Abstract
A limited number of studies suggested that in ischemic stroke patients, the number of bone marrow circulating progenitor cells (CPCs), either endothelial progenitor cells (EPCs) or CPCs, was negatively correlated with the number of infarcts as well as with the outcome. The aim of this study was to simultaneously measure CPCs and EPCs in the acute phase of ischemic stroke, and to establish whether a relationship exists with stroke severity and discharge outcome. In 67 (40 M; 27 F) ischemic stroke patients with a median age of 73 (21 to 91) years, the number of CPCs and EPCs was measured by flow cytometry and analyzed in relation to baseline NIH Stroke Scale score, ischemic stroke syndromes, and discharge outcome. Patients with partial anterior circulation syndrome showed a higher CPCs’ number with respect to patients with total anterior circulation syndrome. Moreover, a negative relationship between National Institutes of Health Stroke Scale score at the admission and CPCs number was observed. When the outcome was considered, patients discharged to home had a higher number of CPCs, but not of EPCs, compared with those moved to a rehabilitation unit. We report an association between the number of CPCs measured in the early phase after stroke presentation, neurologic severity, and discharge outcome.
Introduction
Cells derived from the bone marrow (BM-derived) have been shown to have a function in repair processes in both myocardial infarction and peripheral artery disease (Rafii et al, 2002; Ikenaga et al, 2001) and in neovascularization after brain ischemia (Hess et al, 2002; Priller et al, 2001). Within the central nervous system, BM-derived cells can give rise to all three principal cell lines: neurons, glial cells (both astrocytes and oligodendrocytes) as well as endothelial cells, thus participating in either the homeostasis of the organ or in the revascularization process (Brazelton et al 2000; Eglitis and Mezey, 1997; Zhao et al, 2002; Zhang et al, 2002). In an experimental stroke model, CD34+ cells administration accelerated neovascularization in the ischemic areas (Taguchi et al, 2004a). More recently, it has been shown that CD34+ cells may support neurologic function in conditions of ischemic stress (Taguchi et al, 2008). In one study, the number of immature BM circulating progenitor cells (CPCs), namely CD34+ and CD133+, was negatively associated with the number of cerebral infarcts and positively associated with regional cerebral blood flow (Taguchi et al, 2004b).
Endothelial progenitor cells (EPCs) number has been found low in patients defined as affected by cerebrovascular disease in two studies (Ghani et al, 2005; Chu et al, 2008). In the setting of acute cerebral ischemia, studies focused on EPCs, found an association with stroke severity or outcome (Yip et al, 2008; Sobrino et al, 2007). In one of the two studies higher EPCs’ number, counted 48 h from onset of an ischemic stroke, independently predicted a lower (<12) score on the National Institutes of Health Stroke Scale (NIHSS) (Yip et al, 2008). In the second study, the increased number of circulating EPCs 7 days and 3 months after stroke was associated with improved outcome and reduced brain damage (Sobrino et al, 2007). No study has hitherto evaluated, in the acute stroke phase, both types of BM-derived cells, CPCs and EPCs, in relation to severity and outcome of ischemic stroke.
We studied, in a series of acute ischemic stroke patients, CPCs and EPCs number, counted within 72 h from onset of symptoms, in relation to stroke syndromes and to discharge outcome.
Materials and methods
Study Population
The initial study population consisted of 110 patients who were acutely referred after stroke to the Stroke Unit of Careggi University Hospital of Florence (Italy). Patients with hemorrhagic stroke (n=9) and with a latency between the onset of symptoms and blood collection >72 h (n=34), were excluded from the study.
No patient had hematological, renal, severe hepatic, or infectious disease in the 15 days before the event. The final study population consisted of 67 patients [40 males, 27 females; median age 73 (range, 21 to 91) years].
Patients were defined as having hypertension according to the guidelines of the European Society of Hypertension/European Society of Cardiology (Cifkova et al, 2003) or if they were taking antihypertensive medications. The presence of diabetes was defined in agreement with the American Diabetes Association (Report of the expert committee on the diagnosis and classification of diabetes mellitus, 2003) using laboratory data, charts review, or by medications taken. Dyslipidemia was defined according to the Third report of the National Cholesterol Education Program, based on blood lipids values or on the fact the patients were taking antihyperlipidemic drugs, as verified by the physician (Third report of the National Cholesterol Education Program, 2002). A family history of coronary artery disease was defined as the presence of at least one first-degree relative who had developed coronary artery disease before the age of 55 years for men and 65 years for women.
All subjects gave informed consent; the study complies with the Declaration of Helsinki and was approved by the local ethic committee.
Clinical Variables and Neurologic Assessment
Stroke was defined as sudden onset of loss of global or focal cerebral function persisting for >24 h.
