Abstract
Interaction between angiogenesis and axonal remodeling after stroke was dynamically investigated by MRI in rats with or without sildenafil treatments. Male Wistar rats were subjected to embolic stroke and treated daily with saline (
Keywords
Introduction
The critical step for the treatment of ischemic stroke is early recanalization (Broderick and Hacke, 2002). The recanalization of occluded arteries leads to the recovery of cerebral blood flow (CBF), which helps to prevent further damage of cerebral tissue and to promote brain remodeling after stroke.
The majority of patients with ischemic stroke fail to receive thrombolytic treatment with recombinant tissue plasminogen activator because of its narrow therapeutic window, which is limited to 3 h after the onset of ischemic stroke (Grotta et al, 2001; Zweifler et al, 1998). Neurorestorative therapy, thus, is an alternative treatment for all these patients to maximize neural function of the surviving brain. Neurorestorative treatments of either cell-based or pharmacological therapies promote angiogenesis and neurogenesis in ischemic cerebral tissue after stroke (Chen and Chopp, 2006; Jiang et al, 2006, 2005; Zhang et al, 2005, 2003).
Angiogenesis in ischemic cerebral tissue elevates regional CBF, which may benefit functional outcome. Patients with higher cerebral blood vessel density appear to make better progress and survive longer than patients with lower vascular density (Senior, 2001; Slevin et al, 2000). The elevated CBF, caused by either recanalization of occluded arteries or angiogenesis, improves regional cerebral tissue microenvironments to promote axonal outgrowth, remyelination, and white matter reorganization, which may improve neurologic function recovery (Jiang et al, 2006; Zhang et al, 2005). Therefore, neurorestorative treatments may integrate angiogenesis and brain plasticity after stroke. However, few studies to date have provided evidence to show the association between angiogenesis and brain plasticity via the improved CBF after stroke.
Treatment of experimental stroke with sildenafil, a phosphodiesterase type 5 (PDE5) enzyme inhibitor, initiated at 24 h after onset of ischemic stroke, enhanced angiogenesis, neurogenesis, and synaptogenesis in ischemic rat brain (Zhang et al, 2005, 2002), all of which are associated with improvement of functional recovery compared with saline-treated rats (Li et al, 2007; Zhang et al, 2005). Magnetic resonance imaging (MRI) measurements showed that the cerebrovascular changes caused by angiogenesis in rats treated with sildenafil can be detected by T2*-weighted imaging (T2*WI) and susceptibility-weighted imaging (SWI) starting from 1 week after stroke (Ding et al, 2008). Cerebral blood flow values of the ischemic tissue along the ischemic lesion boundary area measured by MRI were significantly elevated in sildenafil treated rats compared with control rats and were associated with the induction of angiogenesis after stroke (Li et al, 2007).
Diffusion tensor imaging (DTI) of MRI provides a means for delineating the anatomic connectivity of white matter pathways and can be used to detect pathologic tract disruption based on the movement of water. Water in white matter moves more freely in the direction parallel to the tract than perpendicular to it, as axonal membranes and myelin restrict motion transverse to the tract. This diffusional directionality is known as fractional anisotropy (FA) and is directly correlated with histologic markers of myelination (Beaulieu, 2002; Mori and van Zijl, 2002; Sotak, 2002; Watanabe et al, 2001). Furthermore, increased FA appears to correlate with white matter tract integrity, whereas reduced FA is correlated with functional deficits (Beaulieu, 2002; Sotak, 2002; Watanabe et al, 2001).
Thus, MRI techniques were used in this study. Axonal changes of ischemic brain in rat after stroke were detected by DTI and FA, and angiogenic cerebral tissue was identified by T2*WI. We tested the hypothesis, with macroscopic MRI measurements, that angiogenesis in cerebral tissue locally benefits axonal reorganization after stroke in rat.
Materials and methods
Animals and Experimental Protocol
All studies were performed in accordance with institutional guidelines for animal research under a protocol approved by the Institutional Animal Care and Use Committee of Henry Ford Hospital.
