Abstract
Magnetic resonance imaging (MRI) is a powerful tool to assess brain lesions, but currently available contrast agents are limited in the assessment of cellular and functional alterations. By use of the novel MRI contrast agent gadofluorine M (Gf) we report on imaging of transient and widespread changes of blood—brain barrier (BBB) properties as a consequence of focal photothrombotic brain lesions in rats. After i.v. application, Gf led to bright contrast in the lesions, but also the entire ipsilateral cortex on T1-weighted MRI. In contrast, enhancement after application of gadolinium diethylenetriamine-pentaacetic acid (Gd-DTPA), a common clinical indicator of BBB leakage was restricted to the lesions. Remote Gf enhancement was restricted in time to the first 24 h after photothrombosis and corresponded to a transient breakdown of the BBB as revealed by extravasation of the dye Evans blue. In conclusion, our study shows that Gf can visualize subtle disturbances of the BBB in three dimensions not detectable by Gd-DTPA. Upon entry into the central nervous system Gf most likely is locally trapped by interactions with extracellular matrix proteins. The unique properties of Gf hold promise as a more sensitive contrast agent for monitoring BBB disturbances in neurologic disorders, which appear more widespread than anticipated previously.
Keywords
Introduction
There is increasing evidence that focal brain lesions profoundly influence remote brain areas not affected by the primary insult (reviewed in Witte et al, 2000). Pathophysiologic alterations involve edema formation, cortical spreading depression (CSD), diaschisis, and other adaptive responses. Magnetic resonance imaging (MRI) is a powerful tool to assess focal brain lesions, but structural and molecular alterations remote from the initial insult are in general not visualized by conventional MR sequences. Thus, there is high interest in developing novel MR-contrast agents for in vivo assessment of cellular and functional responses in the brain (Artemov et al, 2004; Bulte and Kraitchman, 2004). In this study, we report on in vivo MRI of transient and widespread changes of blood—brain barrier (BBB) properties as a consequence of focal photothrombotic brain lesions by the use of a novel MR-contrast agent Gd-GlyMe-DOTA-perfluoroocytyl-mannose-conjugate (gadofluorine M (Gf); Bayer Schering Pharma AG, Berlin, Germany). Brain photothrombosis (PT) was introduced as a simple model of focal cortical lesions by Watson and Dietrich in the 1980s (Watson et al, 1985; Dietrich et al, 1987a, b ; reviewed in Stoll et al, 1998). Photothrombosis can be induced in rodents by systemic administration of a photosensitive dye such as Bengal rose and subsequent focal illumination of the brain through the intact skull by a light source (Watson et al, 1985; Schroeter et al, 2002). Photothrombosis is minimally invasive and does not inflict significant neurologic deficits assessed behaviorally to animals, thereby allowing long-term observations. Because of its focal nature PT facilitates the distinction between events within the lesion and remote brain areas. Numerous studies have shown that PT induces periinfarct depolarization closely related to CSD (Dietrich et al, 1994; Jander et al, 2000) and other neurophysiologic alterations in the ipsilateral hemisphere such as increased spontaneous activity of neurons (Schiene et al, 1996). Remote molecular alterations encompass upregulation of proinflammatory cytokines and growth factors (Jander et al, 2000; Kleinschnitz et al, 2004), and rearrangement of neurotransmitter receptors (Redecker et al, 2002). Magnetic resonance imaging studies using the contrast agent gadolinium diethylenetriamine-pentaacetic acid (Gd-DTPA) as the conventional in vivo marker for the BBB showed early and long-lasting breakdown of the BBB within the PT lesion (Lee et al, 1996; Schroeter et al, 2001; Kleinschnitz et al, 2003). However, there were no remote contrast enhancement or signal alterations on T2-weighted (T2-w) images. By use of the novel MR-contrast agent Gf we now provide in vivo evidence to confirm a widespread remote alteration of the BBB after PT and a novel MRI tool to visualize these profound and transient BBB disturbances.
Methods
Induction of Photothrombotic Brain Lesions
All animal experiments were performed in accordance with institutional guidelines and were approved by the Regierung of Unterfranken. Focal cerebral lesions were induced in male Wistar rats (200 to 250 g) by PT of cortical microvessels under inhalation anesthesia with 2.5% isoflurane in a 2:1 nitrogen/oxygen atmosphere, as described previously (Watson et al, 1985; Jander et al, 1995; Buchkremer-Ratzmann et al, 1996; Kleinschnitz et al, 2003, 2005). During the operation procedure, rectal temperature was kept between 36.5°C and 37.5°C by a servo-controlled heating blanket. Briefly, a fiber optic bundle of 4 mm diameter of a light source (150W Philips lamp type 6423, beam power 600 lm) was centered stereotaxically 4 mm posterior and 4 mm lateral from Bregma on the intact skull. Thereafter, 0.4mL of a sterile-filtered Rose Bengal solution (10 g/L; Sigma, Deisenhofen, Germany) was administered by a femoral vein catheter, and the brain was illuminated for 20 mins. With this setup used in this and our numerous previous studies there was a modest increase of 1°C to 2°C in the temperature of the brain in the area of illumination (measured with digital computer thermometer CTA 1220 and sensor EB01; Ebro, Ingolstadt, Germany; Jander et al, 1995; Buchkremer-Ratzmann et al, 1996). This procedure resulted in cone-shaped cortical infarctions without neurologic symptoms. At the end of the operation, subcutaneous fat and skin were sutured in anatomic layers and animals were placed in separate cages for recovery. Three animals underwent sham operation and received Rose Bengal without irradiation. Animals underwent MRI examination at different stages of lesion development and in a subgroup vital brain slice preparations were used to follow the spread of the MR-contrast agent Gf ex vivo over time.
