Abstract
Hypothermia (HT) improves outcome after neonatal hypoxia–ischemia. Combination therapy may extend neuroprotection. The noble anesthetic gas xenon (Xe) has an excellent safety profile. We have shown earlier that 3 h of 50% Xe plus HT (32°C) additively gives more protection (72%) than either alone (HT = 31.1%, Xe = 10.2%). Factors limiting clinical use include high-cost and specialist administration requirements. Thus, combinations of 1 h of 50% Xe were administered concurrently for either the first (1 hImmediateXe) or last (1 hDelayedXe) of 3 h of posthypoxic–ischemic HT as compared with 3 h of 50% Xe/HT to investigate how brief Xe exposure with a delay would affect efficacy. An established neonatal rat hypoxia–ischemia model was used. Serial functional neurologic testing into adulthood was performed, followed by neuropathological examination. Xenon with HT was more effective with longer Xe duration (3 h versus 1 h) (
Introduction
Hypoxic–ischemic (HI) brain injury occurs in 1 to 6 per 1,000 live human births with a mortality of 15% to 20%, and a quarter of survivors have severe, permanent disabilities (Perlman, 1999; Volpe, 2008).
The HI insult triggers a cascade of destructive ‘excitotoxic’ processes, which include apoptosis due to prolonged activation of
Xenon (Xe), a noble gas with anesthetic properties, has shown great promise as a neuroprotectant in both
Furthermore, Xe has been approved as an anesthetic drug in Russia since 2002 (Burov et al, 2005) and was licensed for use in Germany and France in 2007, with the rest of Europe expected to follow. It is an NMDA antagonist (Franks et al, 1998; Natale et al, 2006), although its neuroprotective effects appear to exceed those of specific NMDA receptor antagonists (Ma et al, 2003), suggesting additional mechanisms of action, such as inhibition of other subtypes of glutamate receptors (Dinse et al, 2005), a general reduction in neurotransmitter release (Petzelt et al, 2003), and effects on other ion channels (Gruss et al, 2004). The possibility of Xe being involved in a process of ‘defensive’ gene induction has also been suggested as a mechanism (Garry et al, 2004).
We have previously published data from a Xenon study series where, in addition to a juvenile control group, 7-day-old (P7) rat pups were randomized to five experimental intervention groups, each of ∼20 subjects, to recover for 3 h at normothermia (NT, 37°C) or HT (32°C), either with or without 50% Xe (Xe50%) in the breathing gas after exposure to a standard HI insult (Hobbs et al, 2008a). We chose Xe50% despite Xe70% being suggested to be more neuroprotective because Xe70% also inhibits spontaneous ventilation in this model (Dingley et al, 2008). We determined that Xe or HT, in isolation, were protective long term (HT > Xe), whereas the combination of Xe and cooling conferred the greatest protection, their effects being additive. The benefit of a 70% reduction in brain pathology and improved function was sustained long term into adulthood. Functionally, the Xe-HT group did not differ from juvenile controls.
We now present the combined results of a shorter (1 h) Xe exposure starting either immediately after the HI insult or after a 2-h delay, as well as 3 h Xe combined with 3 h HT as compared with 3 h HT (no Xe) or 3 h NT (no Xe). The importance of a shorter period of Xe is financially significant, as Xe is an extraordinarily expensive gas. We found that if combined with HT, as little as 1 h of Xe was still neuroprotective after a 2-h delay in initiation. This has an important clinical implication because HT can effectively be started locally whether after delivery or cardiac arrest followed by transfer to a tertiary center for therapeutic Xe administration.
Herein, we report in a neonatal model that against the background of a 3-h cooling regime, adding Xe either for 3 h or for 1 h (with or without a delay) gives a sustained, duration-dependent histologic neuroprotection as well as corresponding functional improvement.
Materials and methods
We used the ‘Vannucci’ neonatal HI rat model based on ischemia (ligation of the left common carotid artery) followed by hypoxia (90 mins in a hypoxic chamber at 8% oxygen), which has been in use for over 25 years (Rice et al, 1981). The model displays inherent variability in the degree of brain injury between litters and strains. Specifically, within any discrete group of rat pups simultaneously receiving the same insult, there will be a wide distribution of damage, from no injury to severe. Consequently, relatively large numbers of animals must be used to convincingly show any treatment effect (Sheldon et al, 1998). We reduced experimental variability by short (60 to 90 mins) duration between carotid ligation and the start of insult, brief duration of anesthesia (< 6 mins) and well-controlled temperature, CO2 levels, and level of hypoxia by using our specially designed exposure chamber (Hobbs et al, 2008a).
