Abstract
Introduction
Ultramarathon running is increasing in popularity worldwide, as is the growing body of research on these athletes. Multiple studies have examined acute kidney injury through estimated baseline creatinine (Cr) and glomerular filtration rate (GFR). Values are estimated through an age-based formula of GFR and the modification of diet in renal disease (MDRD) equation. However, the accuracy of this practice in a cohort of healthy athletes is unknown.
Methods
A prospective analysis of the first 40-km (25-mi) stage of 6-stage 250-km (155-mi) multistage ultramarathons in the Sahara, Namibia, Atacama, and Gobi Deserts. Runners had prerace measured baseline Cr compared to estimated values through age-based estimated GFR and back calculation of Cr through the MDRD equation.
Results
Forty-eight participants (27% female, age 39±10 y) had Cr values analyzed. The mean measured Cr was 0.99±0.17, which was 11% higher than an estimated Cr of 0.88±0.14 (P<0.01). Estimated age-based GFR was 95.9±5.8 mL⋅min−1 compared to GFR based on measured Cr and MDRD of 86.1±14.6 (P<0.001).
Conclusions
Estimated values of GFR and Cr by standard age-based values and MDRD equation were significantly inaccurate, which would overinflate the incidence of acute kidney injury. Future studies should devise a new model for estimation of baseline Cr that is validated in this population.
Introduction
Endurance running events longer than traditional marathon distances are increasing in popularity worldwide.1,2 Athletes in these events, which often take place in wilderness areas, have been found to be at risk for acute kidney injury (AKI). A single-stage 160-km (100 mi) ultramarathon found that 30 to 42% of participants experienced AKI.3,4 Multistage ultramarathon races show greater rates of AKI with 60 to 80% of participants incurring risk or injury over 6 stages with 250 km (155 mi) of running.5,6 The majority of AKI appears to resolve spontaneously, 5 ,7,8 and although progression to acute renal failure is rare, 9 –11 a recent review documented 27 cases of acute renal failure that required hospitalization. 12 There is an increasing level of interest and body of research in this population with concern that, over time, repeated small increases in serum creatinine (Cr) may lead to long-term renal dysfunction. 12 Furthermore, the accurate diagnosis of AKI in the presence of oliguria and potential impending renal failure is a critical diagnosis in the treatment pathway of the endurance athlete. 4 Alternatively, misdiagnosing AKI and overhydrating an endurance athlete could increase the severity of exercise-associated hyponatremia. 13
The RIFLE (risk, injury, failure, loss, end-stage) criteria 14 are used to both diagnose and quantify the degree of AKI; those with an increase of at least 1.5 serum Cr over baseline are defined as at “risk” of AKI, and those with a twofold increase are described as experiencing “injury.” However, a limitation of the RIFLE criteria is that they require a baseline Cr from which to evaluate change. Attempts to directly measure Cr before events have experienced attrition rates up to 50% because participants are reluctant to alter their prerace routine for scientific investigations. 5 Researchers at popular ultramarathons have found baseline phlebotomy unfeasible in its entirety.4,15 Because of this logistical restraint, a common practice is to estimate the underlying glomerular filtration rate (GFR) based on age and then back-calculate Cr from GFR using the modified diet in renal disease equation (MDRD). 3 ,6,11 This equation was introduced by the creators of the RIFLE criteria in 2004, 14 validated in chronic kidney disease (CKD) patients, 16 and has been used for multiple ultramarathon AKI studies. 3 –5,17 In hospitalized patients, this estimation approach has been found to have poor accuracy with baseline Cr values and alters subsequent rates and severity of AKI.18,19 However, neither the age-based estimation of GFR nor the MDRD equation have been validated in a healthy athletic population. We hypothesized that the estimated Cr would be inaccurate and significantly lower than measured Cr in ultramarathon runners because the MDRD equation is designed for use with CKD patients, who have very different underlying physiology. The objective of this study was to evaluate the accuracy of this estimation method by comparison with directly measured prerace Cr levels in ultramarathon runners.