Etiologic stroke subtypes were classified according to the Trial of Org in Acute Stroke Treatment, and are reported in Table 1.
Clinical characteristics, vascular risk factors, stroke subtype, biochemical parameters, and progenitor cells levels of the study population

CAD, coronary artery disease; COPD, chronic obstructive pulmonary disease; CPCs, circulating progenitor cells; EPCs, endothelial progenitor cells; ESR, erythrocyte sedimentation rate; NIHSS, National Institutes of Health Stroke Scale; POAD, peripheral occlusive arterial disease; TIA, transient ischemic attack.
Values are expressed as median and range.
Therapies known to affect CPCs and EPCs number are reported.
A classification into the ischemic stroke syndromes following the Oxfordshire Stroke Classification was also performed: total anterior circulation syndrome: 14 patients; partial anterior circulation syndrome: 28 patients; Lacunar syndrome: 8 patients; posterior circulation syndrome 11 patients; undetermined: 6 patients.
Neurologic impairment, measured using the NIHSS, was assessed at the admission, during the convalescent phase and at discharge. For comparison with an earlier study (14), the neurologic impairment was defined severe when the NIHSS score at the admission was ≥12.
Blood Collection
Blood sample was taken in the morning after an overnight fasting and was collected from the antecubital vein into evacuated plastic tubes (BD Vacutainer, Becton Dickinson, Plymouth, UK) containing ethylenediaminotetracetate 0.17 mol/L for CPCs and EPCs evaluation.
Flow Cytometric Analysis
The CPCs and EPCs number was assessed contemporarily using flow cytometry, as described earlier (Cesari et al, 2008; Cesari et al, 2009).
Briefly, 200 μL of peripheral venous blood were incubated for 20 min in the dark with:
fluoresceine isothiocyanate-labeled monoclonal antibodies against human CD34 (BD Pharmingen, San Diego, California, USA) allophycocyanin-labeled monoclonal antibodies against human AC133 (Miltenyi Biotec, Bergisch Gladbach, Germany) Phycoerythrin-labeled monoclonal antibodies against human VEGFR2-KDR (R&D Systems Inc, Minneapolis, USA) Allophycocyanin-Cyanin7-labeled monoclonal antibodies against human CD45 (Becton Dickinson, San Jose, USA) LDS751, a nucleic acid dye (Molecular Probes, Invitrogen, Eugene, Oregon, USA)
Mouse isotype-identical antibodies served as controls (Becton Dickinson, San Jose, CA, USA). Red blood cells and platelets were subsequently lysed by NH4Cl lysing solution (Autolyse solution; BioSource International, Camarillo, USA). For analysis, 200,000 cells within the leukocyte gate were acquired using a FACSCanto analyzer (Becton Dickinson, San Jose, USA) and data were processed using BD FacsDiva software. Circulating EPCs were identified through their expression of CD34, KDR, and CD133 and were considered as EPCs cells CD34+/KDR+, CD133+/KDR+, and CD34+/CD133+/KDR+.
By using a modification of the International Society of Hematotherapy and Graft Engineering guidelines (Sutherland et al, 1996), CPCs were defined as cells forming a cluster with low side scatter and low-to-intermediated CD45 staining and positive for CD34+, CD133+, and CD34+/CD133+.
Statistical Analysis
Statistical analysis was performed using the SPSS (Statistical Package for Social Sciences, Chicago, IL, USA) software for Windows (Version 13.0). Values are presented as median and range. Mann–Whitney test for unpaired data was used for comparison between different groups. The outcome was defined good if the patient was discharged to home, poor if, after the acute phase, he was moved to a rehabilitation unit. To study the net contribution of EPC/CPC number as predictor of outcome, two logistic regression models were constructed. In a first model (Model 1), the effect was adjusted for age, gender, smoking habit, diabetes, hypertension, hypercholesterolemia, fibrinogen levels, and leukocyte count. In a further model (Model 2), it was adjusted also for NIHSS score at admission. Odds ratios and 95% confidence intervals are presented. A P-value <0.05 was considered to indicate statistical significance.
Post hoc sample size calculation indicated that a number of 31 and 36 patients for group A and group B, respectively, has a sufficient statistical power (β=0.85) to detect a significant difference in CPC number according to the outcome, with an α coefficient of 0.05.
Results
Baseline Characteristics
Demographic and clinical characteristics of patients and CPCs and EPCs levels are summarized in Table 1.
Significant differences for CPCs number, fibrinogen, and erythrocyte sedimentation rate levels were found between the two age groups, below and above the median age of the study population (73 years): patients older than 73 years had a lower number of CPCs, and higher levels of fibrinogen and erythrocyte sedimentation rate than younger patients (Table 2). No significant difference in EPCs and leukocytes count was observed according to age.
