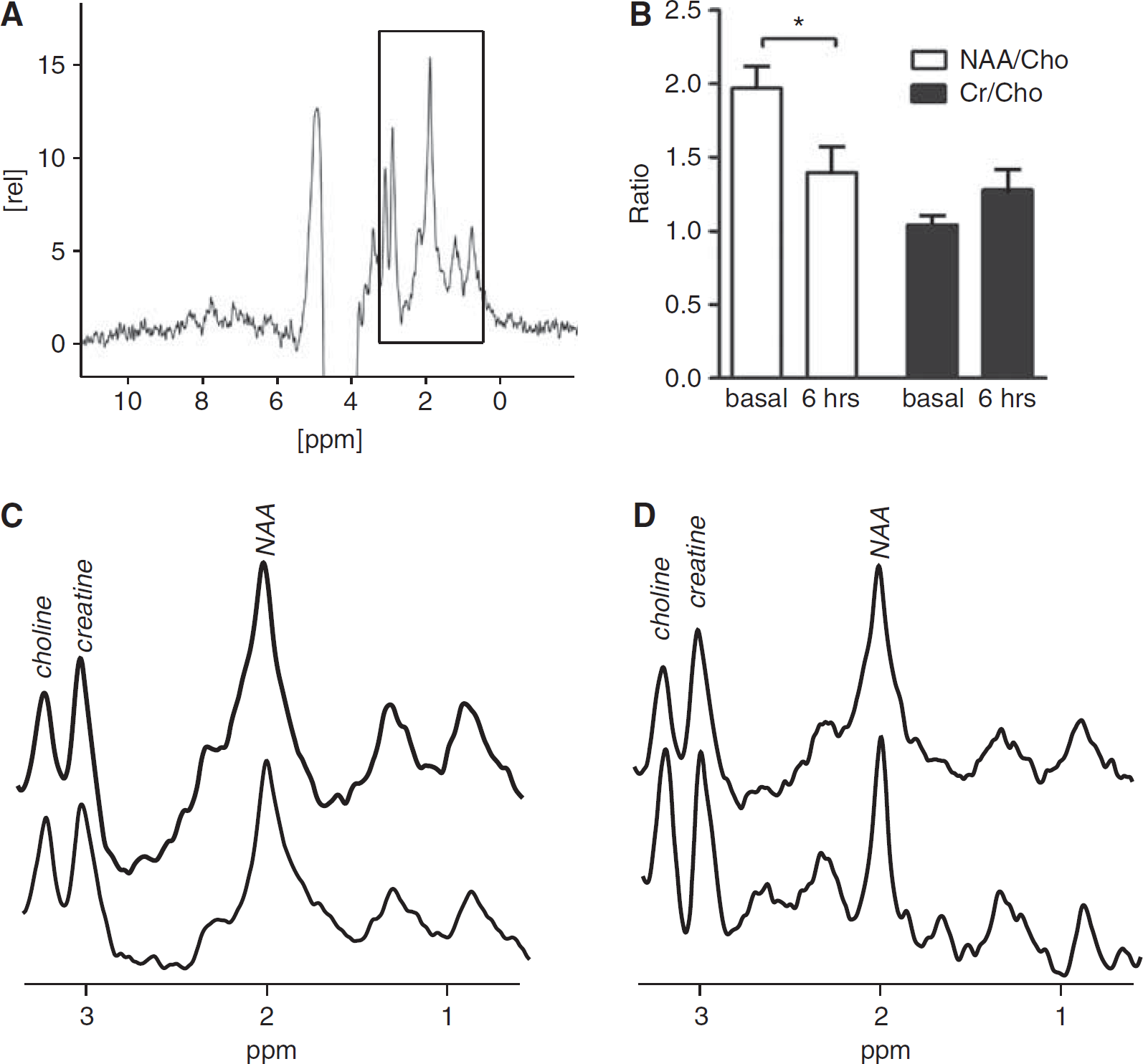
Proton spectra were acquired over a 3 × 3 × 3 mm3 cube region located on the hippocampus. The point-resolved spectroscopy pulse sequence (90°-90°-180°) was used in combination with the variable power with optimized relaxation delays scheme of water suppression. A total of 256 averages were acquired with a repetition time of 2.5 secs and an echo time of 2 × 10 ms over a spectral width of 4,006 Hz (13.3 p.p.m.). The resulting decays were applied a line broadening noise reduction and were Fourier–transformed. The spectra were phased and zero-order baseline was corrected. Because the water peak had been suppressed, the spectra were calibrated with the easily recognizable peak of Ch (3.2 p.p.m.). After ascertaining that the overall aspect of the spectra was similar, analysis was carried out on a 3.5 p.p.m. spectral window where changes were identified, which contained the peaks for Ch compounds (3.21 p.p.m.), Cr compounds (3.03 p.p.m.) and NAA (2.02 p.p.m.) (Pfeuffer et al, 1999). These were fitted against a Gaussian lineshape, which was integrated to yield the peak area. In an effort to limit the sensitivity to phasing artifacts, we computed metabolite ratios for analysis. Although approximately 10 metabolite peaks were routinely identified, only the Cr, Ch, and NAA peaks were analyzed because of their superior peak area-to-noise ratio and clinical relevance.
