Abstract
Introduction
The British Antarctic Survey Medical Unit works in a very remote area of the world, with several Antarctic bases receiving only a single annual resupply of consumable goods. Pharmaceuticals supplied in this manner will often be approaching or past the end of their nominal shelf life before the following year's resupply. Drugs are transported from the UK via ship; the hold is not temperature controlled, and the ship crosses through the tropics (air temperature 25–30°C for approximately 3 wk). The drugs then must be transported from the ship to the base, often in temperatures substantially below freezing. This study assessed the stability of 5 expired drugs (atropine, nifedipine, flucloxacillin, naproxen, and bendroflumethiazide) returned from Antarctic bases.
Methods
Drugs were opportunistically obtained and tested using stability-indicating assays.
Results
All tested drugs were stable.
Conclusions
The results suggest that the studied drugs may be stable beyond expiry, even when not maintained in strictly temperature-controlled conditions.
Introduction
The British Antarctic Survey operates 5 bases and 2 ships within the Antarctic region. Three of the bases and both ships have substantial medical and dental provisions and on-site doctors provided by the British Antarctic Survey Medical Unit. The remaining bases have more limited medical supplies and trained advanced first aiders, with access to remote medical and dental advice by satellite phone or e-mail. Medical provisions are ordered on an annual basis from the UK in approximately May. After delivery to Derriford Hospital Pharmacy in Plymouth (with the usual National Health Service supply-chain time frame, meaning drugs may be many months old before delivery in Derriford), the required pharmaceutical agents are packaged and placed on the supply ships. The ships leave the UK in September and deliver supplies to the bases in approximately December, generally spending 2 to 3 mo on the ship, passing through the tropics for around 3 wk in each direction (from the UK to