Abstract
Our previous studies show that long-term testosterone treatment augments vascular tone under physiological conditions and exacerbates endotoxin-induced inflammation in the cerebral circulation. However, testosterone can be metabolized by aromatase to estrogen, evoking a balance between androgenic and estrogenic effects. Therefore, we investigated the effect of the nonaromatizable androgen receptor agonist, dihydrotestosterone (DHT), on the inflammatory nuclear factor-κB (NFκB) pathway in cerebral blood vessels. Cerebral arteries were isolated from orchiectomized male rats treated chronically with DHT in vivo. Alternatively, pial arteries were isolated from orchiectomized males and were exposed ex vivo to DHT or vehicle in culture medium. DHT treatment, in vivo or ex vivo, increased nuclear NFκB activation in cerebral arteries and increased levels of the proinflammatory products of NFκB activation, cyclooxygenase-2 (COX-2) and inducible nitric oxide synthase (iNOS). Effects of DHT on COX-2 and iNOS were attenuated by flutamide. In isolated pressurized middle cerebral arteries from DHT-treated rats, constrictions to the selective COX-2 inhibitor NS398 or the selective iNOS inhibitor L-nil, [L-N6-(Iminoethyl)lysine], were increased, confirming a functional consequence of DHT exposure. In conclusion, activation of the NFκB-mediated COX-2/iNOS pathway by the selective androgen receptor agonist, DHT, results in a state of vascular inflammation. This effect may contribute to sex-related differences in cerebrovascular pathophysiology.
Introduction
Effects of sex steroids on cerebral vascular function and cerebrovascular pathophysiology are complex. Experimental stroke models in rodents show that androgen treatment exacerbates brain injury after ischemic insult (Cheng et al, 2007; Hawk et al, 1998). In contrast, 17β-estradiol significantly reduces ischemic injury (Alkayed et al, 1988; Simpkins et al, 1997). Studies have shown that 17β-estradiol decreases cerebrovascular tone by enhancing endothelial-dependent pathways (Geary et al, 1998; Krause et al, 2006). However, androgenic compounds such as testosterone have the opposite effect; they enhance tone of cerebral vessels (Gonzales et al, 2004). Another example of different effects of estrogen and androgen is observed with vascular inflammation. Using lipopolysaccharide to induce inflammation, long-term testosterone treatment augments levels of the proinflammatory enzymes, cyclooxygenase-2 (COX-2) and inducible nitric oxide synthase (iNOS), in male rat brain blood vessels (Razmara et al, 2005). In contrast, this same study showed that treatment of male rats with 17β-estradiol significantly suppresses cerebrovascular levels of COX-2 and iNOS protein after an inflammatory stimulus. Taken together, these data suggest that androgens, unlike estrogens, may influence ischemic injury by enhancing proinflammatory processes in the vasculature.
Certainly, the brain vasculature plays a central role in initiation of inflammation after brain ischemia (Del Zoppo and Mabuchi, 2003; Nedergaard et al, 2003), and inflammation is a critical process in the outcome of conditions such as stroke (Dirnagl et al, 1999; Emsley and Tyrrell, 2002). However, it is important to point out that inflammation may have both beneficial and injurious effects (Kempermann and Neumann, 2003). Acute inflammatory responses are part of the normal healing process and may be beneficial. However, in the brain, prolonged inflammation is thought to increase edema, decreasing cerebral blood flow and exacerbating neurologic dysfunction.
Interestingly, virtually no data have been reported on the influence of androgens on inflammatory pathways in the cerebral vasculature under physiologic conditions in the absence of induced inflammation (i.e. ischemic insult or endotoxin exposure). In addition, many of the specific molecular and cellular mechanisms underlying sex differences under both normal and pathophysiological conditions remain unclear; this is especially true of the cerebral circulation. Thus, it is important to determine the contribution of androgen actions on the tissue.
Not all reports agree that androgens exacerbate stroke. For example, clinical studies show that the severity of stroke in aged men is inversely proportional to circulating testosterone (Jeppesen et al, 1996). An important factor that may account for these discrepancies concerns the ability of testosterone to exert effects either directly on the androgen receptor or by means of metabolism to either the potent androgen, dihydrotestosterone (DHT) or aromatization to estrogen. Previously we have shown that the testosterone metabolizing enzymes, 5-α reductase and aromatase are present in cerebral arteries (Gonzales et al, 2007), underscoring the possibility that, because of local tissue differences in the distribution of testosterone metabolizing enzymes, the balance of androgenic and estrogenic effects caused by testosterone may vary regionally.
Therefore, to better understand the nature of androgenic effects on the cerebral vasculature, we investigated the influence of DHT, a testosterone metabolite not subject to aromatization and more potent as an agonist at the androgen receptor, on the nuclear factor-κB (NFκB) pathway and downstream proinflammatory enzymes, COX-2 and iNOS. We first determined whether long-term DHT treatment modulates NFκB activation and levels of COX-2 and iNOS in cerebral arteries. We then evaluated the possible functional consequence of the modulation of these proinflammatory factors by DHT in isolated, pressurized middle cerebral arteries.
Methods
Chronic In Vivo Hormone Treatment
All experimental and surgical protocols were approved by the Institutional Animal Care and Use Committee of the University of California, Irvine. MaleWistar rats (3 months old; Charles Rivers, Raleigh, NC, USA) were orchiectomized by the supplier and delivered 1-week postsurgery. At 1 day after arrival, rats were anesthetized with isoflurane (1.5%) and aseptically implanted with placebo or 5α-androstan-17α-ol-3-one (DHT; 25 or 45 mg per 21 days) pellets (Innovative Research of America Inc., Sarasota, FL, USA). Pellets were tunneled 2 to 3cm underneath the skin, and the incision site was secured with small rodent staples. Postsurgery animals were treated with a single injection (i.m.) of penicillin (penicillin G benzathine/penicillin G procaine, 30,000 U) and a topical triple antibiotic. After anesthesia recovery, rats were returned to animal housing (12:12 h light/dark cycle) with fresh water, food and bedding.
Past studies from our laboratory showed that 3 to 4 weeks of testosterone treatment alters vascular reactivity. In addition, a study by Cheng et al (2007) showed that DHT enhanced ischemic brain injury and altered a number of genes including COX-2 after 1-week of hormone treatment. On the basis of outcome of these past studies, animals were treated with DHT or placebo for approximately 3 weeks (18 to 21 days) to assess the biochemical and vascular effects of this potent androgen receptor agonist on cerebral arteries.
