Abstract
Microscopic polyangiitis is an important and common component of cytoplasmic antibody–associated vasculitides that can lead to serious morbidity and even death. A clear causative etiology has not been identified. Although silica is well known to produce lung damage, the negative renal effects of silica exposure should not be overlooked. We present a case of renal dysfunction associated with silica exposure, its diagnosis by renal biopsy, and the treatment method used. Environmental or occupational silica exposure can cause microscopic polyangiitis. Working in occupations with increased risk of silica exposure may result in serious medical problems.
Introduction
Antineutrophil cytoplasmic antibody (ANCA)–associated vasculitides (AAVs) are a spectrum of potentially serious, multisystemic diseases that consist of granulomatosis with polyangiitis, microscopic polyangiitis (MPA), eosinophilic granulomatosis with polyangiitis, renal-limited vasculitis, and certain drug-induced vasculitis syndromes. Of these, MPA is an important subgroup of AAVs that progresses with small vessel involvement that is often accompanied by necrotizing glomerulonephritis (GN). More than 70% of patients are perinuclear ANCA (myeloperoxidase [MPO])–positive. In individuals with genetic predispositions, specific environmental factors can trigger AAVs with a synergistic effect. In a genome-wide association study, Merkel et al 1 reported a genetic susceptibility loci for MPA: human leukocyte antigen (HLA)-DQB1. Among non-HLA genes, the association of PTPN22 Arg620Trp was remarkable in MPA- and MPO-AAV. 2 A study performed among Japanese individuals revealed that HLA-DRB1∗09:01 and HLADQB1∗03:03 were associated with susceptibility to MPA- and MPO-AAV. 2
Silicosis is an occupational clinical entity that often manifests with pulmonary involvement and may be accompanied by renal injury. Autoimmune events are seen more frequently in the general population in the course of silicosis and crystalline silica exposure.3,4 The exposure potential of silica—a potential T-cell adjuvant—has increased in recent years, with different forms—such as precipitated silica, silica gels, fumed silica, and vitreous silica—being used more frequently in the industrial market (polymer compounds, rubber, sealants, adhesives, ceramics, foundry work, and production of defoamers).
Case Report
A 29-year-old male was admitted to the emergency department, complaining of weakness, cough, hemoptysis on 1 occasion, and darkening of the urine. In his anamnesis, it was learned that he had worked in a quartz mine for 8 y, with maximum unprotected (without silica dust mask) exposure for the first 3.5 y. He had 1 sister who had systemic lupus erythematosus (SLE) and 2 sisters with connective tissue disease, 1 of whom had a daughter with SLE (died). In the physical examination, he was conscious and pale, with an arterial blood pressure of 139/87 mm Hg, a heart rate of 110 beats/min, a temperature of 36.9°C, and an oxygen saturation (SpO2) of 98%. Respiratory sounds were coarse, and the heart was tachycardic, but there was no murmur and no organomegaly, palpable lymphadenopathy, or peripheral edema. On admission, biochemical findings were as follows: serum creatinine concentration 2.9 mg/dL; 24 h proteinuria and albuminuria 1818 mg/d and 891 mg/d, respectively; and erythrocyte sedimentation rate 140 mm/h; urinalysis revealed erythrocyte +3, leukocyte +1, and protein +1. The laboratory values on admission are shown in Table 1. Tuberculosis (sequential sputum acid-fast bacteria negativity and negative culture) and sarcoidosis were excluded in evaluations that had been made for respiratory symptoms 2 mo before admission. As his complaints continued, a wedge biopsy was performed from the right lower lobe superior segment of the lung. Macroscopically, a large number of nodular lesions, with a hard consistency on palpation, were found. Microscopically, many acellular fibrotic nodules in a central hyalinized and condensed collagenous structure were noted. Here, signs of noncaseating granulomatous reactions were detected around these nodules (ie, pancytokeratin+ in the alveoli, with leukocyte common antigen and CD34+ in the granulomatous inflammation foci and vessel walls). Pneumoconiosis secondary to silica exposure was, therefore, the primary consideration (Figure 1). Renal ultrasonography revealed that the kidneys were of normal size and parenchymal thickness, with bilateral echogenicity of grade 1 to 2. In a thorax computed tomography scan, numerous lymph nodes were observed in the mediastinum and hilar areas. Of note, the largest lymph nodes were found in the right upper paratracheal localization, at 20 mm in length, and in the right tracheobronchial bronchovascular and the aorticopulmonary regions, at 23 × 17 mm and 20 × 9 mm, respectively. Also observed was the nodular and fibrotic appearance in both lungs.
