Abstract
Introduction
The potential efficacy of selected plant extracts to counteract the dermal toxicity of jellyfish envenomation was investigated using an in vitro cell culture model.
Methods
We studied plant extracts from Carica papaya, Ananas comosus, and Bouvardia ternifolia, known for their antivenom properties, in pairwise combinations with tissue homogenates of the jellyfish Pelagia noctiluca, Phyllorhiza punctata, and Cassiopea andromeda, to evaluate modulations of jellyfish cytotoxic effects. L929 mouse fibroblasts were incubated with pairwise jellyfish/plant extract combinations and examined by MTT assay (3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide).
Results
C papaya and A comosus significantly lowered the cytotoxicity of P noctiluca and P punctata but induced a slight worsening of C andromeda cytotoxicity. Conversely, B ternifolia was protective against P punctata, ineffective against P noctiluca, and worsened C andromeda cytotoxicity.
Conclusions
Data showed species-specific and contrasting effects of plant extracts, suggesting that those containing protease activities, namely A comosus and C papaya, are more effective in lowering the cytotoxicity of jellyfish venom containing toxic peptidic factors such as phospholipase A. However, all examined plants require further investigation in vivo to evaluate their ability to counteract jellyfish injury to the skin.
Keywords
Introduction
Jellyfish (Cnidaria, Medusozoa) are aquatic organisms that produce various bioactive compounds and are especially known for their cutaneous toxicity. Jellyfish stings are a significant hazard for occupational or recreational activities at sea in various locations. 1 A study conducted in southern Italy estimated that the equivalent of about US $440,000 was spent on jellyfish-related medical services along the Italian coast during a 5-y period. 2 Treatments proposed for jellyfish envenomation include vinegar, warm urine, hot water, baking soda, and herbal products. 3 However, systematic reviews have reported that specific treatments for jellyfish envenomation can produce opposite effects across different jellyfish species. 4 For instance, vinegar is a good remedy to treat stings by jellyfish of the genus Physalia, but with other species it exacerbates pain or the discharge of venom-containing nematocysts.4,5
In this work, we hypothesized that various plant extracts would affect the toxicity of jellyfish species differently because of the different composition of their venoms. Cassiopea andromeda (Forsskål, 1775) (Scyphozoa, Rhizostomeae) is a small medusa known as the “upside down jellyfish” due to the reversed position it maintains in the water, with the umbrella resting on the bottom and the tentacles facing upward to facilitate the photosynthesis of zooxanthellae symbionts. 6 The species is native to tropical seawaters but has also colonized the Mediterranean Sea through the Suez Canal. 7 It is known to produce toxic effects, mainly due to phospholipase A2 contained in its venom. Injury includes neurotoxicity, myotoxicity, vasodilation, hemolysis, and anticoagulation. 8 –10
Phyllorhiza punctata (von Lendenfeld, 1884) (Scyphozoa, Rhizostomeae) is native to the southwestern Pacific and thought to have invaded the Mediterranean Sea through the Suez Canal. 11 Its sting is known to be mild, but a crude tentacle extract has been found to induce neurotoxicity in mice and chickens. 12
Pelagia noctiluca (Forsskål, 1775) (Scyphozoa, Semaeostomeae) is a small, widely distributed, phosphorescent jellyfish, commonly known as the “mauve stinger.” 13 It has nematocyst batteries distributed on tentacles and oral arms and on the bell surface and is considered the most venomous jellyfish in the Mediterranean. 14 In the last decades, outbreaks of this medusa have caused problems among sea bathers and sea workers. 13
The plant species used in this study are generally known for antivenom effects and have been traditionally used for various kinds of snakebites and insect stings. Carica papaya L. (Caricaceae), commonly known as papaya, is native to South and Central America and cultivated in many tropical countries. The fruit provides health benefits due to its nutritional, digestive, and medicinal properties, such as anti-inflammatory and immunomodulatory effects. 15 Papain is a proteolytic enzyme extracted from the fruit and is used as an ingredient in cosmetic and pharmaceutical industries, as well as in beer clarification, meat tenderizing, and dietary supplements. 16 Papain is also useful in treating ulcers, sores, wounds, and eruptions 17 and against injury from various venomous animals, including scorpion stings, snakebites, and jellyfish stings.18,19 The effect of papaya is thought to rely on the capability of papain to break down proinflammatory peptides and venom toxins.20,21 However, an in vivo study showed no evidence of antagonism of bee venom by papain in intradermal lesions, although inactivation occurred when papain was mixed with venom before injection. 22