At the time of euthanasia, animals were deeply anesthetized with sodium pentobarbital (50 mg/kg i.p.), and body weight recorded. Next the thoracic cavity was exposed, and a direct cardiac puncture was used to collect blood samples for serum DHT levels. Samples were stored at −80°C until DHT levels were measured with an ELISA kit (Alpha Diagnostics; San Antonio, TX, USA; DHT ELISA limit of detection = 6 pg/ml). After blood collection, animals were immediately heparinized, exsanguinated and decapitated. For prostate weight measurements, the urogenital complex (prostate—urethra—bladder) was dissected free from the lower abdominal cavity of each animal and placed in a dissection dish containing physiological saline solution (PSS). Next, the dorsolateral and ventral lobes of the prostate were dissected free from the bladder/urethra, and each specimen was placed on wax paper and allowed to air dry overnight before recording dry weight Together with serum DHT levels, prostate weights were used to verify efficacy of androgen treatment. To collect arteries, the brain was removed and placed in a Sylgard-coated dissection dish containing ice-cold PBS. Arterial segments (middle cerebral artery (MCA) and/or pial arteries) were carefully dissected using a dissecting microscope and prepared for Western blot, nuclear isolation/NFκB activation measurements or functional contractile studies.
Ex Vivo Hormone Treatment
Under aseptic conditions, the brain was removed from orchiectomized rats, and pial arteries were dissected and placed in a sterile 12-well incubation plate containing ice-cold Dulbecco's modified Eagle's medium (DMEM; n = 1 per well). Artery segments were then transferred to wells containing a 1:1 mix of pre-warmed (37°C) phenol red free Dulbecco's modified Eagle's medium (Sigma Chemical, St Louis, MO, USA) and serum- and hormone-free Medium 231 (Cascade Biologics; Portland, OR, USA). Arteries were equilibrated for 30mins in a 5% CO2 incubator at 37°C before being transferred to wells containing fresh medium (Dulbecco's modified Eagle's medium/Medium 231) and the indicated hormone, drug or vehicle (VEH) treatment. Artery segments used for NFκB activation studies were incubated for 1h at 37°C whereas artery segments used for Western analysis were incubated for 8 h at 37°C.
NFκB p65 Activation Measurements
Nuclei were isolated from pial artery segments using a nuclear extraction kit following the manufacturer's instructions (Active Motif, Carlsbad, CA, USA). All nuclear fractions were stored at −80°C before analysis. Western blot of the nuclear marker histone-1 (Santa Cruz Biotechnology; Santa Cruz, CA, USA) verified separation of nuclear and cytosolic fractions (data not shown). Protein content for nuclear fractions was determined by the bicinchoninic acid assay (Pierce, Rockford, IL, USA).
DNA binding activity for NFκB p65 was measured in pial artery nuclear extracts. In brief, pial artery nuclear protein samples (10 µg per well) or pial artery nuclear extract samples exposed to cobalt chloride (2.5 µg per well; positive control) were added to 96-well plates labeled with an immobilized oligonucleotide containing the NFκB consensus site (5′-GGGACTTTCC-3′). Activated NFκB was detected after 1 h incubation using an anti-NFκB p65 antibody that recognizes an epitope accessible only when NFκB is activated and bound to its target DNA (TransAM NFκB p65 immunoassay-based kit; Active Motif). Secondary horseradish peroxidase antibody and developing solution exposure completed the reaction. Absorbance values as a result of the colorimetric reaction were measured within 5 mins using a spectrophotometer at 450 nm.
Western Blot
Protein levels for the proinflammatory markers, COX-2 and iNOS, were determined using standard immunoblotting methods. Briefly, pial arteries were gently homogenized in ice-cold lysis buffer (pH 7.4). Lysis buffer contained β-glycerophosphate (50 mmol/L), sodium orthovanadate (100 µmol/L), magnesium chloride (2 mmol/L), EGTA (1 mmol/L) and Triton X-100 (0.5%) and was prepared fresh. Protease inhibitors (DL-dithiolthreitol 1 mmol/L, phenylmethylsulfonyl fluoride 1 mmol/L, pepstatin-A 20 µmol/L, leupeptin 20 mmol/L and aprotinin 0.1 U/mL) were individually prepared as stock solutions dissolved in ddH2O or ethanol and stored according to supplier's suggestion. After 20 mins incubation, homogenates were centrifuged (4500g for 10 mins 4°C), the supernatant drawn off, and a small portion of sample was analyzed for protein content using a bicinchoninic acid protein assay (Pierce). Samples for Western analysis were dissolved in Tris-glycine SDS sample buffer and boiled for 6 mins. Equal amounts of protein (ranging from 30 to 40 mg per lane per experiment) were then loaded and separated in 8% polyacrylamide gels using SDS-polyacrylamide gel electrophoresis. Separated proteins were transferred to polyvinylidene difluoride membranes and blocked overnight at 4°C in PBS (0.1% Tween and 6.5% nonfat dry milk). Membranes were probed with antibodies specific for either COX-2 (1:500), iNOS (1:500) or GAPDH; 1:20,000; Santa Cruz Biotechnology). Proteins of interest were visualized using either enhanced chemiluminescence in conjunction with a horseradish peroxidase-labeled secondary antibody (1:15,000) or infrared imaging system (LI-COR Biosciences, Lincoln, NE, USA) using a fluorescently labeled secondary antibody (1:20,000; IRDye 680 goat anti-rabbit, LI-COR Biosciences). Densitometry of bands was analyzed with UN-SCAN-it software (Silk Scientific, Orem, UT, USA) or the analysis software provided by LI-COR Biosciences. Each blot included samples from all of the relevant groups along with GAPDH-loading controls. Intensity ratio for each band was normalized to the intensity of its corresponding GAPDH-loading control. The normalized values were then expressed as a ratio relative to control (placebo or VEH) within each blot.
Isolated Pressurized Middle Cerebral Artery Preparation
A segment of the second-order MCA (250 µm diameter) was carefully dissected from whole brain, kept in ice-cold PSS containing Ca2+ and aerated with oxygen/carbon dioxide gas mixture. The proximal end of the artery was cannulated with a glass micropipette and secured with a single cotton filament. Residual blood cells were gently rinsed from the lumen, and the distal end of the MCA was cannulated and secured in a small vessel bath (Living Systems, Burlington, VT, USA). Each MCA was slowly pressurized to 60 torr with PSS using a servo-controlled peristaltic pump (Living Systems) whereas superfusing with aerated PSS (21% O2/%6 CO2, balance N2; pH 7.4) at 37°C. Intraluminal diameters were visualized using an inverted microscope (Nikon, USA) equipped with a CCD camera/computer interface system (IonOptix Corp., Milton, MA, USA) and measured using an electronic dimension analyzer (Living Systems). Changes in intraluminal diameter were recorded using a computer-based data acquisition edge-detection system (IonOptix Corp.).