Contrast Agent and Magnetic Resonance Imaging
Gadofluorine M (Bayer Schering Pharma AG) is an amphiphilic Gd complex with a molecular weight of 1,528 Da (Barkhausen et al, 2003; Misselwitz et al, 2004). The contrast medium is synthesized by adding a perfluoroctyl chain to a Gd-containing macrocycle. The complex also contains a sugar moiety, which leads to increased hydrophilicity. In vitro studies have shown that Gf forms micelles in water (approximately 5.5nm in diameter) and binds reversibly to plasma albumin and matrix proteins. After intravenous administration, Gf binds reversibly to plasma proteins leading to macromolecular Gf—protein complexes in the circulation. The plasma half-life T1/2β of these Gf complexes is 15.6h (Raatschen et al, 2006). Gadofluorine M has a high T1 relaxivity in plasma (15.9L/mmol per sec per Gd ion) and in water (15.8L/mmol per sec; Misselwitz et al, 2004). For subsequent localization of Gf accumulation in histologic sections, Gf was prelabeled with a carbocyanine dye allowing detection of Gf by fluorescence.
All measurements were performed on a clinical 1.5T MRI unit (Magnetom Siemens Vison, Erlangen, Germany) as described previously (Kleinschnitz et al, 2003, 2005). Briefly, rats were anesthetized as described above. For all MRI scans, animals were lying in supine position with their heads fixed in a heatable custom-made dual channel surface coil designed for investigations of the rat brain (A063HACG; Rapid Biomedical, Würzburg, Germany). The MR protocol included a scout sequence in three planes, a coronal T1-weighted (T1-w) (repetition time 460 ms, echo time 14 ms) and a coronal T2-w sequence (repetition time 2.500 ms, echo time 92 ms) with a slice thickness of 2 mm. After a nonenhanced MR examination, Gd-DTPA (0.2 mmol/kg body weight) or Gf (0.1 mmol/kg body weight) was applied by the femoral vein. A second MRI was performed always 20 mins after Gd-DTPA at the time points 3, 6, 12, 24, and 72 h after PT in groups of animals. In a first set of experiments MR measurements were performed in separate groups of four animals each at 24, 48, and 72 h after PT (always 24 h after Gf application). In a second set of experiments the spatiotemporal evolution of Gf enhancement was studied: Gf was always given shortly after induction of PT when irradiation was completed and groups of animals underwent two MR examinations at 1 and 3 h (n = 3), 5 and 9 h (n = 3), or 18 and 26 h (n = 3) after PT, respectively. Finally, three animals that had received Gf at the time of PT underwent serial MR examinations at 24 h, 3 and 7 days after photothrombosis.
A subgroup of Gf-injected animals was killed at different time points after PT in deep anesthesia and the brains were quickly removed and snap frozen in isopentane for fluorescence studies. Moreover, some rats were perfused by 4% paraformaldehyde and the brains were embedded in paraffin.
Assessment of Blood—Brain Barrier Patency
To study the patency of the BBB after PT using the Evans blue method (Belayev et al, 1996) a separate group of rats received 80mg/kg of the dye Evans blue (solution 20mg/mL) i.v. immediately after cessation of the illumination for PT induction, or with a delay of 48 h (for time point 72 h). Two animals each were killed shortly thereafter (time point 0) or 1, 3, 6, 9, 12, 24, and 72 h after PT, respectively. Brains were rapidly removed and fixed in 4% paraformaldehyde. Photographs of the entire brain were taken to assess Evans blue extravasation. Then the brains were cut into 1 mm thick coronal slices for further morphologic examination.
Brain Slice Preparations
For preparation of vital brain slices rats were deeply anesthetized with isoflurane and decapitated. A block of tissue containing the lesion was removed and transferred into ice-chilled saline, containing (mmol/L): sucrose, 200; PIPES, 20; KCl, 2.5; NaH2PO4, 1.25; MgSO4, 10; CaCl2, 0.5; dextrose, 10; pH 7.35 with NaOH. Coronal sections (300 µm) through the photothrombosed region were prepared on a vibratome (Gala Instruments, Bad Schwalbach, Germany). Before fluorescence microscopy slices were kept submerged in standard artificial cerebrospinal fluid (mmol/L): NaCl, 125; KCl, 2.5; NaH2PO4, 1.25; NaHCO3, 24; MgSO4, 2; CaCl2, 2; dextrose, 10; pH adjusted to 7.35 by bubbling with a mixture of 95% O2 and 5% CO2. For serial observations slices were transferred to standard cell culture dishes and placed under the microscope (Axioskop FS; Zeiss, Jena, Germany). Under these conditions slices are alive for 6 to 8 h as indicated by previous electrophysiologic recordings (Meuth et al, 2003, 2006).
In one set of experiments animals were killed 1.5 and 5 h after PT and brain slices were analyzed by fluorescence microscopy. In a second set of experiments brain slices were obtained shortly after PT and application of Gf (1 min) and Gf autofluorescence was repetitively followed for 3 h by fluorescence microscopy. In a last set of experiments slices were prepared 30 mins after PT induction and Gf application, and slices were sequentially analyzed up to 2.5 h after the preparation.
N -Methyl-D -Aspartate-Receptor Blockage and Lipopolysaccharide Application
To analyze whether the remote Gf enhancement was mediated by N-methyl-D-aspartate (NMDA)-receptor signaling, rats were injected i.v. with the noncompetitive NMDA-receptor antagonist MK-801 (dizolcipine; Research Biochemicals International, Natick, MA, USA) at a dosage of 2 mg/kg body weight 30 mins before induction of PT. Three MK-801-treated animals received Gf immediately after PT induction and underwent MRI 24 h later.
Systemic application of the proinflammatory lipopolysaccharide (LPS) can alter permeability of the BBB (Xaio et al, 2001) and, additionally, activate inflammatory pathways within the central nervous system (CNS; Tasaki et al, 1997). To address the potential role of these inflammatory processes in the opening of the BBB (Blamire et al, 2000) we aimed to mimic the remote Gf accumulation after PT by systemic LPS application. Groups of three naive rats received LPS (0.5 mg/kg body weight) intraperitoneally and Gf 1 or 48 h thereafter i.v., and underwent MRI at 24 or 72 h, respectively.
Immunohistochemistry and Fluorescence
Thick paraffin (10 µm) sections were cut and stained with peroxidase-conjugated sheep immunoglobulin G antibodies against rat albumin and 3′4′ diaminobenzidine as chromogen (Cappel, Lot 37201). In addition, 10 µm thick sections from snap-frozen brains were cut through the lesion and analyzed for the presence of carbocyanine labeled Gf by red fluorescence on a Zeiss Axiophot microscope (Zeiss, Thornwoods, NY, USA).
