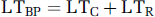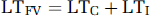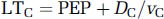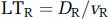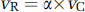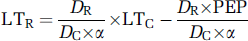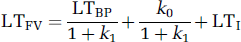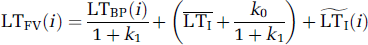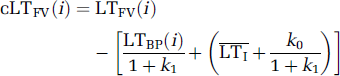Abstract
Changes in cerebral blood flow velocity (CBFV) pulse latency reflect pathophysiological changes of the cerebral vasculature based on the theory of pulse wave propagation. Timing CBFV pulse onset relative to electrocardiogram QRS is practical. However, it introduces confounding factors of extracranial origins for characterizing the cerebral vasculature. This study introduces an approach to reducing confounding influences on CBFV latency. This correction approach is based on modeling the relationship between CBFV latency and systemic arterial blood pressure (ABP) pulse latency. It is tested using an existing data set of CBFV and ABP from 14 normal subjects undergoing pressure cuff tests under both normoxic and acute hypoxic states. The results show that the proposed CBFV latency correction approach produces a more accurate measure of cerebral vascular changes, with an improved positive correlation between beat-to-beat CBFV and the CBFV latency time series, for example, correlation coefficient increased from 0.643 to 0.836 for group-averaged cuff deflation traces at normoxia. In conclusion, this study suggests that subtraction of systemic ABP latency improves CBFV latency measurements, which in turn improve the characterization of cerebral vascular changes.
Introduction
Continuous assessment of the structural and functional status of the human cerebral vasculature is highly desirable in managing cerebral vascular diseases. A prototypical clinical condition requiring a continuous cerebral vascular assessment is cerebral vasospasm (pathologic narrowing of cerebral arteries), which is a frequent and devastating complication after subarachnoid hemorrhage (Mayberg et al, 1994). In the current clinical practice, standard surveillance methods based on sporadic transcranial Doppler (TCD) measurements of blood flow velocity in major cerebral vessels may delay or even miss the detection of important changes in cerebral hemodynamics before cerebral ischemic damage (Aaslid, 2002; Sloan et al, 1989).
To develop clinically viable tools that are capable of continuously assessing the cerebral vasculature, a focus of our laboratory has been to explore novel methods of extracting physiologic information by analyzing continuously acquired signals of an intracranial origin (Hu et al, 2007, 2008). In a recent study (Hu et al, 2008), we have postulated that the latency of the onset of a vascular pulse relative to an extracranial timing signal (i.e., time delay between the electrocardiogram (ECG) QRS peak and the initial inflection in the resulting blood pressure pulse) is a function of several variables of relevance to the cerebral vasculature, including its radius, stiffness, and wall thickness. Our theoretical foundation for postulating that the latency of a pulsatile intracranial signal as a cerebrovascular index is based on the premise that the Moens–Korteweg equation, pulse wave velocity (PWV =√Eh/2ρbR), establishes a deterministic relationship between the velocity at which a blood pressure or a flow velocity pulse travels through the cerebral vasculature and the basic properties of this vascular route, including Young's elastic modulus (E), internal radius (R), and wall thickness (h). The underlying relationship is that changes in PWV should manifest with reciprocal changes in pulse waveform latency. Of the various vascular-related intracranial waveforms available for study, we have previously investigated the latency of the onset of an intracranial pressure (ICP) pulse relative to QRS peak (Hu et al, 2008), primarily from the perspective of developing an algorithm to reliably extract latency measure.
One of the inherent problems with measuring intracranial waveform latency, relative to the QRS peak, is that there are both extracranial and intracranial components to this latency value. From the viewpoint of assessing the cerebrovascular influence on pulse latency, physiologic changes occurring anywhere from the cardiac sinus node to the skull base could confound the ability to infer intracranialonly changes. For example, changes in the preejection period (PEP), which is the delay comprised of both the electromechanical delay and period of isovolumic contraction, could affect the latency of the ascending aorta flow waveform, which would obviously affect a measure taken downstream. The confounding effect of PEP on using pulse transit time for indirectly characterizing PWV has been reported previously (Payne et al, 2006).
On the positive side, choosing the ECG QRS peak as a timing signal is of practical value for developing a clinically friendly tool for continuously assessing the cerebral vasculature. Therefore, we sought to develop methods to extract the component of the pulse latency referable to the intracranial pulse propagation, which would therefore reflect changes originating in the cerebral vasculature. Our approach involves a nontrivial subtraction of latency of a systemic arterial blood pressure (ABP) pulse from the measured intracranial latency. This effort furthers our previous methodological development of the ICP latency extraction algorithm. In a larger context, although extensive applications of the concept of PWV in studying systemic arterial systems have been undertaken (Chang et al, 2006; Davies and Struthers, 2003; Foo and Lim, 2006; Foo et al, 2005; Nichols and Singh, 2002; Willum-Hansen et al, 2006), few studies of its application in the cerebral circulation have been pursued (Giller and Aaslid, 1994; Hu et al, 2008).
The main objective of this study is to investigate the proposed subtraction approach of reducing extracranial influence on intracranial latency. We collaborated with a team of researchers at the University of Colorado Altitude Research Center, who were conducting a study of cerebral blood flow velocity (CBFV) changes associated with sudden changes in systemic hemodynamics. We adapted our previous latency methodology, which was applied to ICP waveforms, to CBFV measurements. We reasoned that the CBFV in the major cerebral arteries, as measured by the TCD (Aaslid et al, 1982), would similarly serve as an excellent candidate hemodynamic signal for the pulse latency analysis. Fundamentally, many of the same principles in terms of latency detection, analysis, and interpretation should apply to both ICP and CBFV pulse waves. We hypothesized that subtraction of ABP latency would enhance the application of CBFV latency in cerebral hemodynamic assessment.
Materials and methods
Before proceeding with detailed methods and data analysis, we provide a summary of the study design here. In the Colorado study, subjects had CBFV (through TCD), systemic ABP, and ECG recordings during a test in which the thigh pressure cuff was inflated and then suddenly deflated using the well-established protocol (Aaslid et al, 1989). With abrupt deflation, volumetric redistribution leads to arterial hypotension and subsequent systemic and intracranial hemodynamic responses. We utilized our previously developed algorithm (Hu et al, 2008) to extract latencies of CBFV pulse measured at the middle cerebral artery and ABP pulse that was measured noninvasively at the radial artery. The proposed subtraction approach was then applied to these two pulse latency measurements to extract a ‘corrected’ CBFV latency measure. The algorithm was then tested by comparing the original and this corrected CBFV latency with regard to their abilities in capturing the acute changes in the cerebral vasculature by cuff inflation/deflation, during both normoxic and hypoxic states.