Procedure
All procedures were conducted under Home Office license in accordance with UK guidelines.
P7 rat pups of either sex were subjected to a standard model of HI injury (Rice et al, 1981). Briefly, 110 pups from 10, size-culled (to 12) litters were anesthetized with halothane (3.5% induction, 1.5% maintenance) in NO2/O2 (2:1). The left common carotid artery was cut between double ligatures of 6.0 silk sutures. After > 90 mins recovery with their dam, pups were exposed to 8% oxygen for 90 mins in a temperature-controlled chamber at a rectal temperature (
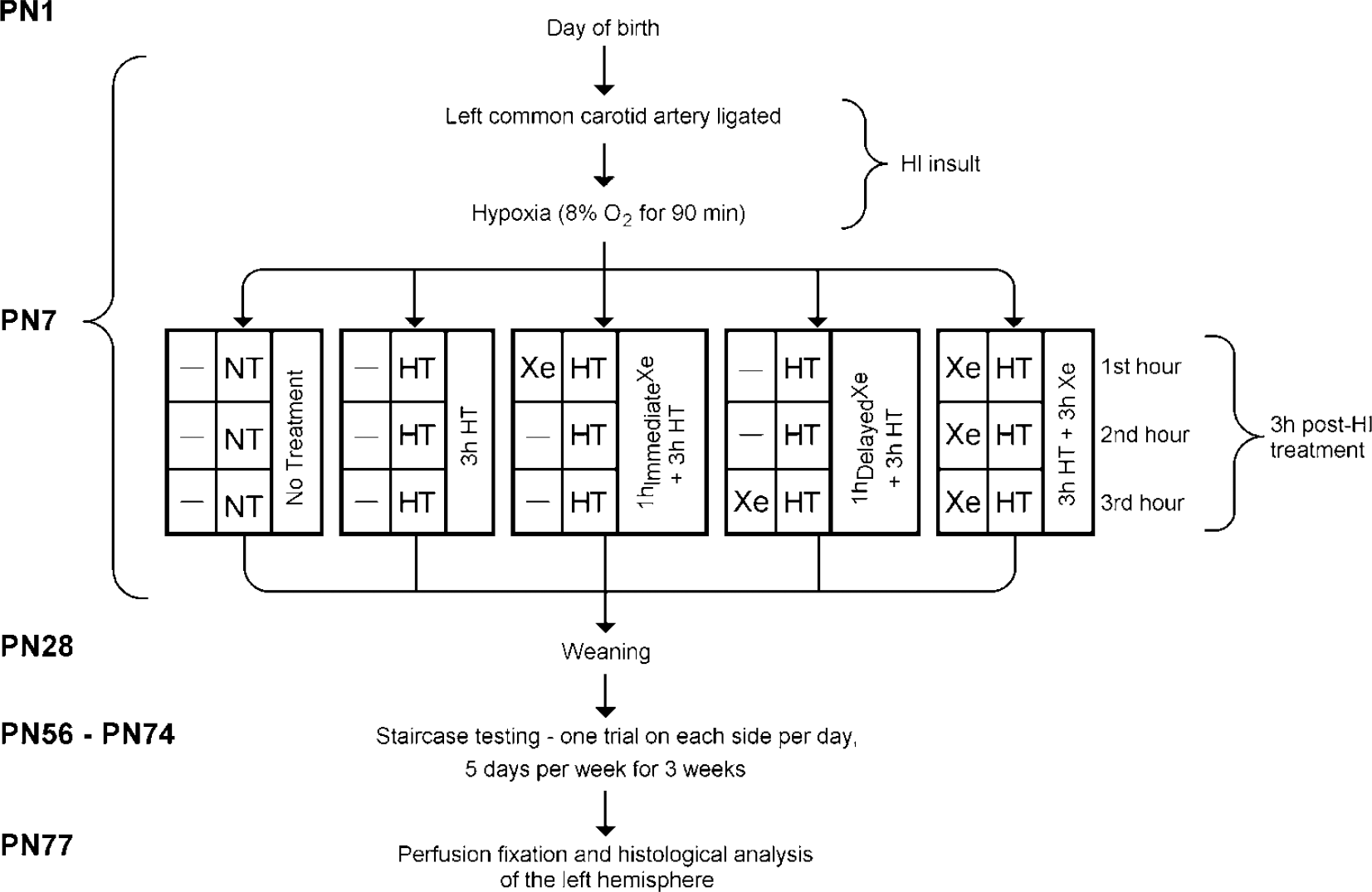
Diagram of experimental protocols during the 10-week long-term survival period.