Results
Gadolininium Diethylenetriamine-Pentaacetic acid-Enhanced Magnetic Resonance Imaging of Photothrombotic Lesions
After application of the conventional MR-contrast agent Gd-DTPA, photothrombotic cortical lesions showed a marked contrast enhancement within the lesions on T1-w (Figure 1) and appeared hyperintense on T2-w MRI (Figure 2A) as described before (Schroeter et al, 2002; Kleinschnitz et al, 2003). Importantly, no uptake of Gd-DTPA on T1-w MRI was seen remote from the lesions in the ipsi- and contralateral cortex at all time points examined, for example, 3, 6, 12, 24, and 72 h after PT. In these experiments Gd-DTPA was applied according to clinical practice always 20mins before MRI, because the contrast agent is rapidly cleared from the circulation resulting in a disappearance of tissue enhancement.
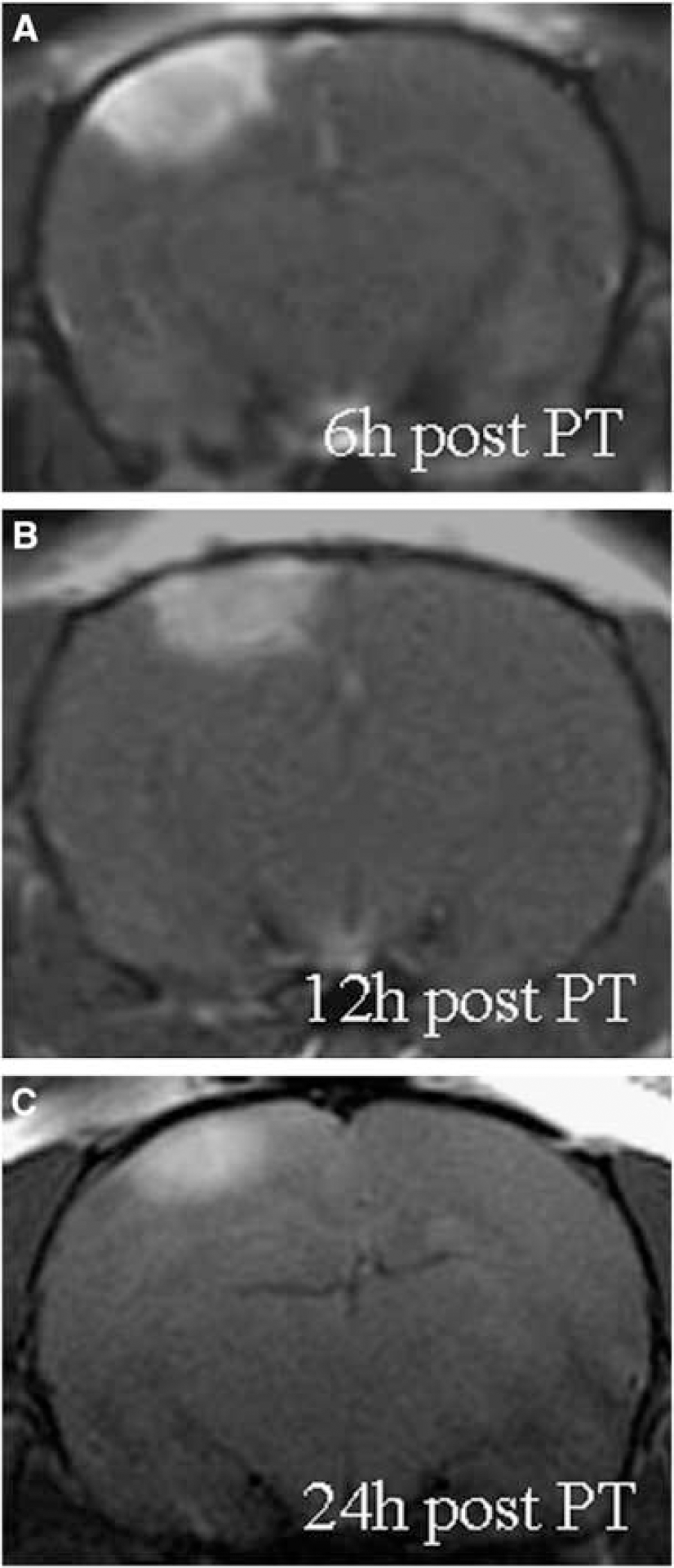
Gd-DTPA enhancement is restricted to photothrombotic (PT) lesions. T1-w MRI of lesions at 6 h (
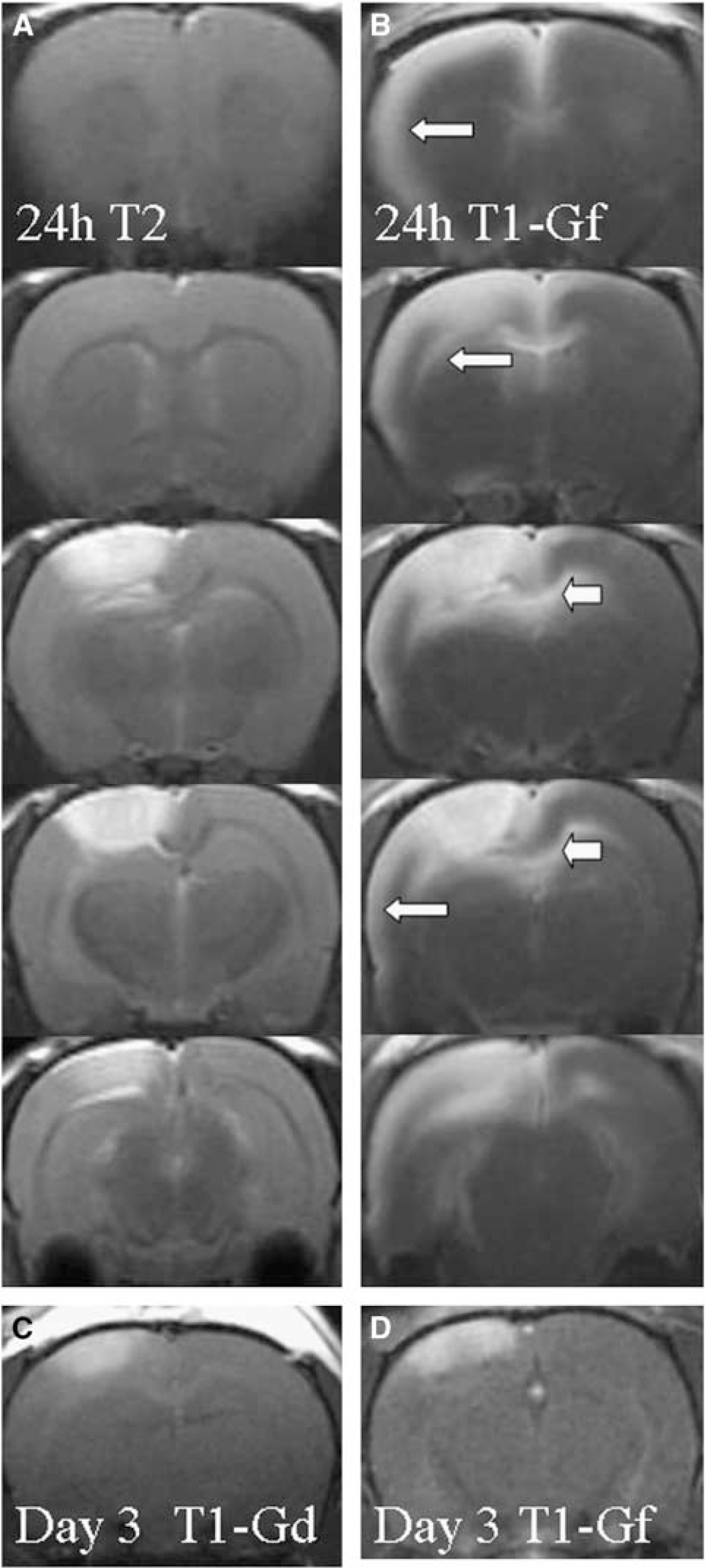
Gf enhancement in relation to tissue damage on T2-w MRI at 24 h after induction of photothrombosis (PT). (
Gadofluorine M-Enhanced Magnetic Resonance Imaging after Photothrombosis
Application of Gf to normal- and sham-operated rats showed contrast enhancement within large arterial and venous vessels on T1-w MRI, but no parenchymal enhancement (not shown). In animals after PT, Gf similarly to Gd-DTPA led to contrast enhancement within the lesion (Figure 2). This lesion-related Gf uptake was present at all stages of lesion development. In contrast to Gd-DTPA, which rapidly disappeared from the lesions again, Gf enhancement persisted in the lesions for several days upon repeated MR examinations (not shown). As our main finding, Gf application led to an additional widespread cortical enhancement involving mainly the entire ipsilateral cortex remote from the lesion and to a lesser extent also the contralateral cortex (Figures 2 and 3). The upper layer of the cerebral cortex frontal, lateral, and