Statistical Analyses
The statistical analysis was performed using the paired two-tailed Student's
Results
The brains of the septic mice were observed before surgical procedures, 6 h, and 24 h after surgery through MRI of axial slices with
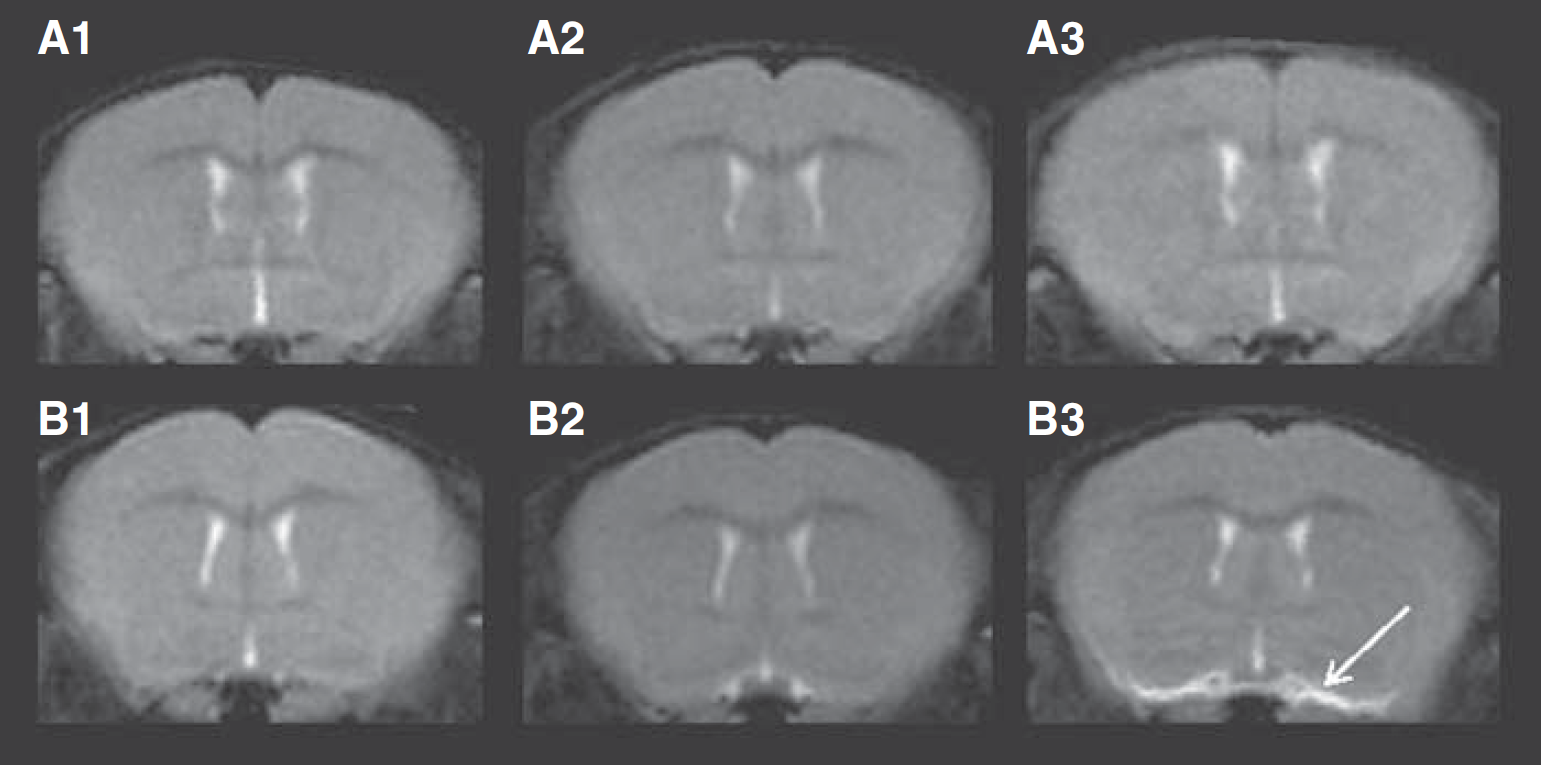
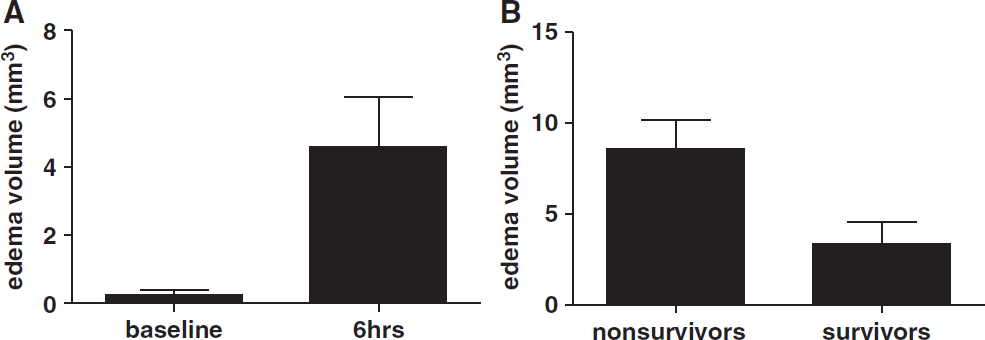
Basal brain-vessel-associated edema volumes (in mm3) as measured on
The ADCs in the brain tissue of septic animals were compared with sham animals. Regions of interest were traced on the cortex and the thalamus using the diffusion-weighted images, and were reported on the ADC maps (Figure 3). The ADCs were averaged over these regions of interest, resulting in the observed values. In general, septic animals were found to have a diffuse decrease in the ADC. When separating CLP-operated animals according to their 24 h survival outcome, an important difference was found in the magnitude of the ADC decrease observed in both cortex and thalamus. Thus, sepsis-related deaths were linked with a decrease in the ADC values in the cortical and thalamic areas. On the ADC maps, the regions of
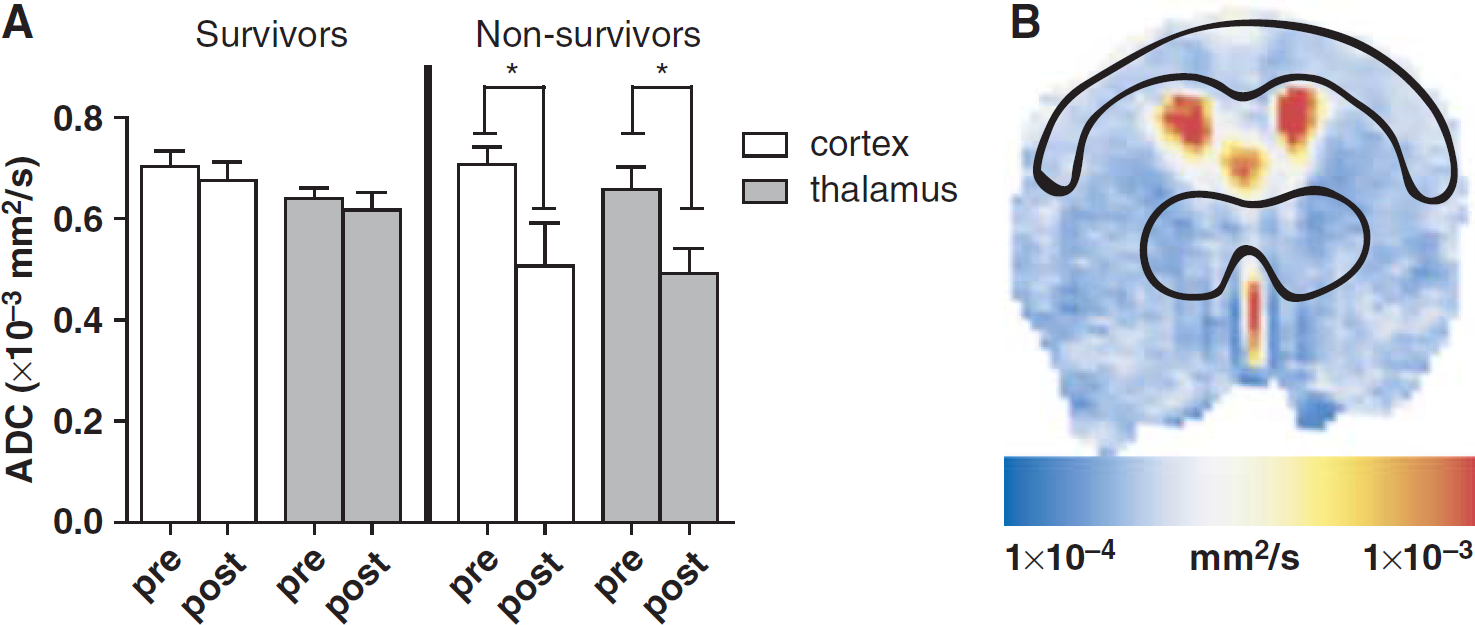