Temperature Monitoring
In each of the five groups, one pup had a calibrated (< 0.1°C deviation) temperature probe (IT-21; Physitemp Instruments, Clifton, NJ, USA) inserted 0.5cm rectally. Rectal temperature was recorded continuously and maintained close to target temperature by varying the chamber temperature (Figure 2). Using Windows HyperTerminal software, mean rectal temperature could be analyzed at 1 min intervals throughout the experiment. These ‘sentinel’ rats were excluded from analysis, as the stress of carrying a probe has been shown to effect outcome (Thoresen et al, 1996a). Rectal temperature correlates well with brain temperature in this model (Thoresen et al, 1996b).
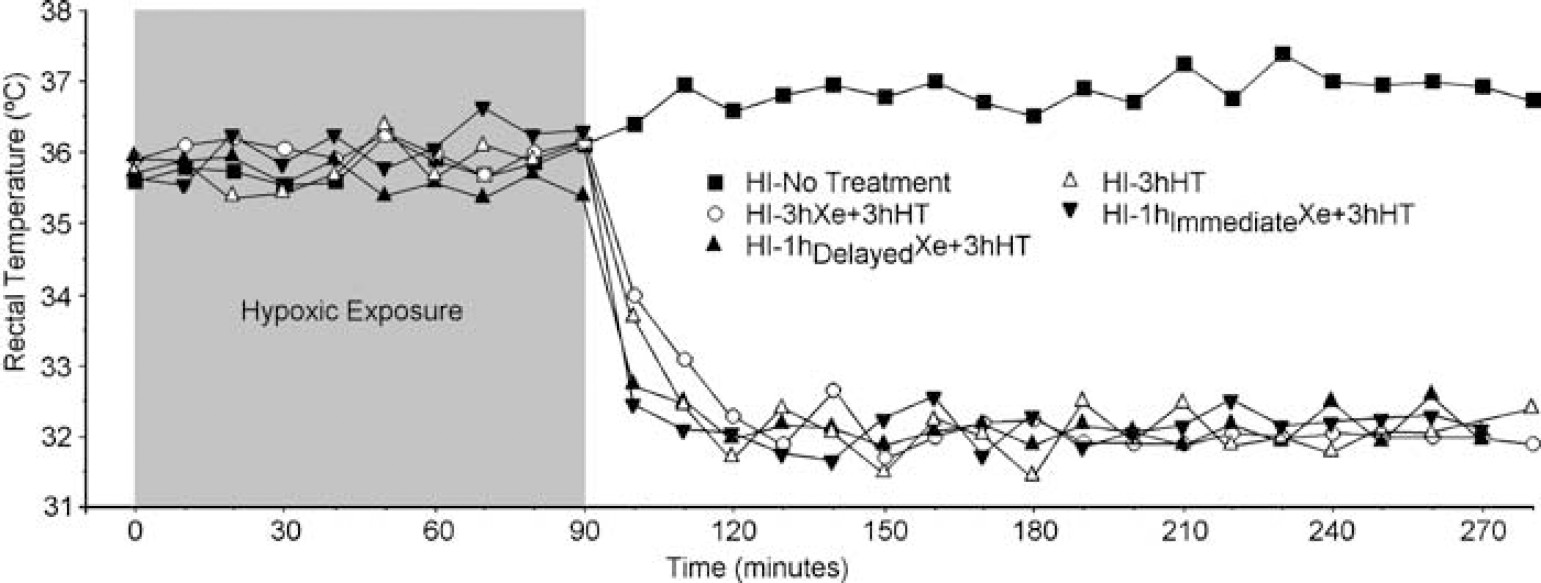
Rectal temperature recordings in the temperature probe rats during the 90-min hypoxic exposure at 36°C and 3 h recovery/treatment period at NT (37°C) or HT (32°C), with or without inhalation of 50% xenon. Data points represent 10 mins epochs.
