Abstract
Ischemic and traumatic brain injuries often induce non-convulsive seizures (NCSs), which likely contribute to the worsening of neurological outcomes. Here, we evaluated the effect of glycyl-
Introduction
Patients who have sustained acute brain injuries, such as those from stroke or brain trauma, are at high risk of developing seizures in the first week after injury. Generalized convulsive seizures are readily recognizable, but non-convulsive seizures (NCSs) occur without motor manifestation and therefore are often undiagnosed. Like convulsive seizures, NCSs are ictal electrographic discharges associated with excess glutamate release, ionic imbalances, and hyperexcitability of neuronal circuits triggered by stroke or brain trauma. In an early study, Jordan (1993) reported that NCS was detected in 34% of patients in the neurological intensive care unit using continuous electroencephalographic (EEG) recording. Subsequent studies yielded similar observations (37%, Privitera and Strawsburg, 1994; 26%, Jordan, 1995; 38%, Bergsneider et al, 1997; 18%, Claassen et al, 2004; and 22%, Vespa et al, 2007), confirming the prevalence of this problem. In addition to the difficulty in detecting NCSs, treatment of NCSs is also a clinical challenge because they are more refractory to conventional anti-epileptic drug (AED) treatments than convulsive seizures (Jordan, 1995; Maganti et al, 2008; Treiman et al, 1998).
Even though NCSs are difficult to detect and treat, prompt medical intervention should be provided to prevent synergistic brain damage and worsening of the prognosis. Clinical evidence has shown that seizures in critically ill patients are harmful and can exacerbate the underlying injury processes, resulting in increasing mortality rate in stroke patients (Waterhouse et al, 1998) and worsening neurological outcomes in patients with intracerebral hemorrhage (Claassen et al, 2007; Vespa et al, 2003). This unanswered medical need to control and treat NCSs effectively in brain-injured patients underscores the importance to develop novel therapeutic drugs with both neuroprotective and antiepileptic properties.
To date, preclinical studies of brain injury-induced NCSs have been sparse despite the availability of a variety of relevant animal models that mimic human acute brain injuries. For decades, middle cerebral artery occlusion (MCAo) has been widely used to study brain ischemia, which is one of the major clinical etiologies of NCS; yet, it was only recently that MCAo-induced NCS was systematically characterized in rats (Hartings et al, 2003). In that report, 81% of the animals experienced NCS after MCAo, which manifested as generalized 1–4 Hz rhythmic spikes or ictal discharge detected by continuous cortical EEG recording. This high incidence of MCAo-induced NCSs was subsequently confirmed in studies designed to control MCAo-induced NCSs with conventional AEDs (Williams et al, 2004, 2006, 2008).
Because of the silent nature of NCS events, continuous EEG recording is required for their detection, which may pose a problem for neurological intensive care unit facilities not equipped with an EEG system. Therefore, developing drugs that are neuroprotective and that have anti-seizure activities would be more beneficial as they may be used prophylactically in the absence of EEG detection. Glycyl-
Materials and methods
Male Sprague–Dawley rats (Charles River Labs, Raleigh, VA, USA) weighing 275–350 g were used in this study. They were individually housed under a normal 12-h light/dark cycle in a well-ventilated vivarium. Food and water were provided ad libitum before and after pMCAo surgery.
All surgical procedures (EEG electrode implantation, jugular vein cannulation, and pMCAo) were performed on isoflurane-anesthetized but spontaneously breathing animals. During the surgery, the core body temperature of the animal was maintained at 37°C using a homeothermic heating blanket (Harvard Apparatus, Holliston, MA, USA).
Electrode Implantation
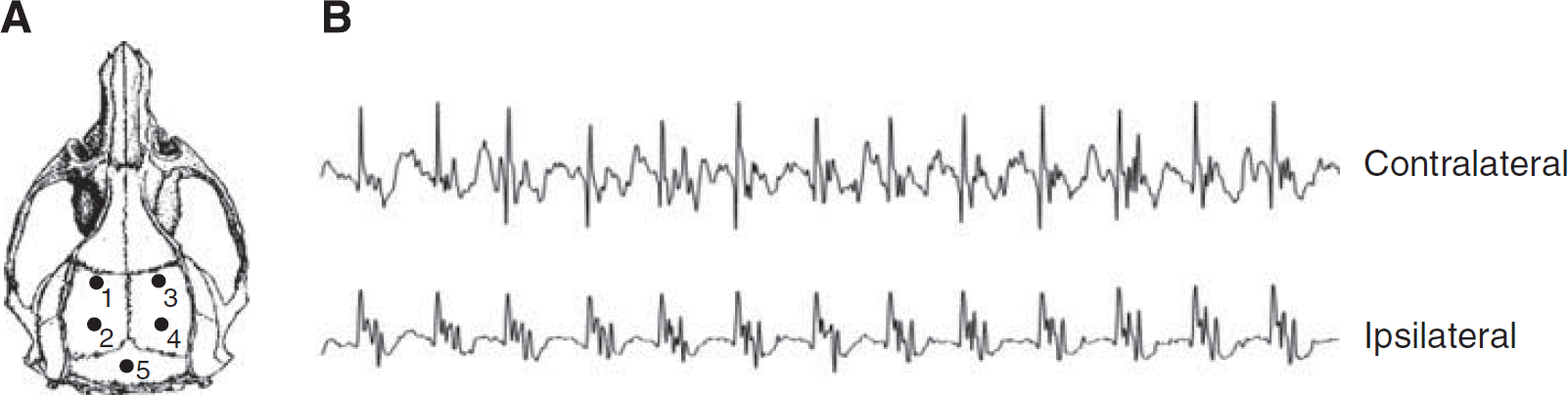
Four screw electrodes were implanted epidurally through burr holes over the bilateral parietal regions of the rat cortex (0 and 4 mm posterior to the bregma, ±5 mm lateral to the midline, Figure 1A) for measurement of brain seizure activity. A ground electrode was implanted posterior to the lambda over the transverse sinus (Figure 1A, electrode 5). Electroencephalographic electrodes were made of an insulated nichrome wire piece (0.2 mm in diameter with both tips un-insulated) soldered to a 080-1/8-inch stainless steel screw. The free end of the wire of each electrode was soldered to a multi-pin connector (March Electronics, West Hempstead, NY, USA) and the entire assembly was secured to the skull with a dental acrylic. The rats were allowed 5 days to recover before receiving the pMCAo injury.
