Abstract
Fire corals (Millepora spp) are the second most common reef-forming organisms and are frequently found in tropical and subtropical waters. Fire corals are not true corals but rather hydrozoans more closely related to jellyfish and sea nettles. Rigidly affixed to the reef and with a branching structure, each fire coral is a colony of numerous individual hydrozoans forming a collective symbiotic organism. It is common for divers to accidentally make contact with fire corals. Fire coral contact is characterized by the immediate onset of burning pain caused by venom discharge from numerous tiny nematocysts located externally on the creature. Treatment consists of saltwater irrigation of the wound, nematocyst removal, and supportive care of the associated symptoms of pain, dermatitis, and pruritus. Rarely, fire coral can cause systemic toxicity. We present a case report of a 30-y-old recreational diver who experienced a fire coral sting of her left anterior thigh and review the recommended prevention and management of fire coral stings.
Introduction
Invertebrates of the genus Millepora, commonly known as fire coral, are frequently encountered marine organisms of coral reefs in the Indian, Pacific, and Atlantic Oceans and the Caribbean Sea (Figure 1). 1 Unlike true corals, fire corals are categorized as hydrozoans. Seventeen species of Millepora are currently recognized. 2
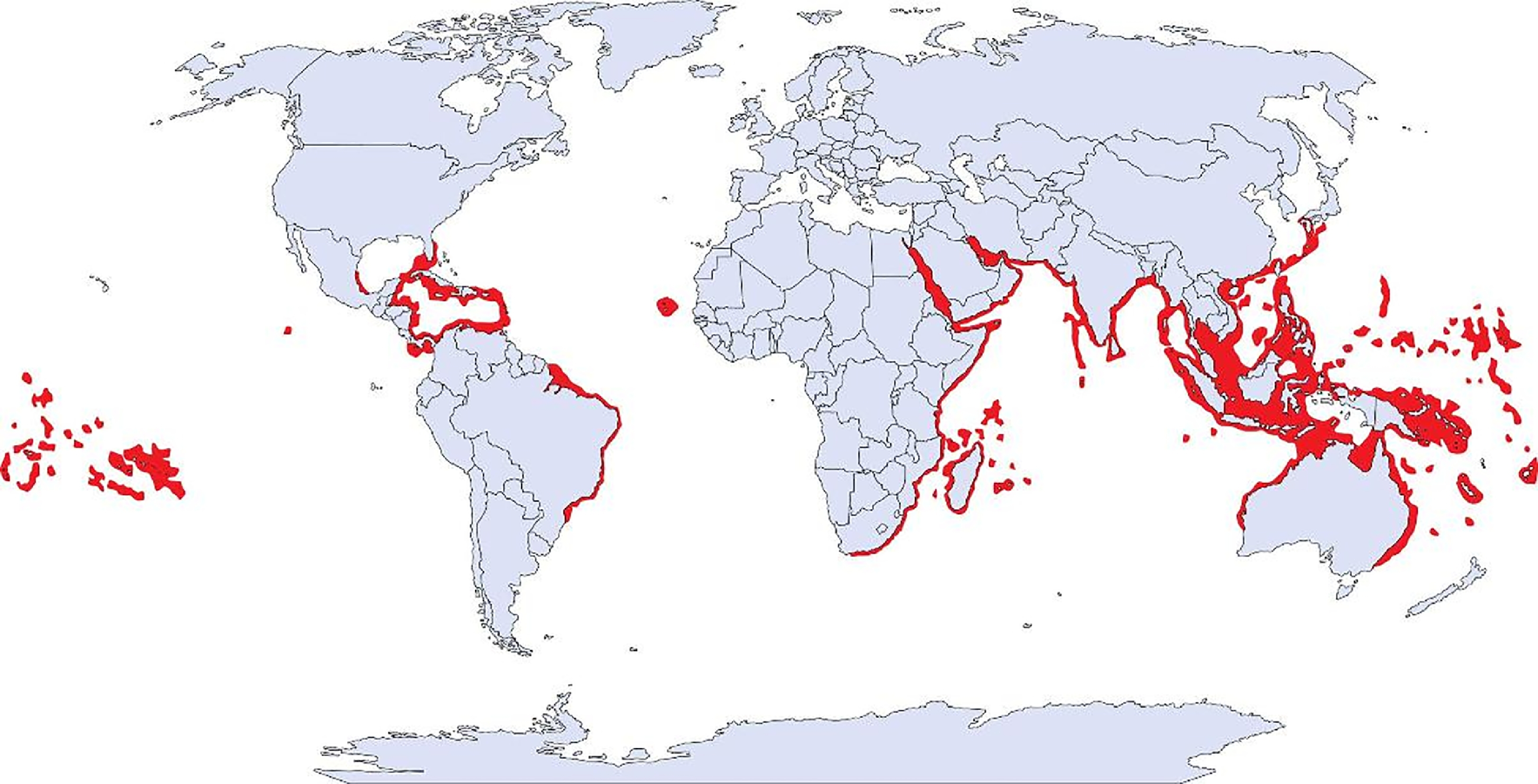
Natural habitat of the genus Millepora. Map by Divers Alert Network. 4
A highly diverse class, hydrozoans are small, carnivorous marine animals. They may be pelagic like sea nettles or affixed like fire coral, individual creatures like jellyfish, or collective structures like the Portuguese man-of-war and fire corals. Direct skin contact with fire coral is characterized by the immediate onset of a painful stinging reaction consequent to the release of countless microscopic barbed threads contained within the nematocysts, or venomous capsules, found on the surface of the creature. 3 Most fire corals are notable for the presence of fine peripheral “hairs” called dactylozooids that are visible to the naked eye on careful inspection. These structures are modified defensive tentacular structures, each of which contains thousands of nematocysts. 