Antarctica and returning for disposal after expiry). The ships’ holds are not temperature or climate controlled. The manufacturers of all 5 tested drug products used in this study advised storage below 25°C. Figure 1 shows an example period of air temperatures and relative humidity (RH) obtained from the data log (located on the foremast) of the Royal Research Ship James Clark Ross over 17 d of the 2017 northward passage. These data are representative of the temperatures to which the tested drugs would have been exposed for a minimum of 3 wk in each direction. 1
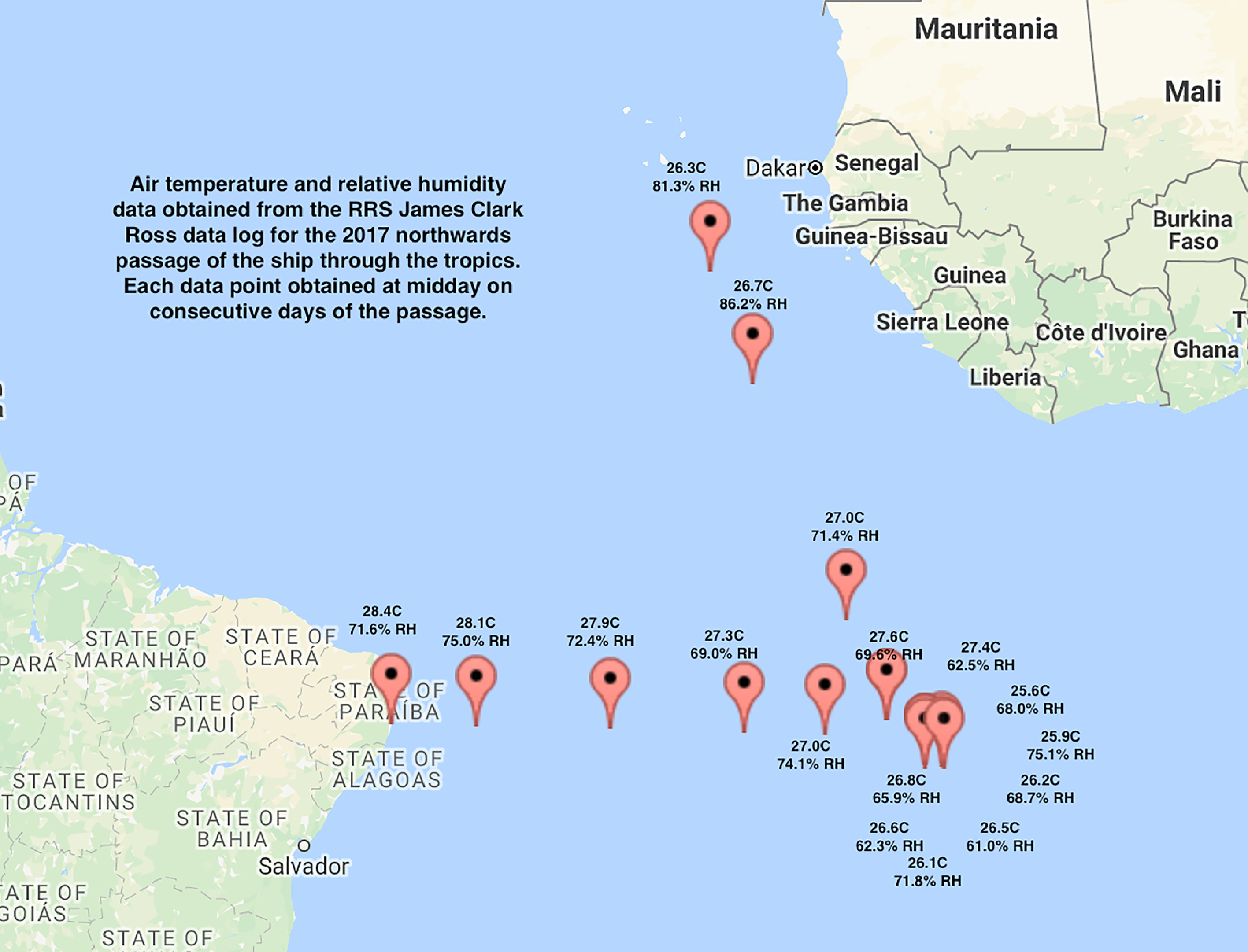
Sample air temperature and relative humidity for a passage through the tropics. Data from the 2017 northward passage of the RRS James Clark Ross.
The lowest average temperature would usually be at Halley Station, which averages -10°C in the summer months and may reach temperatures as low as -55°C during the winter months. Transport between the ship and base may take several hours, exposing the drugs to a single freeze-thaw cycle in each direction. The base buildings are usually maintained between 17 and 21°C, although temperature drops do occur occasionally. Some drugs will be stored in field huts for use by field parties and will regularly be exposed to freeze-thaw cycles; however, the tested drugs are not likely to have been among field supplies.
Most drug products have an expiry between 2 and 3 y from manufacture, which may result in a number of these products expiring before the following year's anticipated resupply date, which occasionally may vary by up to 2 mo. Because the ship spends a short time at each base, the new drug supply will not be unpacked until the ship has left the base, and the return of drugs to the UK will therefore be a year behind the delivery. This means that there are frequently drugs past expiry date available on base. On occasion, the use of a given drug on base may outstrip the within-expiry stock, posing the question of whether it is safer to provide no drug or one that is past the manufacturer's labelled expiry date.