The predicted values for regional and global pathology using the regression model with the best fit are shown
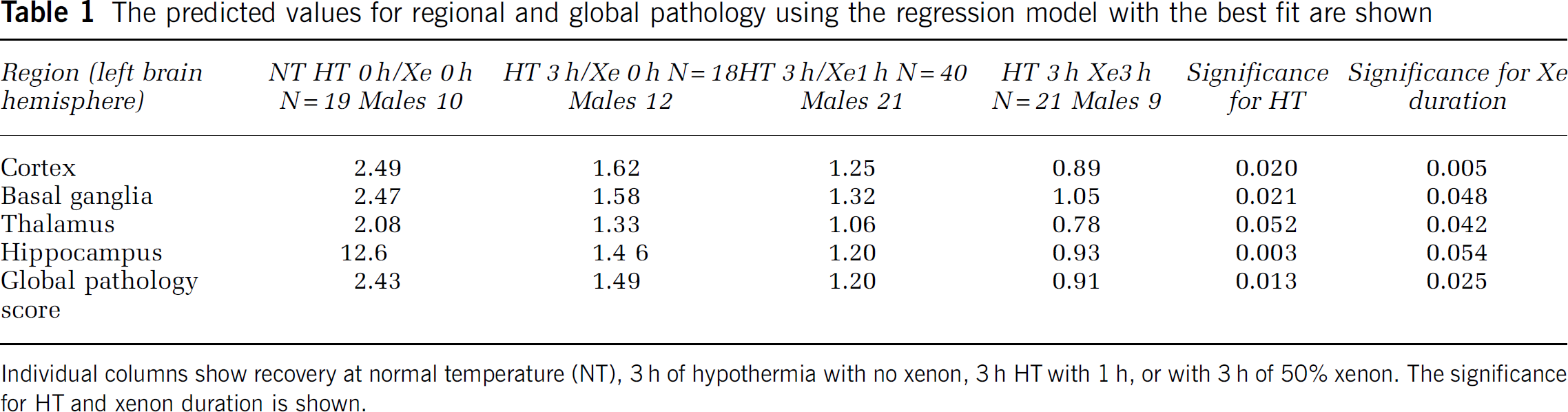
Individual columns show recovery at normal temperature (NT), 3 h of hypothermia with no xenon, 3 h HT with 1 h, or with 3 h of 50% xenon. The significance for HT and xenon duration is shown.
Temperature Monitoring and Chamber Design
Ideally, the core temperature of all rats in any group should be identical as differences in
Long-Term Behavioral Testing (Staircase Test)
Throughout weeks 8 to 11, the rats underwent long-term ‘staircase’ testing. This is a functional test of the ability to pick up and manipulate sugar pellets from descending staircases baited with pellets, where each step is more difficult to reach than the previous one, making this a sensitive test of fine motor dexterity (Montoya et al, 1991). Ligation of the left common carotid followed by hypoxia impairs the function of the right paw. Briefly, sucrose pellets (three, 45mg pellets per step; BioServ, Frenchtown, NJ, USA) were placed on each of the seven descending steps of either one of two staircases straddling a central platform. By baiting one staircase at a time, fine motor dexterity of each forepaw can be assessed independently (Hobbs et al, 2008a). Rats underwent one trial per day with each staircase baited, 5 days a week for 3 weeks. The number of pellets (a maximum of 21 pellets possible) retrieved during this time was recorded.
Histopathology
Transcardiac perfusion with 10% phosphate-buffered (0.1 M) formaldehyde was performed at 11 weeks of age under deep halothane/fentanyl anesthesia. The brains were held in 4% formaldehyde until further processing. Coronal 3mm blocks were cut through the brain using a standard matrix for uniformity (ASI Instruments Inc., Warren, MI, USA) and then embedded in paraffin. Blocks were sectioned at 6 µm and stained with hematoxylin and eosin. Four areas of the brain were examined (cortex, basal ganglia, thalamus, and hippocampus) by an investigator blinded to the treatment allocation. The severity of damage was graded from 0.0 (no injury) to 4.0 (maximum injury), with 0.5 intervals for each of the four regions, giving a nine-step scale adapted from a scoring scale used to assess injury 1 week after HI that has been validated against cell counting previously by us and used by others (Dingley et al, 2006; Haaland et al, 1997; Hobbs et al, 2008a; Thoresen et al, 1996a; Thoresen et al, 1996c; Trescher et al, 1997).