EPCs, CPCs, fibrinogen levels, ESR, and white blood cell count according to age
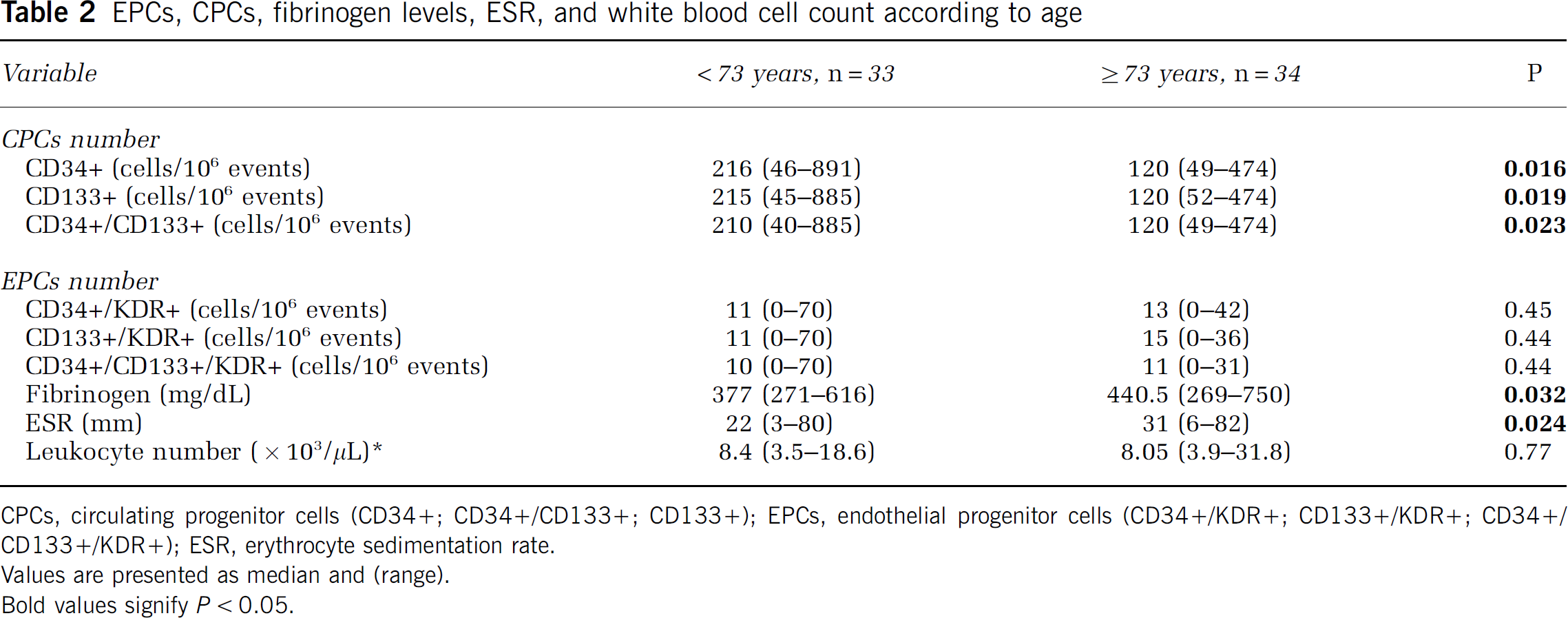
CPCs, circulating progenitor cells (CD34+; CD34+/CD133+; CD133+); EPCs, endothelial progenitor cells (CD34+/KDR+; CD133+/KDR+; CD34+/CD133+/KDR+); ESR, erythrocyte sedimentation rate.
Values are presented as median and (range).
Bold values signify P<0.05.
The presence of any cardiovascular risk factor (diabetes, hypertension, hyperlipidemia, smoking habit, gender) had no effect on either CPCs or EPCs number (data not shown). No difference in CPCs and EPCs was observed in relation to medications known for altering CPC and EPC number (statins, Ace inhibitors, and estroprogestins) (data not shown). Among comorbidities, peripheral occlusive arterial disease and heart failure significantly influenced the number of circulating EPCs: patients with peripheral occlusive arterial disease (n=3) [CD34+/CD133+/KDR+ 0 (0 to 0) cells/106 events versus 11 (0 to 70) cells/106 events; P<0.05] and heart failure (n=4) [CD34+/CD133+/KDR+ 0(0 to 0) cells/106 events versus 11 (0 to 70) cells/106 events P=0.009] had a lower number of EPCs than patients without. No significant difference was observed in relation to other comorbidities for CPCs levels.