Subjects and Data
The data set used in this study was obtained from a study on the influence of hypoxia on cerebral blood flow autoregulation conducted at the Altitude Research Center (University of Colorado, Denver). After approval from the institutional review board, 14 healthy individuals (7 males and 7 females; 24 to 40 years) volunteered and gave written informed consents to participate in the study. All subjects completed a thorough medical examination and were included only if shown to be free of known diseases.
Testing for each individual took place during a single experimental session. The subjects underwent a continuous monitoring of ABP using a tonometer placed over the left radial artery (Model 7000; Colin Medical Instruments Corp., San Antonio, TX, USA) and CBFV in the ipsilateral middle cerebral artery isonated through the temporal window using a 2-MHz transcranial Doppler (Model Multi-Dop T2, DWL Electronic Systems, Singen, Germany). Signals including ABP, ECG, CBFV, and cuff pressure indicator were synchronized and recorded simultaneously at a sampling rate of 400 Hz (Powerlab 16SP, ADInstruments, Colorado Springs, CO, USA).
The subjects were studied in a supine position with their left arm abducted to avoid hydrostatic differences in ABP between the radial and middle cerebral arteries. In a single-blinded design, medical-grade hypoxic (12% O2) or normoxic (21% O2) gas was delivered under normobaric conditions (625mmHg) using compressed gas tanks, a 15-L breathing reservoir and, a Hans Rudolph two-way valve (Hans Rudolph, St Louis, MO, USA) connected in line. Each subject completed a total of six 10-min trials breathing either hypoxic (three trials) or normoxic (three trials) gas, using a Latin squares design to control for order effects. After a 6-min period of resting data collection, large pneumatic cuffs placed around both thighs were inflated to 30mmHg above systolic pressure to occlude blood flow to the lower extremities. After 3 mins of occlusion, the cuff pressure was rapidly released (> 0.1 secs) and changes in ABP and CBFV were followed for the next 60 secs. Subjects rested and breathed ambient air 5 mins between trials. In summary, each of the 14 subjects had three recordings collected under the normoxic state and three recordings collected under the hypoxic state, totaling 84 recordings.
Arterial Blood Pressure and Cerebral Blood Flow Velocity Latency Modeling
Modeling arterial blood pressure and cerebral blood flow velocity latency: Figure 1 illustrates a schematic representation of the components of ABP latency (LTBP) and CBFV latency (LTFV). The LTBP value was decomposed into two components termed LTC and LTR, where LTC represents the time interval from the ECG QRS to the time when blood pressure pulse reaches the juncture point between the extra- and intracranial components. LTC is thus a shared latency component that is common to both the ABP and CBFV waveforms (anatomically, roughly the origin of the common carotid arteries). LTR represents the traveling time of blood pressure pulse from the terminal point of the LTC to the radial artery assessment point. Similarly, LTFV was decomposed into LTC and LTI, where LTI represents the traveling time of blood pressure pulse to reach the site of CBFV measurement from the juncture point. LTI also includes the intrinsic phase difference between pressure and flow velocity pulse measured at the same site (Nichols et al, 2005), which should be a negative value given the fact that flow velocity pulse usually leads pressure pulse. We were, of course, most interested in determining the LTI value, as it should be the closest to reflecting the status of the cerebral vasculature as defined by the Moens–Kortweg equation.
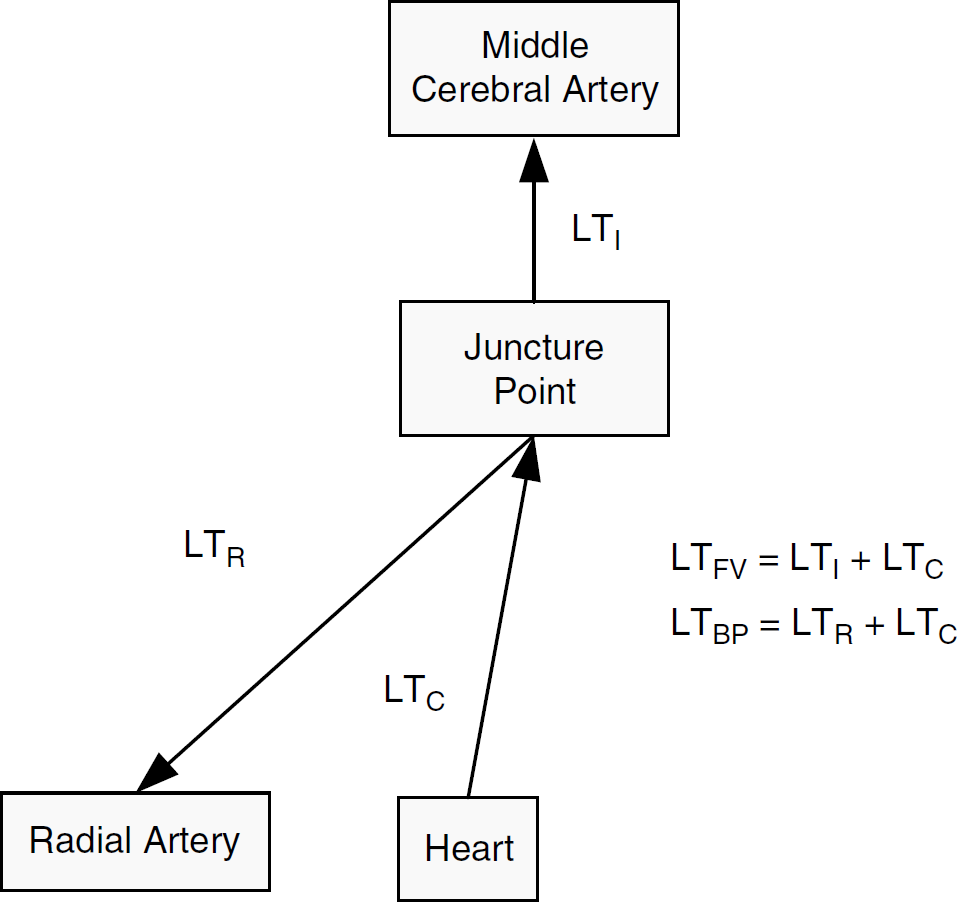
Schematic plot of the components of two latency measures including arterial blood pressure latency (LTBP) and cerebral blood flow velocity latency (LTFV). All latency measures are measured from the onset of pulse to the QRS peak of ECG.