Statistics
In the figures data are given as mean±s.e.m.
Data were checked for normality before being analyzed using SPSS version 15. Variables were compared using analysis of variance with Bonferroni's correction for multiple comparisons. To investigate the nature of any interaction between Xe, HT, and gender, we used a full factorial linear model. We investigated between-subject effects of staircase testing, regional and global pathology, and gender. A
Results
Pathology Results
Four areas in the brain were examined in both frontal and dorsal regions (cortex, hippocampus, basal ganglia, and thalamus) using our validated pathology score (Hobbs et al, 2008a; Thoresen et al, 1996b).
Figure 3 shows regional pathology score as well as the summated ‘global’ pathology score for the five treatment groups. There was little difference in the pattern of neuroprotection between the regions. As previously published (Hobbs et al, 2008a), 3 h of hypothermic recovery exerts a ∼35% improvement compared with animals that recover at normothermia, combining 3 h of Xe with 3 h of HT gives additive neuroprotection of ∼70%. In two more groups, only 1 h of Xe was administered during the 3-h HT period, this 1 h was either administered immediately after the hypoxic period or with a 2-h delay. There was no significant difference in the degree of histologic neuroprotection whether the 1 h of Xe was initiated immediately after the HI insult or after a 2-h delay (
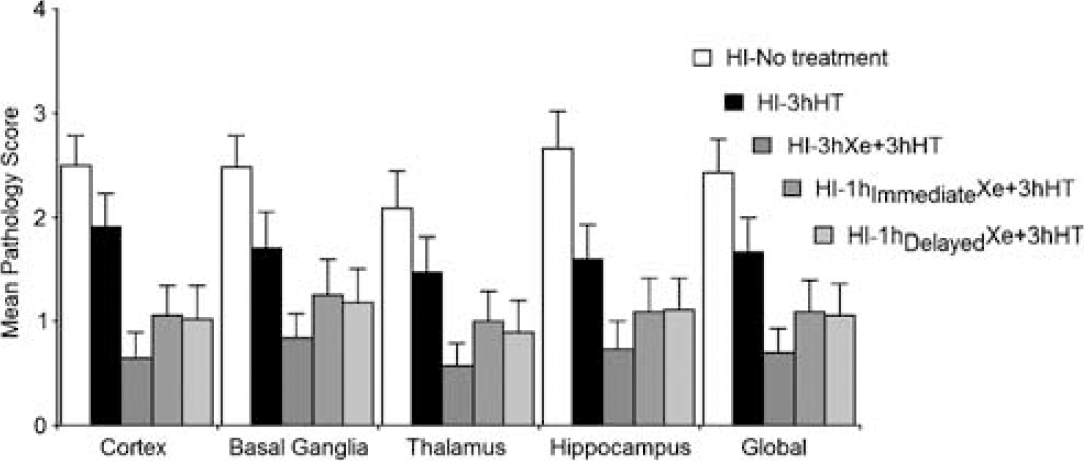
Mean (s.e.m.) pathology scores from different regions of the left hemisphere of haematoxylin and eosin-stained sections evaluated at 11 weeks of age (i.e., 10 weeks after the HI insult). The global (average of all four regions) pathology is shown to the very right.