(
Jugular Vein Cannulation
For intravenous infusion of NNZ-2566, all rats were surgically implanted with a chronic indwelling catheter made of polyethylene-50 tubing with a silastic tip inserted into the right jugular vein. The distal end of the catheter was guided subcutaneously via a trocar to exit at the dorsal neck region. The chronic infusion was implemented by connecting the indwelling intravenous catheter to a swivel via a tether (Instech Laboratories Inc, Plymoth Meeting, PA, USA) allowing free movement of the animals during infusion.
Induction of Non-Convulsive Seizure by Permanent Middle Cerebral Artery Occlusion
Non-convulsive seizures were induced by pMCAo in rats using the intraluminal filament method. Briefly, the right common carotid artery was exposed at the level of external and internal carotid artery bifurcation through a midline incision. The external carotid artery and its branches were cauterized and cut at the lingual and maxillary artery branches. A piece of 3–0 monofilament nylon suture with a round tip was inserted into the internal carotid artery via the stump of the external carotid artery. The filament was advanced approximately 20 mm until a slight resistance was encountered as it lodged in the anterior cerebral artery and therefore blocked the origin of the middle cerebral artery. The suture remained in place for the 24-h duration of the experiment.
Experimental Procedures
Electroencephalographic Recordings
On the day of pMCAo surgery, rats were placed in a custom-designed Plexiglas EEG recording chamber (30 cm × 30 cm × 50 cm L × W × H) equipped with multi-channel gold contact swivel commutators (Dragonfly Inc., Ridgeley, WV, USA). The multi-pin electrode connector on the rat skull was connected to the swivel system via a flexible shielded cable, allowing the free movement of the animals during recordings. The swivel commutator was interfaced with a Grass polygraph amplifier (Model 15, Natick, MA, USA) and a computerized EEG recording and digitizing system (Stellate Harmonie software, Montreal, Quebec, Canada). Bipolar EEG recordings were made from the left and right hemispheres through electrodes 1–2 and 3–4, respectively (Figure 1A). Baseline recordings were made from awake and freely behaving animals for at least 20 mins before pMCAo surgery. Recordings were interrupted during the pMCAo surgery but immediately resumed after the surgery and continued for 24 h. The EEG traces were displayed on a computer monitor adjacent to the EEG recording cages to allow simultaneous monitoring of the animals' behavior and EEG activity. The animals were visually observed for signs of convulsion during the initial 6 h after pMCAo.
Treatment Protocols
Two treatment protocols were used in this study. In Experiment 1, the dose response of NNZ-2566 was determined by intravenous injection of 3, 10, or 100 mg/kg given as a bolus at 20 min after pMCAo, before the first NCS event. Each bolus was immediately followed by an infusion of a fixed dose of 3 mg/kg per hour for 12 h. In Experiment 2, a bolus dose of 100 mg/kg (selected on the basis of the results from Experiment 1) was given immediately after the first NCS event was detected, and was followed by the same infusion dose as that in Experiment 1. During this experiment, if no NCS event was detected during the initial 3 h after pMCAo, the animal was excluded. All injections were delivered via a jugular vein catheter, and a matching vehicle-treated control group was used for each treatment protocol.
Electroencephalographic Data Analysis
The off-line EEG traces were reviewed at a display resolution of 30 mm/sec and analyzed by individuals who were blinded to the treatments using the NCS criteria defined previously (Hartings et al, 2003; Williams et al, 2004). An NCS episode was defined as repetitive spikes or spike-and-wave discharges recurring at frequencies of >1 Hz with amplitude greater than spontaneous activity and duration greater than 10 secs. Non-convulsive seizure episodes with an interseizure interval less than 10 secs were considered as a single event.