Laboratory values on admission
ANA, antinuclear antibody; ALT, alanine transaminase; HbsAg, hepatitis B surface antigen; c-ANCA, cytoplasmic antineutrophil cytoplasmic antibody; Ery, erythrocyte; GBM, glomerular basement membrane; HCO3, carbonate; HCV, hepatitis C virus; Ig, immunoglobulin; INR, international normalized ratio; K, potassium; LDH, lactate dehydrogenase; LDL, low-density lipoprotein; Leu, leucocyte; MCV, mean corpuscular volume; Na, sodium; p-ANCA, perinuclear antineutrophil cytoplasmic antibody; PCR, polymerase chain reaction; PTH, parathyroid hormone; WBC, white blood cell.
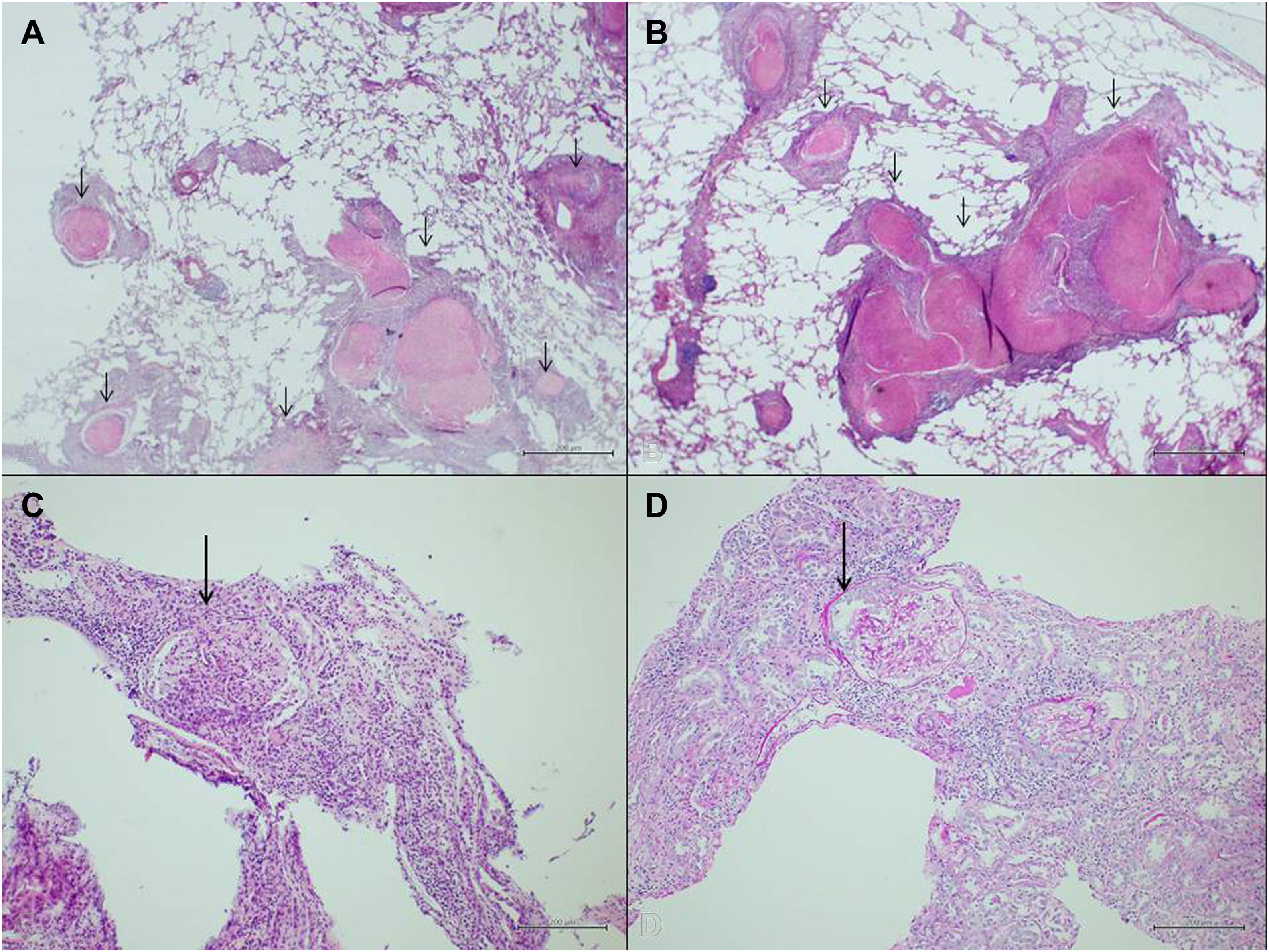
A and B, In lung wedge resection material, emphysematous changes in the surrounding lung with diffuse collagenized nodules (short arrows) in the lung parenchyma (HE, ×40). C, In kidney biopsy, cellular crescent structure in the glomerulus (long arrow) (HE, ×40). D, Crescent (long arrow), stained with periodic acid‒Schiff (PAS) stain (PAS, ×40). HE, hematoxylin-eosin.