Ananas comosus (L.) Merr. (synonym A sativus L.) (Bromeliaceae), the common pineapple, grows in various tropical and subtropical countries and has been used as a medicinal plant in various native cultures. The main active principle is bromelain, a mixture of proteolytic and other enzymes, exhibiting fibrinolytic, antiedematous, antithrombotic, and anti-inflammatory activities.23,24 Main bromelain indications include treatment of bruising, swollen and painful joints, wounds, and burns.25,26
Bouvardia ternifolia (Cav.) Schldtl. (Rubiaceae), popularly known as firecracker bush, is a shrub distributed in Central America and the United States. The plant roots have been traditionally used as remedies for snakebite and scorpion, spider, and centipede stings due to alleged analgesic, sedative, and antiviral properties.27,28 A phytochemical analysis of the hydroalcoholic extract has shown the presence of 3-O-quercetin glucopyranoside as a main component, followed by 3-O-quercetin rhamnopyranoside, chlorogenic acid, rutin, triterpenes (ursolic and oleanolic acids), and coumarin (scopoletin). 27 Ursolic and oleanolic acids have been shown to exert anti-inflammatory activity, 29 and scopoletin has been indicated as a major constituent of the most active fraction of the extract. 28
Cytotoxic effects of jellyfish/plant extract combinations were evaluated in vitro, by using the MTT assay (3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide), on L929 mouse fibroblasts as a skin cell model. Such an experimental model is commonly used in toxicologic and preclinical research. It was chosen here because the skin is the first body district to be involved in jellyfish envenomation, and fibroblasts are the main skin cells involved in inflammatory responses.
Methods
The study concerns plant extracts derived from Carica papaya, Ananas comosus, and Bouvardia ternifolia, in pairwise combinations with homogenates of the jellyfish Cassiopea andromeda, Phyllorhiza punctata, and Pelagia noctiluca, to evaluate modulation of jellyfish cytotoxic effects. Tests were carried out exclusively on in vitro cell cultures.
Chemicals
HPLC-grade methanol and glacial acetic acid were purchased from Merck (Darmstadt, Germany). Scopoletin, HPLC grade (purity ≥99%), was purchased from Extrasynthese (Genay, France). All other solvents and reagents were of analytical grade and were purchased from Sigma-Aldrich (Milano, Italy).
Plant and Jellyfish Materials
Dried aqueous extracts of A comosus fruit stem and of C papaya fruit are from EPO, Istituto Farmochimico Fitoterapico Srl (Milano, Italy). Roots of B ternifolia were obtained from plants acclimated at the Hanbury Botanical Garden, University of Genova (Ventimiglia, Italy), and extracted as previously reported. 30 Roots were ground to fine powder by a blade mill (IKA-Werke GmbH & Co, Staufen, Germany) under liquid nitrogen, and 2 g of frozen powder was extracted 3 times with 40 mL of ethanol/water (80:20 v/v), vortex mixed for 3 min, sonicated in an ice-cold bath for 5 min with a 3 mm titanium probe set to 200 W power and 30% amplitude signal (Vibra CellTM Sonics Materials Inc., Danbury, CT), and centrifuged at 3000×g for 10 min at 4°C. The supernatant was filtered and evaporated to dryness by a rotary evaporator (R-205 Buchi, Flawil, Switzerland) at 37°C.
The specimens of C andromeda and P punctata used for experiments were reproduced and bred at the Aquarium of Genova (Costa Edutainment, Genova, Italy). P punctata was bred in daily-cleaned, acrylic Kreisel tanks with a circular laminar current, using UV-treated, filtered seawater at 24°C, pH 8 to 8.2, salinity 37%, ammonium ≤0.05 mg·L−1, nitrites ≤0.05 mg·L−1, nitrates ≤10 mg·L−1, and 12 h·d−1 photoperiod. Jellyfish were fed twice a day with Artemia salina nauplii enriched with Easy DHA Selco (INVE Aquaculture, Dendermonde, Belgium). C andromeda was bred in 180 L tanks under the same conditions as P punctata, except at a 25°C temperature and fed with fish and shrimp smoothies. Specimens of P noctiluca were sampled from the Ligurian Sea, transferred to the laboratory, and kept frozen until use.