Functional Responses
Middle cerebral artery segments were first equilibrated in PSS for 1 h at 60 torr, and diameter was measured at increasing step pressures (60, 80 and 100 torr) before and after drug testing. At the end of each experiment, arteries were incubated in Ca2+-free PSS containing EDTA (3 mmol/L; 30 mins), and intraluminal step pressure changes (60, 80 and 100 torr) were recorded to obtain passive diameters (Table 2). In this paper, we refer to vascular tone as the difference in arterial diameter relative to the passive diameter in the presence of Ca2+-free PSS. In experiments to measure the effect of COX-2 inhibition, arteries were incubated for 30 mins in the presence of the nonselective NOS inhibitor, NG-nitro-
Body weight, prostate weight and dihydrotestosterone serum levels
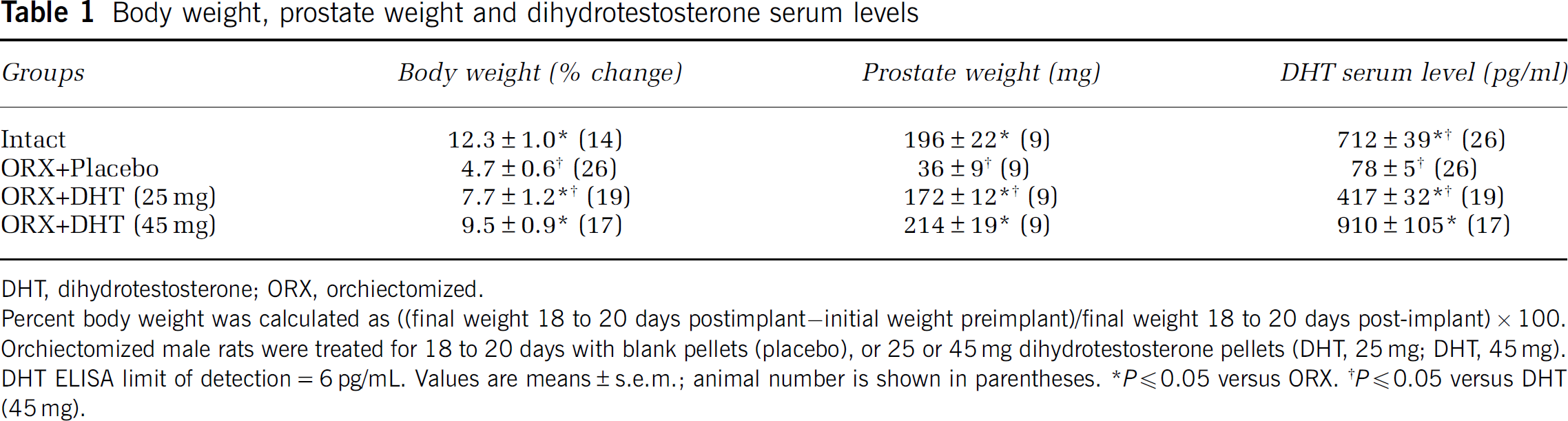
DHT, dihydrotestosterone; ORX, orchiectomized.
Percent body weight was calculated as ((final weight 18 to 20 days postimplant—initial weight preimplant)/final weight 18 to 20 days post-implant)×100.
Orchiectomized male rats were treated for 18 to 20 days with blank pellets (placebo), or 25 or 45 mg dihydrotestosterone pellets (DHT, 25 mg; DHT, 45 mg).
DHT ELISA limit of detection = 6 pg/mL. Values are means±s.e.m.; animal number is shown in parentheses.
P≪0.05 versus ORX.
P≪0.05 versus DHT (45 mg).
Passive diameters in Ca2+-free PSS with EDTA in isolated pressurized middle cerebral arteries
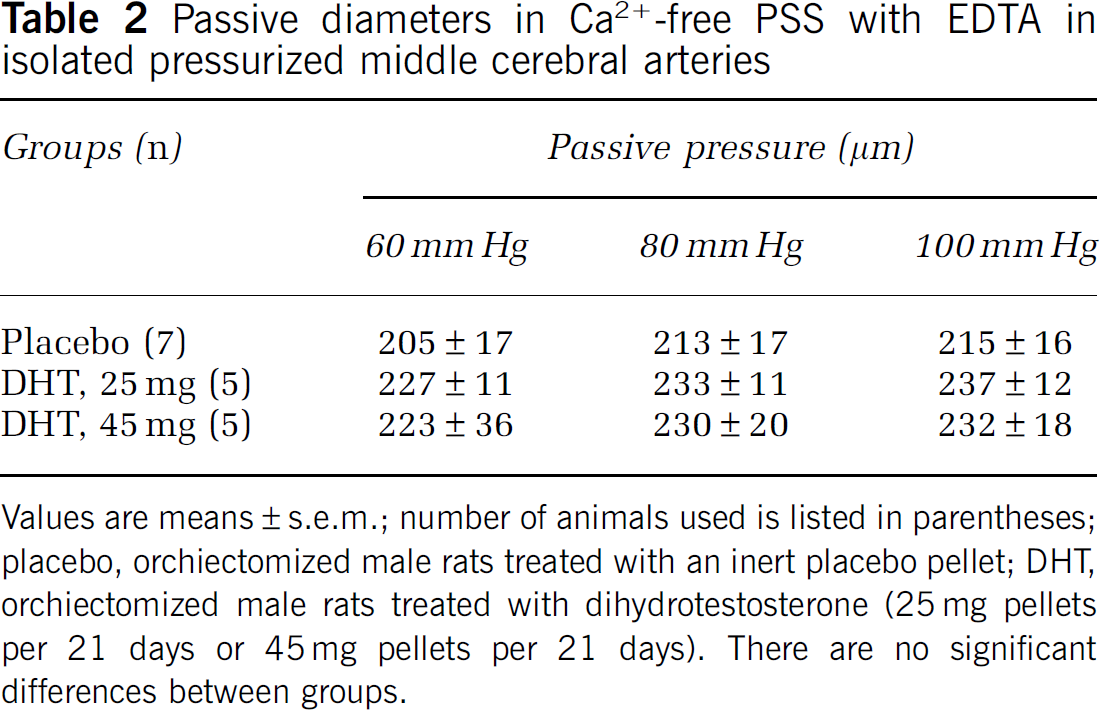
Values are means±s.e.m.; number of animals used is listed in parentheses; placebo, orchiectomized male rats treated with an inert placebo pellet; DHT, orchiectomized male rats treated with dihydrotestosterone (25 mg pellets per 21 days or 45 mg pellets per 21 days). There are no significant differences between groups.