The presence of an earlier stroke in the history determined a significant lower number of CPCs [CD34+/CD133+: 195 (46 to 885) cells/106 events versus 85 (40 to 205) cells/106 events P=0.002], but not of EPCs.
To exclude any possible influence of the different time points of blood collection on CPCs and EPCs’ number in our study population, we performed a further analysis. By dividing our study population into tertiles according to the hours of latency between the onset of the symptoms and the blood collection: 1st tertile :0 to 25 h; 2nd tertile: 26 to 45 h; 3rd tertile: 46 to 72 h, no significant differences in CPCs and EPCs’ number were observed (data not shown).
Circulating Progenitor and Endothelial Progenitor Cells Number According to Stroke Subtype and National Institutes of Health Stroke Scale
Significant difference in CPCs number was observed across subgroups of patients with different stroke syndrome: the lowest number was observed among patients with total anterior circulation syndrome, followed by patients with Posterior Circulation Infarction (POCI), and eventually by patients with partial anterior circulation syndrome or Lacunar Circulation Infarction (LACI) (Table 3).
Differences in CPCs and EPCs number, ESR, fibrinogen and leukocytes in relation to stroke subtype according to the Oxford Stroke Classification
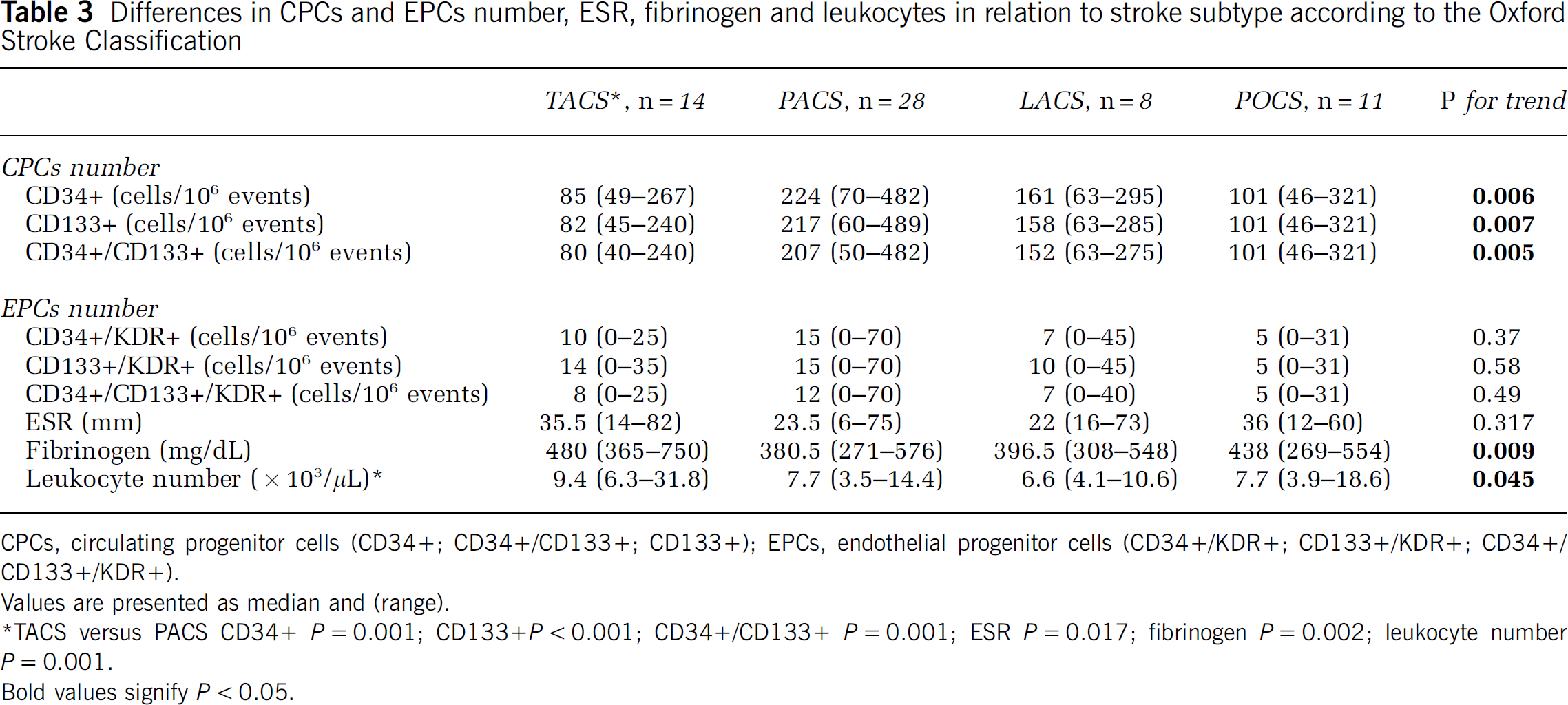
CPCs, circulating progenitor cells (CD34+; CD34+/CD133+; CD133+); EPCs, endothelial progenitor cells (CD34+/KDR+; CD133+/KDR+; CD34+/CD133+/KDR+).