occipital from the PT lesion showed strong Gf enhancement at 24 h (arrows in Figure 2B). Moreover the entire corpus callosum exhibited marked Gf uptake (arrowheads in Figures 2B, 3E and 3F). The spatiotemporal evolution of Gf enhancement was further assessed in separate groups of animals in which two subsequent MR examinations were performed at 1 and 3 h, 5 and 9 h, or 18 and 26 h after PT, respectively (Figure 3A–3F). Thereafter the interval between Gf application and MRI varied between 1 and 26 h, which is feasible because Gf has a much longer plasma clearance time than Gd-DTPA (Misselwitz et al, 2004). At 1 h Gf enhancement was restricted to the PT lesion and spared the center because of restricted access of the contrast medium by ongoing vessel occlusion (Kleinschnitz et al, 2005). At 3 h Gf enhancement extended into the upper cortical layer close to the lesion. In the group of animals studied between 5 and 9 h after PT, cortical enhancement further spread ipsilaterally and now involved the frontal and occipital cortex. Gadofluorine M uptake moreover involved white matter tracts most prominently along the corpus callosum (Figure 3E and 3F). The maximum of Gf enhancement was reached around 24 h after PT (Figures 2B, 3E and 3F). Parenchymal enhancement of Gf remote from the lesion seen within the first 24 h persisted on subsequent examinations when the same animals underwent repeat MR at 3 and 7 days after PT (not shown). At these time points Gf was completely eliminated from the circulation. Gadofluorine M enhancement on MRI corresponded to localization of labeled Gf fluorescence on corresponding cryostat sections (Figure 3G and 3H). Of note, remote Gf uptake was a temporally restricted event: Systemic application of Gf later than 24 h after induction of the PT lesion led to Gf enhancement within the lesion, but not in remote areas (shown for day 3 in Figure 2D). At these later stages, focal Gf uptake within the lesion corresponded to Gd-DTPA enhancement (Figure 2C and 2D).