Based on these criteria, the following NCS parameters were included in statistical analyses: (1) NCS incidence, defined as the percentage of animals in each treatment group that experienced at least one NCS; (2) NCS frequency, defined as the number of NCS episodes experienced by individual animals; (3) NCS episode duration (secs), defined as the average duration between the onset and offset of each NCS episode; (4) NCS total duration (secs), defined as the sum of episode durations for each rat; (5) NCS onset latency (mins), defined as the time between the end of pMCAo surgery and the onset of the first NCS episode; and (6) time course of NCS frequency, defined as the hourly NCS frequency up to 9 h after pMCAo. The NCS incidence, frequency, duration, and latency were calculated as the average for each treatment group.
Histopathology
All animals were euthanized 24 h after pMCAo. The fresh brain tissue was harvested, cut into seven coronal sections, and stained with 2,3,5-triphenyltetrazolium chloride. A tissue completely lacking the red 2,3,5-triphenyltetrazolium chloride stain indicated infarct areas in the brain. The brain sections were digitally imaged using a computerized analysis system (LOATS Inc., Westminster, MD, USA) and the infarct area on each section was traced by an individual who was blinded to the treatments. The total infarct volume of each brain was computed by integrating the infarct areas of sequential brain sections.
Statistical Analysis
The difference proportion test was used to compare the NCS incidence between the vehicle-treated group and each NNZ-2566-treated group. For each NCS parameter and infarct volume, an ANOVA test followed by a Bonferroni post hoc test were used to compare the NNZ-2566-treated groups with the matching vehicle-treated control group. The relationship between infarct volumes and the NCS parameters (NCS frequency, NCS total time, and onset latency) was evaluated using Pearson's correlation coefficient test. Data are presented as the mean±standard error of the mean, and P values <0.05 were considered significant.
Results
Electroencephalographic Analysis
Visual observation of animals during the 6-h period after pMCAo confirmed the non-convulsive nature of the ictal discharges. (No animals exhibited convulsive behavior during the observation period.) As shown in Figure 1B, the electrographic characteristics of the NCSs observed in this study were similar to those previously described by Hartings et al (2003) and Williams et al (2004).
Experiment 1
Table 1 summarizes the NCS data for each treatment group. In the vehicle-treated group, 91% (10/11) of the animals experienced one or more NCS episodes during the 24-h period after brain ischemia. The NCS frequency of these animals was 22.7±6.8 events per rat and the average NCS episode was 53.0±5.6 secs in duration. This yielded an NCS total duration of 1238.3±360.0 secs per rat. The onset latency of the first NCS was 35±7.4 mins after pMCAo. In NNZ-2566-treated groups, 78% (7/9) of the animals that received 3 mg/kg bolus and 80% (8/10) of the animals that received 10 mg/kg bolus (each followed by the infusion) experienced NCS events. When the bolus dose was increased to 100 mg/kg, only 36% (4/11) of the animals experienced NCS events, which was significantly less than the percentage of vehicle-treated animals that experienced the events (Figure 2; confidence interval: 0.16–0.94, P<0.05). Glycyl-
Brain seizure statistics and infarct volume in vehicle- and NNZ-2566-treated groups
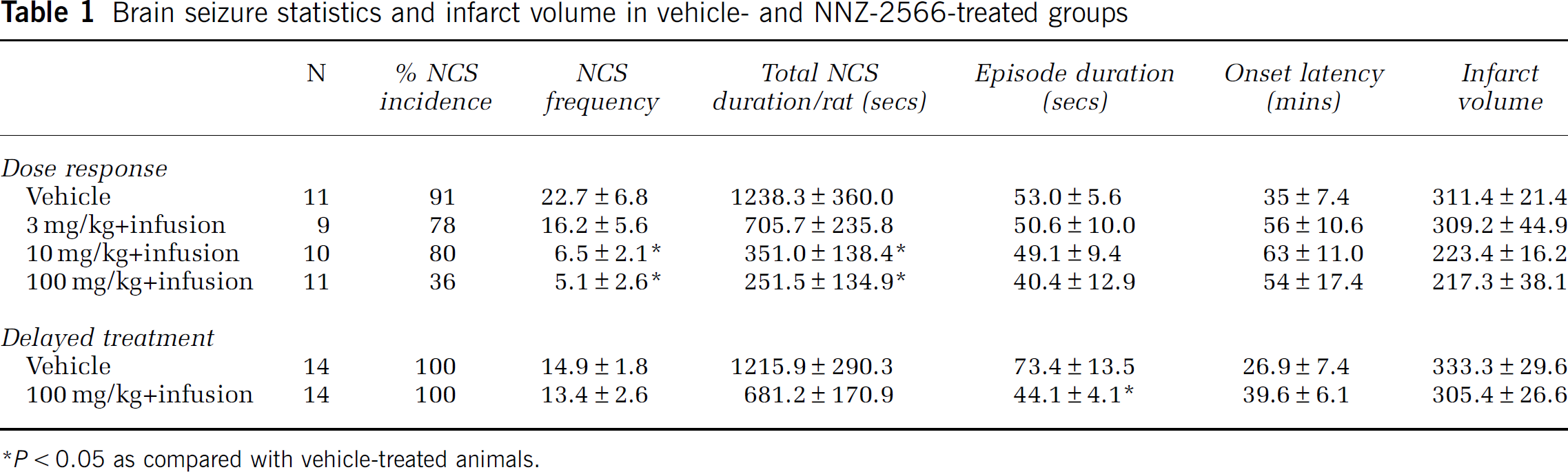
P<0.05 as compared with vehicle-treated animals.