Apparent diffusion coefficients (ADCs). (
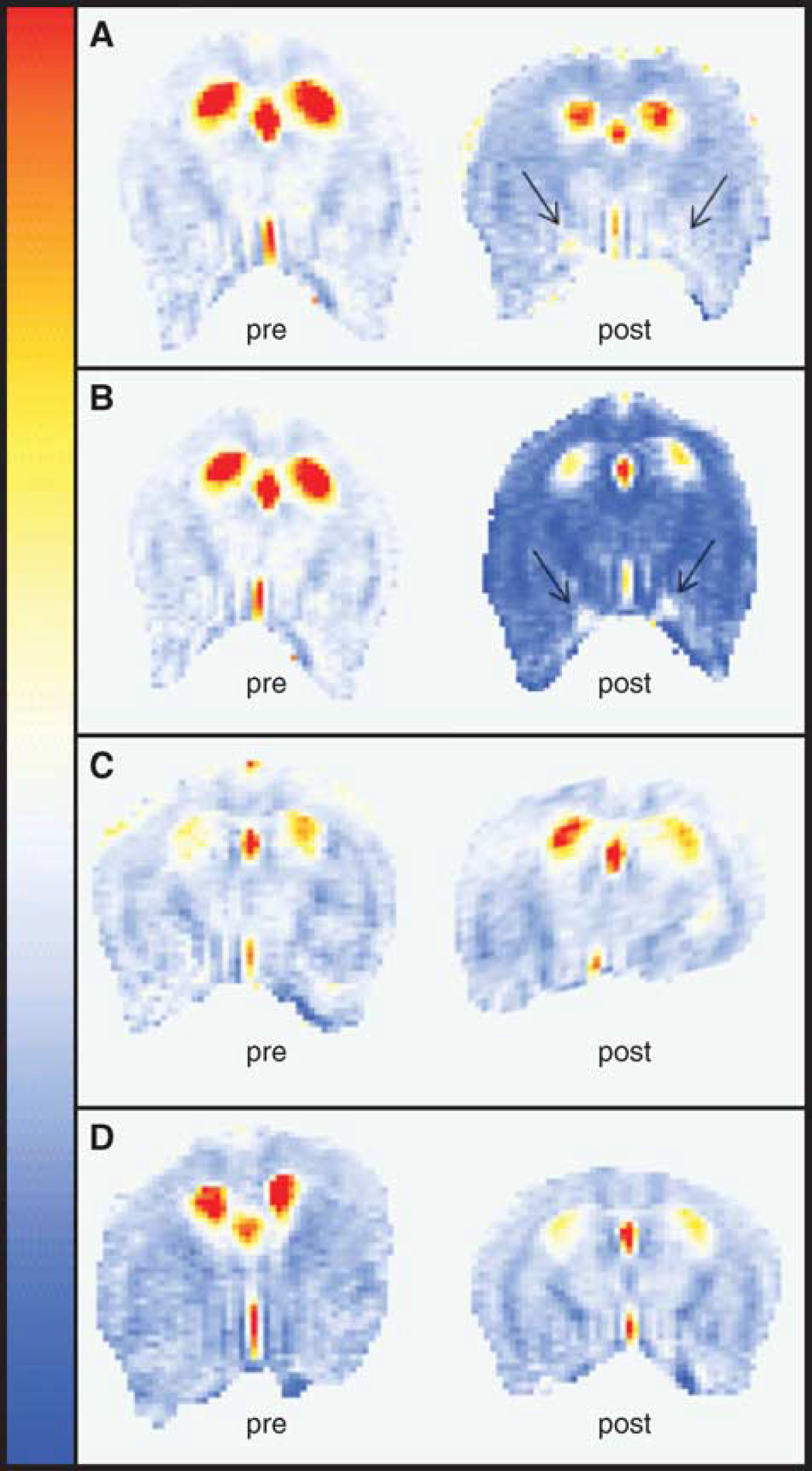
Representative apparent diffusion coefficient (ADC) maps before (baseline, left) and 6 h after (6 h, right) surgery. (
Proton spectra (Figure 5A) were acquired in a cubic volume of 27 μL including the hippocampus, and the relative amounts of the total Cr, total Ch, and NAA compounds were determined after the identification of their respective peaks (Figure 5B). A small increase was detected in the Cr/Ch ratio of septic animals (1.28 ± 0.14) 6 h after surgery compared with sham animals (1.04 ± 0.06). This change was not of sufficient amplitude to reach statistical significance given our sample size. A strong decrease was however seen in the NAA/Ch ratio, from a baseline value of 1.97 ± 0.15 to 1.39 ± 0.18 at 6 h after CLP. Overall, the results of our study could be explained by a decrease in NAA, although an increase in Ch levels concomitant with a greater increase in Cr levels could also result in the ratios that we observed. However, based on the individual aspect of the peaks, a decrease in NAA accompanied by relatively little changes in either Cr or Ch seems more likely.
Discussion