Staircase Testing
Figure 4 shows the mean number of pellets retrieved during the last 3 days of testing at 10 weeks of age for each treatment group. The results from juvenile controls are included in the graph for clarity; there is no gender difference in healthy pups (Hobbs et al, 2008a). We found no gender difference in the group that was subjected to the insult followed by recovery at normal temperature. There was, however, a large (females picked up 3.2 more pellets) and significant (
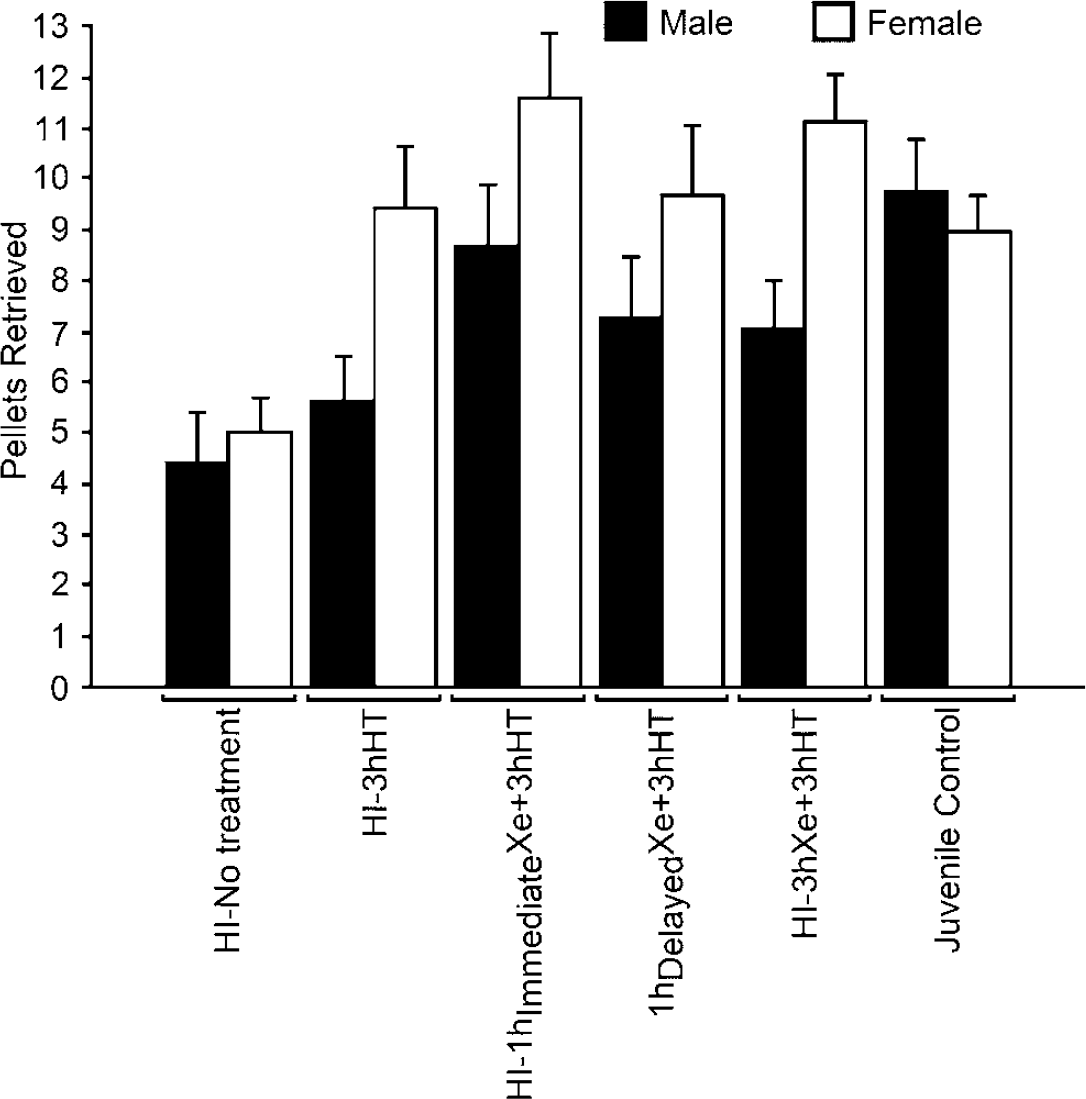
Mean (s.e.m.) number of pellets retrieved during the last 3 days of testing with the right forepaw from steps of increasing distance in the staircase test, 10 weeks after the hypoxic–ischemic insult (adulthood). Data are split on gender.