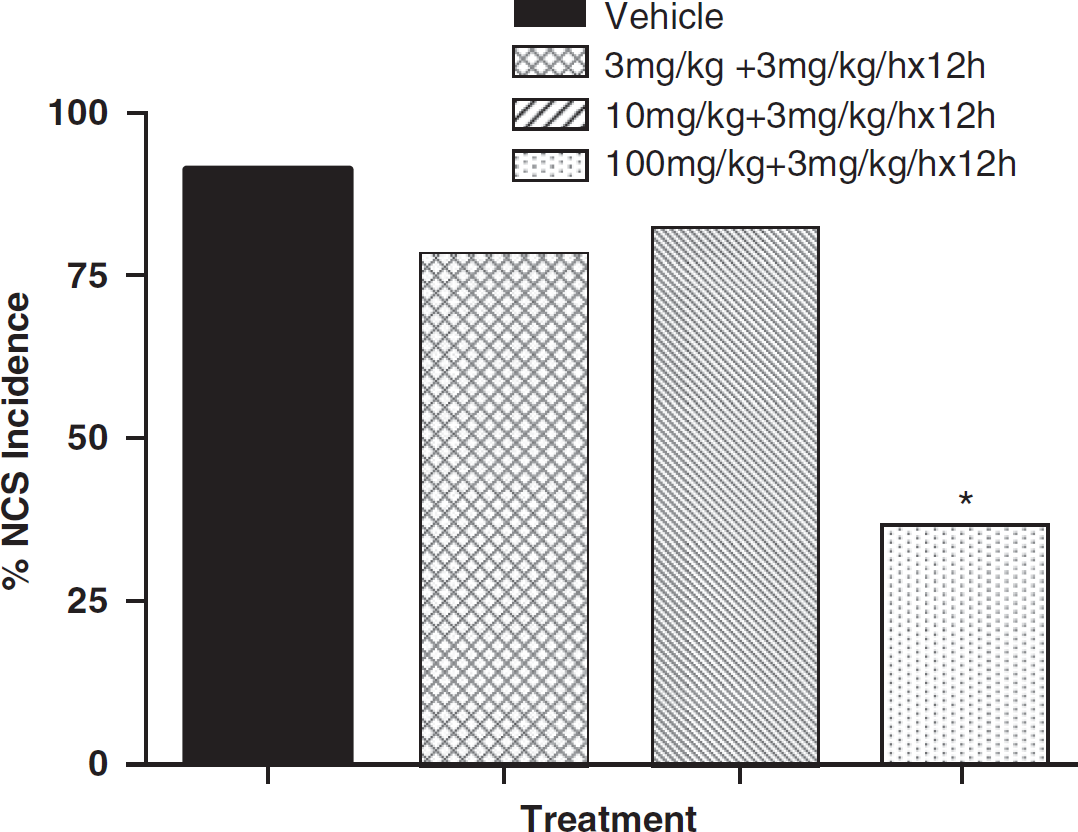
Effect of NNZ-2566 on non-convulsive seizure (NCS) incidence (percentage of animals experiencing NCS per treatment group). ∗P<0.05 as compared with the vehicle-treated group.