Tissue homogenates of jellyfish were prepared as previously reported.31,32 Briefly, jellyfish tissue was homogenized for 24 h by magnetic stirring in distilled water and sonicated in ice for 45 min to induce nematocyst discharge and venom release. The resulting homogenates were stored at −20°C until use. Protein was determined in homogenate samples by the Bradford method. 33
Before cell treatments, A comosus and C papaya extracts were resuspended in water, and the extract of B ternifolia was resuspended in 80% hydroalcoholic solution. Plant extracts and jellyfish homogenates were sterilized by disposable syringe filters and then diluted with cell culture medium to reach final concentrations.
Titrations of Extracts
Scopoletin titration of B ternifolia root hydroalcoholic extract was conducted by HPLC-DAD analysis. 34 Chromatographic analysis was carried out with a 5 μm ODS3 reversed-phase Prodigy column (250 mm × 4.6 mm; Phenomenex, Torrance, CA), using solvent A (water/acetic acid, 97/3 v/v) and solvent B (methanol). The following elution program was used: 0 to 3 min, 0% B; 3 to 9 min, 3% B; 9 to 24 min, 12% B; 24 to 30 min, 20% B; 30 to 33 min, 20% B; 33 to 43 min, 30% B; 43 to 63 min, 50% B; 63 to 66 min, 50% B; 66 to 76 min, 60% B; 76 to 81 min, 60% B; 81 to 86 min, 0% B and equilibration for 4 min, for a total run time of 90 min. Flow rate, injection volume, and column temperature were 1.0 mL·min−1, 20 μL, and 25°C, respectively. Scopoletin was recognized by comparing the retention time and relative UV-Vis spectrum of the sample (range 190–400 nm) with those of the commercially available standard and quantified at 330 nm by an external standard calibration curve (range 1–20 μg·mL−1).
Papain titration of the C papaya dry extract was carried out according to a previously reported method. 35 Briefly, a 50 mM phosphate buffer solution, pH 6, containing L-cysteine (30 mM) and EDTA (2.5 mM), was used to dissolve papain reference standard (30,000 USP-U·mg−1, range 4000–500 USP·mL−1), N-benzoyl-DL-arginine-p-nitroanilide (BAPA) (2 mM), and 50 mg of dry extract, the latter sonicated for 15 min and filtered by a syringe filter (0.22 μm) before analysis. During preparation, all reagents were kept on ice and covered with aluminum foil. The solutions were transferred to a 96-well plate in the following order: BAPA (120 μL), papain or sample (100 μL), and acetic acid 30% v/v (50 μL), which was added to the wells every 15 min for 45 min to cease the reaction. The experiment was carried out at 40°C, and the enzyme activity was measured according to the ρ-nitroaniline absorbance values estimated at 405 nm using a Multiskan GO reader plate (Thermo Fisher Scientific, Waltham, MA).
Cell Culture and Viability Assay
The L929 mouse fibroblast cell line 36 was purchased from the Istituto Zooprofilattico Sperimentale della Lombardia e dell’Emilia Romagna, Brescia, Italy. Cells were cultured in DMEM, with 5% FBS, 1% penicillin (100 U·mL−1), 1% streptomycin (100 μg·mL−1), and 1% L-glutamine, in a 5% CO2, humidified atmosphere.
The effects of plant extracts and jellyfish homogenates on L929 cell viability were determined using the MTT assay, as previously reported. 37 Briefly, cells were settled in 96-well plates, exposed for 48 h to increasing concentrations of plant extracts or to increasing concentrations of jellyfish homogenates in the absence or presence of fixed concentrations of plant extracts, processed for MTT assay, and read at 570 nm in a BioTek microplate reader (BioTek Instruments, Winooski, VT).