On the basis of the above decomposition, we have the following equations
If just one sample of each LTBP and LTFV has been given, these two equations will not be sufficient to recover LTI as we have only two known measurements for three unknown variables. However, a relationship between LTC and LTR can be derived using the following equation:
where DC is the length of the vascular segment from the heart to the origin of the juncture node; DR the length of the vascular segment from this juncture node to the location of the radial artery where ABP is measured; vc and vR are the pulse wave velocities at these two vascular segments, respectively; α is a constant ratio between vR and vC. Combining Equations (3–5), we have the following equation:
By defining k1 = (DR)/(DC × α) and k0 = (DR × PEP)/(DC × α), Equation (6) leads to
By combining and re-arranging the above equations, we obtain the following equation:
Suppose we are given a set of paired samples of LTBP(i) and LTFV(i), where i=1, 2… is merely a symbol for indexing samples so that Equation (8) can be written as
where we separate LTI into two components:
With the above theoretical setup, we can proceed to discuss the situation where samples of LTFV and LTBP are from the same subject. Given k1 = (DR)/(DC × α) and k0 = (DR × PEP)/(DC × α), it is fairly valid to expect that k0 and k1 should be constant for samples obtained in a short period of time. Therefore, a linear relation between the measured LTFV and LTBP would indicate a cerebrovascular steady-state (CV-SS) condition, where
Estimating
Equation (10) resembles a subtraction procedure where scaled systemic pulse latency and a constant term were extracted from the measured CBFV latency. Although a positive linear relationship between CBFV latency and ABP latency samples used in the model fitting process is required, the applicability of Equation (10) does not depend on this prerequisite. Indeed, the essence of this correction is to look for variations that deviate from a linear model. On the basis of our modeling analysis, this corrected CBFV latency represents the information more directly related to the sample-to-sample changes in the cerebral vasculature by essentially removing an extracranial confounding influence.
Data Analysis Protocol
Data segmentation: Figure 2 illustrates the notations used in this study for denoting different segments of data from the start of measurement to the end. We manually mark the starting points of cuff inflation (t0) and deflation (t1) based on the pressure indicator signal recorded. On the basis of t0 and t1, a recording was divided into four segments denoted as I, II, III, and IV, respectively. It is interesting to include Segments I, III, and IV in this study. Segment I includes data from the start of recording to t0. A 50-sec segment after t0 and t1 was considered as the cuff inflation (II) and cuff deflation period (IV), respectively. Acute changes in the cerebral vasculature are expected to occur during Segments II and IV.
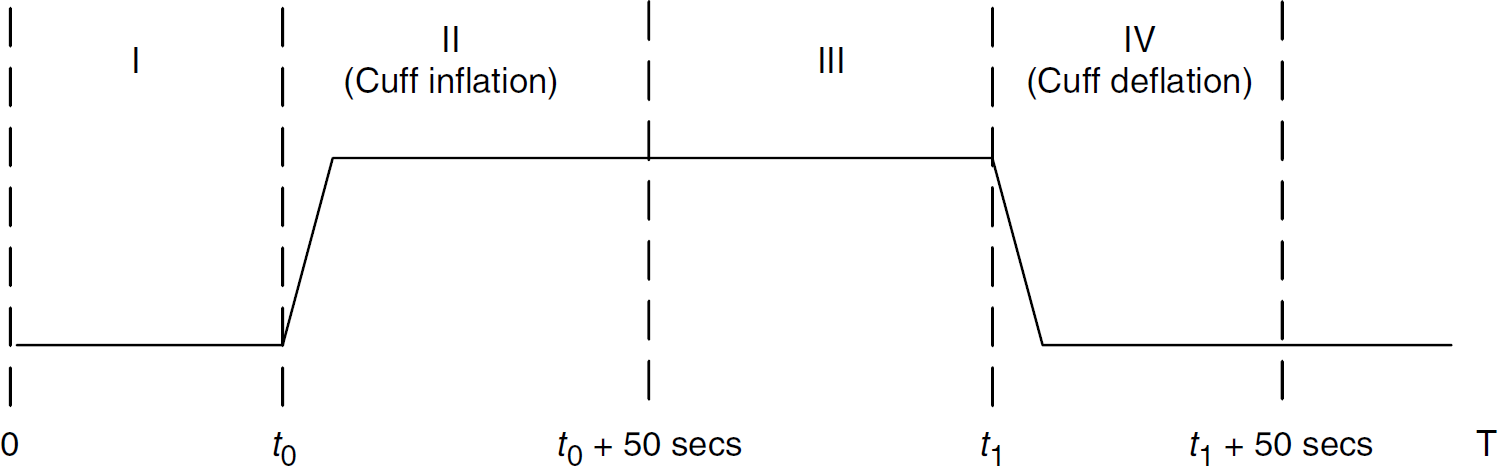
Diagram defining the notations of four signal segments from the start of the experiment to the end of the experiment, where t0 is the starting moment of cuff inflation and t1 the starting time of cuff deflation.
Pulse detection and manual editing: An ECG-aided pulse detection algorithm (Hu et al, 2008) was used to delineate each pulse of ABP and CBFV, using lead II of ECG. In addition, each detected pulse was saved and visualized using the custom software developed in-house to screen obvious noise or artifacts, so that only clean beats were further processed. Algorithm parameters for the pulse detection were the same as those used for ICP (Hu et al, 2008). ECG QRS detection was performed using a previously published algorithm (Afonso et al, 1999) on lead II of the ECG.
Calculation of the autoregulation index and conventional cerebral vascular measures: We calculated an autoregulation index (ARI; Aaslid et al, 1989) for each trial of the cuff deflation. We report the average value of this index and an intersubject correlation coefficient between ARI and LTFV. In addition, we used the FFT-based method to calculate critical closing pressure (CCP) and resistance area product (RAP) (Panerai, 2003). We also report the correlation analysis of corrected CBFV latency and RAP.
Latency extraction: Latency was derived for beats of ABP and CBFV that passed the screen test mentioned above. Latency was measured from the onset of each pulse relative to ECG QRS. The onset was automatically located using the tangent intercept method (Chiu et al, 1991).
Correction of flow velocity latency: To derive cLTFV, pairs of ABP and CBFV latency samples extracted from the beats of Segment I of each trial were used to fit a linear model to estimate the slope and the intercept coefficients using Equation (9). A total of 84 linear models were obtained for the whole data set. Later the cLTFV of a trial was calculated, using Equation (10), for each trial's Segments of II and IV using the estimated coefficients from the corresponding Segment I of the same trial.