Drugs and Chemicals
PSS bicarbonate/phosphate buffer stock solution contained (in mmol/L): 122 NaCl, 5.1 KCl, 1.2 MgSO4, 1.2 KH2PO4, 25.5 NaHCO3, and 0.03 EDTA, 11.5 mmol/L glucose and 1.6 mmol/L CaCl2. NG-nitro-
Statistical Analysis
For group comparisons, all Western blots included samples from each relevant group so that these were analyzed on a single blot. Band densities were compared using analysis of variance with repeated measures, and the Student—Newman—Keuls test was used post hoc when analysis of variance indicated statistical differences. Change in body weight was calculated as: ((final weight postimplant—initial weight preimplant)/final weight postimplant) ×100). Changes in body weight were compared using analysis of variance and Student—Newman—Keuls post hoc test. P≪0.05 was considered statistically significant for analysis.
Results
Dihydrotestosterone Increased Body and Prostate Weights
Increase in body weight over 18 to 20 days was greater in intact males and orchiectomized males receiving long-term in vivo DHT treatment (25 or 45 mg) compared with animals treated with placebo pellets (Table 1). Measurements for prostate weight and DHT serum levels in intact animals were used as an index for a physiological range of DHT in the hormone replacement groups. Orchiectomy resulted in a significant decrease in both prostate weight and serum DHT levels compared with levels in intact males. Replacement of DHT dose dependently increased both prostate weight and DHT serum levels; the 45 mg DHT dose resulted in prostate weights comparable with those recorded in intact males. Similar to prostate weight, long-term DHT treatment dose dependently increased DHT serum levels in the physiological range (Table 1). Therefore, in the remainder of this study, we assessed the effects of DHT itself by comparing orchiectomized animals treated with either DHT or placebo pellets.
Dihydrotestosterone Enhanced NFκB Activation
We determined whether long-term DHT treatment, in vivo or ex vivo, activated the NFκB pathway in cerebral arteries. Figure 1A illustrates that long-term treatment with DHT (in vivo) increased nuclear NFκB DNA-binding activity in cerebral artery nuclear homogenates compared with nuclear homogenates from placebo-treated rats. In DHT groups, NFκB DNA-binding activity was increased by 26% (DHT 25) and 32% (DHT 45) compared with placebo. Cerebral arteries removed from orchiectomized rats and then incubated with DHT in culture (ex vivo) for 1 h also showed significant increases in nuclear NFκB DNA binding compared with VEH treatment (Figure 1B). For ex vivo studies cobalt chloride (CoCl2)-stimulated vessels were used as a positive control for NFκB pathway activation.
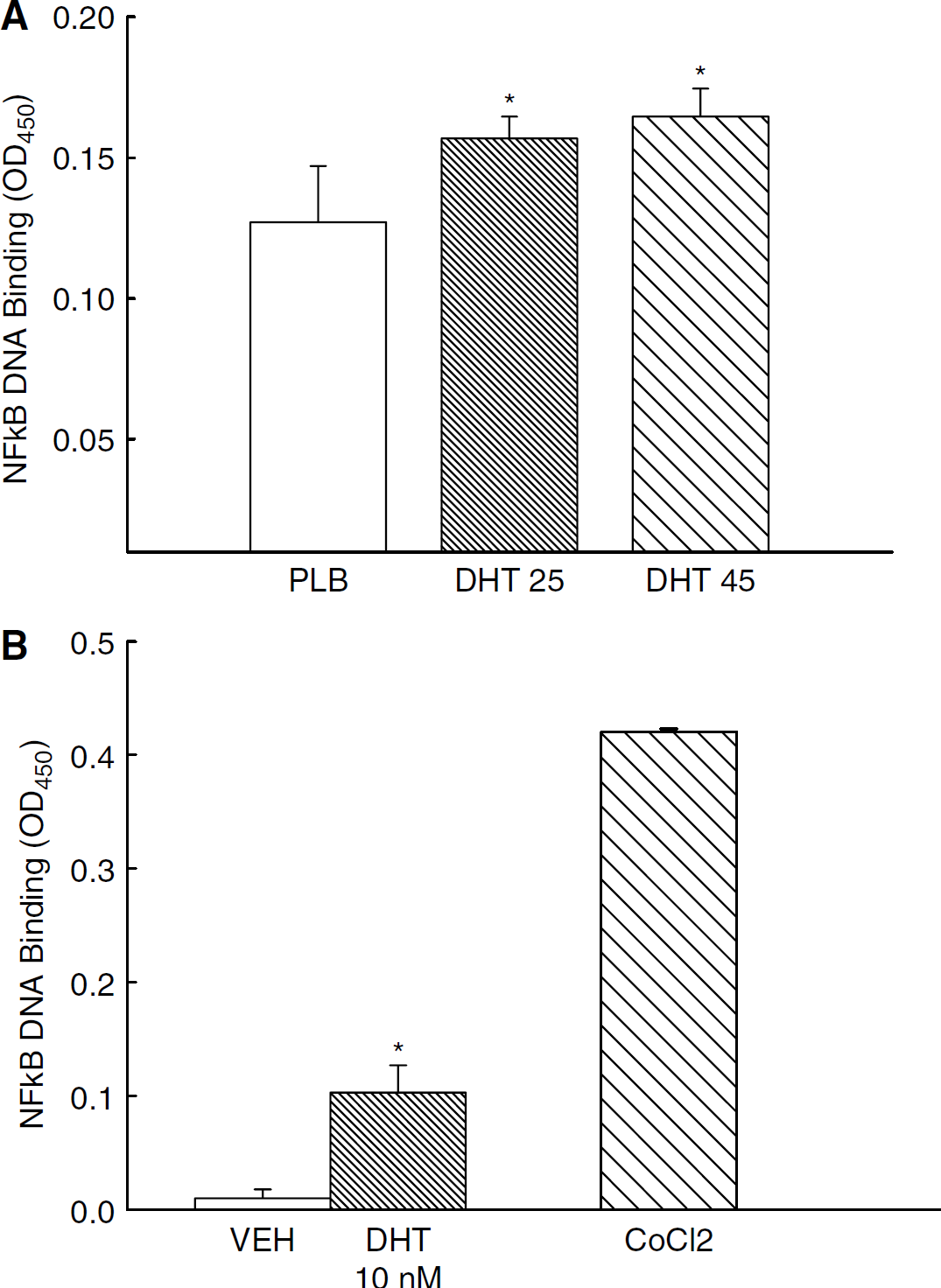
Effect of in vivo or ex vivo dihydrotestosterone treatment on NFκB activation. Nuclear NFκB DNA binding activity was measured in pial arteries isolated from orchiectomized male rats (
Dihydrotestosterone Increased Cyclooxygenase-2 Protein Levels
Because DHT increased NFκB activation, we assessed whether DHT also increases COX-2, a proinflammatory product of the NFκB transcription pathway. Figure 2A depicts a representative Western blot and levels of COX-2 protein in cerebral arteries of rats treated extensively with DHT. In vivo DHT treatment administered at doses of 25 and 45 mg per pellet significantly increased levels of COX-2 in cerebral arteries compared with animals receiving placebo (Figure 2A). Consistent with studies of in vivo treatment, ex vivo administration of 10nmol/L DHT for 8h also increased COX-2 protein levels in cerebral arteries (Figure 2B). Together these data suggest that this increase in COX-2 is an androgenic response localized within the cerebrovasculature.