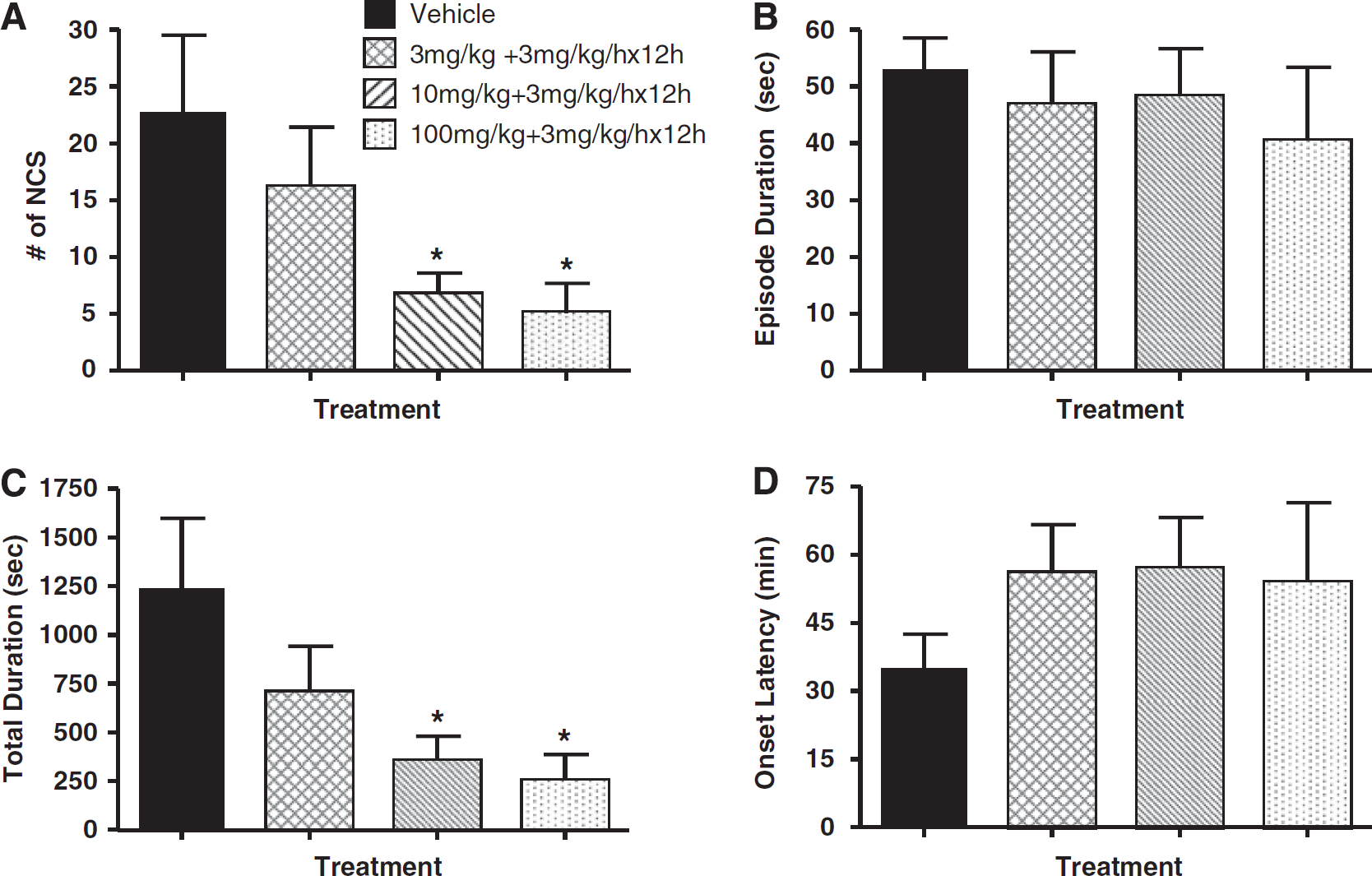
Effects of NNZ-2566 on non-convulsive seizure (NCS) frequency (
The temporal distribution of NCS events is shown in Figure 4 as the number of NCS/h per rat up to 9 h after injury (95% of all NCSs occurred during the first 9 h after injury). In the vehicle-treated animals, most of the NCS events occurred during the initial 4 h after pMCAo with an average of 3–5 episodes/h per rat. This was followed by an apparent remission period until 7 h after injury, at which time the NCS activity re-emerged at 3 episodes/h per rat. The high doses of NNZ-2566 (10 and 100 mg/kg bolus followed by continuous infusion) attenuated NCS occurring beyond 2 h after pMCAo. All three doses of NNZ-2566 completely suppressed the delayed occurrence of NCS as compared with the vehicle-treated animals.
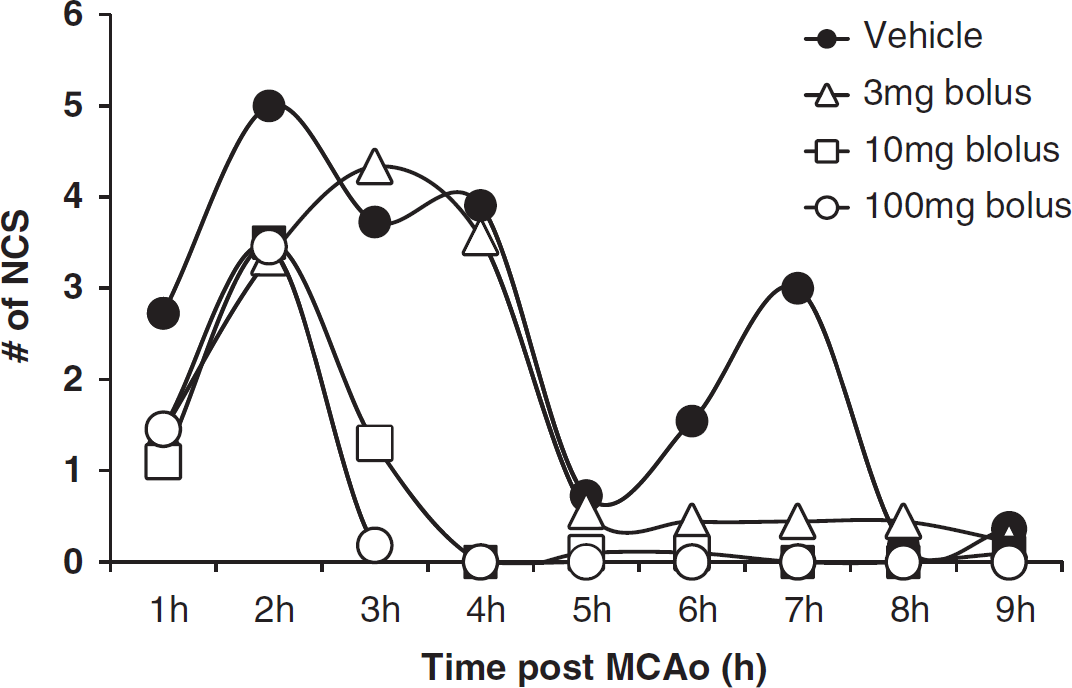
Temporal distribution of non-convulsive seizure (NCS) events as the number of NCS episodes per hour per rat over 9 h after injury.