Pharmaceutical products are regulated by statutory regulatory bodies in each country. This is the Medicines and Healthcare Products Regulatory Agency (MHRA) in the UK and the Food and Drug Administration (FDA) in the United States. The International Council for Harmonization of Technical Requirements for Pharmaceuticals for Human Use was incepted in 1990 and aims to achieve worldwide harmonization of drug safety and quality, as do branches of the World Health Organization. The main role of the MHRA and other regulatory bodies is to ensure that medicines (including devices and blood products) meet standards of safety, quality, and efficacy.2,3
Regulatory bodies will usually stipulate that a drug contain 90 to 115% of the labelled active pharmaceutical ingredient (API), with no substantial degradation products, but many other variables including potency, impurities, water content, color, particulates, dissolution, pH, preservative content, appearance, and storage environments will be assessed. Expiry dates are usually based on stability data for the product in real-time analysis (usually a year) and extrapolation of data from accelerated conditions, which may allow the drug company to provide an expiry date up to 3 y from manufacture. The expiry date may be as long as 5 y, but it is conventionally considered unacceptable to use accelerated conditions to extrapolate an expiry date longer than 3 y, and expiry dates longer than this must therefore be ascertained from real-time data. The expiry date is the last day that the pharmaceutical company will guarantee the safety and potency of the product when stored under stipulated conditions, but it does not necessarily mean that the product is unsafe or lacks effectiveness beyond this point. The true shelf life of a product may be much longer than the marked expiry date. This fact has been recognized, and there are cases in which the FDA will extend the expiry date of certain stockpiled agents when correctly stored and rigorously tested because the expense of replacing these stockpiles would add an enormous cost to the annual federal drug bill. Regardless of whether the date could be extended, it is unlikely to be in the interest of the drug company to do so because this would likely reduce sales of the product. The required stability testing may delay the initial introduction of a new agent to the marketplace. Additionally, conservative dating could allow a window of safety for many medications if stored outside recommended conditions (although this would be outside the manufacturer's guidelines). 4 –7
In usual medical practice, it would be entirely unacceptable to use drugs once they have passed their expiration date; however, when working as a medical practitioner in a remote environment such as Antarctica, many of the normal rules of practice must be pragmatically adapted.
Objective
The objective of this study was to assess the stability of 5 expired drugs (atropine, nifedipine, flucloxacillin, naproxen, and bendroflumethiazide) returned from Antarctic bases that had not been maintained in environmentally controlled conditions.
Methods
Five expired agents, returned from Antarctica, were obtained from those returned to Plymouth in summer 2016 and analyzed using stability-indicating, reverse-phase, high-performance liquid chromatography (HPLC) methods by the Pharmacy Research Laboratory at Derriford Hospital. Because of the opportunistic nature of this study, the exact storage conditions of the drugs were unknown. These products were unlikely to have been used in field kits; therefore, they would have been unlikely to have been exposed to repeated or prolonged freezing but may have been exposed to subzero temperatures on transport from ship to base and back. They were transported by sea through the tropics twice (spending at least 6 wk between 25 and 30°C) and were between 15 and 51 mo past the expiry date at the point of testing. The drugs all had a shelf life of 36 mo (bar nifedipine, which had an expiry date of 24 mo), and the variability in time postexpiry reflects the variability of when individual base physicians returned the drugs to the UK. As previously described, return of drugs lagged a year behind delivery. Furthermore, agents may be kept in situ for training purposes or occasionally for use in exceptional circumstances, such as limited emergency supplies remote to the main stores.
Stability-indicating reverse-phase HPLC analysis was used for each agent; the exact methodology of each analysis is listed. Each drug was compared to a within-date batch of the drug; where a within-date version of the drug with the same formulation could not be obtained, an equivalent drug formulation and a pure drug sample were used.
Ethical approval was granted by British Antarctic Survey Medical Unit and Plymouth University.