In the biopsy that was performed due to renal dysfunction and proteinuria, cellular crescents were observed in all glomeruli, with intense mixed inflammation in the interstitial area, tubulitis, and marked thickening of the vessels (Figure 1). There was no amyloid accumulation and no specific accumulation detected by the direct immunofluorescence method; neither was granulomatous inflammation detected. With all these findings, a diagnosis of MPA associated with silicosis was made. Since our patient was young, single, and male, cyclophosphamide was not preferred as the first-line treatment due to its gonadotoxic effect. We used rituximab (RTX) in the first-line treatment and achieved effective results. RTX (375 mg/m2) and methylprednisolone 500 mg IV/d were started in divided doses for 3 d and then prednisolone 1 mg/kg/d in divided doses. In addition, ramipril, proton pump inhibitor, oral calcium, trimethoprim-sulfamethoxazole, and metoprolol were added to his treatment. On day 82 of follow-up, after 4 cycles of RTX, serum creatinine was 0.9 mg/dL and spot urine protein/creatinine was 0.759 mg/d (Figure 2).
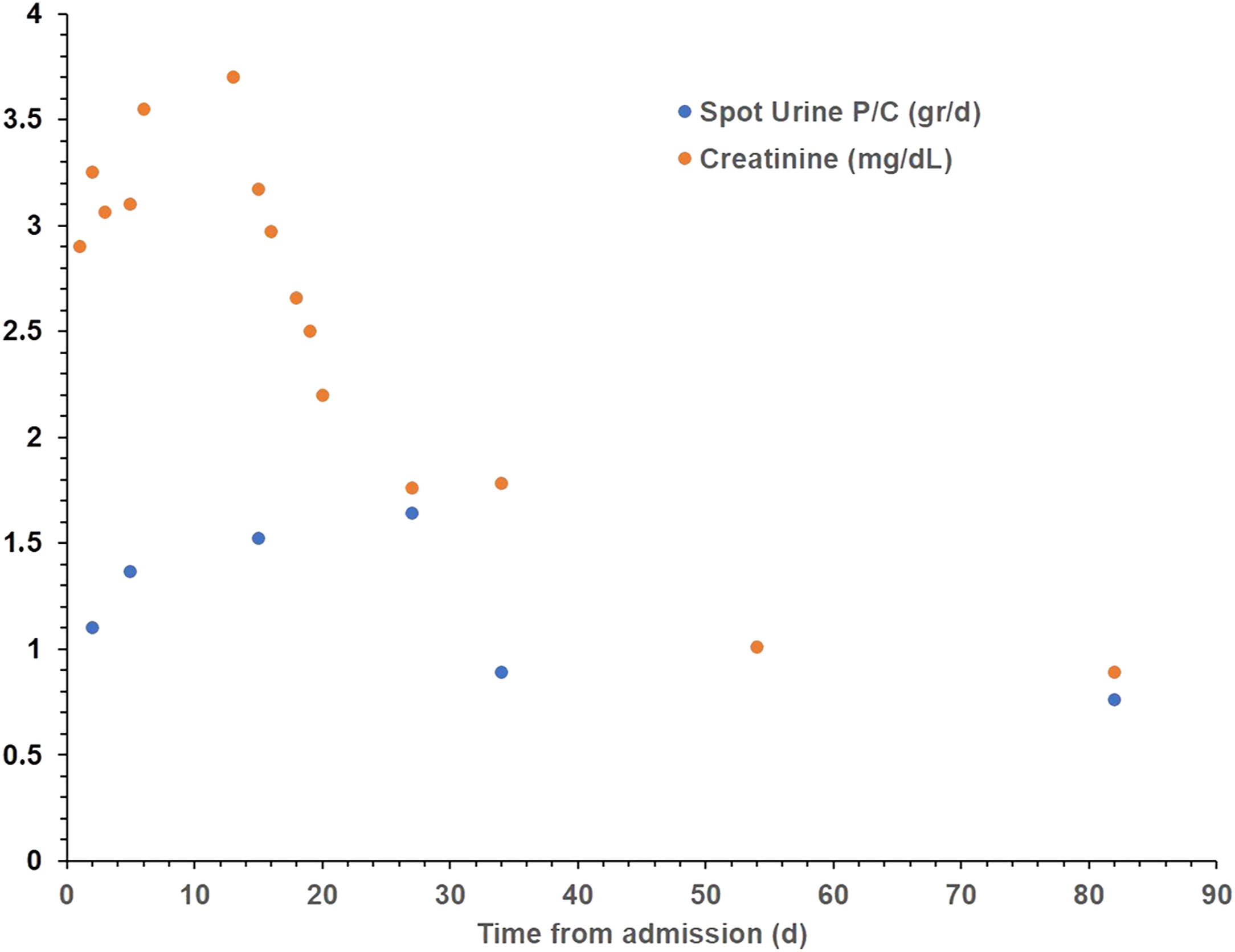
The course of serum creatinine and spot urine protein/creatinine levels in the case.