Generation of group average: The group average of data from all subjects was obtained by first fitting a cubic spline to the beat-by-beat time series of a variable to unify the time axis. The resulting time series were then re-sampled at 2 Hz starting from t0 to t1 for Segments II and IV, respectively. Then the group average can be readily obtained from these re-sampled time series with effective anchor points at t0 and t1. Two different physiologic states within the same individuals were investigated. Data were analyzed separately as hypoxia was expected to impose mild cardiovascular stress, including increased heart rate (decreased R-R interval) and cardiac output.
Comparison of two latency extraction methods: This study focuses on correlating changes in CBFV after cuff inflation/deflation with those in the uncorrected and corrected CBFV latency because CBFV changes depict clearly the physiologic response of the cerebral vasculature to cuff inflation/deflation. This is advantageous for investigating whether the corrected CBFV latency could describe the same cerebral vascular process in a way compatible to what is shown by CBFV. Therefore, we chose the correlation coefficient between group-averaged CBFV latency and CBFV after cuff inflation/deflation, as a measure of the fidelity of the reflection of the CBFV change pattern by CBFV latency extracted using different methods. The significance of the difference in the correlation coefficient will be tested by a method of comparing the in-sample correlation coefficient proposed by Cohen and Cohen (1983).
Results
Figure 3 shows a scatter plot of ABP latency and CBFV latency extracted from the pulses in Segment I for one of the three normoxia recordings of each subject. A rectangle was used to enclose the scatter plot of each subject. It can be seen from this figure that, in general, a strong linear correlation exists between ABP latency and CBFV latency. An average correlation of 0.791±0.104 was achieved for measuring the intrapatient correlation of the beat-by-beat time series of these two variables shown in the figure.
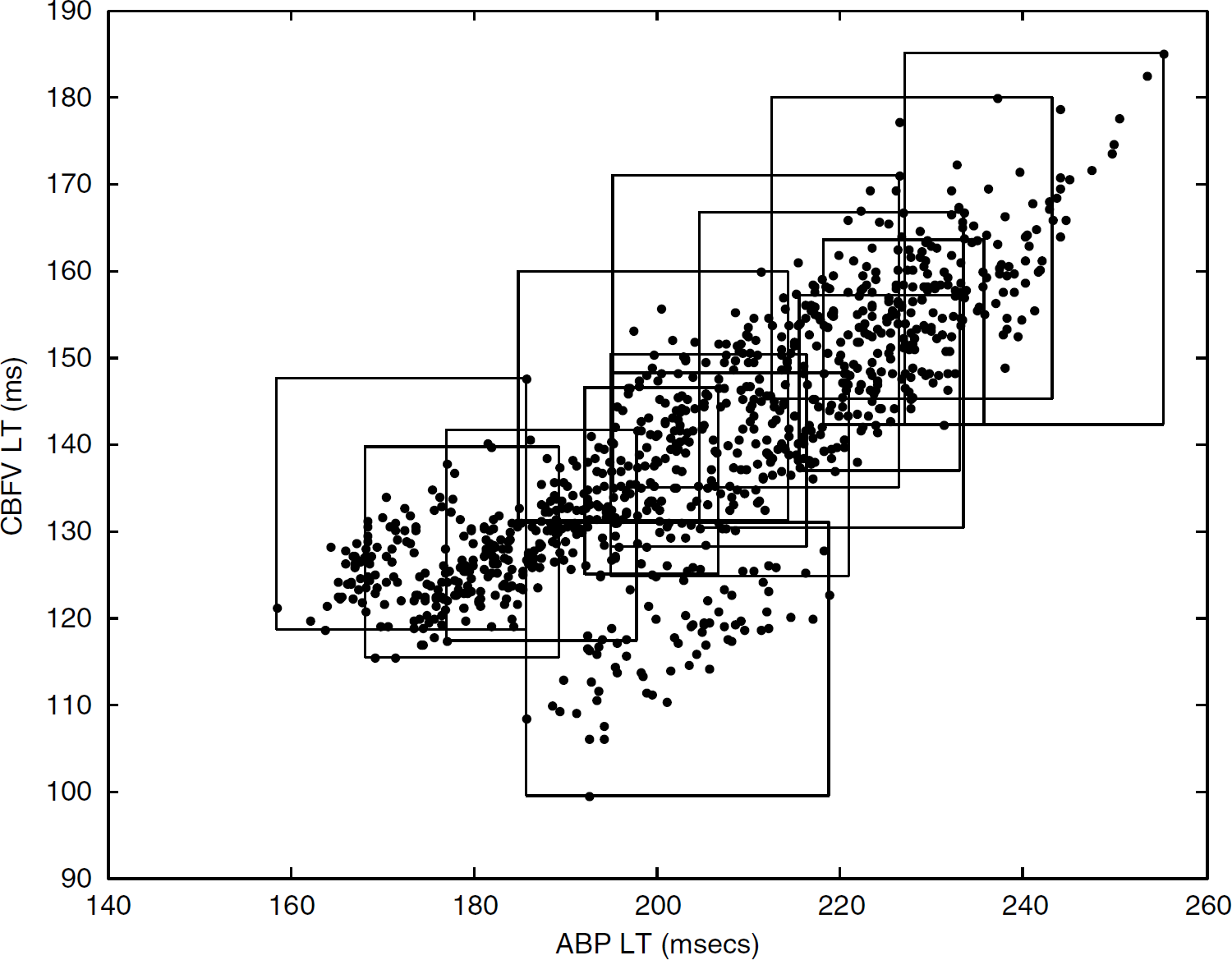
The scatter plot of the ABP latency versus CBFV latency for Segment I of 14 subjects' normoxia data. Each subject's data are enclosed with a rectangle.
Figure 4 shows the group average of beat-by-beat mean ABP and CBFV time series for Segment II, which correspond to cuff inflation, and Segment IV, which correspond to cuff deflation. An average was calculated on all 42 traces of normoxic and hypoxic states. To help assess the timing of the signals, a vertical line is positioned at the peak of ABP for the cuff inflation cases and positioned at the nadir of ABP for the cuff deflation cases. The plots in Figure 2 represent typical responses that have been reported in the literature (Aaslid et al, 1989). A clear earlier start of the return of CBFV toward the baseline, as compared with that of ABP, is shown in all panels. This has been considered as a signature pattern of cerebral blood flow autoregulation (Aaslid et al, 1989).