Coefficients found for the linear regression model with predicted number of pellets retrieved by the right paw
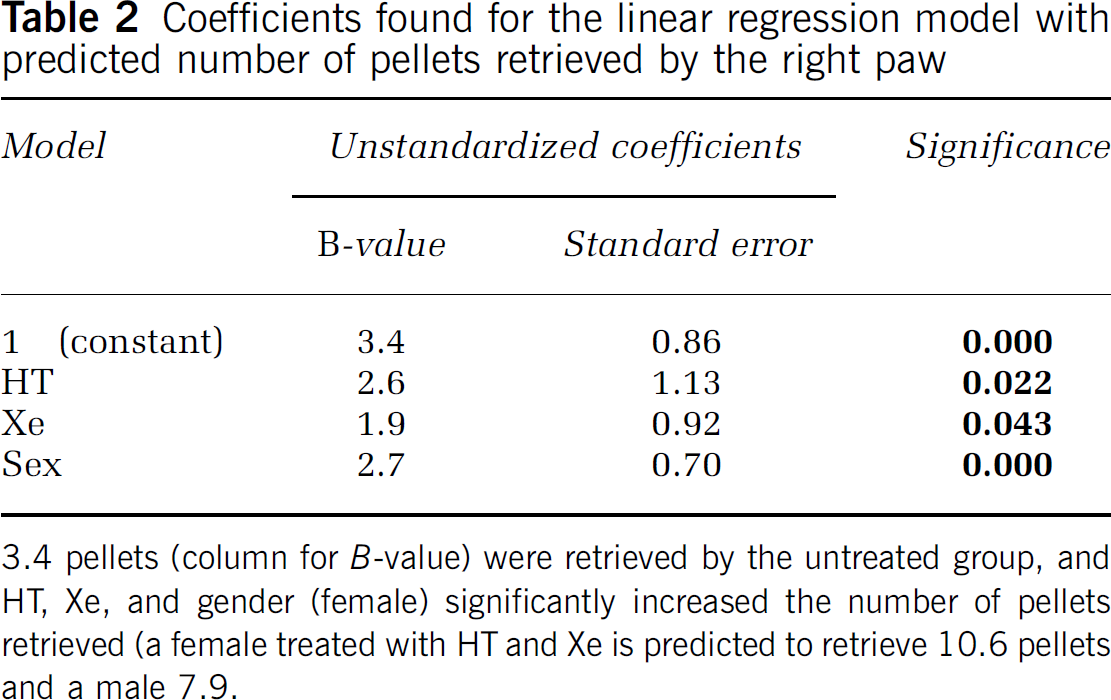
3.4 pellets (column for
Discussion
Previously, we have shown that applying a combination of 50% Xe inhalation and HT to 32°C for 3 h offers better protection than HT alone as compared with no treatment (Hobbs et al, 2008a). The known safety profile, lack of toxicity, rapid brain penetration, and easy reversal options make Xe a uniquely suitable therapy to combine with HT. This combination has the potential to improve the otherwise bleak outcome after perinatal asphyxia. Considering that HT is currently becoming standard of care after HI injury in both adults after cardiac arrest (Polderman, 2008) and newborns after perinatal asphyxia (Hoehn et al, 2008) and that Xe significantly enhances this effect (Dingley et al, 2006; Hobbs et al, 2008a; Ma et al, 2005; Martin et al, 2007), we have compared three different Xe delivery strategies in combination with the established HT treatment.
As Xe is very expensive and is likely only to be delivered in specialist centers for combination with HT, patient transport is required and two important questions arise:
Is a shorter duration of Xe than 3 h still effective? Is a short duration of Xe still effective if started after a delay?
Adding 1 h of Xe to 3 h HT gave better protection than 3 h HT alone, and adding 1 h of Xe was equally effective whether the Xe was started early or after a 2-h delay. Three hours of HT with 3 h of Xe gave the best neuroprotection protection as well as functional preservation compared with either HT alone or 1 h Xe treatment. All groups were well balanced for sex, and there was no difference in pathologic outcome between the sexes in any of the groups. This contrasted with the results from the functional test where females did significantly better than males. This difference was seen in the four groups with effective neuroprotection—HT alone or any combination of HT and Xe. There was a significant negative correlation between the staircase test and pathology. This correlation was stronger in the groups with more severe global pathology ranging from
In recent studies, we have shown that immediate delivery of 3 h Xe at normothermia was very neuroprotective in the short term (1-week survival) (Dingley et al, 2006), but not after long-term survival (Hobbs et al, 2008a). Therefore, we did not include a group of immediate or delayed Xe at normothermia only in this study.