Although systematic MRI brain studies of sepsis are not yet available, recent preliminary investigations carried out in a small number of patients (Sharshar et al, 2007) and on animal models of endotoxemia (Rosengarten et al, 2008) highlighted the potential applications of MR techniques to determine the mechanisms of sepsis-associated encephalopathy. The noninvasiveness of MR techniques represents an important advantage to study temporal evolution of disease processes. In addition, these techniques also offer the ability to acquire
The data presented here also indicate that the brain damage in sepsis is characterized by disruption in the normal function of the blood–brain barrier and in cellular function as well. This was clearly apparent in the vicinity of blood vessels at the base of the brain as early as 6 h after disease onset, when edematic fluid accumulated therein. Also, fluid accumulation in the brain was strongly associated to the outcome at 24 h, although we could not establish a direct cause–effect relationship. We propose that this phenotype is a reliable marker of sepsis severity. The increase in vascular permeability observed here seems to be related to the systemic inflammation and the intensity of septic insult. Although animals were placed in a prone position, fluid accumulation was attributed to a leakage out of the vessels because it was systematically present in their vicinity, even when their volume was still small. However, we cannot discard a drainage effect from brain interstitial fluid to the base by gravity. In the most prominent cases, fluid was visible on axial slices ranging from the most caudal section of the brain to the ventral side of the olfactory bulb. The development of cerebral edema often involves a sharp decrease in blood–brain barrier function (Papadopoulos et al, 2000). Compromised barrier integrity was evidenced in animal model and human sepsis by monitoring the levels or transport of various markers in the brain parenchyma (Jeppsson et al, 1981; Siami et al, 2008). Various cytokines released by activated leukocytes were shown to induce increased pinocytosis by endothelial cells (Anda et al, 1997; Huynh and Dorovini-Zis, 1993) and recently a central role of complement components (C5a) in blood–brain barrier dysfunction in sepsis was shown (Flierl et al, 2009).