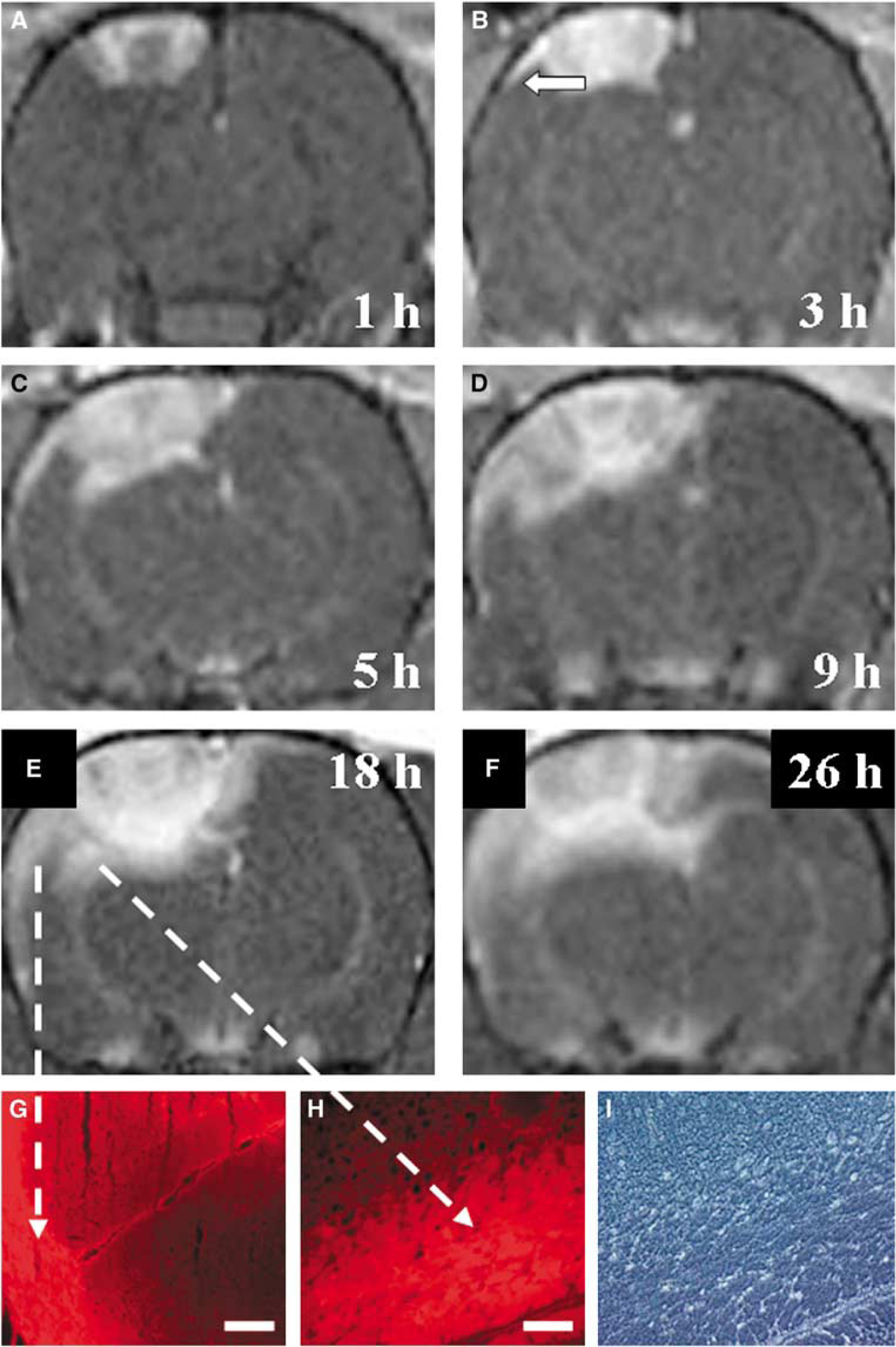
Spatiotemporal evolution of Gf enhancement. (
Transient Breakdown of The Blood—Brain Barrier as Assessed by Evans Blue Extravasation
To further elucidate the processes underlying remote Gf uptake after PT, animals received Evans blue i.v. immediately after irradiation was completed and were killed at different time points thereafter to localize regions showing extravasation of the dye (Figure 4). Immediately after PT the blue dye was localized intravascularly. However, as early as 1 h after induction the lesions appeared blue diffusely and between 3 and 9 h blue staining further extended to the neighboring intact cortex (Figure 4) corresponding to the MRI findings shown in Figure 3. When Evans blue was given with a delay of 48 h and brains were removed at day 3 after PT, only the lesions were stained indicating that the breakdown of the BBB in the remote cortex and corpus callosum is transient and fully reversible further confirming our MRI findings. Staining of paraffin sections with antibodies against rat albumin revealed albumin extravasation within the PT lesions and the remote ipsilateral cortex during the first 24 h after PT (Figure 5A) further substantiating our MRI findings.
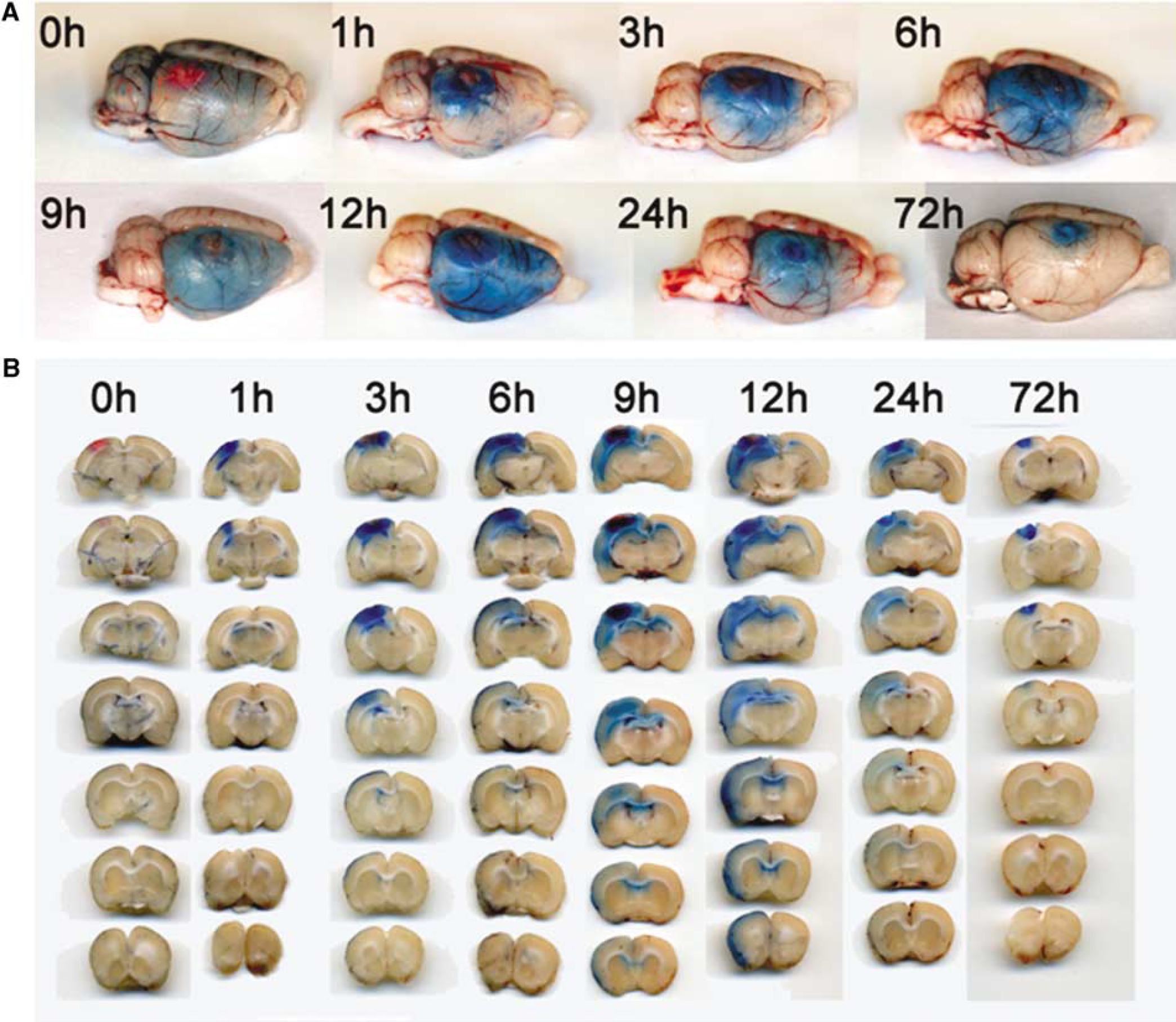