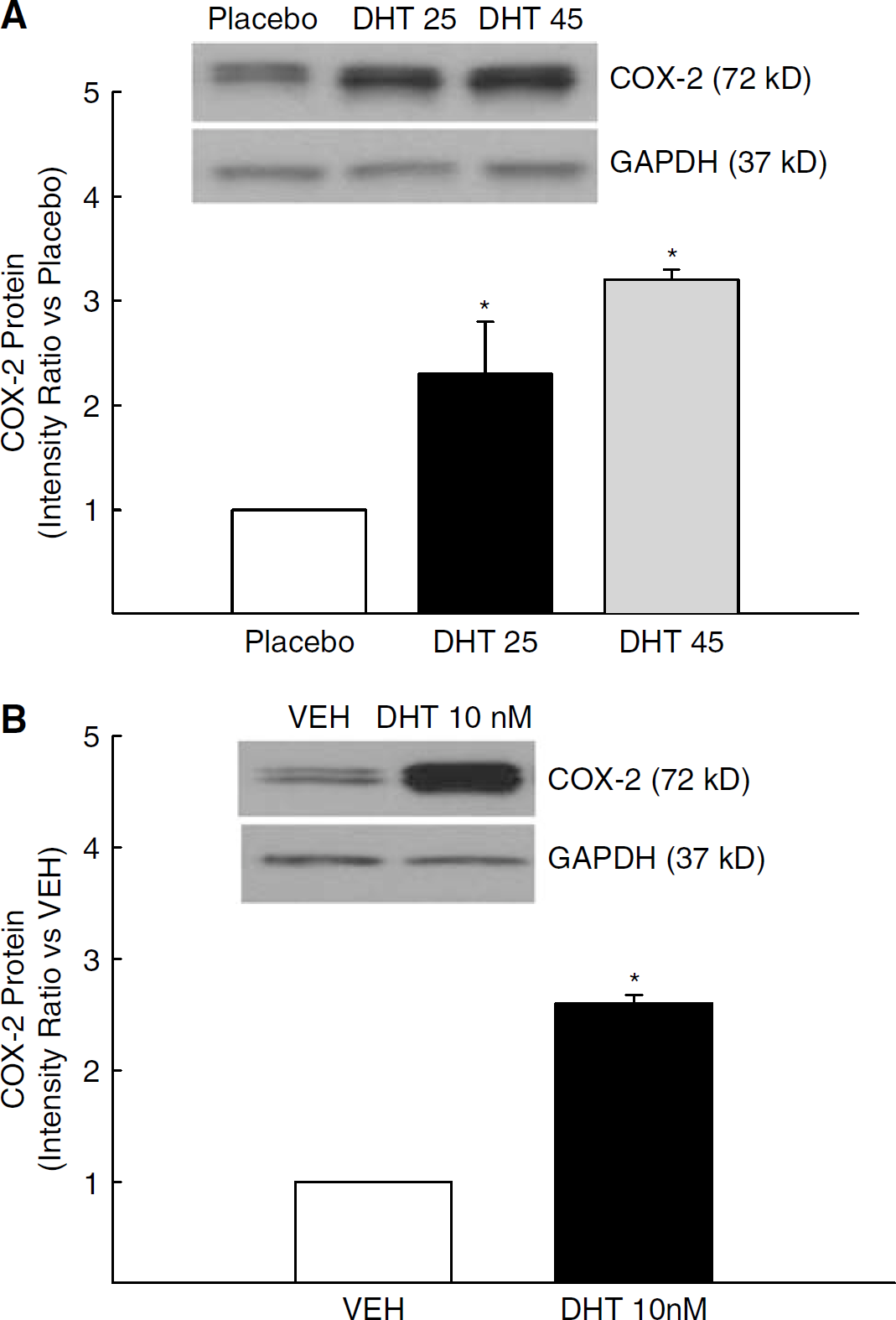
COX-2 protein levels in pial arteries isolated from orchiectomized rats treated (
Flutamide Blunted Dihydrotestosterone-Mediated Increases in COX-2 and iNOS Protein
Because DHT (in vivo or ex vivo) increased levels of the proinflammatory marker, COX-2 (Figure 2), we also investigated whether DHT alters iNOS, another proinflammatory marker known to be regulated by the NFκB pathway. In addition, the effect of DHT to alter COX-2 and iNOS levels via the androgen receptor was assessed using the androgen receptor antagonist, FLU. Figure 3A illustrates a representative Western blot for both COX-2 and iNOS proteins in pial arteries that had been incubated ex vivo (8 h) with VEH, DHT 10 nmol/L, VEH+ FLU(10 nmol/L) or DHT+ FLU. In each experiment, GAPDH (37 kDa) was visualized to verify equal sample loading. Intensity ratio analysis showed that both COX-2 and iNOS levels were significantly increased in cerebral arteries after ex vivo DHT treatment compared with VEH (Figure 3B). Treatment with FLU had no effect by itself, but FLU significantly blunted DHT-induced increases in protein levels of both COX-2 and iNOS.