Apparent diffusion coefficients measured in the cortex and thalamus of septic animals indicated alterations in this parameter, which were related to the outcome. Indeed, we observed that an ADC decrease is more evident in animals that would not survive the septic challenge than in surviving animals (Figure 3). This indicates that the induction of cytotoxic edema in the cortical and thalamic areas of the brain is an important component of the pathogenesis of brain dysfunction in sepsis, as shown earlier in the hippocampal region using optical microscopy (Kafa et al, 2007). However, we cannot rule out that the cytotoxic edema is present also in other areas of the brain that were not studied in this protocol. Conceivably, the biophysical consequences of this cellular damage alone could be responsible for the decrease in ADC through the nonspecific release of intracellular components in the interstitial milieu. In addition, damaged cells would also be unable to maintain proper regulation of endothelial function. The recent identification of mitochondrial dysfunctions occurring in the septic brain (d'Avila et al, 2008), that is, the disruptions in energy metabolism and impaired chemical potential gradients across the mitochondrial membrane, may be mechanistically related to the osmotic imbalance leading to ADC perturbations and thus cytotoxic edema.
Recently it was revealed that little to no changes in ADCs could be detected in early (up to 3.5 h) periods of lipopolysaccharide-induced endotoxemia in rats (Rosengarten et al, 2008). This apparent discrepancy could arise from the differences between experimental models (Remick et al, 2000). The inflammatory processes mediated by purified lipopolysaccharide cannot
Despite the novelty of our findings, we acknowledge some limitations to our work. Our approach could become more informative if it were merged with hemodynamic observations of brain. One could argue that the observed effects of sepsis in the brain could be secondary to the hemodynamic changes frequently found in sepsis. Despite the fact that we could not monitor blood pressure during the MRI procedure, we think that brain dysfunction has a multifactorial pathophysiology and the precise role of each component, such as, inflammation, apoptosis, hypotension, metabolic alterations, and others is presently uncharacterized. Also, using a pig model of sepsis, Bogdanski et al (2000) showed brain perivascular and cytotoxic edema and neuronal death. It is important to notice, however, that hemodynamic and ventilatory parameters were strictly controlled in such a way that oxygen delivery to tissues was not limited (Bogdanski et al, 2000). Recently, it was shown that rats subjected to CLP (with a mortality rate of 43%) did not have alterations in mean whole or local brain blood flow and the authors concluded that brain dysfunction is not a consequence of changes of cerebral blood flow during severe sepsis (Hinkelbein et al, 2007).
Previous studies by Messaris et al (2004) have shown cells with apoptotic morphology in pyramidal cells of the CA1 region of the hippocampus, the cells of the choroid plexus, and the Purkinje cells of the cerebellum by hematoxylin and eosin staining and transmission electron microscopy. These modifications started at 6 h after CLP and increased in the late phases of sepsis. In fact, these authors also showed an increase of bax immunoreactivity in CA1 region of the hippocampus, in choroids plexus, and in Purkinje cells of the cerebellum early at 6 h after CLP and concluded that Bax seems to have an important role in this process, probably by leading cells to death by releasing cytochrome
Despite these limitations, the present results highlight the potential that magnetic resonance techniques have in determining the fundamental processes associated with sepsis as well as possibly its response to various therapeutic agents. The development of new magnetic resonance technique of molecularly targeted contrast agents could also be used to monitor the expression of a variety of markers of interest in sepsis pathogenesis. This approach was especially useful in determining the expression profile of the inflammatory mediator, c-Met in the brain of tumor-bearing rats (Towner et al, 2008), and could in principle be used in sepsis with only minor modifications. In particular, the acquisition of data regarding oxidative stress markers sensitive to mitochondrial metabolism is currently underway in our group, as the results presented here corroborate earlier findings about sepsis-induced mitochondrial dysregulations (d'Avila et al, 2008).
Taken together, these observations indicate that MRI represents a suitable modality to investigate inflammation-mediated complications of the cerebral microvasculature and more generally for the study of brain complications in septic mice. Significant quantitative changes were detected in the metabolic profile, brain basal vasculature integrity, and tissue ADC of septic animals. These previously unreported end points add insight into sepsis pathologic processes, and their quantitative nature may ultimately result in improvements to the accuracy of sepsis diagnostic and severity assessment in patients.
Footnotes
Acknowledgements
FAB, MFO, and HCCFN are research scholars from CNPq.
The authors declare no conflict of interest.