Adult male Wistar rats at ages of 8 to 12 weeks, weighing 300 to 350 g, were subjected to embolic stroke by placement of an aged white clot, that is femoral arterial blood from a donor rat was withdrawn into 20 cm of PE-50 tubing and retained in the tube for 24 h (2 h to clot at room temperature and subsequently for 22 h at 4°C), at the origin of the middle cerebral artery (Zhang et al, 1997) and were randomly assigned to treatment (
Magnetic Resonance Imaging Measurements
Magnetic resonance imaging measurements were performed using a 7 Tesla system with a Bruker console (Bruker Biospin Inc., Billerica, MA, USA). A radio frequency saddle coil was used as the transmitter and an actively radio frequency decoupled surface coil as the receiver. Stereotaxic ear bars were used to minimize movement during the imaging procedure. During MRI measurements, anesthesia was maintained using a gas mixture of nitrous oxide (69%), oxygen (30%), and halothane (0.75% to 1.00%). Rectal temperature was kept at 37°C ± 1.0°C using a feedback-controlled water bath.
A tri-pilot imaging sequence was used for reproducible positioning of the animal in the magnet at each MRI session. A complete set of MRI images was performed before ischemia, repeatedly at 24 h, and weekly up to 6 weeks after embolization for all animals.
T2-weighted imaging and T2*WI were performed by multiple slices (13 slices, 1 mm slice thickness), multiple spin, or gradient echoes sequence, respectively, using a 32 × 32 mm field of view (FOV) and 128 × 64 image matrix, where all six echoes were acquired with equal interval of echo time (TE) and under the same readout gradient polarity in T2*WI. The TE was 3.6 or 15 ms for gradient or spin-echo sequence, respectively. The repetition time (TR) was 8 secs. The total time for the each sequence was approximately 9 mins.
The arterial spin labeling technique (Williams et al, 1992) was used for quantifying CBF in cerebral tissue. The adiabatic inversion of arterial water protons (Dixon et al, 1986) was accomplished via an axial gradient of 0.3 kHz/mm, and a continuous radio frequency power wave of approximately 0.3 kHz at a frequency offset of 6 kHz. This was followed by a spin-echo imaging sequence with TR/TE = 1,000/20 ms. The labeled slice was 2 cm distal from the imaging slice with 1 mm thickness. To remove the asymmetry of gradients in the axial direction, an image average was applied by switching around the gradient polarities. Field of view is 32 × 32 mm, matrix is 64 × 64. The scan time was 18 mins.
The DTI uses a two-dimensional multislice spin-echo diffusion-weighted imaging sequence. Seven diffusion-weighted imaging images were acquired for each slice, including one baseline with
Histology
Animals were anesthetized with ketamine (44 mg/kg, intraperitoneally) and xylazine (13 mg/kg, intraperitoneally), and were transcardially perfused with heparinized saline followed by 10% neutral-buffered formalin. The brain was removed and immersed in formalin solution for 1 h, after which a total of seven 2-mm-thick blocks of brain tissue were cut, processed, and embedded in paraffin.
MCID image analysis system (Imaging Research Inc., St Catherines, ON, Canada) with a × 40 objective (Olympus BX40) and a 3-CCD color video camera (Sony DXC-970MD) was used for histologic immunohistochemical measurements. Coronal sections (6 μm thick) were cut from each block and stained with hematoxylin and eosin for the evaluation of cerebral infarction. Immunostaining with antibodies against the endothelial barrier antigen was performed for quantification of cerebral vessels (Rosenstein et al, 1992). To measure myelinated axons, double Bielschowsky's silver stain for axons (modified) and Luxol fast blue (B&LFB) staining was performed (Wakefield et al, 1994). Three coronal sections and four locations of each coronal section were used for quantification. The immunoreactive areas in each location (percentage to FOV) were measured under a × 40 objective of optical microscope in the ischemic lesion boundary area and the homologous area in the contralateral side of the rat brain, and area sizes were digitized for assessing axonal density using an average of two FOVs for each location.
Data Analysis and Statistics
Image analysis of MRI was performed with homemade software, ‘Eigentool,’ in Sun workstations (Jacobs et al, 1999; Peck et al, 1992; Windham et al, 1988). All images were reconstructed using a 128 × 128 matrix. The different MRI maps and histologic section images were coregistered and analyzed using the Eigentool package. T2-weighted imaging images are referred to as reference images in coregistration, warping, and lesion size detection. Ischemic lesion size was determined by T2 maps acquired after stroke with values above mean plus two standard deviations (s.d.) of contralateral measurements (Ding et al, 2004; Hoehn-Berlage et al, 1995). The difference of the ischemic lesion sizes in T2 maps acquired at 24 h (Figure 1 A) and 6 weeks (Figure 1B) after stroke is referred to as the recovery area (Figure 1C).