Discussion
Silica (silicon dioxide) is an abundant mineral in the Earth’s crust, especially in rock, sand, and soil structures. 5 It is most commonly found in the form of quartz, whose content of crystalline silica is nearly 90%. 6 An artificial (processed or regenerated) form of silica stone is being used more frequently in industry, and the occupational groups with high risk due to exposure include miners, construction workers, ceramic workers, dental prosthesis manufacturers, electrical cable manufacturers, maritime sector workers, and denim grinders. 7
Silicosis is a pattern of pulmonary syndromes that occurs as a result of the inhalation of silica in a free crystalline form. The cumulative dose (respirable dust concentration × crystalline silica content × exposure time) is the most important factor, and it has been found that silicosis is observed in 47 to 77% of patients if they are followed up for a sufficient period after exposure. 8 In addition to inhalation, there is a risk, albeit lower, from consuming contaminated groundwater or skin exposure. As a full cure cannot currently be achieved, it can have serious clinical consequences, such as lung cancer and chronic obstructive pulmonary disease.
In addition to readily diagnosed silica-related lung injuries, another important complication is acute or chronic kidney injury. It has been suggested that this damage may be secondary to inflammation and fibrosis, resulting from the transport of silica to the kidneys by the blood flow and from long-term renal retention. Sponholtz et al, 9 in an article analyzing cohort studies, reported that silica exposure has a risk potential of 55% for the development of chronic kidney disease (CKD) and a risk of 22% for CKD-related deaths. In a newly published study, the incidence of autoimmune disease or abnormal autoimmune serology in engineering stoneworkers was 21% (33 of 154) and CKD was identified in 10%. 10
It is thought that in some patients with CKD of unknown origin, the disorder is caused by nephrotoxic agents that are present in higher concentrations in some specific geographical regions. 11 Here the renal injury may be associated with silica exposure rather than the silica itself. 12 Etiologically, silica exposure has been associated with chronic tubulointerstitial nephritis accompanied by proximal tubular injury, tubular atrophy, and interstitial fibrosis. 13 This type of damage is similar to long-term exposure to environmental factors, such as heavy metals and pesticides. In Mascarenhas et al’s 14 detailed pathophysiological study, oxidative stress caused by silica exposure of 100 mg/L or more in the proximal tubules of rats was shown to be similar to the oxidative damage caused by heavy metal–related renal toxicity. Prolonged mitochondria-mediated apoptotic cell death and sustained intracellular inflammation (inappropriate reactive oxygen species levels and DNA damage) and consequent tubular atrophy and fibrosis were hypothesized as the main nephrotoxic molecular mechanisms. 14
Silica exposure can trigger autoimmune diseases, such as SLE (approximately 10 times more common than in the general population), systemic sclerosis, rheumatoid arthritis (approximately 3 times higher risk), and, less frequently, AAVs. 8 These silica-related clinical conditions are, contrary to the female gender predominance of nonsilica etiologies, more common in exposed men. 3 Underlying this diversity are both the apparent genetic contribution and the interaction of environmental and genetic factors. In a case-control study in which 31 patients with AAVs were evaluated using a structured questionnaire, occupational exposure to chemicals containing silica was found in 23% of the patients but in 0% of the control group. It has therefore been suggested that silicosis may play a role in the pathophysiology of AAVs. 15 Gómez-Puerta et al, 16 in a meta-analysis, identified a significant correlation between AAVs and silica exposure (over any time period; pooled OR, 2.57; 95% CI, 1.51–4.36). It has also been shown that the risk of developing ANCA-associated vasculitis is more than doubled in those exposed to silica. In contrast, it has been suggested that AAVs are less common in MPO-silica–related renal injury; in a study by Blanco-Pérez et al, 17 which evaluated 489 silicosis patients, AAVs were detected in only 2 patients (0.4%).