Statistics
Cytotoxicity experiments were done in duplicate, using 8 replicates for each condition. Data were used to derive dose-response curves, expressed as mean±SEM. Nonlinear regression analysis was conducted by using the Prism software package (GraphPad Software Inc, San Diego, CA) using a downhill logistic curve, to obtain IC50 and IC05 values (50% and 5% inhibitory concentrations, respectively) and their 95% confidence intervals. Confidence intervals at 95% indicate a significant difference between a pair of IC50 values in case of no overlap (P<0.05)—that is, a significant difference in cytotoxicity (lower IC50=higher cytotoxicity and vice versa). In experiments concerning extract titrations, not involving statistical inference, data were expressed as mean±SD.
Results
Plant extracts were titrated for the presence of active compounds possibly involved in antivenom activities: bromelain in A comosus, papain in C papaya, and scopoletin in B ternifolia. Titration experiments showed the presence of 0.15±0.004% papain in the fruit extract of C papaya and 0.06±0.001% scopoletin in the root extract of B ternifolia, and the presence of 0.3% bromelain in the fruit stem extract of A comosus was certified by the manufacturer. Titrations are expressed as percentage on a dry extract weight basis.
Preliminary cell viability tests on L929 fibroblasts, conducted with plant extracts, were aimed at finding nontoxic extract concentrations to be used in subsequent experiments with jellyfish homogenates. The results of these analyses are shown in Table 1, which reports IC50 values for the inhibition of fibroblast viability by extracts, together with IC05 values that are usual toxicity thresholds, indicating the upper limit of the nontoxic concentration range. Data revealed a very weak cytotoxicity of C papaya and A comosus, both showing IC05 values above 1000 μg·mL−1. Conversely, B ternifolia showed stronger cytotoxicity, with an estimated IC05 of 27 μg·mL−1 (Table 1). On the basis of these results, nontoxic concentrations of 100 μg·mL−1 C papaya, 100 μg·mL−1 A comosus, and 15 μg·mL−1 B ternifolia (lower than the lowest IC50 95% confidence limit) were chosen for use in combination with jellyfish homogenates.
IC50 and IC05 values at 48 h of different plant extracts (in μg·mL−1), obtained by MTT assays on L929 fibroblasts
Data derive from 2 separate experiments, each with n=8. Values in brackets represent 95% confidence intervals (see Methods).
Thereafter, the effects of different jellyfish homogenates on L929 cell viability were evaluated. To this aim, homogenates were standardized in terms of protein content. Protein determination by the Bradford assay showed 3.2 mg·mL−1 in P noctiluca, 2.6 in P punctata, and 4.3 in C andromeda homogenate. Table 2 reports IC50 values for the inhibition of fibroblast viability upon exposure to jellyfish homogenates alone or in combination with each of the plant extracts (see the Statistics paragraph in the Methods). Based on these values and their overlapping or not overlapping 95% confidence intervals, we could infer not significant or significant differences, respectively, between pairs of dose-response curves (ie, between the strengths of cytotoxic effects). Cell exposure to homogenates alone revealed a very similar cytotoxicity for P noctiluca and P punctata and a significantly lower one for C andromeda (Table 2, Figure 1). Cell exposure to pairwise combinations of homogenates and plant extracts showed that C papaya almost abolished the cytotoxic effect of P noctiluca and significantly reduced that of P punctata; conversely, it increased the toxic effect of C andromeda. The extract from A comosus induced modulatory effects on homogenate cytotoxicity similar to those of C papaya, though to a lesser extent on P punctata. Finally, the extract from B ternifolia was completely ineffective on P noctiluca; induced a significant decrease in the cytotoxicity of P punctata, similar to that of A comosus; and exacerbated the effects of C andromeda (Table 2, Figure 1).
IC50 values and 95% confidence intervals of jellyfish homogenates (in μg protein·mL−1) derived from MTT assays on L929 fibroblasts
Cells were exposed for 48 h to increasing concentrations of jellyfish homogenate in the absence or presence of fixed concentrations of extracts from Carica papaya (100 μg·mL−1), Ananas comosus (100 μg·mL−1), and Bouvardia ternifolia (15 μg·mL−1). Data as in Table 1.