Experiment 2
In Experiment 2, NNZ-2566 treatment was delayed until immediately after the first NCS event occurred, and only the highest dose of NNZ-2566 (100 mg/kg bolus injection followed by an infusion of 3 mg/kg per hour for 12 h) was used. As shown in Table 1, in this experiment, the vehicle-treated animals (n=14) experienced 14.9±1.8 NCS events per rat, with an average episode duration of 73.4±13.5 secs and an NCS total duration of 1215.9±290.3 secs per rat (Figure 5). Although the number of NCS events experienced by NNZ-2566-treated animals (n=14) was similar to that of the vehicle-treated animals (13.4±2.6 per rat, Figure 5A), the NCS episode duration was significantly shortened by 40% (P<0.05) with NNZ-2566 treatment (Figure 5B). As a result, the NCS total duration was greatly decreased by 44% (P>0.05, Figure 5C) as compared with the vehicle-treated animals. The onset latency of NCS before treatments was similar between vehicle- and NNZ-2566-treated groups, and the NNZ-2566 treatment failed to delay the onset of the subsequent NCS events (data not shown).
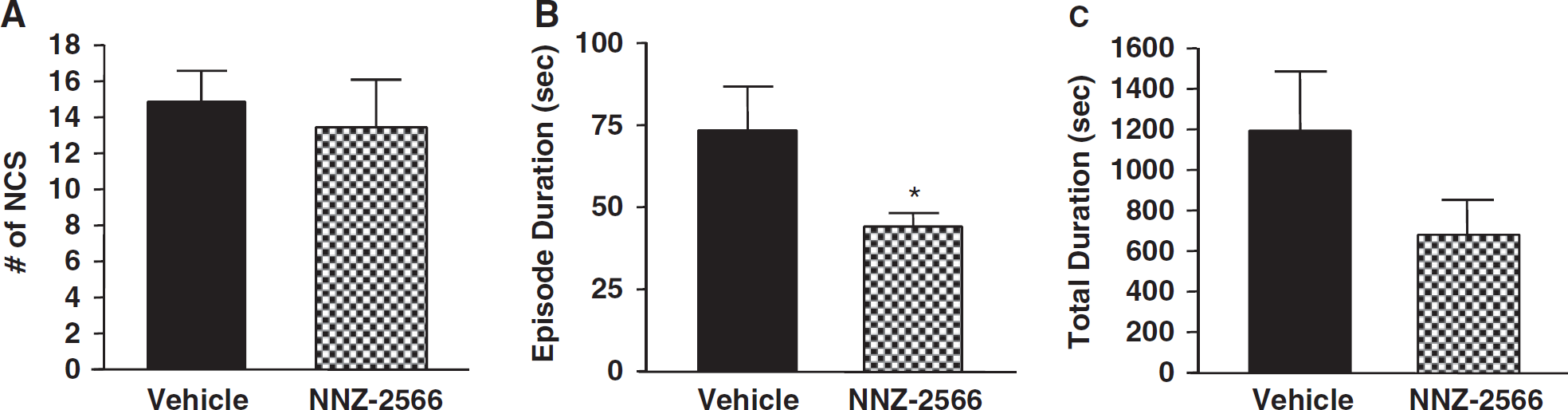
Effects of delayed treatment of NNZ-2566 on non-convulsive seizure (NCS) frequency (
Brain Infarction and NCS
Permanent MCAo resulted in severe cerebral infarction. Infarct volumes of the two vehicle-treated groups from Experiments 1 and 2 were 311.1±21.4 and 333.3±29.6 mm3, respectively (Figure 6A and 6B). In Experiment 1, treatment with NNZ-2566 significantly reduced the infarct volume by 29% (P<0.05) for the 10 mg/kg dose followed by the infusion and by 30% (P<0.05) for the 100 mg/kg dose followed by the infusion (Figure 6A). When the treatment was delayed until after the NCS occurred, as in Experiment 2, NNZ-2566, even at the highest dose, failed to reduce the infarct volume when compared with its matching control group (Figure 6B).