Values are presented as median and (range).
∗TACS versus PACS CD34+ P=0.001; CD133+P<0.001; CD34+/CD133+ P=0.001; ESR P=0.017; fibrinogen P=0.002; leukocyte number P=0.001.
Bold values signify P<0.05.
The EPCs showed a similar trend, but the differences did not reach the statistical significance (Table 3). No significant difference in CPCs and EPCs number was observed (data not shown) across the Trial of Org in Acute Stroke Treatment subgroups.
By analyzing the relationship among CPCs, EPCs, and NIHSS score at admission, a significant negative correlation between NIHSS and number of CPCs was observed [CD34+ and NIHSS r=−0.36 P=0.003; CD133+ and NIHSS r=−0.36 P=0.002; CD133+/CD34+ and NIHSS r=−0.37 P=0.002]. No association was detected between EPCs and NIHSS score. Dividing patients by the cutoff < or ≥12 on NIHSS score, significantly (P=0.003) less CPCs were counted among patients with NIHSS ≥12 compared with patients with NIHSS ≤12 (Table 4).
Differences in CPCs and EPCs according to NIHSS score
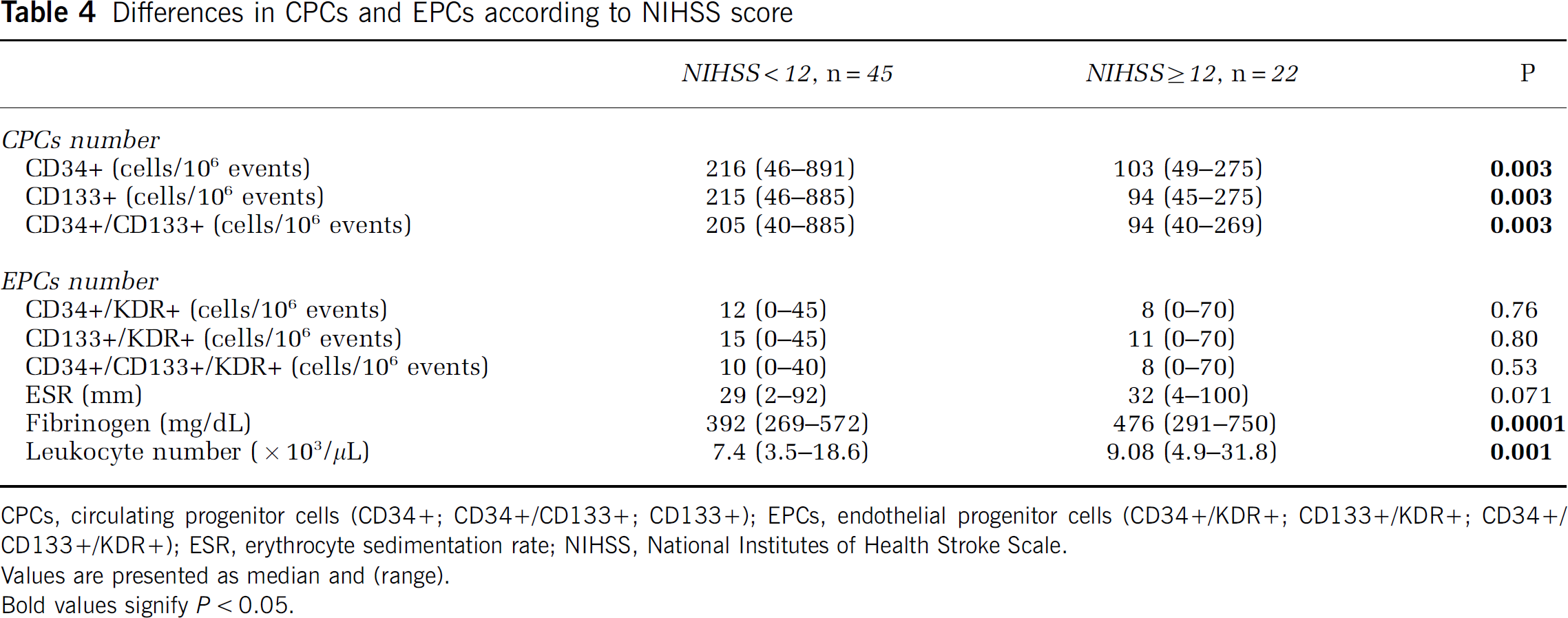
CPCs, circulating progenitor cells (CD34+; CD34+/CD133+; CD133+); EPCs, endothelial progenitor cells (CD34+/KDR+; CD133+/KDR+; CD34+/CD133+/KDR+); ESR, erythrocyte sedimentation rate; NIHSS, National Institutes of Health Stroke Scale.
