Abstract
Various cardiovascular operations are performed during conditions of deep hypothermic circulatory arrest. Here we investigated the effects of deep hypothermia on the small ubiquitin-like modifier (SUMO) conjugation pathway using a clinically relevant animal model of deep hypothermic cardiopulmonary bypass (DHCPB). Deep hypothermic cardiopulmonary bypass induced a marked activation of the SUMO conjugation pathway and triggered a nuclear translocation of SUMO2/3-conjugated proteins. Furthermore, DHCBP significantly modified gene expression. Activation of the SUMO conjugation pathway is believed to protect neurons from damage caused by low blood flow. This pathway may, therefore, play a key role in defining the outcome of cells exposed to DHCPB.
Introduction
Various pediatric and adult cardiovascular operations involve use of a cardiopulmonary bypass (CPB) that requires a period of circulatory arrest. To protect organs from ischemic damage, surgery is usually performed during deep hypothermia. Deep hypothermic circulatory arrest or low-flow CPB has long been practiced in the repair of congenital heart disease in neonates, and of aneurysms of the thoracic aorta and giant intracranial aneurysms in adult patients (Mahle et al, 2000; Gega et al, 2007; Levati et al, 2007; Tabbutt et al, 2008). Although the protective potential of deep hypothermia is unquestionable, little is known about the mechanisms through which it protects organs or how to maximize its efficacy. Elucidating the mechanisms underlying protection of organs by deep hypothermia is, therefore, of tremendous clinical interest. Recently, deep hypothermia of 5°C during hibernation has been found to activate the small ubiquitin-like modifier (SUMO) conjugation pathway, and evidence has been presented suggesting that this process may shield cells from damage caused by low blood flow during the torpor phase (Lee et al, 2007). This profound reduction in body temperature reduction would be lethal in most mammalian species. Whether the marked increase in levels of SUMO-conjugated proteins observed in ground squirrels during the torpor phase reflects a unique physiologic adaptability exclusive to hibernating animals and whether it only occurs at the low temperatures associated with the torpor phase remains to be established. The present series of experiments was designed to investigate whether deep hypothermia of 18°C, as used for operations involving CPB and deep hypothermic circulatory arrest, activates the SUMO conjugation pathway.
Materials and methods
This study was approved by the Duke University Animal Care and Use Committee. Normothermic (37°C) or deep hypothermic (18°C) CPB experiments were performed on anesthetized male 12- to 14-week-old Sprague-Dawley rats (n = 3 to 5/group), as described before (Jungwirth et al, 2006). Blood gases and glucose levels were recorded at various time points and did not differ significantly in animals subjected to 37°C or 18°C CPB. After 1h normothermic or deep hypothermic CPB, animals were decapitated. Brains and organs were quickly harvested, immediately frozen and stored at −80°C, before being processed for SUMO conjugation western blot analysis, as described elsewhere (Yang et al, 2008a; see also Supplementary material). Control animals were anesthetized with 5% isoflurane and decapitated, and organs were harvested and processed as for experimental animals. Brains to be used for microscopy were perfusion fixed in situ by intraaortic infusion of buffered 4% paraformaldehyde solution. After embedding in paraffin, brains were sliced. Processing of sections for confocal microscopy was performed, as described elsewhere (Yang et al, 2008b; see also Supplementary materials). The effects of transient deep hypothermia on gene expression were evaluated using Affymetrix GeneChip Rat Genome 230 2.0 Arrays (n = 3/group). For selected genes, microarray data were verified by quantitative polymerase chain reaction. Western blot analysis and polymerase chain reaction data are presented as means ± s.d. Statistical analysis of western blot data and quantitative polymerase chain reaction results were performed using analysis of variance followed by Fisher PLSD test. An extended Materials and methods section can be found at the Journal of Cerebral Blood Flow & Metabolism website.