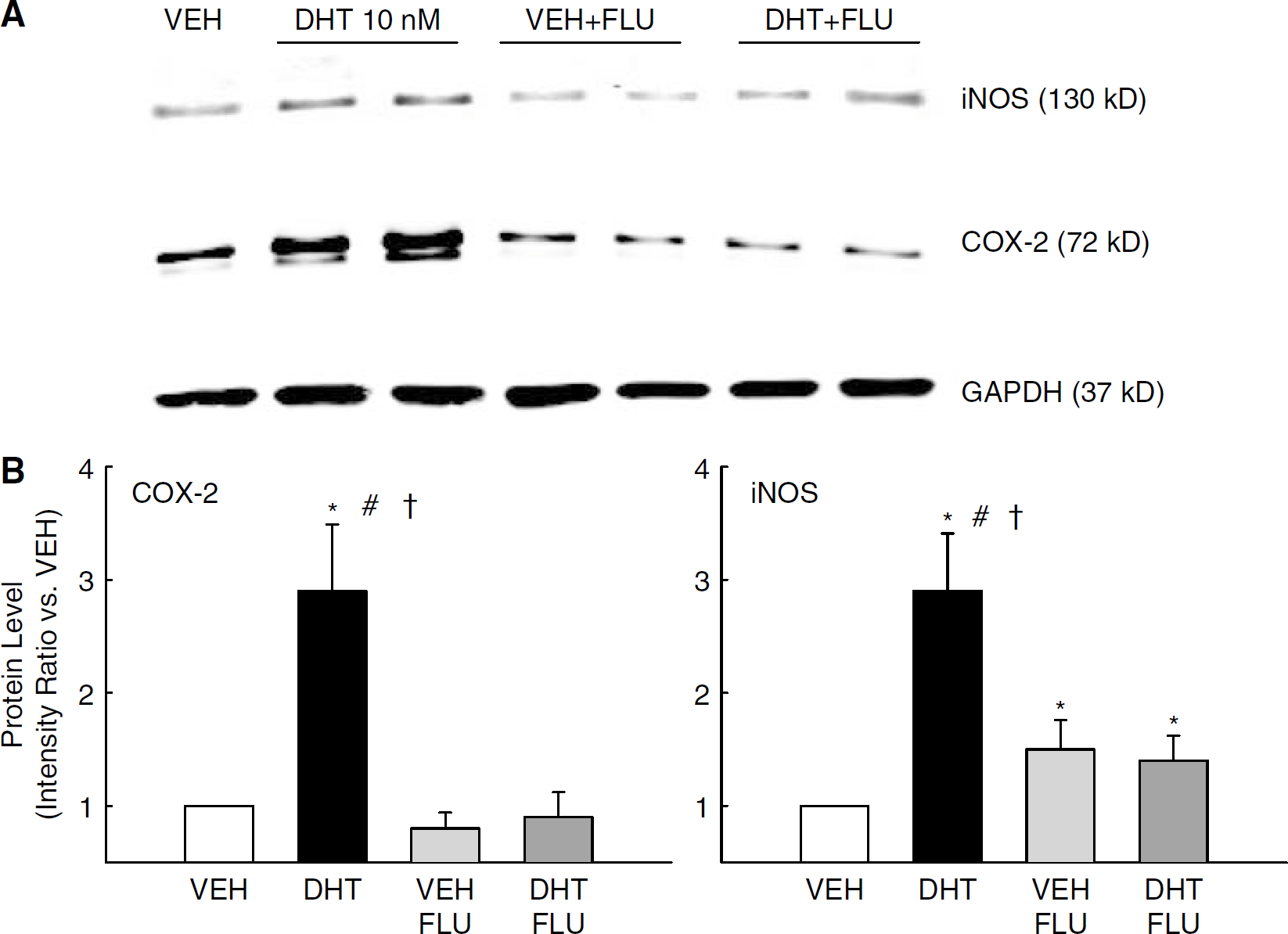
COX-2 and iNOS protein levels in pial arteries incubated ex vivo with DHT (10 nmol/L), vehicle (VEH), VEH plus flutamide (10 nmol/L; VEH+FLU) or DHT plus flutamide (DHT+FLU) for 8 h. GAPDH was used to verify equal protein loading in each lane. (
Dihydrotestosterone Increased Constriction to a COX-2 Inhibitor in Pressurized Middle Cerebral Artery
As long-term treatment with DHT increased COX-2 protein levels (Figure 2), functional experiments were designed to investigate whether vasoactive products of the COX-2 pathway contribute to MCA tone after long-term
in vivo treatment with DHT. Passive diameters of the arteries, measured in the presence of Ca2+-free PSS, were not different among the animal groups (Table 2). To assess COX-mediated contributions to vascular tone, diameter measurements were made in the presence of
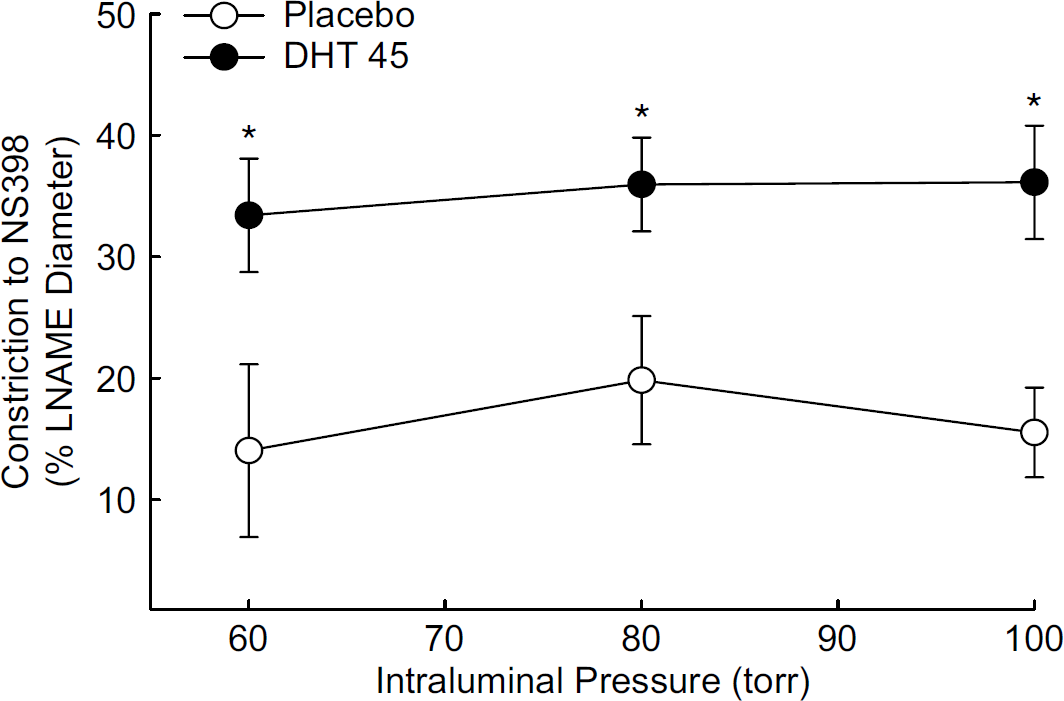
Effect of long-term dihydrotestosterone treatment on constriction to selective cyclooxygenase-2 (COX-2) inhibition with NS398 (10 µmol/L). Pressurized middle cerebral artery segments from orchiectomized rats treated with placebo (PLB) or dihydrotestosterone 45 mg pellets (DHT 45 mg) were studied in the presence of the nonselective NG-nitro-
Chronic Dihydrotestosterone Increased Constriction to an iNOS Inhibitor in Pressurized MCA
As treatment with DHT also increased iNOS protein levels (Figure 3), we used the selective iNOS inhibitor,
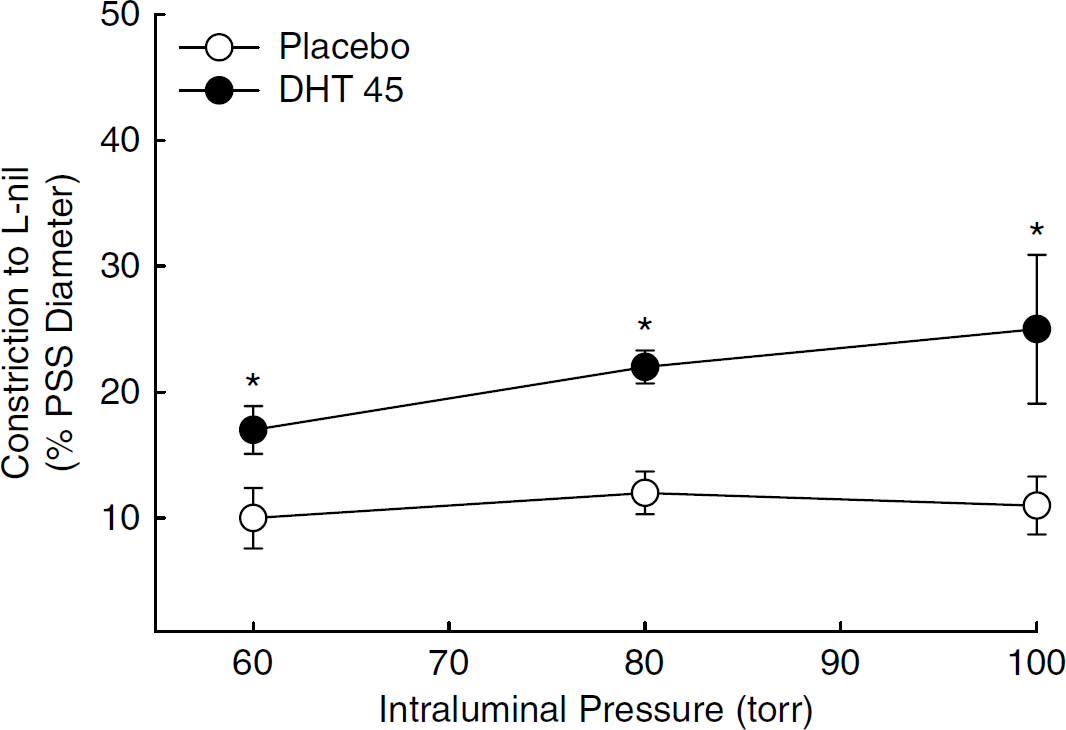
Constriction to inhibition of inducible nitric oxide synthase (iNOS) with L nil (5 µmol/L) in pressurized middle cerebral artery segments isolated from orchiectomized rats treated with placebo or dihydrotestosterone 45 mg pellets (DHT 45 mg). Constriction to
Discussion
The main objective of this study was to investigate the influence of an androgenic stimulus, DHT, on the NFκB inflammatory pathway in the cerebral vasculature of male rats. We observed that a physiological level of DHT promotes an inflammatory response in cerebral arteries in the absence of another inflammatory stimulus. Specifically, we found that both in vivo and ex vivo DHT treatment enhanced nuclear NFκB DNA binding in cerebral arteries and augmented levels of COX-2 and iNOS, two well-known proinflammatory products of NFκB activation (Baeuerle and Henkel, 1994). The effect of DHT to increase COX-2 and iNOS levels was attenuated by the androgen receptor antagonist, FLU. These effects of DHT were shown to have functional consequences for vascular tone of cerebral arteries. Selective inhibitors of either COX-2 or iNOS produced constriction in isolated, pressurized MCA segments, and this effect was greater for arteries taken from orchiectomized rats treated extensively with DHT. These data suggest that enhanced COX-2/iNOS levels produced by DHT results in increased basal production of vasodilatory factors that modulate vascular tone.