ATROPINE
An analysis method for atropine was developed based on the stability-indicating HPLC method for the determination of morphine and atropine in morphine-atropine injections. The assay was shown to be stability-indicating, and the calibration plot was linear over the range of 5 to 40 micrograms per mL (y = 80,178x – 6187; R2 = 0.999; n = 8). Precision at 5, 20, and 40 micrograms per mL was 1.6, 1.4, and 1.2%, respectively (n = 5). Samples were diluted 1:10 in water for HPLC analysis (n = 3). Quantitation was performed using the calibration plot prepared from a pure atropine standard (Sigma-Aldrich Chemical Co, St. Louis, MO) and verified using atropine injection 1 mg⋅mL−1. Sample: Atropine injection BP Minijet 1000 microgram/10 mL, batch no. AS129BO, expiry date 1/12 (International Medication Systems [UK] Ltd), analyzed 04/2016. Standard: Sigma lot no. MKBR0023V, expiry date 10/04/2017. Atropine injection: Batch no. 0045328, expiry date 03/2018 (Martindale Pharmaceuticals). 8
NIFEDIPINE
An analysis method for nifedipine was developed based on a previously described stability-indicating HPLC method. The assay was shown to be stability-indicating, and the calibration plot was linear over the range of 0.75 to 5 micrograms per mL (y = 6,977,401x + 1, 742,125; R2 = 0.999; n = 7). Precision at 0.75, 2, and 5 micrograms per mL was 0.3, 0.2, and 0.1%, respectively (n = 5). Capsules were cut open, and capsule and contents were sonicated in 20 mL methanol for 10 min before being diluted to a final dilution of 1:2500 (v/v) in methanol for HPLC analysis (n = 5). Quantitation was performed using the calibration plot prepared from a pure nifedipine standard (Sigma) and verified using nifedipine injection 1 mg⋅mL−1. Sample: Nifedipine 10 mg capsules, batch no. D15091, expiry date 11/2014 (TEVA UK Ltd), analyzed 06/2016. Standard: Fluka lot no. LRAA0488, expiry date 05/16/2017. 9
FLUCLOXACILLIN
An analysis method for flucloxacillin was developed based on stability-indicating HPLC. The assay was shown to be stability-indicating, and the calibration plot was linear over the range of 0.5 to 5 micrograms per mL (y = 117,034.20x – 1,788.50; R2 = 0.99; n = 7). Precision at 0.5 and 2 micrograms per mL was 7.7 and 0.6%, respectively (n = 5). Flucloxacillin vials were opened and vial contents were mixed with 5 mL water before being further diluted 1:50,000 (v/v) in water for HPLC analysis (n = 3). Quantitation was performed using the calibration plot prepared from a pure flucloxacillin standard (Sigma). Sample: Flucloxacillin injection 500 mg, batch no. OP21HY, expiry date 09/2013 (Bowmed Ltd), analyzed 08/2016. Standard: Fluka lot no. SZBE090XY, expiry date 03/31/2017. 10
NAPROXEN
An analysis method for naproxen was based on the stability-indicating HPLC method. The assay was shown to be stability-indicating, and the calibration plot was linear over the range of 2 to 20 micrograms per mL (y = 2,293,707x – 1,078,938; R2 = 0.999; n = 5). Precision at 2, 10, and 20 micrograms per mL was 0.2, 0.3, and 0.4%, respectively (n = 5). Five naproxen tablets from each batch were each weighed and then ground together with a pestle and mortar. Three samples of approximately 75 mg were taken (equating to approximately 50 mg naproxen), and 50 mL methanol was added before sonication for 30 min. The resulting solutions were then diluted to a final concentration of 10 micrograms per mL naproxen for HPLC analysis (n = 3). Quantitation was performed using the calibration plot prepared from the solution obtained using the in-date naproxen batch, as above. Sample: Naproxen 500 mg tablets, batch no. PP 01441, expiry date 07/2015 (Accord Healthcare Ltd), analyzed 10/2016. Standard: Naproxen 500 mg tablets, batch no. PS 02261, expiry date 07/2017 (Accord Healthcare Ltd). 11
BENDROFLUMETHIAZIDE
An analysis method for bendroflumethiazide was based on stability-indicating HPLC method. The assay was shown to be stability-indicating and the calibration plot was linear over the range of 1.25 to 5 micrograms per mL (y = 147,506x + 10,743; R2 = 0.999; n = 5). Precision at 1.25, 3.125, and 5 micrograms per mL was 1.3, 0.3, and 0.4%, respectively (n = 5). Five bendroflumethiazide tablets from each batch were each weighed and then ground together using a pestle and mortar. Three samples of approximately 70 mg were taken (equating to approximately 2.5 mg bendroflumethiazide), and 20 mL methanol was added before sonication for 30 min. The resulting solutions were then diluted to a final concentration of 3.6 micrograms per mL bendroflumethiazide for HPLC analysis (n = 3). Quantitation was performed using the calibration plot prepared from the solution obtained using the in-date bendroflumethiazide batch, prepared as above. Sample: Bendroflumethiazide 2.5 mg tablets, lot no. BNDA 2006, expiry date 02/2015 (Bristol Laboratories Ltd), analyzed 12/2016. Standard: Bendroflumethiazide 2.5 mg tablets, lot no. CBU015021, expiry date 02/2018 (Bristol Laboratories Ltd). 12
Results
A summary of the results is shown in Table 1.