3 The degree of pain inflicted and the subsequent reaction correspond to the amount of skin surface area and the sensitivity of the location in direct contact with the nematocysts. Fire coral stings are a common occurrence in coastal regions adjacent to coral reefs, and their management is primarily based on experiential reports and extrapolation from other hydrozoan-related injuries. We present the case of a 30-y-old woman who unintentionally contacted a fire coral while snorkeling in the Atlantic waters offshore Key West, FL, and provide a review of the medical management of fire coral stings.
Case Report
A 30-y-old Caucasian woman was snorkeling over a coral reef offshore of Key West, FL, in the early afternoon in the month of July. She reported good visibility and swam to a depth of approximately 4 m to view marine life. While maintaining her position with gentle kicks, she felt her left anterior thigh lightly contact an immobile structure. Immediately afterward, she developed an intense pain isolated to the uncovered skin of the region that made contact. She described the structure as a bright yellow, multidimensional coral similar to the species depicted in Figure 2. After observing numerous thin hair-like barbs measuring about 1 cm in length attached to her left anterior thigh, she used her gloved hand to brush the barbs away. The pain worsened despite removal of the thin barbs. The pain was described as 8 out of 10 in severity with a constant, burning, searing sensation localized to the left anterior thigh. The pain did not radiate beyond the area that had made contact with the fire coral. Soon after exiting the water, the wound developed confluent urticaria in addition to raised, palpable wheals over the area of exposure. There were no signs or symptoms of anaphylaxis, systemic symptoms apart from the urticaria, or residual barbs. The wound was rinsed with household vinegar (dilute acetic acid) 45 min after exposure with no discernible effect. Oral acetaminophen 1000 mg and ibuprofen 600 mg were taken for pain control with minimal relief. Oral diphenhydramine 50 mg was given and resulted in resolution of the raised wheals and flattening of the painful, erythematous, dry, excoriated rash within approximately 20 min (Figure 3).

Millepora complanata, also known as “blade fire coral,” on a reef offshore Key Largo, FL. Photograph by Don Rhodes.