Results and discussion
Deep hypothermic cardiopulmonary bypass triggered an activation of the SUMO conjugation pathway, as illustrated by a marked increase in the levels of SUMO1- and SUMO2/3-conjugated proteins in all organs investigated (Figure 1A; Supplementary Figure S1). The changes were more prominent for SUMO2/3- compared with SUMO1-conjugated proteins, with the cerebral cortex exhibiting the most pronounced activation of SUMO2/3 conjugation. Normothermic CPB did not induce any major change in SUMO conjugation (Supplementary Figure S1), indicating that the effect of DHCPB on SUMO conjugation was indeed caused by hypothermia. However, we cannot completely rule out a yet not identified interaction between CPB and hypothermia that could contribute to the observed findings. To evaluate the possible impact of activation of the SUMO conjugation pathway on the functional state of cells exposed to DHCPB, we investigated the subcellular location of SUMO2/3-conjugated proteins by confocal microscopy. Recently, we have found that in the brain, SUMO2/3 immunoreactivity was confined to neurons (Yang et al, 2008b). Here we illustrate that in the cerebral cortex (Figure 1B) and hippocampal CA1 and CA3 subfields (Supplementary Figure S2A+B) of control animals, SUMO2/3 immunoreactivity was present in the cytoplasm but not in the nucleus, as identified by DAPI staining. One hour of DHCPB induced a dramatic nuclear translocation of SUMO2/3-conjugated proteins in all brain structures investigated.
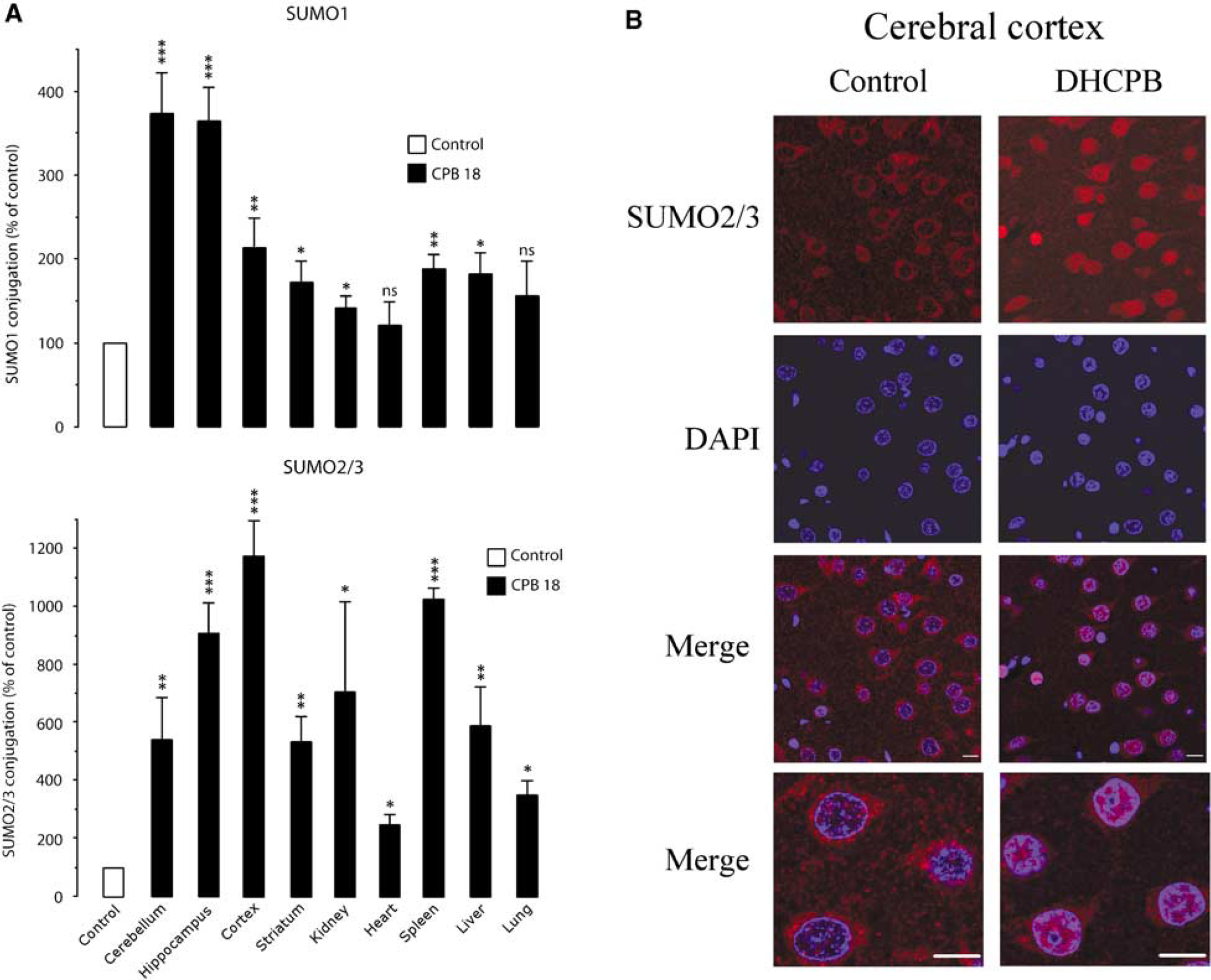
Effect of deep hypothermic cardiopulmonary bypass on levels and subcellular localization of SUMO and SUMO-conjugated proteins. (
Several SUMO conjugation target proteins identified so far have been shown to be transcription factors, and SUMO conjugation of most of these factors has been found to suppress transcription of target genes. In neurons, many SUMO conjugation target proteins have been found to be cytosolic or membrane proteins controlling important processes including kainate receptor-mediated currents, axonal mRNA trafficking, and G protein or phosphatase signaling (Martin et al, 2007). To evaluate whether DHCPB modifies gene expression, rats were subjected to 1h 37°C or 18°C CPB, and samples were taken from the cerebral cortex for microarray analysis. Expression of 43 genes was differently