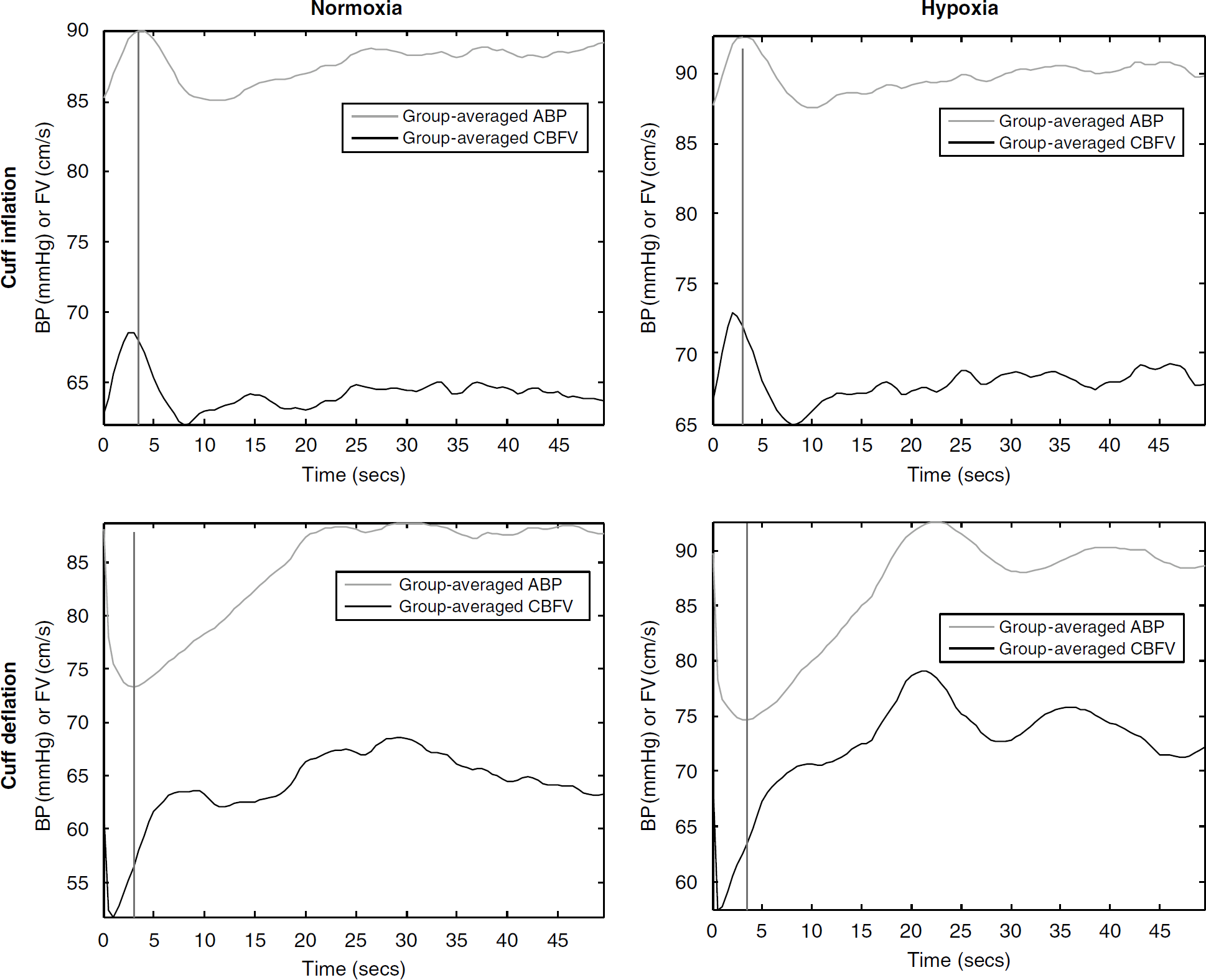
Overlaid plots of group-averaged beat-to-beat ABP and CBFV. The top panels display data for cuff inflation at normoxia and hypoxia states, respectively. The lower panels display similar data for cuff deflation.
Table 1 lists the mean and the standard deviation of basic hemodynamic variables and the four parameters obtained from using Equation (9) to fit samples of CBFV latency and ABP from Segment I data. In addition, we compared the mean values of these variables at the normoxic and the hypoxic states using the paired t-test. Hemodynamic variables that are significantly different at the two states include systolic blood pressure, heart rate, end-tidal CO2, and both latency measures (P > 0.05). We did not obtain a significant correlation between ARI and LTFV with a correlation coefficient of 0.07 for the normoxic state and a value of −0.01 for the hypoxic state, indicating that ARI and CBFV latency are independent assessments of the cerebral vasculature. The four parameters from the model fitting include the goodness of fit (R2), Pearson's correlation coefficient (ρ), the slope coefficient, and the intercept coefficient in Equation (9). R2 and ρ are significantly higher for the hypoxic data with a P-value of 2.1 × 10−4 and 4.4 × 10−4, respectively. However, no significant difference of slope and intercept parameters was detected between the normoxic and hypoxic states. It should also be noted that the intercept parameters of most subjects are negative. This is possible because of the intrinsic negative-phase difference between the pressure pulse and the flow velocity pulse that are measured at the same site, as discussed in the Section ‘Modeling arterial blood pressure and cerebral blood flow velocity latency.’