Summary of results
ATROPINE
A Minijet of atropine 1 mg/10 mL that was 51 mo postexpiry was compared with an external standard and a within-date Minijet and had 102% API, and no degradation peaks were observed. Particulate count was not performed, but theoretically this drug was stable and could be used clinically.
NIFEDIPINE
Capsules of nifedipine 10 mg, 19 mo postexpiry, contained 101% API with no degradation products.
FLUCLOXACILLIN
Flucloxacillin 500 mg powder for injection, 35 mo postexpiry, contained 559±27 mg. For the 3 vials tested, there was an 8% variance, with 1 vial containing 105% and the other 2 containing 111% API. The vial with the slightly lower API also had some changes in the chromatogram. Although these drugs were all still within acceptable limits, the variability found should induce some caution when interpreting these results.
NAPROXEN
Naproxen 500 mg tablets, 15 mo postexpiry, contained 111% API, and no degradation peaks were identified.
BENDROFLUMETHIAZIDE
Bendroflumethiazide 2.5 mg tablets, 22 mo postexpiry, contained 101% API, and no degradation products were identified.
Discussion
ATROPINE
Several studies have reported on the stability of atropine. The US FDA and Department of Defense shelf-life extension program of pharmaceutical products (SLEP) aim to maintain a strategic stockpile of rigorously stored and tested drugs for use in emergency situations. In 2014, the SLEP reported on the stability of many agents, one of which was the Mark I Nerve Agent Antidote Kit Atropine (maintained within strictly controlled environmental conditions), which were extended between 12 and 135 mo. When atropine failed to meet standards, over 80% of failures were due to polymerisation of contents. The data from this study are large scale and support the idea that this drug may be stable beyond expiry while in a controlled environment. 6
Several smaller scale, short-term studies have attempted to assess the stability of within-date atropine stored in heat, cold, or cycled temperatures outside the manufacturer's guidelines. 13 –17
The US National Park Service (NPS) employ rangers who are trained to advanced emergency medical technician level and, due to the remote environment, carry drugs that cannot be stored as recommended. Temperature extremes in the parks range between -21 and 46°C. This NPS study chose parenteral medications carried by park rangers that do not require reconstitution. The samples were within date and had not been used previously in the field. The drugs were divided into 4 study arms (hot 45°C, cold -20°C, hot then cold, and cold then hot alternating weekly) and stored for 4 wk in amber glass vials before analysis. Atropine was stable at -20°C but significantly degraded in all arms with heat exposure. The study methodology included removing the drugs from the original package and placing them in amber vials, which may affect results. 13
A similar study from 2008 looked at 23 within-date, nonnarcotic, aqueous drugs carried by emergency medical service (EMS) for IV administration without reconstitution. These drugs were kept in the original packaging unless the container was irreversibly opened, and those agents were transferred to sealable glass vials. Drugs were cycled between -6 and 54°C every 12 h and sampled every week for a month. This study did not have a control arm, and samples were compared with FDA standards. Atropine remained stable throughout the month-long study. 14