Using immunostaining and biochemical analysis, past studies from our laboratory show that the cerebral circulation is certainly a target for androgens. We have previously shown that androgen receptors are expressed in both vascular smooth muscle cells and endothelial cells of the cerebral circulation (Gonzales et al, 2007). Other investigators have shown similar findings in other vascular tissues such as aorta (Fujimoto et al, 1994) and coronary blood vessels (Liu et al, 2005). Functional studies show that after long-term in vivo treatment with testosterone, the precursor molecule to DHT, vascular tone in small resistance-sized cerebral arteries is increased (Gonzales et al, 2004). In addition, a portion of this enhanced tone appears to result from suppression of an endothelial vasodilator component resembling EDHF (endothelial-derived hyperpolarizing factor) and augmentation of the thromboxane synthase pathway (Gonzales et al, 2004, 2005). Other investigations from our laboratory have shown that long-term in vivo testosterone treatment enhances endotoxin-induced inflammation in cerebral blood vessels (Razmara et al, 2005). Taken together, these findings suggest that androgens may contribute to a predisposed risk for vascular diseases such as stroke.
To date, no published studies have investigated the influence of androgens on the cerebrovascular inflammatory response in the absence of an inflammatory stimulus. In addition, there are very few studies that have investigated effects of the more potent androgen, DHT, on vascular function and inflammation. However, one recent investigation by Cheng et al (2007) showed that DHT enhanced ischemic brain injury after long-term hormone treatment (1 week). They also showed that in a stroke model, MCA occlusion/reperfusion, a number of genes are regulated by DHT, including the gene responsible for encoding COX-2.
Interestingly in contrast to our current study using the potent androgen DHT, our previous studies on intact male rats showed very low basal levels of COX-2 and iNOS in cerebral arteries (Razmara et al, 2005). Moreover, long-term testosterone treatment of orchiectomized males did not cause any significant effect on markers of inflammation (COX-2 or iNOS) in the absence of an inflammatory stimulus. One explanation for this difference with the present findings using DHT might be that effects of testosterone reflect a balance between androgenic and estrogenic actions. Estrogen treatment of males suppresses the inflammatory response of cerebral vessels (Razmara et al, 2005), and DHT, which is not converted into estrogen by aromatase, has purely androgenic effects. As mentioned above, we have previously shown that enzymes responsible for metabolizing testosterone to DHT or estrogen, 5-α-reductase and aromatase, are present in cerebral arteries (Gonzales et al, 2007). Thus testosterone can metabolically be converted to estrogen and testosterone, possibly explaining differences between our previous studies of intact and testosterone-treated males and the current study of DHT. Moreover, we would not predict that DHT treatment would return the orchiectomized animals to a normal, physiological baseline, because the estrogen component is missing. In previous work on cerebrovascular contraction, we have found that neither androgen nor estrogen fully restores orchiectomized responses to those of an intact animal. Instead, the intact animal appears to exhibit a response that is midway between androgen-mediated constriction and estrogen-mediated dilation (Geary et al, 2000).