List of basic hemodynamic parameters and four derived parameters obtained after fitting a linear model according to the Equation (7) to samples of CBFV latency and ABP latency
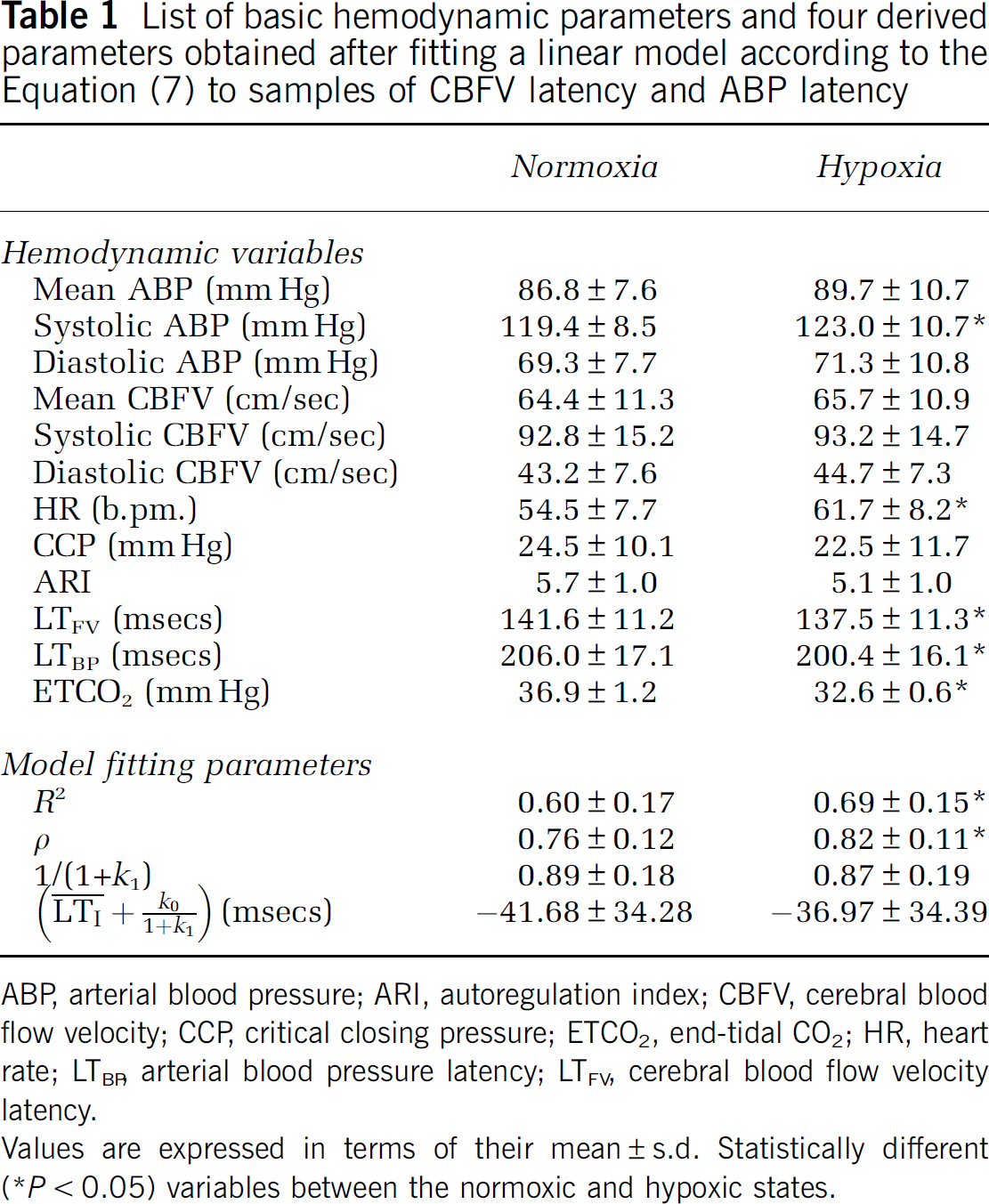
ABP, arterial blood pressure; ARI, autoregulation index; CBFV, cerebral blood flow velocity; CCP, critical closing pressure; ETCO2, end-tidal CO2; HR, heart rate; LTBP, arterial blood pressure latency; LTFV, cerebral blood flow velocity latency.
Values are expressed in terms of their mean±s.d. Statistically different (*P<0.05) variables between the normoxic and hypoxic states.
As a typical example, Figure 5 shows the group averaged CBFV in panel A, RAP in panel B, LTBP in panel C, LTFV in panel D, and cLTFV in panel E for normoxia traces aligned at the starting point of cuff deflation. A vertical line is positioned at the nadir of the CBFV so that timing of different signals can be readily assessed. It can be seen that an apparent delay exists between the first start of CBFV increase after the initial drop and the start of CBFV latency increase. However, this delay was significantly reduced when comparing CBFV and cLTFV traces. This is a consistent pattern for all hypoxia and cuff inflation cases as well. This reduction of delay improved the correlation between the corrected CBFV latency and the CBFV as shown in Table 2, where the linear correlation coefficients between the trace of CBFV and those of original and corrected CBFV latencies are listed. The results in Table 2 suggest that the corrected CBFV latency measures correlate better with the CBFV than does the original CBFV latency in a statistically significant manner (P<10−6).
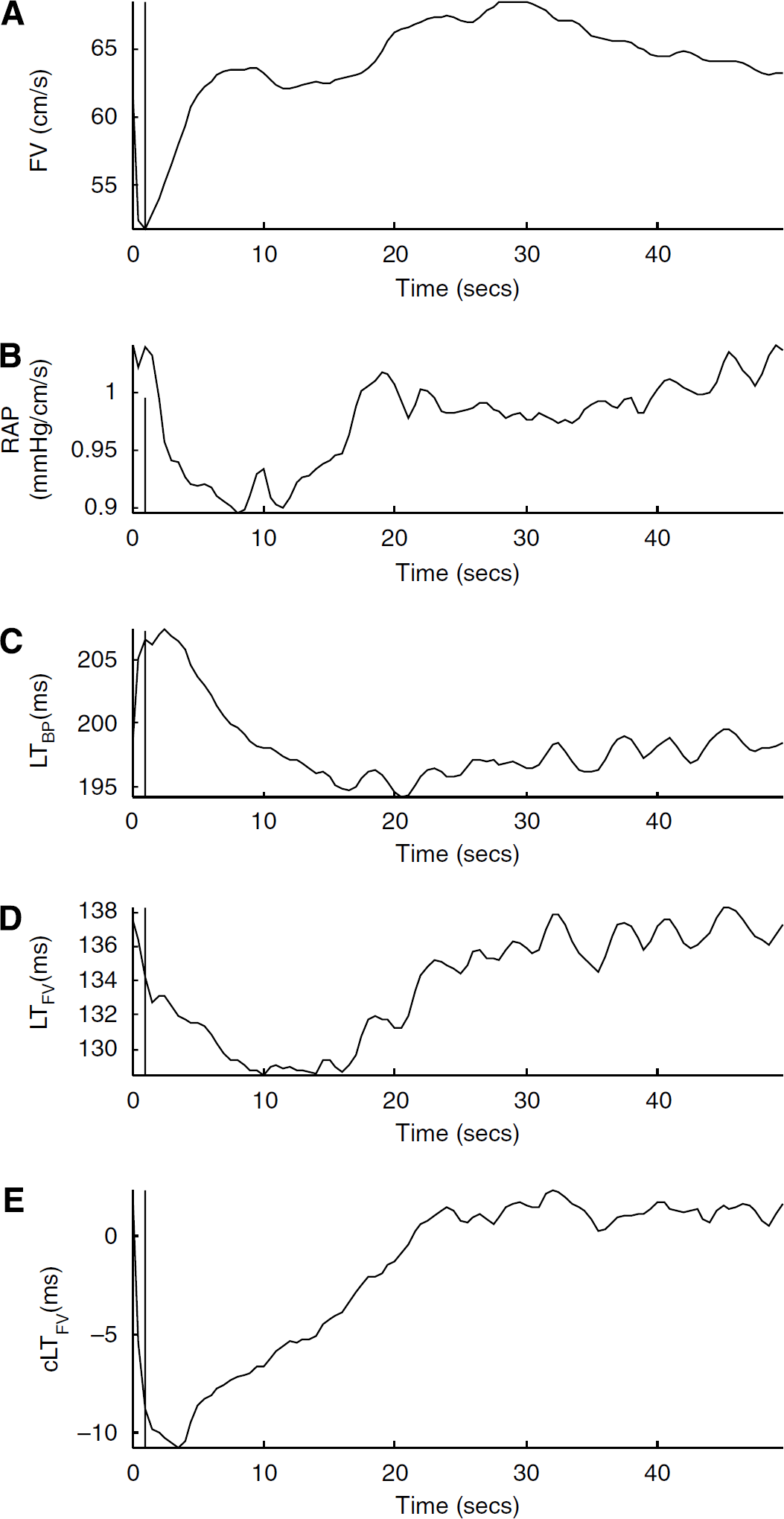
Group averaged beat-to-beat CBFV (
Similar patterns also exist in Figure 5 when comparing the timing of the start of the RAP decrease with the timing of the start of the latency increase, that is, cLTFV trace shows a much shorter lag between the two as compared with LTFV. Cerebral blood flow autoregulation after cuff deflation is usually considered to start at the point when RAP starts decreasing. Owing to this shortening of the lag, the cLTFV time series, beginning at the starting point of RAP decrease, have a negative correlation with the corresponding RAP time series, whereas LTFV has a positive correlation with RAP. Quantitative results of this correlation analysis are also shown in Table 2. However, the correlation of RAP with both cLTFV and LTFV after cuff inflation is both negative and close to each other.