MPA is a necrotizing vasculitis without immune deposit accumulation, and it progresses with small vessel involvement. Necrotizing arteritis, rapidly progressive pauci-immune GN, and pulmonary capillaritis or alveolar hemorrhage are common, and granulomatous inflammation is not usually present.
18
It is accepted that environmental factors, such as exposure to
It has been speculated that crystalline silica triggers autoimmune events by activating the innate immune system, causing inflammation and the activation of antigen-presenting cells. 20 Chronic activation of T and B lymphocytes may stimulate the autoimmune response, and the autoimmune balance may then be disrupted, especially with the dominance of regulatory T (Treg) lymphocyte apoptosis. Silica has been shown to increase interleukin-1 production and secretion, leading to inflammation, free radical formation, and fibroblast proliferation, and silica also prevents apoptosis of responder T cells in CD4+ peripheral T cells by stimulating anti-Fas autoantibodies. 21 In addition, with increased Fas expression in Treg cells, activation of responder T cells increases with Treg apoptosis; responder T cells thus become more dominant than Treg cells. Crystalline silica can also induce inflammasome activation by stimulating antigen-presenting cells such as dendritic cells and macrophages. It is thought that inflammasome activation may also potentially cause development of this clinical scenario. 22
Eleftheriadis et al 20 showed that crystalline silica stimulates both CD4+ T cells (even without antigen-presenting cell) and B-cell proliferation (even without the need for CD4+ T-cell activation) in cell cultures. In the same study, therapeutic concentrations of tacrolimus (10 ng/mL) significantly decreased silica-induced cell proliferation in both CD4+ T cells and B cells. It is thought that silica crystals cause crystalline silica–induced autoimmune diseases by directly activating CD4+ T cells and B cells by exceeding their self-tolerance.
In animal experiments, the relationship between silica exposure, autoimmune diseases, and serum autoantibody positivity has been shown in many studies. 23 Proteinuria, renal immunoglobulin G, and C3 deposits were detected after exposure of lupus-prone NZM2410 mice with crystalline silica; positive markers, such as antinuclear antibody (ANA) and anti-DNA, were observed after subcutaneous administration of sodium silicate to a group of rats; 8 and silicosis exposure in mice infected with lymphocytic choriomeningitis virus has been shown to induce severe GN with elevated autoantibody levels. It has, therefore, been suggested that autoimmunity may be activated by silicosis exposure in the case of chronic viral infection as part of the pathogenesis of a prototype autoimmune disease like SLE. 24
The interaction between silica and cell membranes can induce MPO expression on the cell surface of leukocytes and monocytes, making it the starting point of an autoimmune process with ANCA formation. 25 In a case-controlled study by Beaudreuil et al, 26 a significant correlation was found between silica exposure and ANCA positivity. Hypergammaglobulinemia associated with B-cell activation, even with no obvious autoimmune disease, ANA, rheumatoid factor, or immune complex, was observed in about a third of the silicosis patients, even without an overt autoimmune disease. 20 In a study by Fernández et al, 27 27% of patients had positive autoimmunity markers, the 2 most common being ANA (89%) and anti-Ro (22%), and it was reported in yet another study that ANA positivity could be seen with a frequency of 34% and hypergammaglobulinemia with a frequency of 65%. 8 In our case, anti-Ro was also positive, but its clinical significance was uncertain.
The case presented here is unique in that both lung biopsy and renal biopsy were performed in a patient with silicosis exposure. The combination of lung and renal biopsies gave us a more complete histopathological evaluation. Miranda et al 28 reported a similar association between MPA and silicosis, but in that case, MPA appeared first and then pulmonary silicosis was diagnosed. In a case report by Lee et al, 29 remission was achieved in a 56-year-old man using cyclophosphamide + steroid + plasma exchange induction and maintenance with azathioprine. In the treatment of MPA, RTX is primarily recommended in the guidelines because it causes less toxicity and is similar in efficacy. 30
Conclusion
Considering that the use and therefore frequency of exposure to silica is increasing with industrial development, awareness should be raised of not only the pulmonary effects of silicosis but also the renal damage. Silica exposure should be avoided, especially in patients with a familial history of autoimmune diseases, as in our patient. In individuals who cannot avoid exposure, routine screenings for renal function should be conducted in addition to other systemic screenings and algorithmic evaluation that should be performed.
Footnotes
Acknowledgements
Author Contributions: All authors contributed equally to the writing of this manuscript and take responsibility for the final form of the manuscript.
Financial/Material Support: None.
Disclosures: None.