Values are presented as median and (range).
Bold values signify P<0.05.
Circulating Progenitor and Endothelial Progenitor Cells Number and Discharge Outcome
Patients who were discharged to home (group A) compared with patients who were moved to a rehabilitation hospital (group B) had, in the acute stroke phase, a significantly (P=0.002) higher number of CPCs [CD34+: 229 (63 to 482) cells/106 events versus 119 (46 to 891) cells/106 events; CD133+: 219 (63 to 489) cells/106 events versus 119 (45 to 885) cells/106 events; CD34+/133+: 215 (63 to 482) cells/106 events versus 119 (40 to 885) cells/106 events] (Figure 1). A higher (albeit nonsignificant) number of EPCs was also observed in group A with respect to group B [CD34+/KDR+: 15 (0 to 45) cells/106 events versus 5 (0 to 70) cells/106 events P=0.16; CD133+/KDR+: 15 (0 to 45) cells/106 events versus 5 (0 to 70) cells/106 events P=0.19; CD34+/CD133+/KDR+ 15 (0 to 40) cells/106 events versus 5 (0 to 70) cells/106 events P=0.71] (Figure 1).
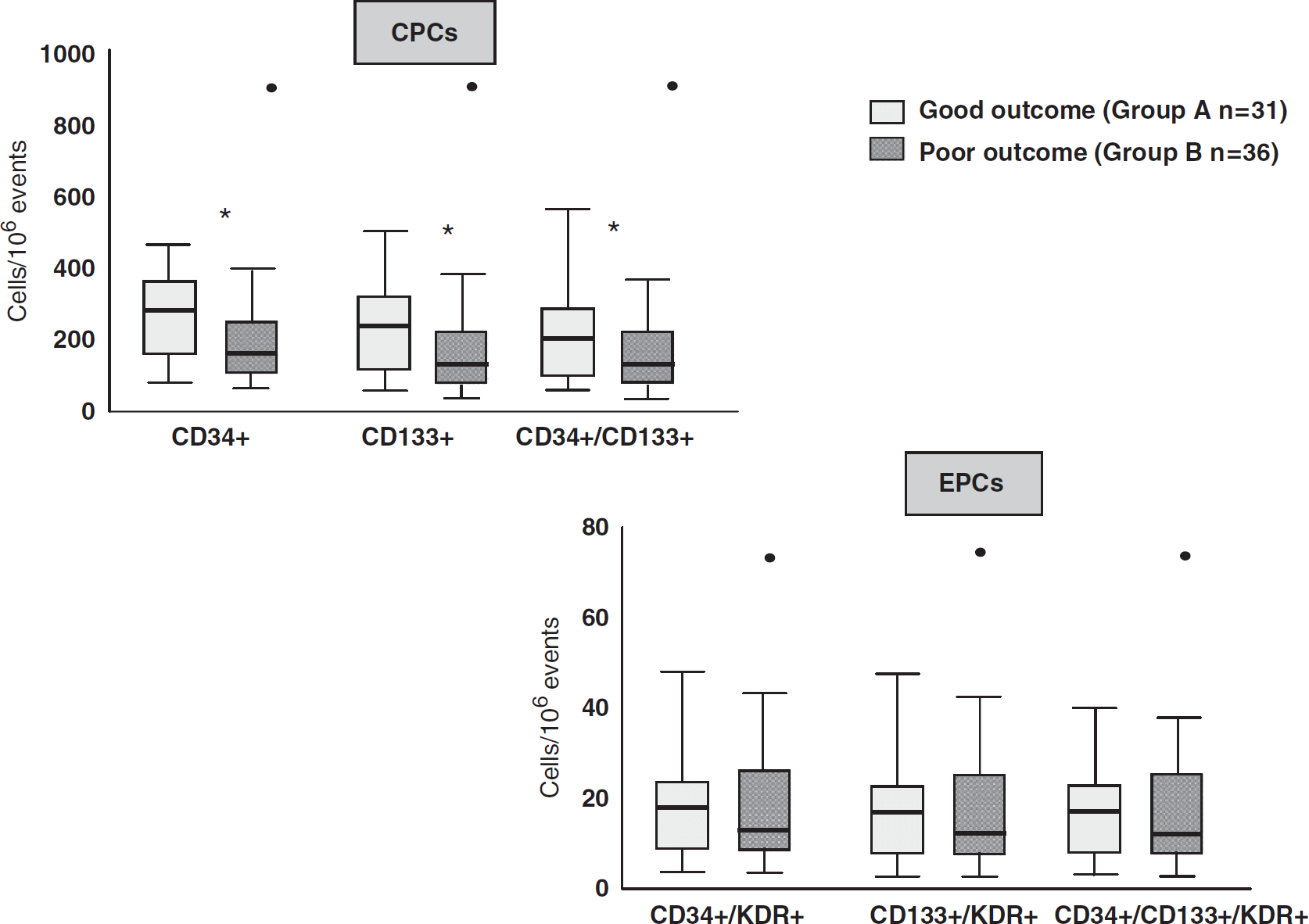
Differences in CPCs and EPCs number between group A (discharge to home) and group B (discharge to a rehabilitation hospital). ∗P<0.01. CPCs, circulating progenitor cells; EPCs, endothelial progenitor cells.