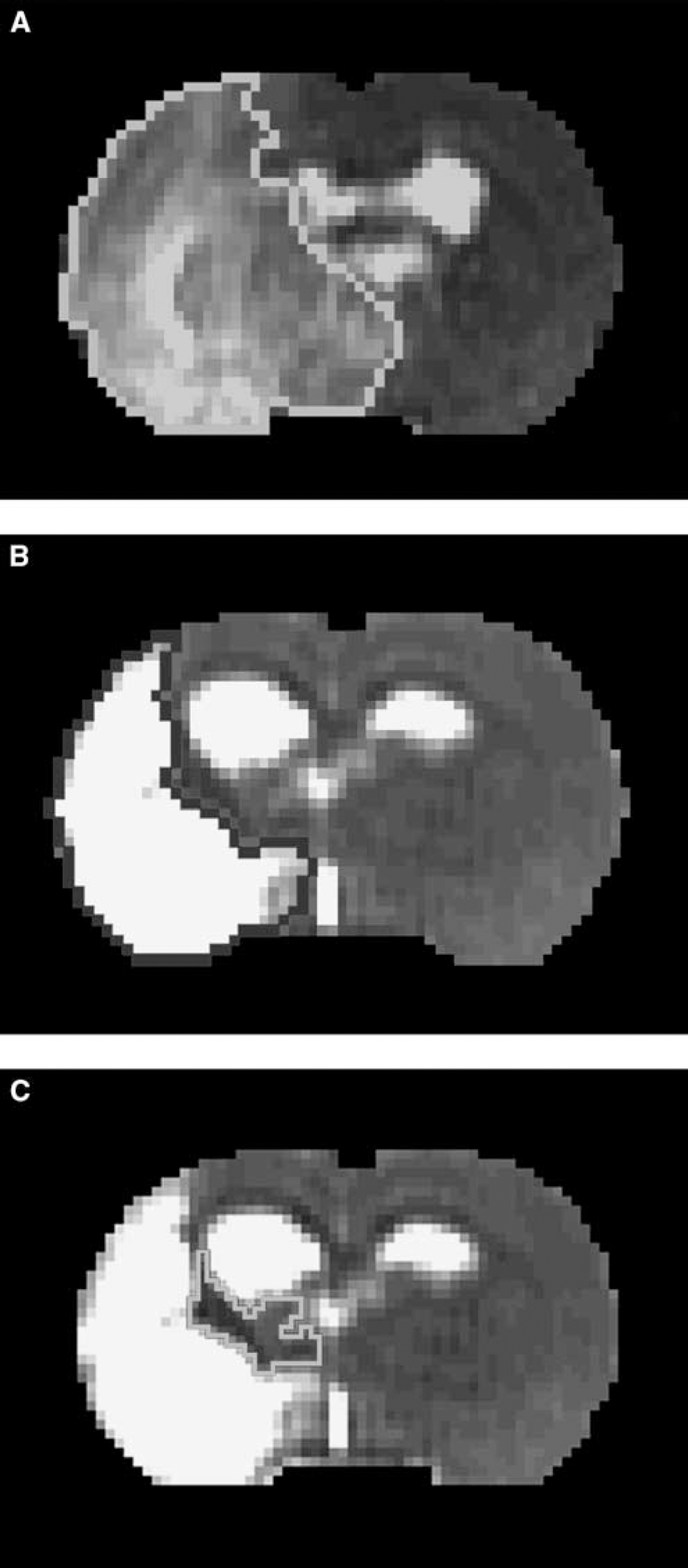
T2 map acquired at 24 h after stroke (
To quantitatively analyze MRI data, a region of interest (ROI) for recovery cerebral tissue was determined as follows: first, we measured FA values of homologous tissue in the mirror image of the recovery area on FA maps acquired at 6 weeks after stroke. Then, we acquired the ROI composed of pixels in the recovery area whose FA values are above the mean plus two s.d. This ROI was animal dependent and used to measure all MRI metrics for all time points in each animal. Using the ROI and its mirror image, MRI values of recovery ischemic tissue and homologous tissue in contralateral hemisphere were measured and used to obtain ratios. In the same way, ratios for irreversibly damaged tissue were obtained, where the ipsilateral ROI of the ischemic core was a small area located in the center of the infarct area based on a T2 map acquired at 6 weeks after stroke.
Diffusion tensor imaging data were analyzed using DTI studio software (Mori and van Zijl, 2002). Fiber tracking using DTI data started with an FA value > 0.15 and stopped with an FA < 0.15 or the turning angle greater than 65°. Fractional anisotropy maps were also produced from the conventional diffusion-weighted imaging data in three perpendicular directions (van Gelderen et al, 1994).