Confirmation of a transient remote disturbance of the blood—brain barrier by Evans blue extravasation. Rats received Evans blue i.v. immediately after irradiation for photothrombosis (PT) induction or after 48 h (for assessment at 72 h). Individual animals were killed at the indicated time point extending from induction of PT up to 72 h. (
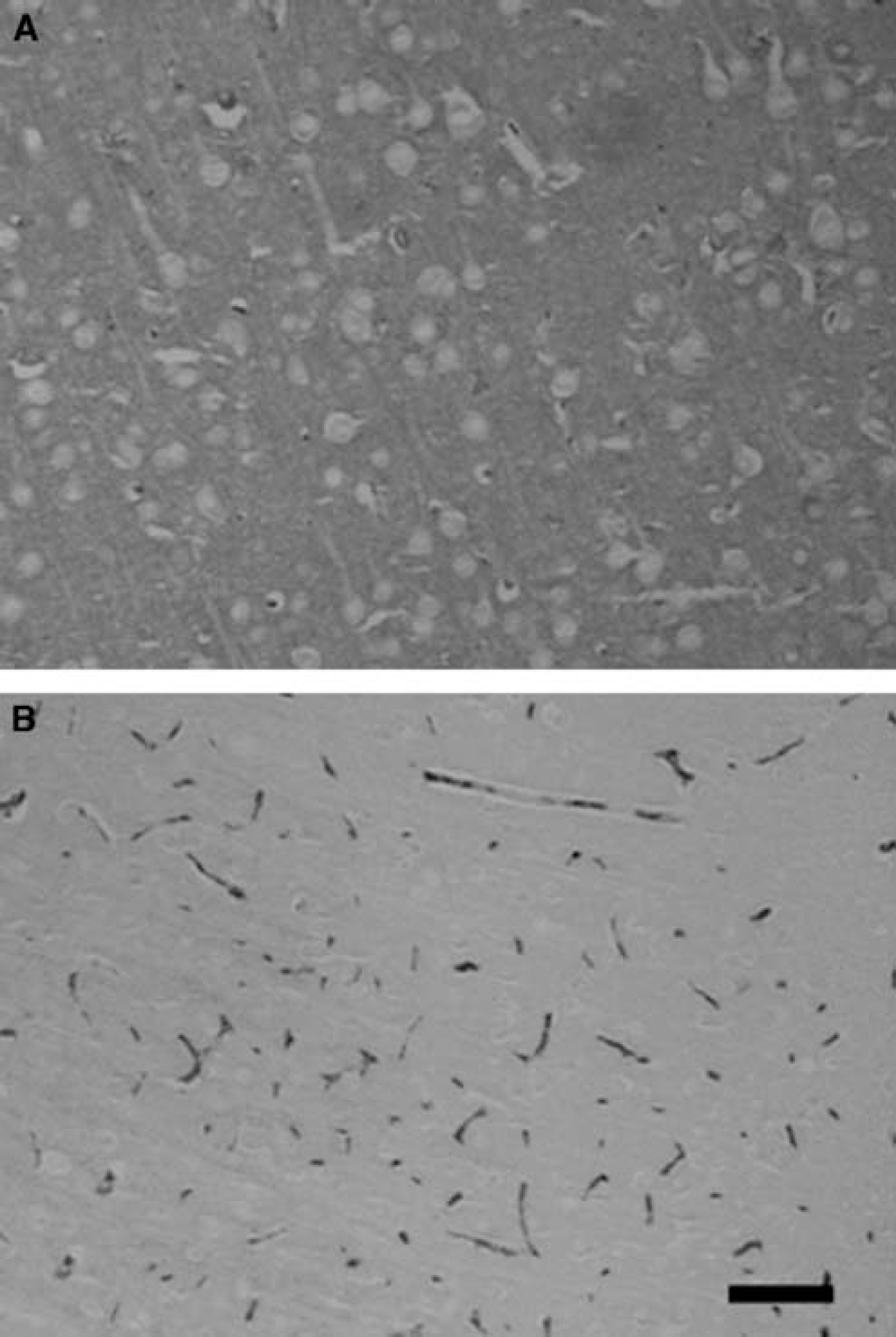
Immunohistochemical demonstration of albumin extravasation. (
Possible Mechanisms of Remote Gadofluorine M Leakage After Photothrombosis
Mechanistically, Gf could have reached the remote cortex by two different ways: (1) it could have leaked out of the PT lesion by diffusion into neighboring normal cortex, or (2) Gf gained direct access into the remote cortex. This latter possibility is suggested by our Evans blue studies showing leakage of the BBB also remote from the PT lesion. To answer this important question brain slices were prepared and kept alive in artificial cerebrospinal fluid containing culture dishes. Brain slices removed immediately after PT and Gf application showed strong Gf enhancement restricted to the PT lesion (Figure 6A–6C). Gadofluorine M was often associated with vessels at the lesion borders initially. Brain slices removed at 1.5 h after PT/Gf application showed additional cortical Gf fluorescence outside the lesion precisely corresponding to MR enhancement (Figure 6D and 6F). In slices prepared at 5 h after PT/Gf application, further spread of Gf into the remote cortex and subcortex was noted, again corresponding to MR findings (Figure 6E and 6G).