regulated in animals subjected to DHCPB. Deep hypothermic cardiopulmonary bypass resulted in down-regulation of several genes, where expression is associated with the pathologic process triggered by transient cerebral ischemia and culminating in neuronal cell death, including chemokines, intracellular adhesion molecule-1, interferon regulatory factor-1, and interleukin-1β and interleukin-6 (Figure 2; Supplementary Table S1).
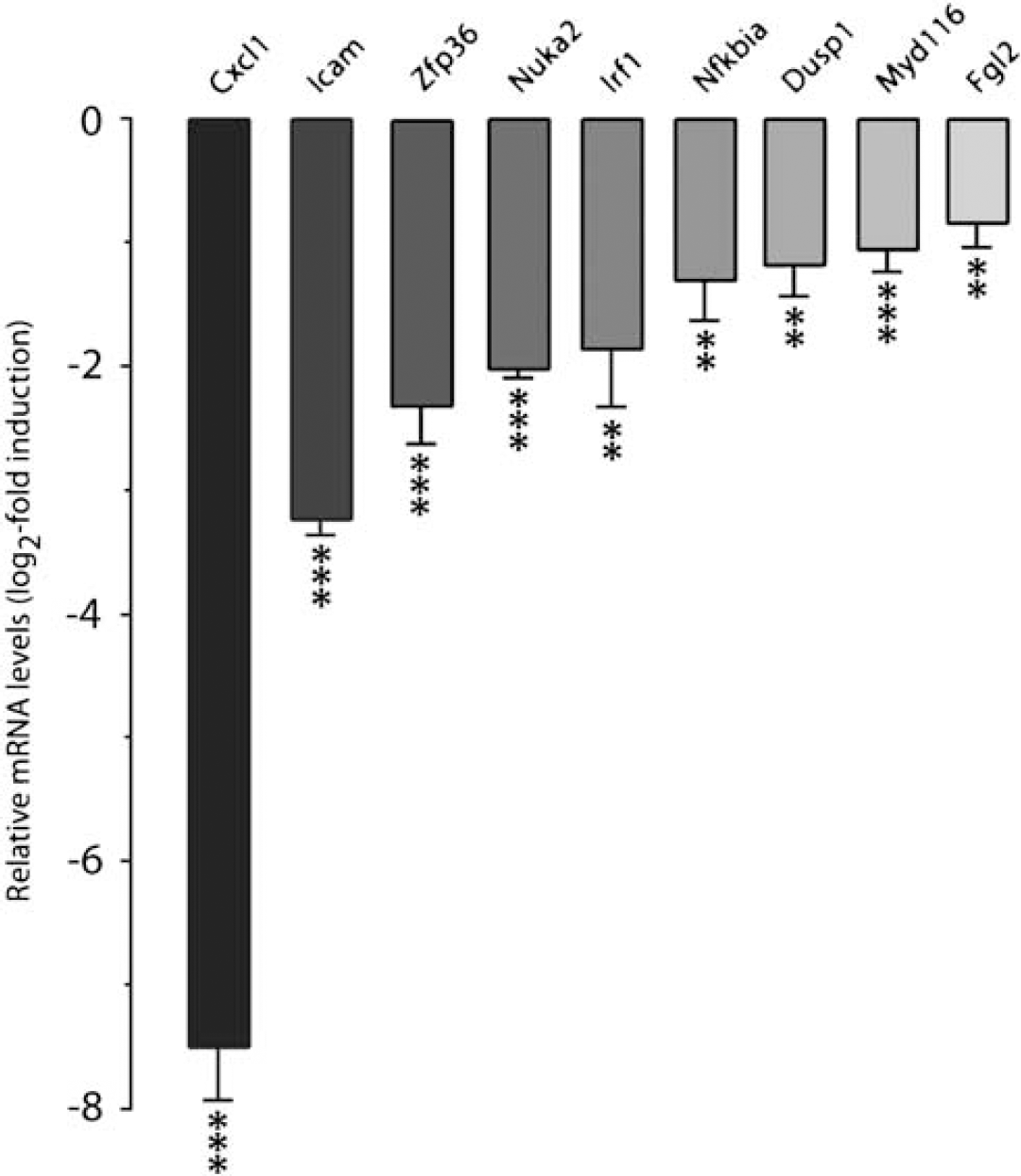
Deep hypothermic cardiopulmonary bypass (DHCPB) markedly modifies gene expression. Anesthetized rats were subjected to 18°C or 37°C CPB for 1 h. Transient hypothermia-induced changes in mRNA levels, identified using the microarray approach (Supplementary Table S1), were verified by quantitative polymerase chain reaction. DHCPB induced down-regulation of the expression of several genes associated with cell death induced by transient cerebral ischemia. The data are presented as means ± s.d. (n = 3/group). Statistically significant differences between groups were evaluated by analysis of variance, followed by Fisher PLSD test. **P < 0.01, ***P < 0.001.
The protective effects of hypothermia are best documented in experimental models of cerebral ischemia. It has been shown that even a small reduction in intraischemic temperature markedly limits the extent of ischemic neuronal injury (Busto et al, 1987). One plausible explanation for the neuroprotective effects of hypothermia could be that it protects cells from ischemic damage by retarding the rate of energy depletion during ischemia. Hypothermia has indeed been found to depress the tricarboxylic acid flux (Kaibara et al, 1999) and, thus, to preserve cerebral energy metabolism during ischemia (Laptook et al, 1995), resulting in delayed anoxic depolarization (Kaminogo et al, 1999). These observations imply that hypothermia-induced neuroprotection is a passive process whereby the rate of glucose metabolism is lowered and the time to terminal depolarization consequently increased. This would shorten the period of terminal depolarization and, thus, mitigate all pathologic processes triggered during the state of energy depletion and manifested after recovery from ischemia. However, transient hypothermia has been shown to induce rapid and delayed forms of tolerance to ischemic injury, suggesting that it can trigger an active