Pearson's linear correlation coefficient between group-averaged CBFV (RAP) and two CBFV latency measures
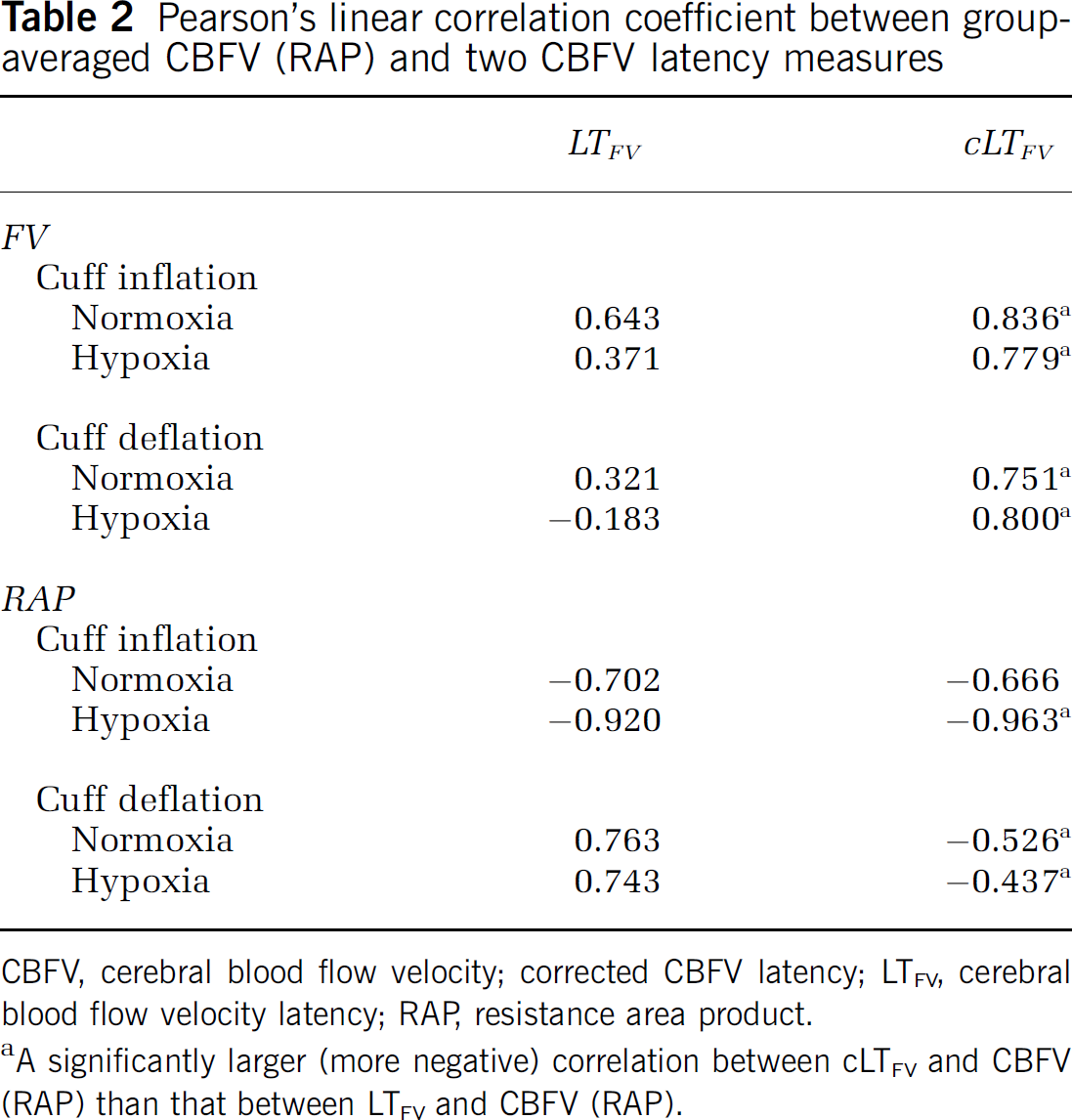
CBFV, cerebral blood flow velocity; corrected CBFV latency; LTFV, cerebral blood flow velocity latency; RAP, resistance area product.
A significantly larger (more negative) correlation between cLTFV and CBFV (RAP) than that between LTFV and CBFV (RAP).
Discussion
Pathologic changes in cerebral vasculature, such as the development of cerebral vasospasm, are not unusual during the intensive care of stroke and brain trauma patients. Therefore, the state of cerebral vasculature should ideally be monitored in a continuous fashion to support timely clinical treatment and management of critical conditions related to cerebral vasculature changes. In the current clinical practice, intermittent assessments of cerebral hemodynamics using TCD or cerebral blood flow measurement are often used for such a purpose, but may miss important hemodynamic events. The sound physics and physiology behind the concept of PWV and its many successful applications in systemic circulation support the potential usefulness of the signal processing technique of extracting beat-to-beat latency of cerebral hemodynamic signals developed in our previous study. Timing latency relative to ECG QRS is of practical value, but unavoidably confounded by influences of extracranial origins. The main motivation behind this study was therefore to develop an algorithm to improve PWV analysis in the cerebral circulation.