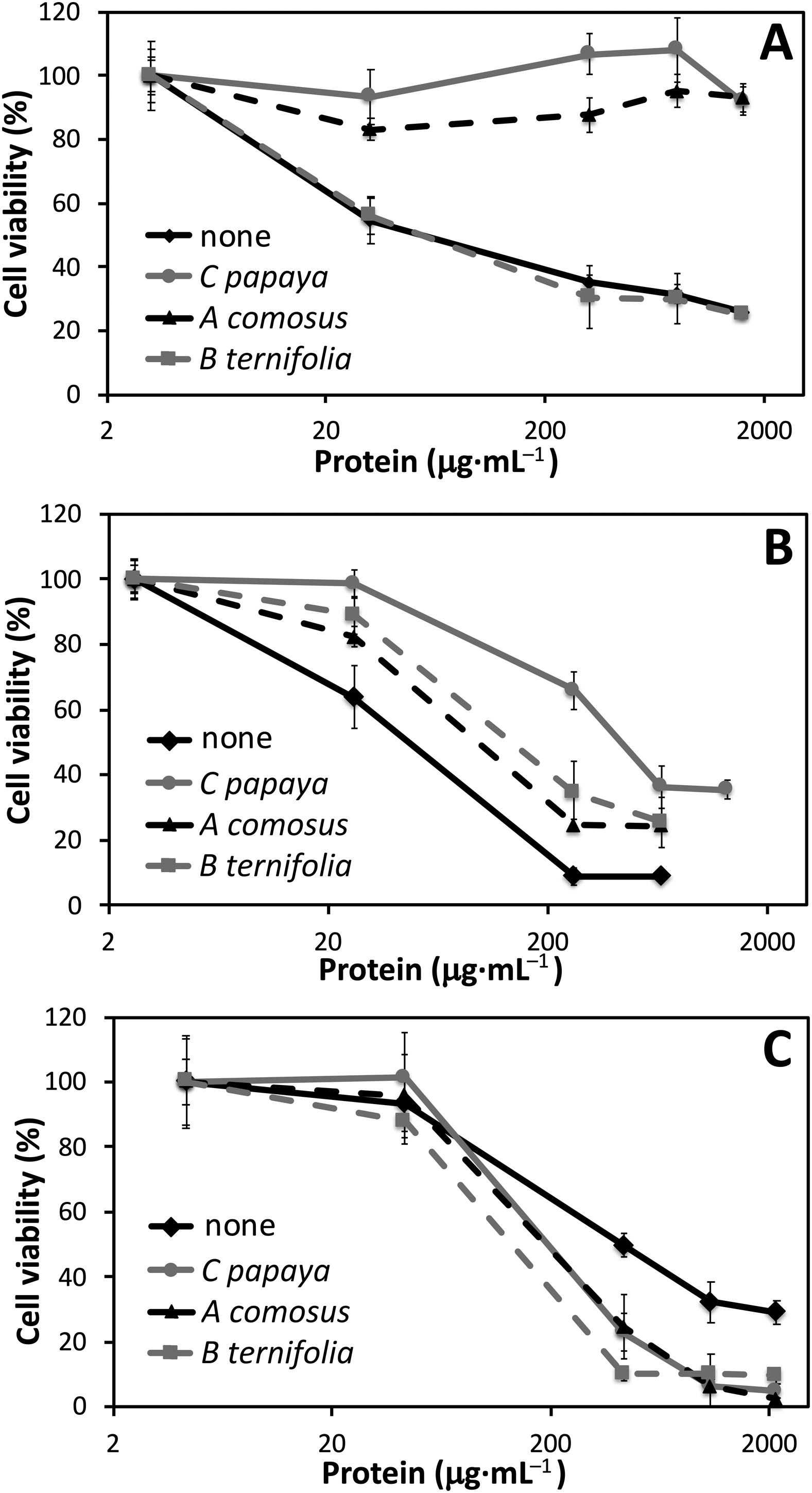
Dose-response variations of cell viability in L929 fibroblasts exposed for 48 h to increasing concentrations (μg protein·mL−1) of jellyfish homogenates obtained from (A) Pelagia noctiluca, (B) Phyllorhiza punctata, and (C) Cassiopea andromeda. Homogenates were administered in the absence or presence of extracts from Carica papaya (100 μg·mL−1), Ananas comosus (100 μg·mL−1), and Bouvardia ternifolia (15 μg·mL−1). Data are expressed as mean±SEM, deriving from 2 independent experiments, each with 8 replicates. The IC50 values determined from these curves are reported in Table 2.