The area with elevated CBF at 6 weeks after stroke was identified as that above the threshold of 50 mL/100 g per min (Ding et al, 2005) within the original low CBF area measured at 24 h after stroke, because mean CBF value of recovered cerebral tissue was usually lower than that of homologous tissue in the contralateral side.
Magnetic resonance imaging observations and histologic measurements are summarized as mean value with s.d., but the group average data for MRI measurements are presented as mean value with standard error (s.e.). The differences of MRI and histologic measurements between two groups were analyzed by
Results
The quantitative MRI data of group averaged measurements (ratios of ipsilateral ROI with higher FA values to its contralateral ROI), as shown in Figures 2A to C, showed the temporal features of T2* (Figure 2A), CBF (Figure 2B), and FA (Figure 2C) for cerebral tissue with axonal changes, that is higher FA values, in stroke rats with or without sildenafil treatments. Compared with the control group, tissue with axonal changes in the sildenafil-treated group had relatively lower T2* values, and higher CBF and FA values at 1 to 6 weeks after stroke. The differences were significant (
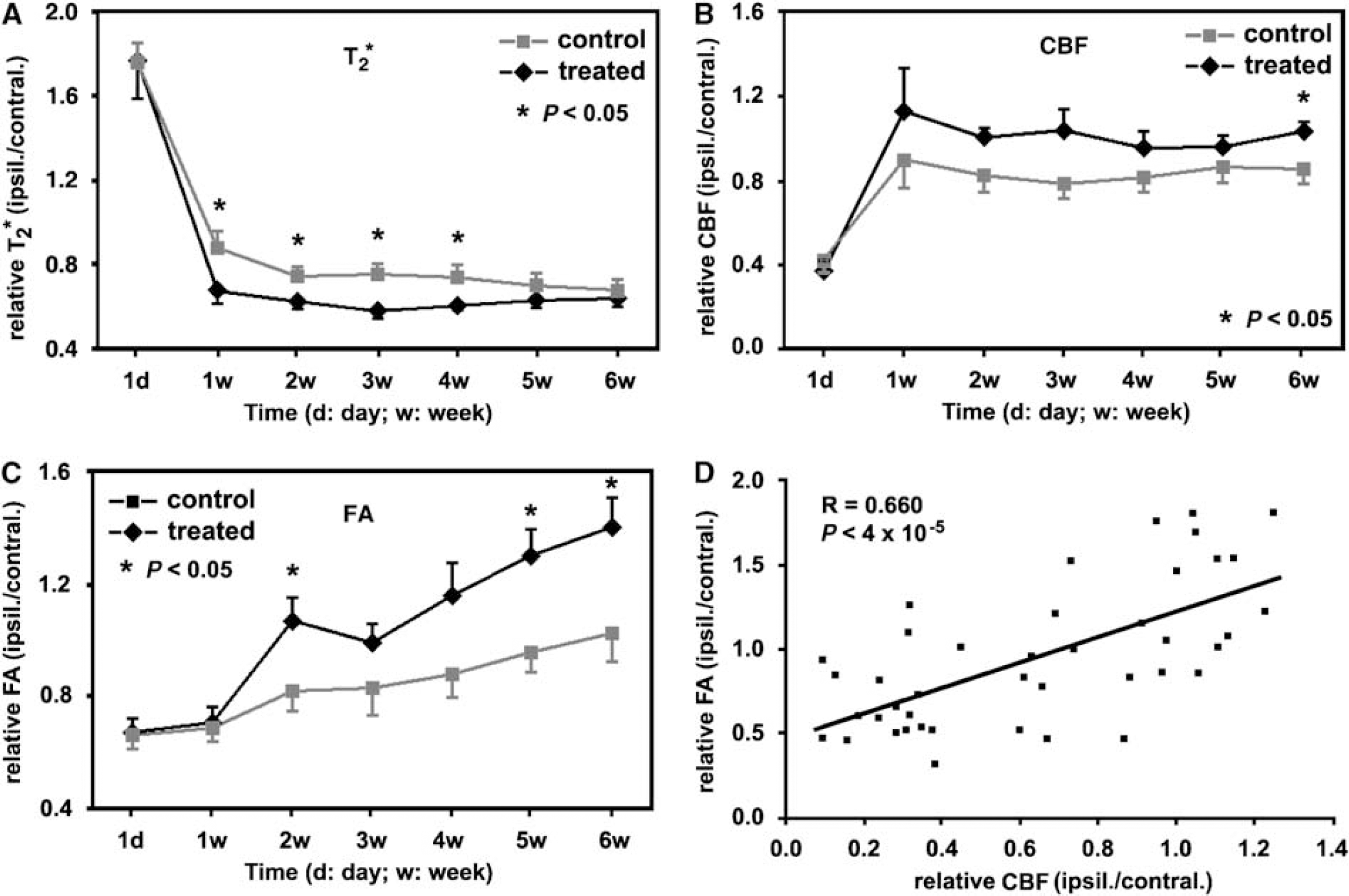
With the ROI identified hyperintensity in FA map acquired at 6 weeks after stroke, the group averaged ratios, measurements from the ROI to its contralateral ROI, showed the temporal features of T2* (
In a representative rat treated with sildenafil after stroke, areas with lower T2* values, high CBF, and FA values (indicated by red arrows in A2, B2 and C2 of Figure 3) appeared in T2*, CBF, and FA maps starting from 1 week after stroke, respectively. These areas, located in the recovery region, exhibited individual temporal profiles (red arrows in Figures 3A to C) up to 6 weeks after stroke. These temporal maps