Group B showed significantly higher leukocyte count, fibrinogen levels, and NIHSS score at admission than group A [leukocyte number: 8.8 (4.1 to 31.8) versus 7.6 (3.5 to 11.8) × 103/μL P=0.045; fibrinogen 443 (280 to 750) versus 370(269 to 579) mg/dL P=0.009; NIHSS 13.5 (1 to 37) versus 3 (0 to 12) P<0.0001].
After adjustment for age, sex, hypertension, smoking habit, diabetes, hypercholesterolemia, fibrinogen levels, and leukocyte number (Table 5, Model 1), CPCs number remained significantly associated with the outcome. By adding the NIHSS score at the admission as a further covariate (Table 5, Model 2) the association with the outcome lost the statistical significance.
Univariate and multivariate logistic regression analysis in relation with discharge outcome
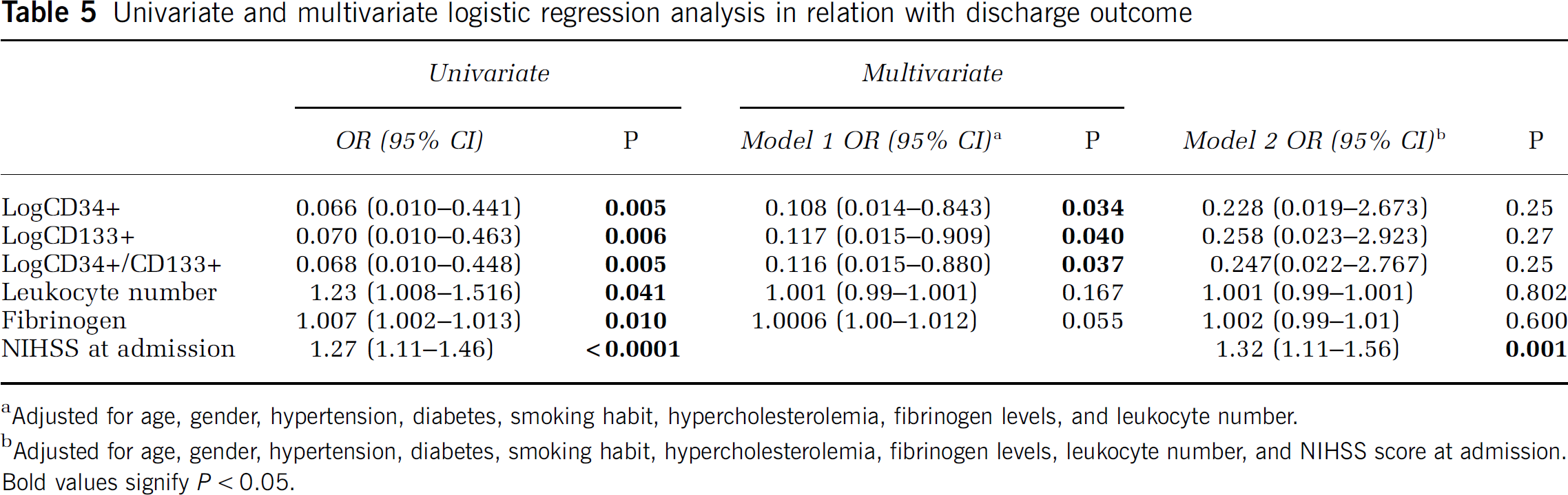
Adjusted for age, gender, hypertension, diabetes, smoking habit, hypercholesterolemia, fibrinogen levels, and leukocyte number.
Adjusted for age, gender, hypertension, diabetes, smoking habit, hypercholesterolemia, fibrinogen levels, leukocyte number, and NIHSS score at admission.
Bold values signify P<0.05.