As shown in Figure 3, there is, in general, a strong positive linear correlation between CBFV latency and ABP latency. On the basis of the modeling analysis presented in the Section ‘Modeling arterial blood pressure and cerebral blood flow velocity latency’ and Equation (9), a positive linear correlation between intrapatient samples of ABP latency and CBFV latency is expected if sample-to-sample variation of the pulse propagation time from the extracranial/intracranial juncture node to the site of CBFV measurement is small. Despite this high positive correlation between ABP latency and CBFV latency, it was also observed that CBFV latency could change in an opposite direction to that of ABP latency in the initial seconds after cuff deflation, as evidenced in Figure 5 (comparing panels C and D). This shows the power of simultaneous measurement of CBFV latency and ABP latency for detecting changes that cannot be explained by alternations in the common components of both latency measures. This observation is indeed supportive of the concepts of CV-SS and CV-DS. A positive linear relationship between CBFV latency and ABP latency exists at CV-SS, whereas such a relationship may not hold at CV-DS.
The challenge was then to reduce the extracranial confounding influence on CBFV latency by exploring the relationship between ABP latency and CBFV latency, as modeled in the Section ‘Modeling arterial blood pressure and cerebral blood flow velocity latency.’ This modeling process led to a practical CBFV latency correction algorithm, as proposed in the Section ‘Estimating gLTI from a set of cerebral blood flow velocity and arterial blood pressure samples: deriving a corrected cerebral blood flow velocity latency.’ Using a set of ABP and CBFV signals from the thigh cuff test experiment, we were able to collect evidence supportive of the validity of the proposed algorithm. The correlation between beat-to-beat CBFV and corrected CBFV latency time series is greater than that between CBFV and the original CBFV latency. This improvement of correlation was mostly achieved by reducing the time delay between the start of CBFV recovery and the start of latency increase, which is shown for the cuff deflation case shown in Figure 5, where it showed that a large time delay existed between the start of CBFV recovery and the start of CBFV latency recovery.
Cerebral blood flow velocity recovery is presumably caused by a vasodilatation in the cerebral vasculature to compensate for the decrease in cerebral blood perfusion pressure as a result of cuff deflation, which would have also led to a decrease in pulse wave velocity in the cerebral vasculature and, thus, an increase in CBFV latency. However, increase in CBFV latency may be masked by a simultaneous decrease in LTc because of an increase in systemic blood pressure that increases pulse wave velocity in the systemic arterial vasculature (Nichols et al, 2005). This explanation is supported by the observation that the period of decreasing ABP latency overlaps that of decreasing CBFV latency, as shown in Figure 5. Improved correlation between the corrected CBFV latency and CBFV also occurs for the cuff inflation data as well as for both normoxic and hypoxic states, as reported in Table 2.
When replacing CBFV with an existing cerebral vascular resistance metric (RAP) for conducting the correlation analysis with latency, we observed that RAP started to decrease before LTFV started to increase after cuff deflation. However, this time lag between RAP and cLTFV is greatly reduced. The start of RAP decrease should ideally coincide with the start of CBFV latency increase because of a slower PWV caused by the vasodilatation. The reduced time lag between RAP and cLTFV lends further support to the conclusion that the proposed latency correction approach is effective in reducing extracranial confounding influence on characterizing cerebral vascular changes.
This study focuses on correlating changes in CBFV and RAP after cuff inflation/deflation with those in CBFV latency extracted by different methods. In the future study, several existing cerebral vascular indices, including cerebral CCP, RAP, CBFV pulsatility index (PI), should be studied more thoroughly, together with CBFV latency because there exists a large body of literature pertaining to their role as cerebrovascular indices (Czosnyka et al, 1999; Hsu et al, 2004; Michel et al, 1997; Panerai et al, 1993, 1999; Panerai, 2003), which would provide additional corroboration needed for further illustrating whether CBFV (or ICP) latency may be useful for characterizing pathologic cerebral vascular changes. In addition, it will be worthwhile to further derive cerebral blood flow autoregulation indices from latency measurements and to evaluate them with established scores such as ARI. As we briefly reported, LTFV and ARI are not correlated. This is not surprising because LTFV reflects changes in the cerebral vasculature, whereas an ARI reflects how effective the cerebral vascular changes are at stabilizing the cerebral blood flow. Therefore, directly correlating latency measures with ARI is not expected to reveal a correlation. Instead, metrics have to be derived from latency time series to quantify the amount or the speed of latency changes, which may be more appropriate for comparing with autoregulation indices.
Some remaining issues need to be taken into consideration when interpreting the results obtained in this study. No effort has been made to account for the individual difference in the distance from the branching node in Figure 1 to the middle cerebral artery before taking group average of latency measures. In addition, the assumption of stability of PEP from beat to beat is needed for fitting a straight line to obtain the model coefficient in Equation (9). Furthermore, the assumption of a constant PEP is also needed for the applicability of the model coefficients estimated using Segment I data to Segments II and IV data. This may not pose a serious problem to the current data set because the duration of the whole experiment is about 10 mins. In such a short period, these assumptions may not be seriously violated given that the reported coefficient of variation of PEP is about 8.3% (Payne et al, 2006). However, for the intended use as a continuous cerebral vasculature indicator over a long period of time, the validity of these assumptions has to be checked for corrected CBFV latency by inspecting the adequacy of model fitting. Alternatively, one would resort to using a different signal other than the ECG QRS as a timing landmark, for example, the second heart sound to directly measure PEP in a beat-by-beat manner.
Conclusion
In conclusion, CBFV latency alone (relative to ECG QRS peak) is inadequate as an indicator for characterizing cerebral vascular changes because of the extracranial confounding influence on this measure. However, we showed that CBFV latency can be used continuously to track cerebral vascular changes for an individual patient in situations where continuous ABP latency or latency of a systemic pulse is available. This allows the cerebral vascular dynamic state to be detected by simultaneously monitoring both the cerebral pulse and the systemic pulse latency. In addition, the extracranial confounding influence on CBFV latency was reduced using the proposed correction method. Future studies will be required to determine whether the results regarding the use of CBFV latency and ABP latency to derive a corrected latency measure can be extrapolated to ICP latency as well.