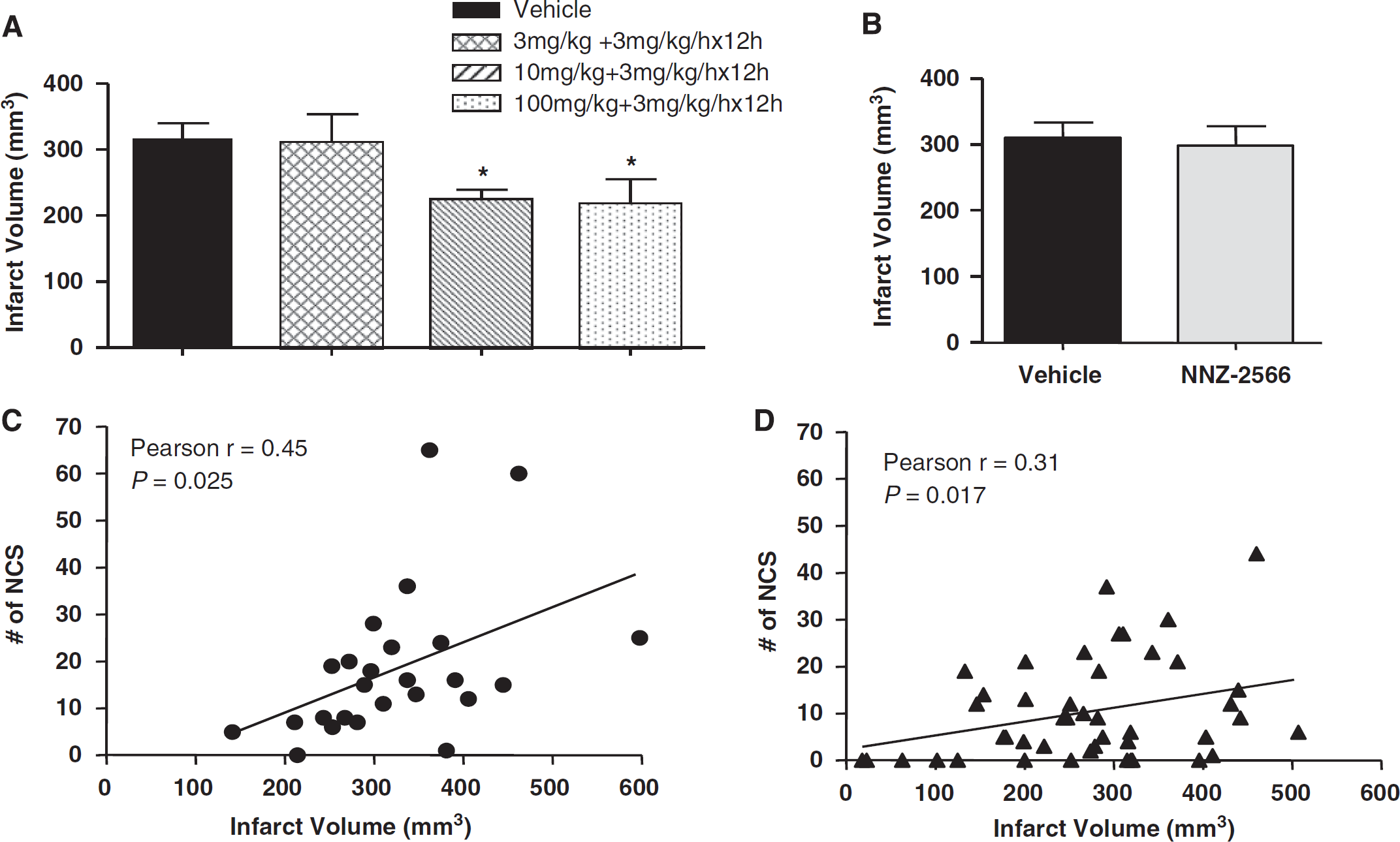
(
To determine whether the injury severity was associated with NCS severity, Pearson's correlation coefficients between infarct volumes and major NCS parameters, NCS frequency, NCS total duration, and NCS onset latency, were calculated. The results revealed that the infarct volume was positively correlated with NCS frequency in both vehicle- and NNZ-2566-treated animals when the data of all vehicle-treated animals from both Experiments 1 and 2 were combined, and the same analysis was applied to the NNZ-2566-treated animals (vehicle: Pearson's coefficient r=0.45, P<0.05, n=25, Figure 6C; NNZ-2566: Pearson's coefficient r=0.31, P<0.05, n=45, Figure 6D). However, the infarct volume did not significantly correlate with the NCS duration or onset latency.
Discussion
In this study, injury-induced NCSs were successfully inhibited or significantly attenuated by treatment with NNZ-2566. The effects of NNZ-2566 on NCS incidence, frequency and duration appeared to be dose-dependent, with significantly efficacious doses ranging from 10 to 100 mg/kg followed by a continuous infusion of 3 mg/kg per h for 12 h. The highest dose, 100 mg/kg bolus followed by the infusion, when given at 20 mins after pMCAo, inhibited NCS occurrence in 64% of the animals, significantly reduced the NCS frequency and total duration by 78% and 80%, respectively, and concomitantly reduced the infarct volume by 30%. More importantly, this dose also significantly attenuated NCSs when the treatment was delayed until after the onset of the first NCS. These brain ischemia-induced NCS activities are likely to exacerbate the injury severity because there was a moderate but significant positive correlation between the infarct size and the NCS frequency in both vehicle- and NNZ-2566-treated animals.