process protecting cells from damage caused by ischemia (Nishio et al, 2000; Yunoki et al, 2002, 2003). The magnitude of tolerance induced by hypothermic preconditioning was found to be dependent on the depth of hypothermia with hypothermia set at 25.5°C producing the most pronounced effects (Yunoki et al, 2002). Therefore, it is conceivable to conclude that an even further decrease in body temperature, as performed in the present study and in pediatric and cardiovascular operations that involve CPB and require a period of circulatory arrest, will induce more pronounced tolerance towards transient ischemia. We hypothesize that hypothermia-induced activation of SUMO conjugation to target proteins may play a key role in the protection conferred on organs by deep hypothermia. If our hypothesis proves valid, that is, deep hypothermia does indeed induce an active process resulting in a rise in levels of SUMO-conjugated proteins and activation of the SUMO conjugation pathway does play a key role in organ protection provided by deep hypothermia, it could revolutionize our understanding of the mechanisms underlying the protective effects provided by deep hypothermia. If we understood the mechanisms underlying organ protection by deep hypothermia, we would be able to design therapeutic strategies to activate these processes and, thus, induce a state of tolerance to transient ischemia without risking the adverse effects associated with deep hypothermia. The SUMO conjugation could represent an exciting new target for therapeutic intervention by providing a means of increasing the resistance of organs to a transient interruption of blood supply. One way to uncover the role of SUMO conjugation in the protection provided by deep hypothermia is by elucidating the effects of silencing or activating SUMO expression on deep hypothermia-induced organ protection. Experiments are under way to verify whether activation of SUMO conjugation is indeed involved in the protective effects induced by deep hypothermia and, thus, to support our hypothesis.
Footnotes
Acknowledgements
The authors are grateful to Dr John M. Hallenbeck, National Institute of Neurological Disorders and Stroke, for providing SUMO1 and SUMO2/3 polyclonal antibodies. This study was supported from funds provided by the Department of Anesthesiology, Duke University Medical Center.