Methods
SETTING AND SELECTION OF PARTICIPANTS
RacingThePlanet events are 7-d, 6-stage, 250 km (155 mi) ultramarathon foot races held throughout the world. Participants in these events are generally experienced ultramarathon runners who have completed multiple endurance running events in the past.6,17 This research was a reanalysis of data collected from prospective observational studies on AKI incidence in multistage ultramarathon runners.5,6 All athletes who understood English and registered to compete in a RacingThePlanet event were invited to participate. Participants were excluded if they were currently taking cardiac medication, had a history of cardiac arrhythmia or myocardial infarction, were pregnant or thought to be pregnant, were under 18 y of age or older than 75, had a level of cognitive impairment that would prohibit understanding of an informed consent document, had language impairment in reading/speaking English, or had hearing impairment not alleviated by the use of a hearing aid. One additional participant was excluded because he had a single kidney. Participants from the Atacama Desert, Chile (2012); Sahara Desert, Namibia (2008, 2009); and Gobi Desert, China (2008) were included. Each participant was only entered in the study once. At the time of race registration, interested healthy participants reviewed the study and gave informed consent. Approval was obtained from the institutional review board at Stanford University School of Medicine and University of Washington.
RESEARCH DESIGN
Participants were enrolled the day before the race start to obtain baseline demographic data and provided written informed consent. Before race start, measured Cr was obtained from fingertip blood samples using lancet and capillary collection tube and immediately analyzed at time of collection using an iSTAT point-of-care analyzer (Abbott, East Windsor, NJ). 20
For age-based baseline GFR estimation, when the individual's age was less than 40 y, a GFR of 100 mL⋅min-1 was assigned, and 140 mL⋅min-1 minus age was applied when they were 40 y or older. 21 Estimated Cr was back-calculated from this baseline GFR using the standard MDRD equation as shown in Figure 1.
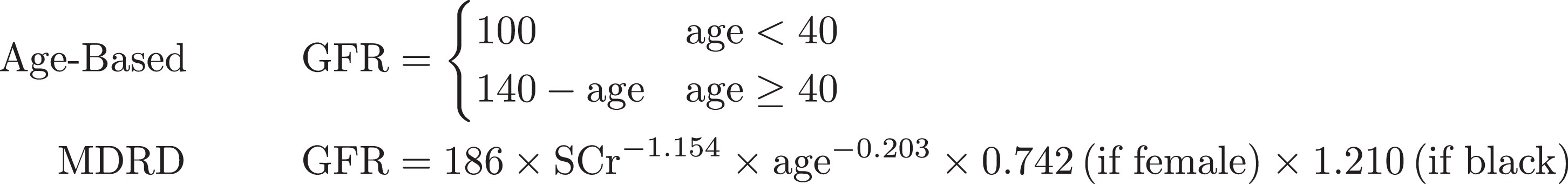
Equations used to calculate glomerular filtration rate (GFR). First is the age-based estimation typically used by researchers when baseline creatinine (Cr) is not available. Second is the standard modification of diet in renal disease equation typically used to calculate GFR from Cr but for our purposes used to back-calculate Cr from GFR.
STATISTICAL ANALYSIS
Estimated Cr was compared to measured Cr using the Mann-Whitney U (Wilcox) test because our data are not normally distributed. Data were presented as mean±SD. Statistical significance was set at P<0.05. Data analysis was performed using R version 3.2.2 (Microsoft Windows 64-bit binaries).
Results
Creatinine levels from 48 study participants were analyzed with all racers combined as a single cohort for analysis because there were similar distances and logistical demands between stages (Table 1). 15 ,17,22,23 Mean Cr and GFR are shown in Figure 2. Estimated Cr was 11% lower than measured (P<0.01). Figure 3 shows the disparity between measured and estimated Cr, with a significant underestimation of Cr using the standard estimation technique.
Study participant characteristics broken down by individual race
BMI, body mass index.

Comparison of measured creatinine (Cr) to estimated Cr. Comparison between estimated and measured glomerular filtration rate (GFR) and Cr. Differences between GFR are significant at P<0.001 and between Cr are significant at P<0.01.