One further study from 1989 looked at 23 drugs used by EMSs. In this study, the drugs had a control batch of drugs maintained at room temperature, and 4 batches were placed in cases in a white metal shed for a month in the summer of 1987 to simulate an uncontrolled ambulance environment. One batch was removed each week and placed in a -4°C freezer until analysis. Temperatures within the cases were measured at 26 to 38°C. This study used mass spectrometry and gas chromatography, and atropine was deemed to be stable. However, the method used was stability-indicating only and did not quantify API; although degradation products were not identified, this does not necessarily mean that the product remained at adequate potency. 15
Over the summer of 1999, within-date, triplicate sets of autoinjectors of cardiac drugs, including atropine, were distributed to 14 Los Angeles fire station units, including desert, marine, and helicopter units, and a fourth set was kept at room temperature in the laboratory. Samples were taken regularly and placed on ice for transport to a -70°C freezer for storage before testing. Temperatures varied between 19.4 and 52°C, with 7 sites exceeding 40°C for between 30 and 795 min. The mean kinetic temperature, which is a calculation used to express the overall cumulative thermal stress experienced by a product over a period of time, was >28.3°C in 6 sites. There was no significant decrease in the drug concentration of atropine at any site over the 45-d study. 16
A 1993 article reported a very brief study of emergency drugs including atropine, which were either exposed to heat (4 h at 70°C, 2 h at room temperature, 4 h at 70°C, 14 h at room temperature twice over a 48-h period), freezing (4 h at -20°C, 2 h at room temperature, 4 h at -20°C, 14 h at room temperature twice over a 48-h period), or heat and cold alternated for 48 h. No statistical difference in concentration of any of the drugs occurred in the study period. This study is useful in noting that heat or freezing does not cause any immediate degradation, but because of its very short time frame it is not applicable to most cases that this question is considering, where drugs used by EMS units or expeditions would undoubtedly be exposed to uncontrolled environments for far longer than 48 h at a time. 17
In the current study, a returned Minijet of atropine 1 mg/10 mL that was 51 mo postexpiry was compared with an external standard and a within-date Minijet and had 102% API, and no degradation peaks were observed. Particulate count was not performed, but theoretically this drug was stable and could be used clinically. The previously reported studies suggest that atropine maintained within manufacturer's recommended conditions would be likely to be stable; however, the brief studies considering temperature excursions had mixed results and suggest that heat or possible freeze-thaw cycles may lead to degradation of atropine. In addition to being over 4 y past expiry, this sample has endured at least 2 periods of 3 wk between 25 and 30°C (on the ship while passing through the tropics) and potentially a number of freeze-thaw cycles and appears to have remained stable.
NIFEDIPINE
The aforementioned 1989 EMS ambulance simulation study reported that the nifedipine tested was deemed to be stable. However, the method used was stability-indicating only and did not quantify API. Although degradation products were not identified, this does not necessarily mean that the product remained at adequate potency. 15
An accelerated study of nifedipine tablets placed in amber glass containers at 75% RH and 40°C for 180 d showed no degradation products on differential scanning calorimetry and thermogravimetry. 18
Returned capsules of nifedipine 10 mg, 19 mo postexpiry, contained 101% API with no degradation products. The results suggest this agent is stable after expiry after storage in an uncontrolled environment.