Discussion
An increase in jellyfish bloom frequency has been documented worldwide, possibly related to climate change and anthropogenic causes and involving direct negative effects on human health and enterprise. 1 The problem of finding remedies for skin injury caused by jellyfish venom is therefore a controversial topic, especially because it is difficult to convert traditional methods into standardized pharmaceutical formulations. Herbal products are attracting increasing interest for the presence of numerous bioactive substances that are potentially useful for the treatment of various ailments and health problems.
In this study, we have evaluated 3 herbal extracts from plants that have a popular pedigree as antivenom remedies. These extracts have been combined with homogenates from venomous jellyfish to evaluate the modulatory effects of extracts on jellyfish cytotoxicity to fibroblasts.
Preliminary cytotoxicity assessment conducted on herbal extracts showed significant cell viability reduction only by B ternifolia. Thereafter, pairwise all-vs-all combinations of plant extracts and jellyfish homogenates allowed us to compare the efficacy of plant extracts in preventing adverse jellyfish actions. The complex of data highlighted a marked species specificity in the interaction between plant extracts and jellyfish, including in some cases a worsening of toxic effects. The C papaya extract was able to significantly counteract the cytotoxicity of P noctiluca and P punctata, but it slightly enhanced the effect of C andromeda. Similar results were found with A comosus, which compared to C papaya was only statistically less protective against P punctata. Conversely, B ternifolia was only protective against P punctata, whereas its effect was null against P noctiluca, and worsened C andromeda cytotoxicity.
These findings probably reflect a complex set of interactions between different plant extracts and the variable composition of venoms from different jellyfish species. Plant extracts containing protease activities, such as A comosus and C papaya, are more suitable to counter jellyfish toxicity. This could be due, at least in part, to the toxic relevance of the peptidic fraction of jellyfish venom. It is well known that phospholipase A2 is a major component of C andromeda and P punctata venoms,8,12 whereas the toxicity of P noctiluca has been attributed to a protein fraction with hemolytic activity. 38 However, nonpeptidic factors are also known to be present in jellyfish venom, such as a complex of neurotoxic compounds. 12 Such venom constituents could synergistically interact with molecules of the herbal phytocomplex, producing the observed worsening of toxic effects. In particular, no extract was protective against the venom of C andromeda, suggesting the need for individuating species-specific antivenom products through a screening of potential candidates.
Conclusions
This study evaluated the modulatory activities of 3 plant extracts (Carica papaya, Ananas comosus, and Bouvardia ternifolia) using an in vitro cell culture model in which mouse fibroblasts were exposed to various jellyfish venoms (Pelagia noctiluca, Phyllorhiza punctata, and Cassiopea andromeda). Our experiments have disclosed protective effects of herbal extracts against jellyfish cytotoxicity, and these extracts may be potentially useful if in vivo studies demonstrate their efficacy. However, data indicate that there could be issues with regard to both efficacy and safety, depending on the specific plant extract and the envenomating jellyfish species. Therefore, based on the cell culture results of this study, further animal or human testing is warranted to study the potential efficacy and safety of these plant extracts, either alone or combined, against various envenomating jellyfish species.
Footnotes
Acknowledgements
Acknowledgments: We thank Helan Cosmesi di Laboratorio, Casella, Italy, for providing the EPO extracts. We are also grateful to Monica Montefalcone and Federico Betti, DISTAV, University of Genova, for providing a photograph of P noctiluca.
Author Contributions: Conceptualization (BB, LC); methodology (GLM, SL, LG, AS, DT); data analysis (PG, BB); experiments (GLM, LG, AS, DT); draft preparation (BB, LC, GLM, SL, AS); review and editing (BB); supervision (BB, LC); final approval of manuscript (LC, GLM, PG, AS, DT, LG, SL, BB).
Financial/Material Support: LC and BB were financially supported by the Italian Ministry of Education, University and Research (FFABR Grant MIUR Funds, 2017).
Disclosures: None.