Discussion
In recent years, BM-derived cells proved to have a definite function in stimulating angiogenesis and repair of ischemic tissues in myocardial infarction and peripheral artery disease (Orlic et al, 2001; Kalka et al, 2000). However, research on CPCs and EPCs in the stroke setting is still limited.
In this study, we evaluated both CPCs and EPCs measured early after an acute ischemic stroke in relation to stroke severity and functional outcome. We observed a relationship only for CPCs, not for EPCs: patients with less severe strokes, that is, presenting at admission with a lower NIHSS score and with a clinical syndrome indicating a smaller infarct had a higher number of CPCs. Moreover, CPCs count predicted a better functional outcome at discharge.
Although some pharmacological treatments as well as physical exercise have been shown to increase BM-derived progenitor cells number and functionality (Vasa et al, 2001; Heeschen et al, 2003; Pistrosch et al, 2005; Laufs et al, 2004), older age and conventional risk factors have been associated with low, both CPCs and EPCs levels (Taguchi et al, 2004; Cesari et al, 2008; Cesari et al, 2009). In keeping with earlier studies (Taguchi et al, 2004), there was an inverse relationship between CPCs levels and age also in our study. Indeed, a low number of circulating CPCs in relation to the increasing age is likely associated with limited vascular renewal in older individuals due to a possible BM exhaustion.
The association between lower CPCs or EPCs number and peripheral artery disease, heart failure, or previous stroke, may be attributed to a major ‘sequestration’ of these cells: in fact, an earlier ischemic event or a condition of chronic ischemia may represent a continuous stimulus for CPCs’ homing from BM into vascular wall and may determine a reduction in the number of circulating cells.
In a study performed in 25 patients, Taguchi et al (2004) observed an inverse correlation between CD34+ and CD133+ cells and the number of cerebral infarctions in ischemic stroke patients. In our study, we confirmed this finding in a larger clinical setting in relation to a validated ischemic stroke syndromes classification. Indeed, patients with a total anterior circulation syndrome and a larger infarct showed a lower CPCs number with respect to patients with partial anterior circulation syndrome and a smaller cerebral infarct. Moreover, the negative association between CPCs number and NIHSS score on admission suggest that CPCs number could provide an index of neurologic damage.
Regarding BM-derived progenitor cells, examined in the acute ischemic stroke in relation to neurologic severity and functional outcome, two studies focused on this issue, providing information on EPCs only. In the first study, conducted in 138 patients with ischemic stroke, a low number of EPCs at 48 h after the event turned out to predict a more severe neurologic impairment, defined as score ≥12 on the NIHSS, and major adverse outcomes on days 21 and 90 (Yip et al, 2008).
In the second study, Sobrino et al (2007) demonstrated that an increase between day 1 and day 7 in EPCs count was independently associated with a good functional outcome at 3 months. In that study, EPCs’ number at baseline had no prognostic value.
In keeping with Sobrino et al, in our study EPCs measured in the acute phase showed no prognostic value. Instead, we found a relationship between CPCs’ number and the outcome at discharge. Albeit we are not able to demonstrate a different timing in CPCs and EPCs mobilization from the BM, this finding might support the hypothesis that, in the early phase after stroke CPCs are mobilized before EPCs to mediate processes of acute repair and revascularization. In fact, it has been reported earlier that BM-derived CPCs can give rise, within the central nervous system, to the four main cell types including neurons (Brazelton et al, 2000), astrocytes (Eglitis and Mezey, 1997), oligodendrocytes (Zhao et al, 2002), and endothelial cells of brain vessels (Zhang et al, 2002) so providing not only revascularization of the damaged organ but also a trophic support.
Moreover, in a recent study of patients with cerebral ischemia, CPCs showed a positive impact on the course of the disease in terms of maintenance of neurologic function (Taguchi et al, 2009).
Some limitations of our study have to be acknowledged. First, the limited number of patients investigated and the short duration of observation do not allow us to obtain data on the long-term outcomes and to evaluate whether any gradient existed between number of cells and degree of functional recovery. Second, the lack of measurement of progenitor cells in our study beyond 72 h does not permit us to exclude a possible and useful role for a time-extended evaluation of progenitor cells after stroke.
These last issues, as well as others relating to the important relationship possibly existing between CPCs and brain repair in acute ischemic stroke, need to be expanded on either experimental or clinical grounds. The corroboration of this information might open insight into potential therapeutic interventions.
In conclusion, our data support and extend the role of CPCs as a potential marker of ischemic stroke severity and outcome. Larger studies with prolonged follow-up are needed to confirm our observations.
Footnotes
Acknowledgements
This work was supported by grants from ‘GENOPOLIS
The authors declare no conflict of interest.