showed that areas with lower T2* values, elevated CBF, and higher FA values synchronously developed from 1 to 6 weeks after stroke. As areas destined for angiogenesis or axonal changes can be characterized by lower T2* values or higher FA values in T2* or FA maps, respectively, Figures 3A to C indicate that the cerebrovascular changes caused by angiogenesis and axonal changes were synchronous at 1 week and were MRI detectable during 1 to 6 weeks after embolic stroke in the sildenafil-treated rat, whereas the CBF value of the area was elevated at 1 week and remained so to 6 weeks after stroke. In each map acquired at 6 weeks after stroke, areas with lower T2* values, elevated CBF, and increased FA values were outlined in individual maps, as shown in Figures 3D to 3F, and all projected onto a T2 map (Figure 3G). This overlapped image showed that the locations of the areas with lower T2* values, elevated CBF and higher FA values were highly spatially correlated each other. Fiber tracking of this animal using DTI showed that the directions of axonal projections in the fiber tract at 6 weeks after stroke were highly consistent with each other and present along the ischemic boundary (Figure 3H), which was coincident with the result of hyperintensity area in the FA map (Figure 3,C7). In this case, the DTI analysis gave means of fiber length (
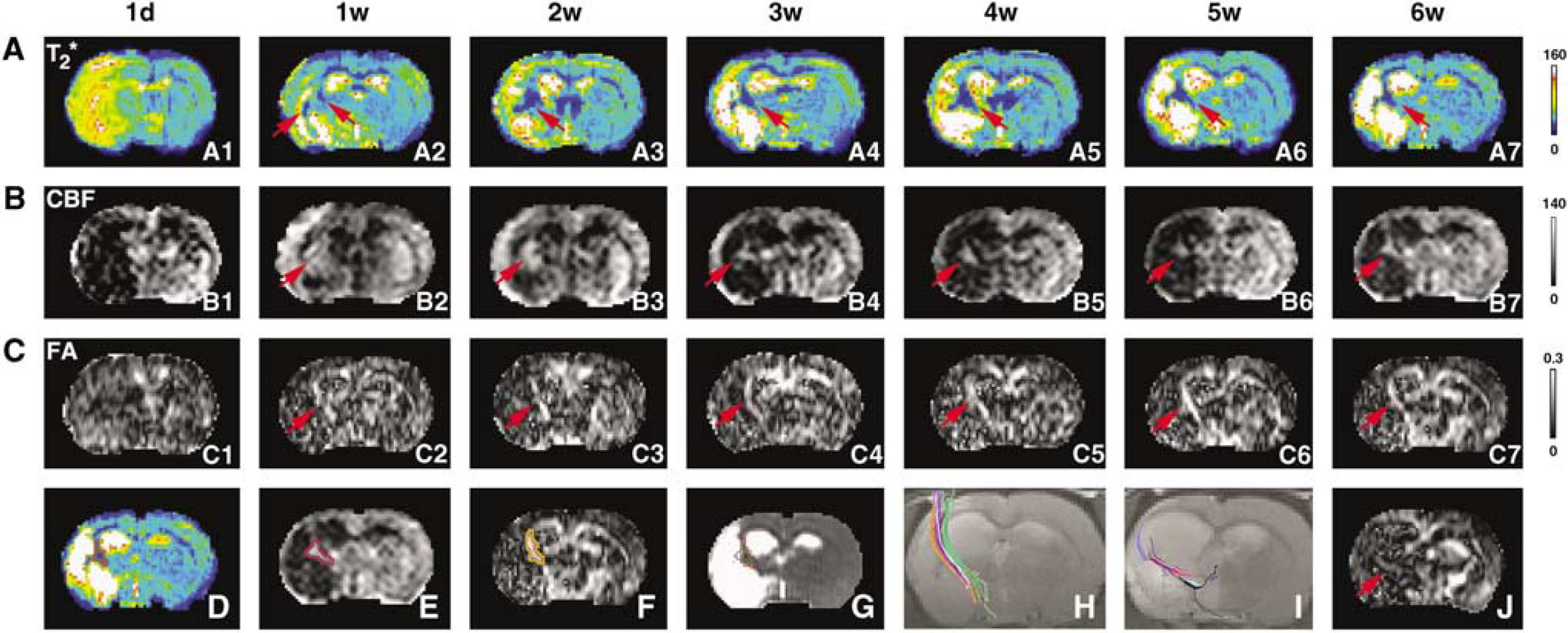
In a representative sildenafil-treated rat, T2* (
In control rats treated with saline after stroke, areas with lower T2* value, high CBF, and FA values in T2*, CBF, and FA maps along ischemic boundary, respectively, developed slower than sildenafil-treated rats, as showed with quantitative data in Figures 2A to C. The fiber tracking at 6 weeks after stroke of a control rat showed that the directionality of fibers paralleled the ischemic boundary (Figure 3I). However, the length and density of fibers tracked by the DTI data acquired and analyzed with same parameters were shorter (