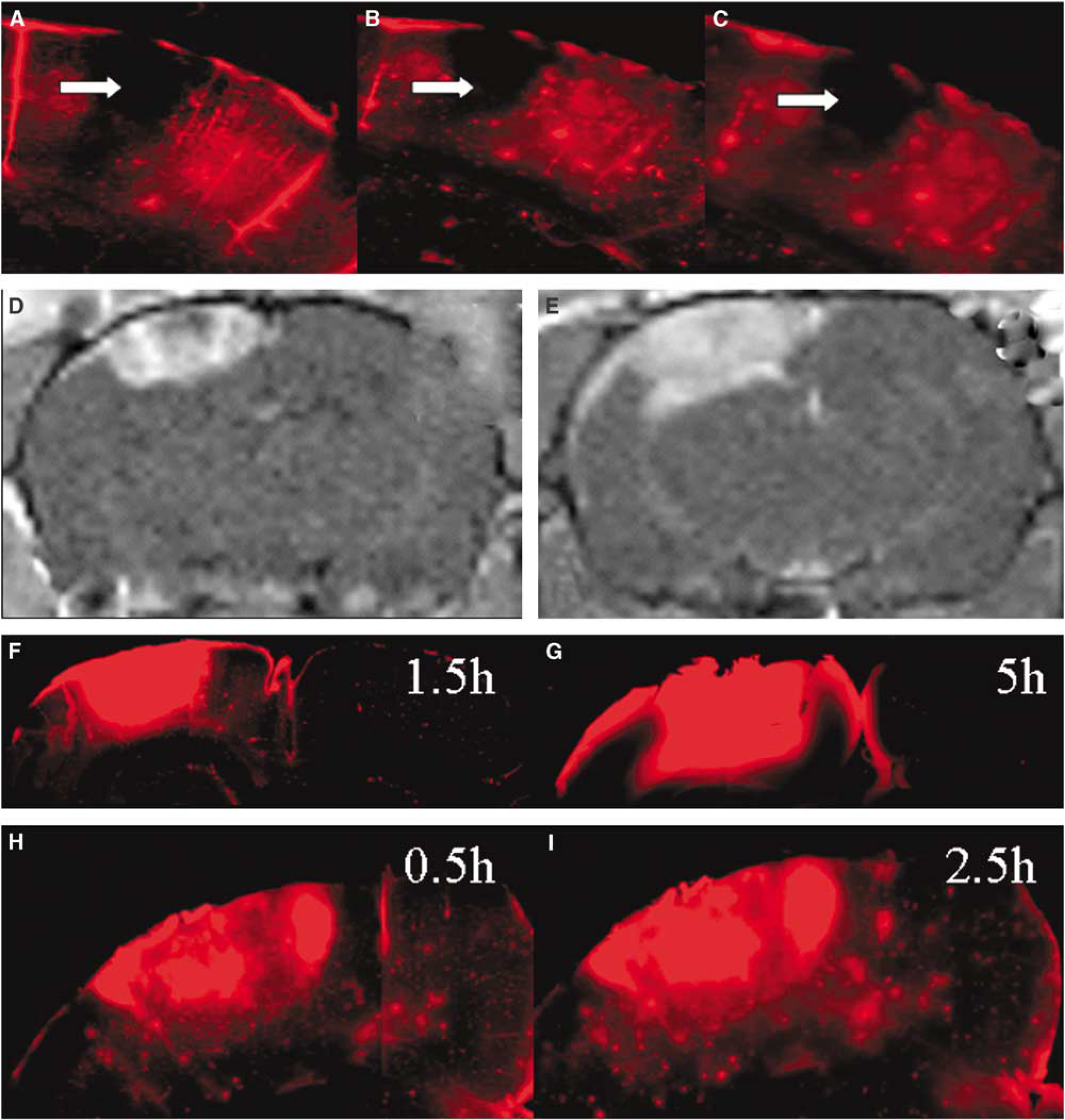
Combined MRI and fluorescence microscopy for labeled Gf in vital brain slices. Brain slices obtained immediately after PT/Gf application (
To further differentiate whether remote Gf enhancement was because of Gf leakage from the PT lesion into the surrounding cortex or by entry by the circulation, Gf was administered i.v. at the time of PT, but brain slices were prepared at 30 mins after PT, and followed by fluorescence microscopy for additional 2 h. In vital brain slices no spread of Gf was seen from the lesion into the surrounding remote cortex in the observation period between 30 mins and 2.5 h after PT (Figure 6H and 6I) whereas in vivo Gf enhancement already involved remote cortical areas at 1.5 h (Figure 6D and 6F). This finding indicates that the widespread Gf enhancement as shown by MRI (Figures 2 and 3) is because of direct entry of Gf from the circulation into the remote cortex, and not merely because of passive Gf diffusion out of the lesion.
The spatial distribution of Gf enhancement and the time frame limited to the first 24 h was reminiscent to the occurrence of CSD. Cortical spreading depression travel over the ipsilateral cortex within the first 24 h after PT, and spontaneously cease thereafter (Dietrich et al, 1994; Jander et al, 2000). To further analyze possible molecular mechanisms of remote Gf enhancement after PT, animals received the NMDA-antagonist MK-801 shortly before PT thereby blocking CSD (Marrannes et al, 1988; Jander et al, 2001). When MK-801-treated and naive rats with PT lesions were compared by T1-w MRI after Gf application, no difference was seen (not shown) indicating that remote Gf enhancement was not a consequence of CSD-related events.
In a further attempt to identify the nature of remote Gf enhancement, rats received intraperitoneal injections of LPS and underwent MRI at days 1 and 3 always 24 h after Gf application. Systemic LPS is known to induce widespread molecular changes in the intact cortex (Singh and Jiang, 2004) including alterations of BBB properties. However, no pathologic signal alterations on T1-w and T2-w MRI were seen before and after Gf application (not shown).