FLUCLOXACILLIN
A 1997 study from New Zealand examining the stability of several drugs, including flucloxacillin capsules, was identified. Expired drugs were sourced from around the country and had been stored between 20 and 23°C and 45 to 70% RH. Once received by the authors, the most recent batch of each of the drugs was subjected to 168 d of stress testing at 40°C and 75% RH. The remaining, older drugs were kept in ambient conditions. The agents were assessed by HPLC for drug content and breakdown products over the 168 d and compared with a standard solution. Flucloxacillin contained 104% API 50 mo postexpiry. Under stress conditions, flucloxacillin underwent significant deterioration: 89% API after 56 d. Dissolution testing failed for flucloxacillin at 50 mo postexpiry. 19
Returned flucloxacillin 500 mg powder for injection, 35 mo postexpiry, contained 559±27 mg. For the 3 vials tested, there was an 8% variance, with 1 vial containing 105% and the other 2 containing 111% API. The vial with the slightly lower API also had some changes in the chromatogram. Although these drugs were all still within acceptable limits, the variability found should induce some caution when interpreting these results.
NAPROXEN
A 2006 summary article reporting data from the US SLEP reported that environmentally controlled naproxen tablets were extended for a mean of 52 mo postexpiry, with a range of 46 to 52 mo. 20
Returned naproxen 500 mg tablets, 15 mo postexpiry, contained 111% API, and no degradation peaks were identified. This suggests these tablets, over a year postexpiry date and not maintained in temperature-controlled conditions, are stable.
BENDROFLUMETHIAZIDE
No useful published data on bendrofulmethiazide stability were identified. Samples of bendroflumethiazide 2.5 mg tablets, 22 mo postexpiry, returned from Antarctica contained 101% API, and no degradation products were identified.
FURTHER DISCUSSION
All of the agents tested appear to be stable. Unfortunately, because of the opportunistic nature of this study, the exact conditions to which the sample had been exposed are unknown. As described earlier, however, they will have been exposed to at least 2 periods of 3 wk between 25 and 30°C (on the ship while passing through the tropics) and may have undergone a number of freeze-thaw cycles.
Where previous studies of expired drug or temperature excursions are available, comparisons with those results are made.
Previous studies suggest that atropine appears to be stable over time but tends to degrade with relatively short durations (days to weeks) of high-temperature (38°C+) exposure or possibly because of temperature cycling. In our study, the returned sample tested showed no signs of degradation but was not exposed to the same degree of heat excursion as in the previous studies (likely 6 wk of 25 to 30°C, compared with shorter, hotter excursions in identified prior studies).
For the other tested agents, fewer pre-existing stability studies were identified. Naproxen tablets and flucloxacillin were expected to be stable when stored within recommended conditions to up to 4 y beyond expiry. However, flucloxacillin had previously been shown to degrade under accelerated conditions. Nifedipine that had been exposed to moderate heat for up to a month appeared to be stable in previously reported studies. For each of these agents tested after return from Antarctica, no significant degradation was identified.
These results need to be confirmed on a larger scale and more robust study but have potentially significant implications for both the ethics and finances of remote expeditions, including those undertaking longer-duration trips, such as space ventures.
LIMITATIONS
This study was a small-scale pilot study, so no consideration of sample size or power has been considered. The opportunistic nature by which the drugs returned from Antarctica were obtained means that the exact storage conditions cannot be ascertained, and only an approximation of environmental temperature can be considered. Only temperature and expiry date have been considered, and other conditions such as radiation, vibration, moisture, and packaging may have affected the stability of drug products.
Conclusion
This small-scale pilot study suggests that certain drugs, up to 51 mo postexpiry, exposed to up to a few weeks of moderate heat excursions (4–6 wk of 25–30°C) appear to be stable with no significant breakdown products.
Footnotes
Acknowledgments
This research was supported and aided financially by the British Antarctic Survey Medical Unit as a part of an MSc project with Plymouth University. We particularly thank Dr Anne Hicks, Clinical Lead of BASMU, for all her assistance with this project.
Author Contributions: Wrote the manuscript and undertook background research (EB); reviewed and revised the manuscript from a pharmacological perspective (FP); undertook the testing of the drugs and collated the results (MP); organized drug testing, provided background information, and reviewed manuscript from a logistics and organizational perspective (PTM).
Financial/Material Support: The costs of testing of the drugs was covered by BASMU.
Disclosures: None.