Histologic measurements were consistent with MRI results. Using the endothelial barrier antigen-stained section (Figure 4A) of the representative rat treated with sildenafil, the microvascular density in the red box, which matched the area demarcated with low T2* value along ischemic boundary in the T2* map (Figure 3D) at 6 weeks after stroke, was higher (Figure 4B) than that of the homologous tissue (Figure 4C) in the contralateral hemisphere. Using the section with B&LFB staining (Figure 4D), the axonal density (Figure 4D) in the red box, which was at the same location as the red box in endothelial barrier antigen section and the area characterized by hyperintensity in FA map along the ischemic boundary (Figure 3F) at 6 weeks after stroke, was higher than that of the homologous tissue in contralateral hemisphere (Figure 4F). The 2 × enlarged image of B&LFB-stained section (Figure 4G) also showed a high density of axonal projections that paralleled the rim of the ischemic boundary in the treated rat. For the control rat, the enlarged B&LFB-stained section (Figure 4H) showed a line along the lesion border with shorter and lower density of axonal projections (Figure 4I) than the sildenafil-treated rat, and was consistent with DTI results (Figure 3I). Quantitatively, histologic data of B&LFB staining showed that axonal density increased in the ischemic boundary area compared with the contralateral side for both control and treated rats at 6 weeks after stroke. However, the increase is 46.8% ± 18.8% for treated animals and 24.3% ± 14.7% for control rats, and the difference was significant (
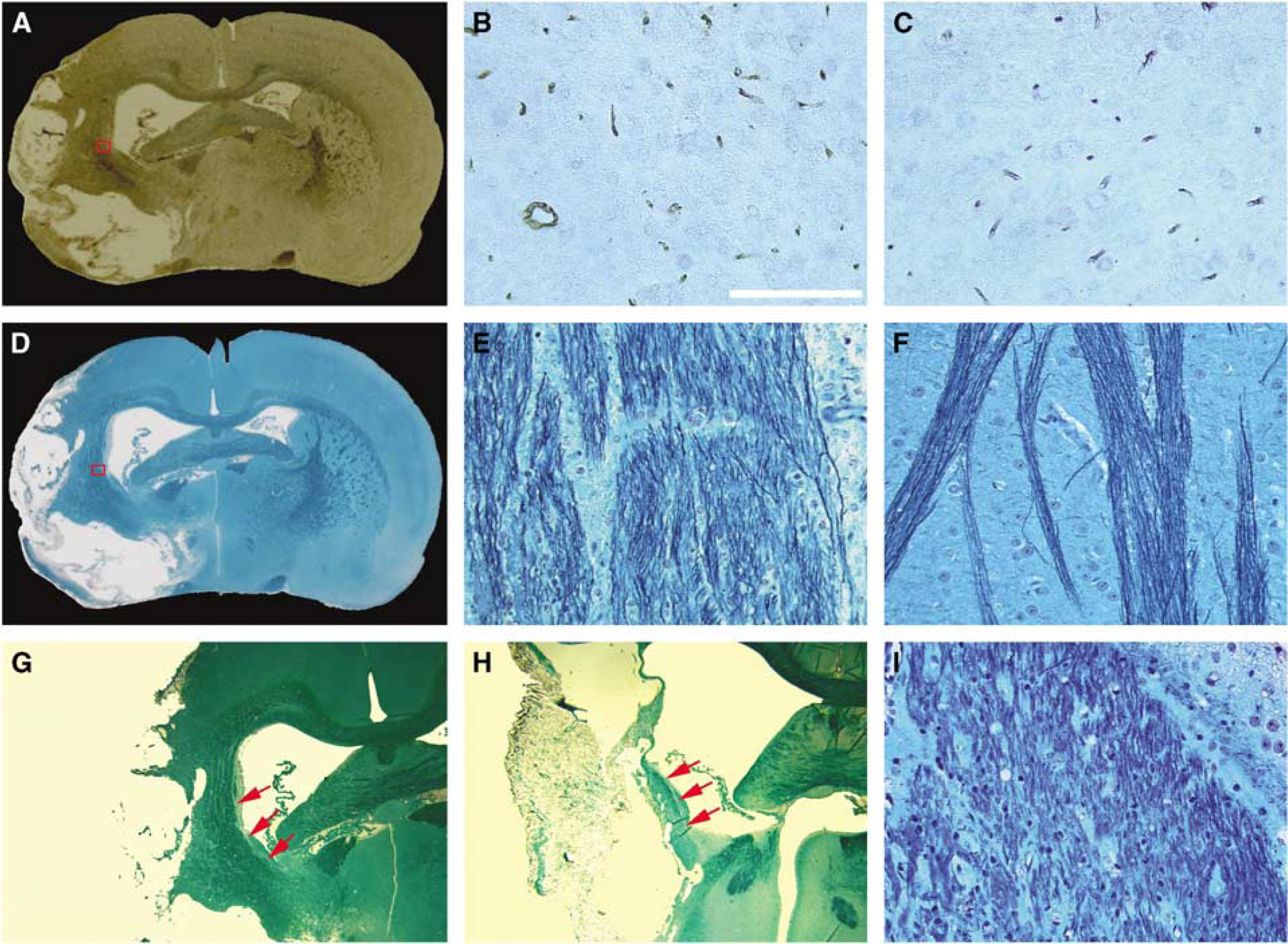
With EBA (