Currently, the optimal period of exposure to Xe and HT is not known in animal models let alone humans. Similarly, the length of the therapeutic window has not been investigated; hence, it remains unknown what duration of delay could be introduced before efficacy is lost. Also, the data that are currently available is largely restricted to rodent models. This leads to the question of how to transfer ‘time’ in a rat model to larger animal models and to humans. With our neonatal HT research that led to effective clinical trial outcome, we found in the rat that 5°C to 6°C cooling for 3 h (Thoresen et al, 1996b) or 5 h (Bona et al, 1998) was protective, in the pig 24 h of HT (combined selective head cooling and body cooling by 4.5°C) was neuroprotective, in the sheep 76 h cooling was effective even after a delayed start of 5.5 h (Gunn et al, 1998), and in humans 72 h of 3.5°C cooling was neuroprotective (Gluckman et al, 2005; Shankaran et al, 2005) in clinical trials where cooling on average started 4.5 h after birth.
It has been suggested that Xe is neuroprotective by several incompletely understood mechanisms of action. For example, it is known that over stimulation of NMDA receptors by excessive transmitter release after HI injury causes the so-called excitotoxic apoptosis as part of a cascade of neuronal damage. It has been shown that Xe reduces overall neurotransmitter release (Petzelt et al, 2003) and also that it is an NMDA receptor antagonist (Franks et al, 1998); these being proposed mechanisms by which it is directly neuroprotective in a dose-related manner.
There is also evidence that Xe can have a preconditioning effect if administered before a neuronal insult. Xenon appears to induce a prosurvival response in cells that lasts longer than the period of Xe delivery, and it has been demonstrated that protein synthesis is a necessary step for this protection to occur, as protein synthesis blockade can inhibit these effects (Ma et al, 2006). Some suggested preconditioning mechanisms include increased gene transcription of activity-dependent neuroprotective protein (Cattano et al, 2008), enhanced phosphorylated cyclic adenosine monophosphate response element-binding protein signaling (Luo et al, 2008), and induction of hypoxia-inducible factor-1α, which is known to promote expression of several prosurvival genes. Gene induction may emerge as an important mechanism for Xe neuroprotection (Garry et al, 2004); so it is possible that a long duration of Xe exposure may not be necessary. The shorter the Xe exposure required, the smaller the demand on equipment and human resources required per treatment. Our finding that a single hour of Xe offers persistent functional and histologic neuroprotection is consistent with the idea that it may be the presence of Xe in itself that is important, for example, if Xe were to influence preconditioning gene induction.
The optimum period for delivery of Xe as a neuroprotectant is not yet known and neither is the maximum ‘window of opportunity’ post-HI insult after which useful damage limitation is no longer possible. Xenon may be neuroprotective both by direct immediate mechanisms by and induction of prosurvival mediators and genes. As the relative importance of each is at present unclear, it may in the future be the case that a short exposure or exposures may suffice.
Our finding that a single hour of Xe offers persistent functional and histologic neuroprotection is consistent with the idea that it may be the presence of Xe itself that is important, for example, if it were exerting its effects by triggering expression of prosurvival genes.
We now report long-term (into adulthood) functional and pathologic protection by Xe combined with HT, even when the Xe administration is delayed and applied for as little as 1 h in an experimental model of HI. We found no gender effect on the neuropathological outcome; however, on functional testing, females did significantly better than males. These findings are crucial in the context of clinical utility, as they suggest that Xe is both a financially and practically viable adjunct to HT treatment.
Footnotes
Acknowledgements
We thank Professor Lars Walloe for help with statistical analysis, SPARKS (UK), The Laerdal Foundation for Acute Medicine (Norway) for funding the study, and Dr Geoff Lloyd of the British Oxygen Company for making xenon available. The protocol was conducted under Home Office license according to UK guidelines.
Individual contributions
CEH, MT, TW, EC, and JD all performed the experiments. JD, MT, and CEH wrote the paper. MT, CEH, and JD designed the protocol, and MT is responsible for funding and running of the laboratory. JD designed and constructed the apparatus used, temperature-controlled chambers, xenon gas delivery system, and measurement/monitoring. CEH, TW, and EC performed the behavioral testing and CEH performed the histopathology examinations.
The authors declare that Dr. Dingley is currently a Board member of a University of Wales College of Medicine spin-out company that is involved in the development of clinical delivery systems for medical gases, including xenon, with relevant intellectual property. None of these systems were used in this study. No other author has conflict of interest.