Discussion
As principal finding we show that after a focal brain lesion the novel contrast agent Gf allows visualization of cerebral responses far beyond the lesion site. Our brain slice experiments support the notion that remote Gf enhancement was because of entry of the contrast agent from the circulation rather than leakage from the PT lesion itself: In Gf-labeled vital brain slices with locally intact neuronal networks the location of Gf outside the lesion did not change over time. In contrast, after in vivo application animals accumulated more Gf in remote areas between two subsequent MR examinations performed at 1/3, 5/9, and 18/26 h, respectively. The distribution of Gf enhancement on MRI within the first 24 h after PT exactly matched to the pattern of Evans blue extravasation as shown in our study and similarly described by Dietrich et al (1987a) 20 years ago. Diffuse blue staining was found within cortical and subcortical regions remote from the lesion, and within the underlying corpus callosum. This, moreover, corresponded to the leakage of albumin. We cannot exclude that our method of generating the PT lesion may have exceeded the bounds of standard PT as originally described by Watson et al (1985), but this does not affect the content of our study: lesions showed both Gf- and Gd-DTPA enhancement on T1-w MRI, but remote responses could only be visualized by Gf-enhanced MRI.
Gadofluorine M features an increased molecular weight compared with Gd-DTPA (1,528 versus 940 g/mol; Misselwitz et al, 2004). Therefore, one would expect to see Gd-DTPA enhancement rather than Gf enhancement. It appears, however, that Gf has unique binding properties (Misselwitz et al, 2004; Giesel et al, 2006; Raatschen et al, 2006; Meding et al, 2007). In contrast to Gd-DTPA, Gf binds reversibly to plasma proteins mainly albumin and avidly forms Gf—protein complexes (95% binding in serum). Albumin is a large, negatively charged globulin protein (molecular weight 66 kDa). The exact mechanism of Gf entry into the CNS is unclear, but it is conceivable that the majority of Gf gains access to the brain parenchyma by protein (albumin) extravasation and is locally captured by subsequent local interactions. In support of this notion Gf enhancement persisted for a long time period in remote areas when given within the first 24 h after PT. In contrast, enhancement of Gd-DTPA is a transient event of passive diffusion against a concentration gradient along the disrupted BBB. Recently, in vitro studies have revealed a strong binding of Gf to the extracellular matrix proteins collagen, proteoglycans, and tenascin (Meding et al, 2007). These proteins could locally trap Gf within the CNS after its entry through a transiently defective BBB after PT.
Importantly, active Gf accumulation remote from the lesion was temporally restricted to the first 24 h after focal cerebral ischemia. This indicates a transient, reversible leakage of the BBB in remote brain areas. Although the precise mechanisms for this transient alteration of BBB properties are unknown yet, it is already well established in the PT model that the ipsilateral hemisphere shows a multifactorial response within distinct time frames (reviewed in Stoll et al, 1998; Witte et al, 2000). On the cellular level microglia reversibly upregulate surface molecules such as complement receptor 3, and astrocytes exhibit an activated phenotype by increased expression of the structural protein glial fibrillary acidic protein (Schroeter et al, 1995, 1999). These morphologic changes are most evident after 3 days and spontaneously disappear after day 7. Functionally, astrocytes are involved in the local microcirculation within the brain by controlling cerebral blood flow (Takano et al, 2006). Besides endothelial cells, astrocytes are also an integral part of the BBB separating the circulation from the cerebral parenchyma (Ballabh et al, 2004; Hawkins and Davis, 2005). Astrocytes are interconnected by gap junctions, which consist of several channel proteins including connexin 43 (Rouach et al, 2000). Recent evidence supports the notion of a disturbance of gap junctional intercellular communication in remote areas after PT. Connexin 43 expression was decreased in postischemic cerebral cortex after PT (Haupt et al, 2007). Astrocytes and gap junction proteins, moreover, are instrumental in the propagation of CSD (Martins-Ferreira et al, 2000). In fact, CSD-like depolarizations could be registered in the vicinity of PT lesions within the first 24 h (Dietrich et al, 1994). The propagation of CSD can be inhibited by the noncompetitive NMDA-receptor antagonist MK-801 (Marrannes et al, 1988; Lauritzen and Hansen, 1992). To delineate a role of NMDA-receptor signaling in remote BBB disturbance, animals received an intravenous injection of MK-801 (2 mg/kg body weight) to block CSD (Jander et al, 2001). However, in these animals MRI revealed similar Gf enhancement as in naive animals after PT. This is surprising because CSD have been shown to induce BBB dysfunction by activation of the matrixmetalloprotease-9 (Gursoy-Ozdemir et al, 2004). As an alternative explanation widespread inflammatory responses such as upregulation of the proflammatory cytokines interleukin-1-β and tumor necrosis factor-α in the surround of PT brain lesions affecting endothelial cell function (Blamire et al, 2000; Jander et al, 2000; Ballabh et al, 2004) could account for Gf leakage. Systemically applied LPS can mimic proinflammatory cytokine induction in the normal CNS without structural damage (Singh and Jiang, 2004) and, moreover, can alter BBB properties by binding to its receptor on endothelial cells (Xaio et al, 2001). We therefore injected LPS intraperitoneally and studied Gf uptake in the brain by MRI in these animals without PT lesion. However, no Gf extravasation into the neocortex was observed.
In conclusion, our study shows that Gf can visualize subtle disturbances of the BBB not covered by the conventional MR-contrast agent Gd-DTPA. We moreover add evidence to the concept that focal ischemic lesions induce widespread functional alterations of the brain. Although the molecular mechanisms of the remote BBB disturbance await further clarification, brain PT served as a valuable CNS lesion model to elucidate the unique properties of Gf. Depending on further clinical development, Gf holds promise as a novel MR-contrast agent for more sensitive detection of areas with an impaired BBB and may help to elucidate the extent and functional impact of remote reactions to focal brain lesions.
Footnotes
Acknowledgements
We thank Dr B Misselwitz, Bayer Schering Pharma AG, for providing Gf and helpful comments on the article. We thank the expert technical assistance of Melanie Glaser, Gabi Köllner, Tanja Horn, and Virgil Michels (Würzburg). MB held a professorship donated by the Bayer Schering Pharma AG, Berlin, Germany, to the University of Würzburg (2002–2007).
Disclosure/conflict of interest
The authors declare no conflict of interest.