Performed with threshold in FA map acquired at 6 weeks after stroke and then histologically confirmed by B&LFB-stained section under a light microscope, pixels with remarkably increased FA values in the recovery area along the ischemic boundary were found in 3 out of 10 control rats and 9 out of 11 sildenafil-treated rats. The difference of axonal changes identified by higher values in FA maps between the control and treated groups of animals was significant (Fisher's test;
Neurologic functional test indicated that the treatment of stroke with sildenafil significantly (
Discussion
On the basis of the MRI data of DTI fiber tracking at 6 weeks after stroke, orientation of axonal projections in rat brain was reorganized to parallel the rim of the ischemic boundary, and FA values were increased after stroke along the ischemic boundary, which may result from the increase of both density and directionality of axonal projections. In corresponding areas, histologic data of B&LFB staining in the study confirmed that the axonal density was increased, and the orientation of axonal projections was reorganized along the ischemic boundary after stroke. Therefore, we refer to such changes of axonal density and directionality after stroke as axonal remodeling.
We recently reported that sildenafil treatment of embolic stroke in rats enhances angiogenesis in ischemic cerebral tissue after stroke and that sildenafil affects CBF differences between the ischemic core and boundary, which may contribute to the neurologic function improvements after stroke in treated rats (Li et al, 2007). To show the associations among angiogenesis, CBF, and axonal remodeling after stroke in rats, we used the same cohort of animals. Here, we show novel findings that sildenafil treatment of embolic stroke in rats amplifies axonal remodeling along the ischemic boundary after stroke, and angiogenesis, CBF, and axonal remodeling are significantly correlated with spatial and temporal consistencies.