NFκB has been studied extensively since its first description by Sen and Baltimore (1986) for its role in immunity and stress responses. NFκB is thought to be a central transcriptional mediator of inflammation and can be activated in numerous cell types either by classic cytokine stimulation or in response to oxidative stress (Chen et al, 1999; Matsui et al, 1999; Schemedtji et al, 1997). NFκB is normally stabilized in the cytosol when bound to the repressor phosphoprotein, IκB. However in the presence of an appropriate stimulus, IκB is targeted for ubiquitination and degradation leaving NFκB available for translocation to the nucleus and activation of target genes. Some target genes for NFκB include vascular endothelial growth factor, heme oxygenase-1, COX-2 and iNOS, all capable of modulating blood flow and vascular inflammation. Although NFκB is a transcription factor, it has the ability to interact with multiple other transcription factors and transcriptional cofactors.
The data of the current study indicate the effects of DHT are mediated by the androgen receptor. Experimental evidence suggests that sex steroid hormones in combination with their receptors can act as transcription factors to modulate induction of inflammation-induced proteins. However, more is known about the effects of estrogens on inflammatory pathways than is the case for androgens. For example, NFκB-dependent inflammation was shown to be inhibited in brain endothelial cells by estrogen without affecting IκB degradation (Galea et al, 2002). Similarly, DNA binding assays revealed that long-term estrogen treatment in ovariectomized female rats suppresses NFκB-dependent cerebral vascular inflammation induced by IL-1-β (Ospina et al, 2004).
Several studies suggest that androgens can modify NFκB transcriptional regulation, but unlike estrogen that appears to inhibit NFκB, androgens have been suggested to have both pro- and anti-inflammatory actions. An in vitro study using electrophoretic mobility shift assay showed physical association of the NFκB p50 and p65 subunits with a promoter region on the androgen receptor gene (Supakar et al, 1993), suggesting interactions between androgen function and inflammation. However, mechanisms associated with the signal between androgen receptor stimulation and NFκB activation are not well understood. Some studies suggest that steroid hormone receptors in general can physically interact with p65 subunit of NFκB and repress NFκB translocation (McKay and Cidlowski, 1998). In one study of human umbilical vein endothelial cells stimulated with IL-1β, DHT enhanced NFκB activation leading to increased vascular cell adhesion molecule-1 promoter activity (Death et al, 2004). Using a 5′-deletion analysis, they showed that NFκB sites within the VCAM-1 promoter region were responsible for the DHT-mediated increase in VCAM-1 protein levels. They proposed that this interaction does not involve a direct interaction between AR and NFκB but instead is modulated by suppression of the NFκB inhibitory protein. These studies suggest a novel finding showing a positive regulation between androgen receptor stimulation and NFκB-mediated mechanisms associated with inflammation. In the current study, we observed that DHT increased nuclear NFκB DNA binding in cerebral arteries suggesting that androgens act via NFκB to contribute to a state of proinflammation. However, this effect has not been found in all studies. For example, another study in cultured human umbilical cord endothelial cells found DHT decreased endotoxin-induced inflammation by downregulating levels of adhesion molecules, chemokines and protease (Norata et al, 2006). The reasons for these discrepancies are not clear but may relate to different conditions of the cells in culture.
None of the previous studies showing DHT can modulate NFκB activation determined a physiological consequence of this effect. Therefore, we investigated the effect of DHT on COX-2 and iNOS protein and function in cerebral arteries. As mentioned before, previously we have shown that testosterone, an androgenic precursor to DHT, further augments endotoxin-mediated induction of proinflammatory enzymes iNOS and COX-2 in cerebral blood vessels (Razmara et al, 2005). In the current study, however, we show that, independent of any inducer of inflammation, long-term administration of physiological doses of DHT by itself augments levels of proinflammatory enzymes in cerebral arteries. This effect is mediated by activation of the androgen receptor as evidenced by use of the androgen receptor antagonist, FLU. As expected, there was also a DHT-induced change in vasoactive factors produced by the inflammatory enzymes, indicating a functional consequence on cerebrovascular tone.
Clinical studies suggest that testosterone replacement therapy in aged hypogonadal men can modulate immune system responses. One study reported that testosterone replacement to hypogonadal patients has anti-inflammatory properties, resulting in significant reductions in circulating levels of TNF-α and IL-1-β when compared with placebo (Malkin et al, 2004). An explanation for the reduced levels of these proinflammatory modulators observed in the above study may be in part due to testosterone being further metabolized to estrogen, and this may explain the anti-inflammatory effects. In experimental animal models, discrepancies in the effects of androgens on inflammation may result from a variety of sources. In the case of the nonaromatizable DHT, differences may arise from different experimental models (in vivo versus in vitro) as well as the concentration of DHT used (10−7 versus 10−10 M). In our case we show that, in the absence of stimulated inflammation, DHT, whether administered in vivo (physiological dose) or ex vivo (10−8 M), increased NFκB DNA binding in cerebral arteries suggesting that DHT alone can elicit an inflammatory response by activating the NFκB pathway.
In summary, activation of the NFκB-mediated COX-2/iNOS pathway by androgens such as DHT elicits a state of vascular inflammation independent of cytokine or any other outside inflammatory stimulus. Clinically, the effects of androgens on proinflammatory enzymes may contribute to further worsening of the detrimental influence of androgenic stimulation on ischemic brain injury. This is critical, because the impact of cerebrovascular inflammation is highly relevant during pathophysiological conditions related to endothelial dysfunction, oxidative stress, hypoxia and stroke. Taken together these data suggest that, in young adults who choose to use androgen supplementation recreationally or for performance enhancement, predisposition for the onset of inflammation may be increased, with significant possible consequences for development of both cerebral and cardiovascular disease.
Footnotes
Acknowledgements
We thank Ms Bebe Ehsan and Mr Peter Horvath for their technical assistance with Western blot. This work was supported by the American Heart Association Scientist Development Award (RJG) and by NIH RO1 NHLBI-50755.