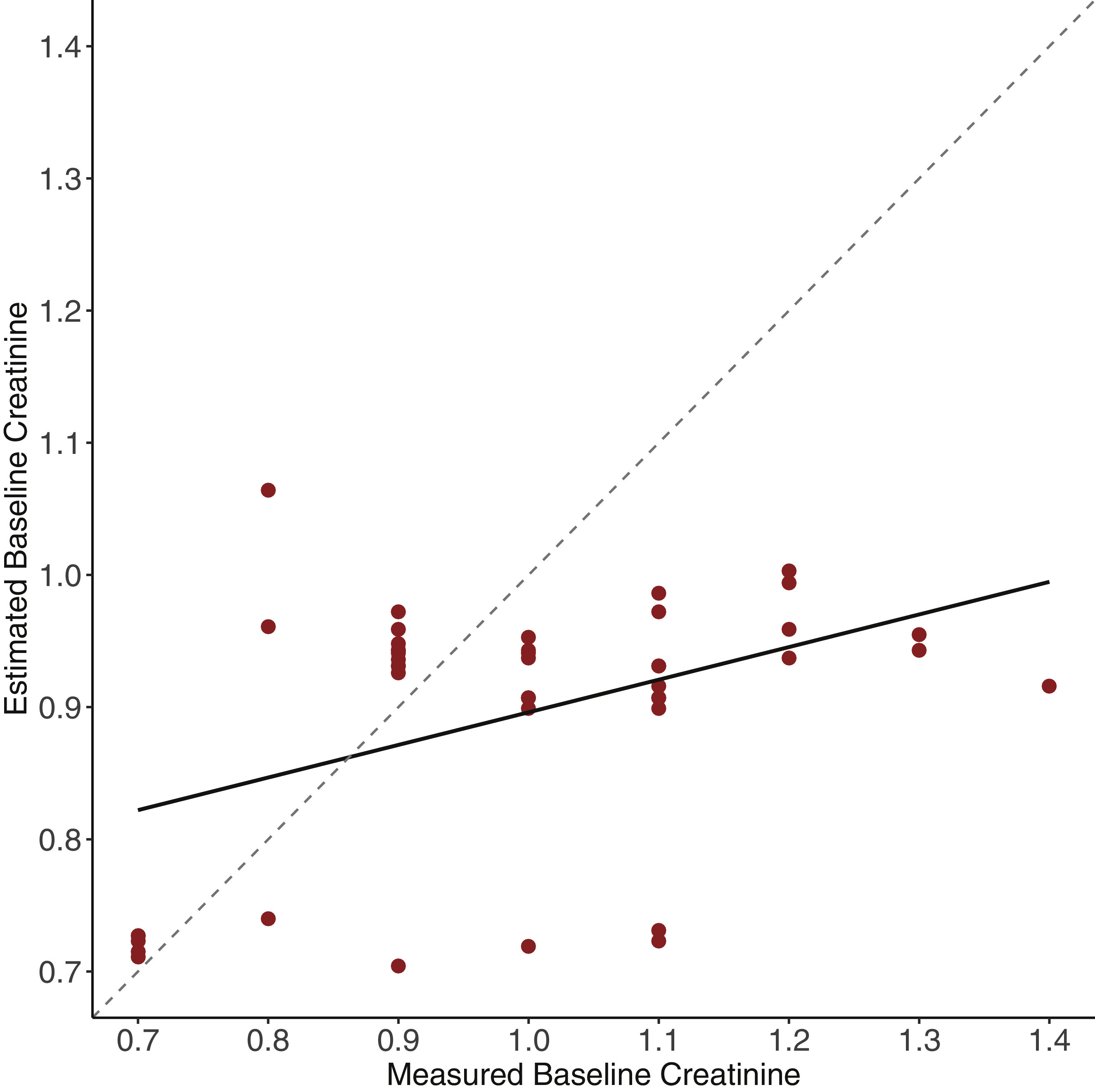
Scatterplot of back-calculated creatinine (Cr) from age-based glomerular filtration rate (GFR) using the modification of diet in renal disease equation vs measured Cr. The dotted line shows a perfect match between the 2, whereas the red linear regression line is evidence of a mismatch between measured and back-calculated Cr. Estimation using this method underestimates Cr and therefore overestimates true baseline renal function.