The ability of NNZ-2566 to reduce ischemic infarction has been reported in a recent study (Bickerdike et al, 2009), which also showed a time-dependent brain bioavailability of NNZ-2566 (30 mg/kg per hour) when the compound was infused through the jugular vein for 3 h. The peak concentration was 0.4 μg/ml at the time when the infusion was terminated and the half-life was 74 mins. The ability of NNZ-2566 to cross the blood–brain barrier (BBB) is important for its anti-seizure and neuroprotective effect; yet, its relatively short half-life makes it necessary to use the paradigm of a bolus dose followed by a continuous infusion for the full effects of NNZ-2566 to be realized. The bolus dose was very important for anti-seizure and neuroprotective effects, because the dose response effects were primarily associated with the changes in the bolus dosing while the infusion dose was fixed.
Glycyl-
Inflammation, particularly the activation of microglia and astrocytes and their production of inflammatory cytokines and related molecules, has been well documented as causing major secondary injuries after ischemic or traumatic brain injury (Amantea et al, 2009; Kadhim et al, 2008; Vezzani et al, 2008b). There is also compelling evidence that inflammatory cytokines or immune reactions after brain injury have an important role in neuronal excitability and epileptogenesis (Choi and Koh, 2008; Fabene et al, 2008; Vezzani et al, 2008a). Studies have shown that soon after an acute brain injury, activated microglia and astrocytes provide a rich source of cytokines such as interleukin (IL)-6, IL-1β, and tumor necrosis factor α (Choi and Koh, 2008; Ridet et al, 1997; Rizzi et al, 2003; Viviani et al, 2003), which promote neuronal excitability by modulating extracellular glutamate levels (Ravizza and Vezzani, 2006; Vezzani et al, 2008b). IL-1β was reported to be responsible for increasing N-methyl-
Although the effect of NNZ-2566 on BBB permeability was not investigated in this study, several previous studies have suggested that NNZ-2566 protects against hypoxic/ischemic and traumatic brain injury through attenuation of brain injury-related cerebral inflammation, i.e. reduced activated microglia cells (Guan et al, 2004; Lu et al, 2009), decreasing the up-regulation of pro-inflammation genes IL-1β, tumor necrosis factor α, and IL-6 (Wei et al, 2008) and preventing leukocyte infiltration (Lu et al, 2009), all of which are likely to be associated with preventing epileptogenesis. Therefore, it is possible that the anti-seizure activity of NNZ-2566 observed in this study was at least in part attributed to its anti-inflammation mechanisms.
An advantage of using NNZ-2566 to treat brain injury-induced NCSs is that it has been shown to be safe and neuroprotective. Many AEDs cause side effects such as hypotension, cardiac depression, and sedation, which may worsen the prognosis of some critically ill patients (Litt et al, 1998). Therefore, the benefits of using these AEDs must be weighed against the detrimental effects on the patient's condition, particularly when used prophylactically. Furthermore, not all established AEDs are neuroprotective and only a few, such as phenytoin, phenobarbital, and carbamazepine, have shown neuroprotective activities in animal models of hypoxia/ischemia (Willmore, 2005). Glycyl-
In summary, in this study we confirmed previous observations of the prevalence of NCSs after ischemic brain injury in a rat model of pMCAo. Their acute onset (<30 mins), early manifestation (within 4 h after injury), and positive correlation with the subsequent development of brain infarction warrant an emergent treatment of NCSs to prevent or ameliorate the augmentation of the brain damage enhanced by these silent seizure events. Although previous studies using clinical AEDs have shown positive results to attenuate pMCAo-induced NCS, the effects of some AEDs were confounded by their sedative actions (Williams et al, 2004). In this study we showed that NNZ-2566, a neuroprotective agent, successfully lessened the NCS severity at doses that were safe and neuroprotective. It was effective even when given after the NCS occurred. The possible mechanism of the anti-seizure effects of NNZ-2566 is likely to be attributed in part to its anti-inflammatory action, providing evidence of successful seizure control with a non-traditional AED.
Footnotes
Acknowledgements
We would like to thank Ms Angela Fleming, Ms Xiaofang Yang, Mr Paul Guevarra, and Ms Ying Cao for their invaluable assistance in the technical aspects of this study, and Ms Christine Gallo for her review and comments on the paper. These studies were supported in part by a CRADA (Cooperative Research and Development Agreement) with Neuren Pharmaceuticals Ltd. (W81XWH-05-0074).
All procedures described in this article were approved by the Animal Care and Use Committee of Walter Reed Army Institute of Research. Research was conducted in compliance with the Animal Welfare Act and other federal statutes and regulations relating to animals and experiments involving animals, and adhered to the principles stated in the Guide for the Care and Use of Laboratory Animals (NRC). The animals were housed in a facility accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International. The material for the study has been reviewed by the Walter Reed Army Institute of Research. There is no objection to its presentation and/or publication. The opinions or assertions contained herein are the private views of the authors, and are not to be construed as official or as reflecting the true views of Department of the Army or Department of Defense.