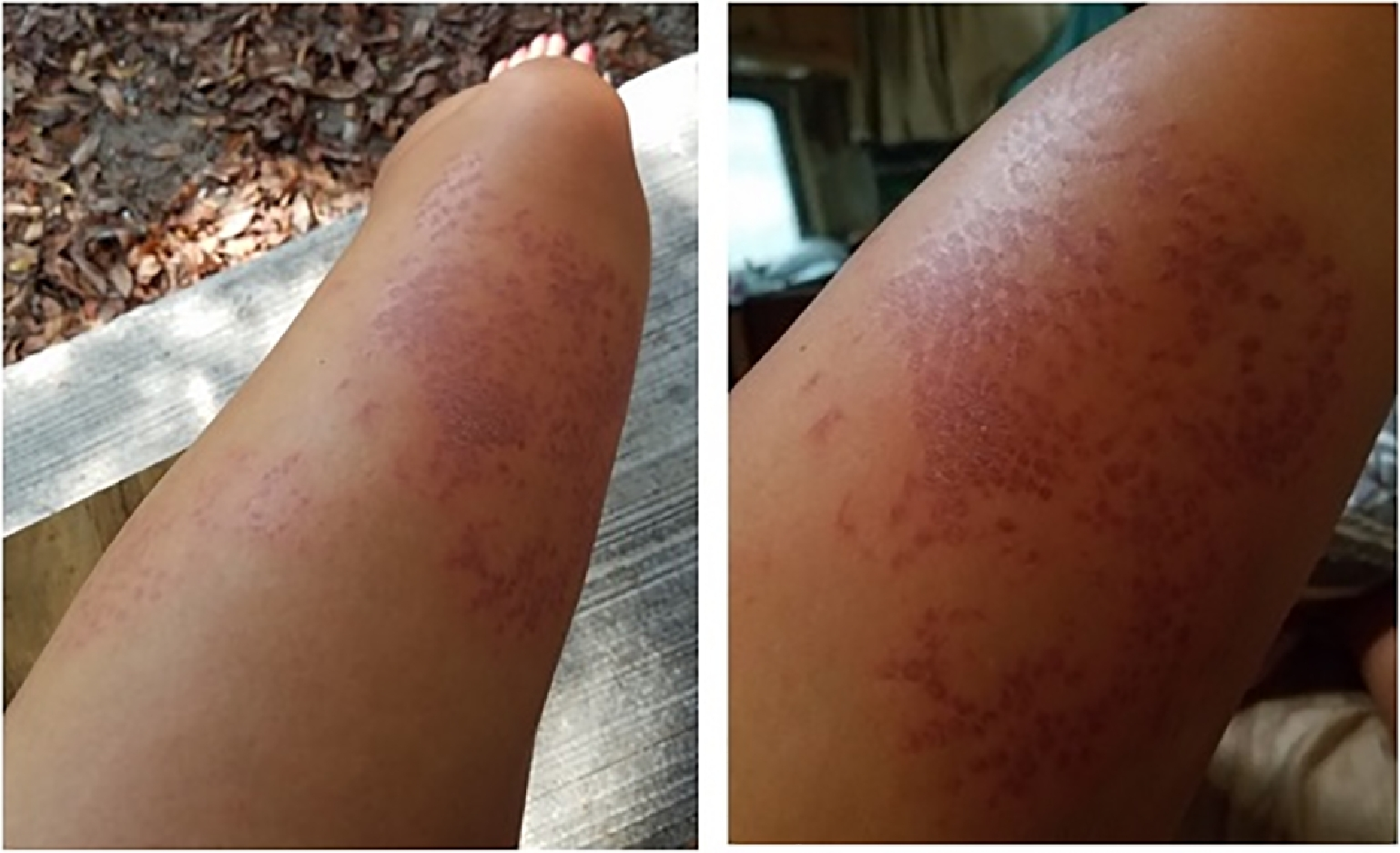
Left anterior thigh, site of sting at 4 h postenvenomation. Symptoms included intense burning pain without radiation and tactile hypersensitivity.