Discussion
This is the first study to attempt to validate the accepted method of an estimated baseline Cr in an athletic population. We found significant differences between measured and estimated baseline Cr, which would result in large disparities and overinflated rates of AKI. These discrepant values between measured and estimated Cr stem from 2 potential sources: estimation of GFR and back-calculation of Cr.
Direct measurement of GFR is impractical outside of research settings because it is a time-intensive undertaking that is expensive and involves a cumbersome intravenous infusion. 21 In hospitals, the standard method of determining renal function is to measure Cr and compute GFR using either the MDRD equation or the Cockcroft-Gault equation. However, in the wilderness setting of most ultramarathon research, direct measurement of Cr before the race start is infeasible, which has prompted researchers to estimate GFR and subsequent baseline Cr based on a participant's age. Early studies determined normal GFR values in healthy 20- to 30-y-olds to be approximately 118 mL⋅min-1 for females and 127 mL⋅min-1 in males. 24 These values have been reproduced multiple times, demonstrating normal values of 109 to 121 mL⋅min-1.24,25 Later studies have also demonstrated and reproduced an expected decline in GFR with age at a rate between 0.35 and 1.0 mL⋅min-1 per year. 24 –26 The standard practice used in ultramarathon research of an age-based estimation of GFR stems from these original studies.
The standard MDRD equation used to back-calculate Cr from GFR has been found to underestimate GFR by 26%, compared to age- and sex-matched healthy adults without CKD. 16 Because ultramarathon runners are likely healthy with excellent GFR, we would expect similar inaccuracies when using the MDRD to back-calculate from GFR to Cr. Our hypothesis was supported with an estimated baseline Cr 11% lower than measured. Because of this, subsequent measurement of end-of-race Cr would result in a larger increase from baseline and would artifactually inflate the subsequent prevalence of AKI. A future direction for research would be to create a more accurate estimation of renal function. Ideally this would avoid the complexity of estimating GFR and back-calculating Cr using equations such as MDRD, which were not designed for use in this population and would instead directly estimate baseline Cr using available data such as age, sex, race, weight, and height.
LIMITATIONS
This study was limited by a relatively small sample size, and although our results were statistically significant, the lack of athletic heterogeneity in our study population may have biased results. For example, a purely endurance running population may have different demographics than a triathlete cohort. Our population was exclusively nonblack and therefore may not be generalizable to more racially diverse groups. However, the healthy MDRD equation was based on a predominantly white population, 16 so these homogeneity concerns are not unique to ultramarathon research.
We could not control for factors that could have affected the athletes’ Cr at time of measurement, such as hydration status, recent medication use, or training runs before the race. This could account for some of the discrepancy between the estimated and measured baseline Cr. That being said, renal studies in ultramarathons have consistently assumed a normal Cr at baseline. 3 ,6,17,27
Conclusions
The accepted method of using age-based estimation of GFR and back-calculation of Cr using the MDRD equation resulted in a significant underestimation of baseline Cr. This would lead to an overestimation of AKI in ultramarathon runners. Further research is needed to develop a new model of baseline Cr estimation in this population, which would allow more accurate identification of AKI for both research and patient care.
Footnotes
Acknowledgements
Author Contributions: Study concept and design (CL, GL); obtaining funding (GL); acquisition of the data (DY, CL, GL, BK); analysis of the data (CL, GL); drafting of the manuscript (CL, DM, BK, GL, DY); critical revision of the manuscript (CL, GL, DY, DM, BK); approval of final manuscript (CL, GL, DY, DM, BK).
Financial/Material Support: This work was supported by RacingThePlanet with a research stipend. i-STAT machines were provided by Abbott.
Disclosures: None.
Presented as an abstract at the Western Regional SAEM Conference, April 2016, San Diego, CA; the SAEM National Conference, May 2016, New Orleans, LA; and the World Congress of Wilderness Medicine, August 2016, Telluride, CO.