By using MRI to simultaneously monitor angiogenesis and axonal remodeling after embolic stroke in rats, we found in this investigation that the temporal profiles of angiogenic area and axonal remodeling along the ischemic boundary were temporally and spatially coincident after stroke. Treatment of stroke with sildenafil in rats augmented angiogenesis and axonal remodeling in ischemic rat brains after embolization, as measured by T2* and FA maps, respectively, and confirmed by histology. The tractography using DTI or immunohistochemical staining of axon and myelin showed that reorganized nerve fibers after stroke was monotonically orientated along the ischemic boundary. Because CBF correlated with FA at 6 weeks after stroke, this study indicated that angiogenesis, via elevating CBF, may coordinate axonal remodeling later after stroke.
Previous studies have shown that treatment of embolic stroke in rats with sildenafil, administered subcutaneously at a dose of 10 mg/kg and initiated 24 h after stroke and continuing daily for 7 days, significantly increased the cortical cGMP (cyclic guanosine monophosphate) level by 60% in adult rats compared with saline-treated ones (Zhang et al, 2005). The elevated cGMP concentration evokes and enhances neurogenesis, angiogenesis, and synaptogenesis (Zhang et al, 2005, 2002). Administration of sildenafil 24 h after stroke also increases synthesis of vascular endothelia growth factor and enhances angiogenesis in ischemic brain via the nitric oxide-/cGMP-dependent pathway (Zhang et al, 2003). Vascular endothelia growth factor also regulates neurogenesis (Greenberg and Jin 2005). Angiogenesis and neurogenesis are linked in the adult brain via vascular endothelia growth factor and brain-derived neurotrophic factor (Palmer et al, 2000; Leventhal et al, 1999). Experiments with adult female canarys suggested a causal interaction between the testosterone-induced angiogenesis and neurogenesis in forebrain of the songbird (Louissaint et al, 2002).
In this study, T2* and FA maps detected angiogenic areas with low T2* values in the ischemic lesion (Ding et al, 2008) and axonal remodeling with high FA values along ischemic boundary (Jiang et al, 2006) starting from 1 week (Figure 2A1 and 2C1) after stroke, respectively. These MRI data indicate that angiogenesis and axonal remodeling after ischemia may be simultaneously promoted and amplified by the sildenafil treatment in rats in an early stage of recovery from stroke. Thus, in an angiogenic area where growth factors (e.g., vascular endothelia growth factor) were enhanced by sildenafil treatment (Zhang et al, 2002, 2003, 2005), axonal remodeling was also amplified. As a result, the areas with angiogenesis and axonal remodeling enhanced by sildenafil treatment spatially matched each other in later stages of stroke, for example, at 6 weeks after stroke.
With the significant development of angiogenesis (Figure 2A) that increases the density of vessels by sprouting from existing vessels and by expanding the volume of existing vessels (Figure 4B), local CBF around the ischemic boundary was significantly increased in sildenafil-treated rats compared with control animals at 6 weeks after stroke (Figure 2B).
The angiogenic area (Figure 3A7) identified by the T2* map matched the area with elevated CBF values around the ischemic border (Figure 3B7) at 6 weeks after stroke. The CBF of the treated group was consistently higher than the control group at all time points. The
Preclinical studies indicate that angiogenesis enhanced by pharmacological treatments or cell therapy of stroke promotes neurologic improvement after stroke (Jiang et al, 2005; Wei et al, 2001). In the clinic, patients with higher cerebral blood vessel density appear to make better progress and survive longer than patients with lower vascular density (Senior, 2001; Slevin et al, 2000). In this study, sildenafil treatment of embolic stroke showed that it can enhance angiogenesis to improve CBF, and then benefit axonal remodeling after stroke in the ischemic boundary area. All of these changes may contribute to the significant improvement of functional recovery in stroke rats treated with sildenafil compared with control animals up to 6 weeks after ischemia.
Axonal density (percentage of FOV) in the ischemic boundary measured by histology at 6 weeks after stroke was significantly increased (
In summary, our data in this study showed that daily treatment of embolic stroke in rats with sildenafil initiated 1 day after embolism and continued 7 days significantly augmented axonal remodeling in the ischemic boundary zone. Dynamic MRI measurements indicated that the cerebrovascular changes caused by angiogenesis and axonal remodeling were synchronously promoted and amplified by sildenafil treatment and were detectable from 1 week after stroke by T2* and FA maps. Because areas demarcated by MRI as angiogenic, with elevated CBF and axonal remodeling, were histologically confirmed, and spatially and temporally matched each other, sildenafil may independently augment angiogenesis, axonal remodeling, and CBF. In addition to the significant correlation between the FA values and the local CBF at 6 weeks after stroke (
Footnotes
The authors state no conflict of interest.