The intense pain and tactile hypersensitivity persisted until 8 d postexposure, at which time the pain lessened and the dried crusty exudate developed an extreme pruritus (Figure 4). Moderate relief of symptoms was obtained with oral hydroxyzine (25 mg every 6 h), topical 0.1% triamcinolone cream twice daily, and topical application of a mixture of lidocaine 4% liquid with petroleum jelly. After 11 d, the pruritus gradually diminished as the eschars spontaneously resolved to reveal healthy underlying skin with hypopigmentation at the site of injury (Figure 5). By 15 d postexposure, the eschars had resolved, and a mild residual anterior leg soreness and tactile hypersensitivity persisted for several weeks (Figure 6). At 6 mo postenvenomation, the patient reported hypopigmented skin at the site of the injury and mild tactile hypersensitivity. At 12 mo postenvenomation, the injured area remained sensitive to touch, and the skin discoloration had resolved with no residual scarring.
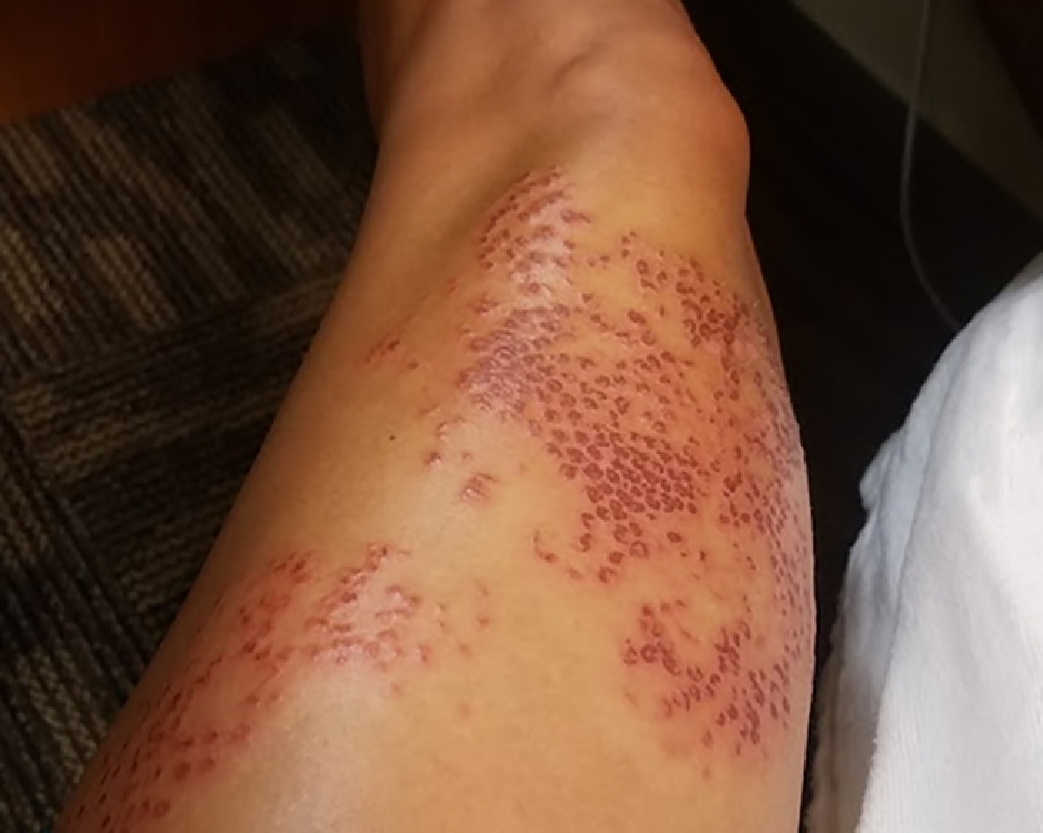
Left anterior thigh, site of sting at 8 d postenvenomation. As the burning pain began to resolve, an intense pruritus developed while tactile hypersensitivity persisted.
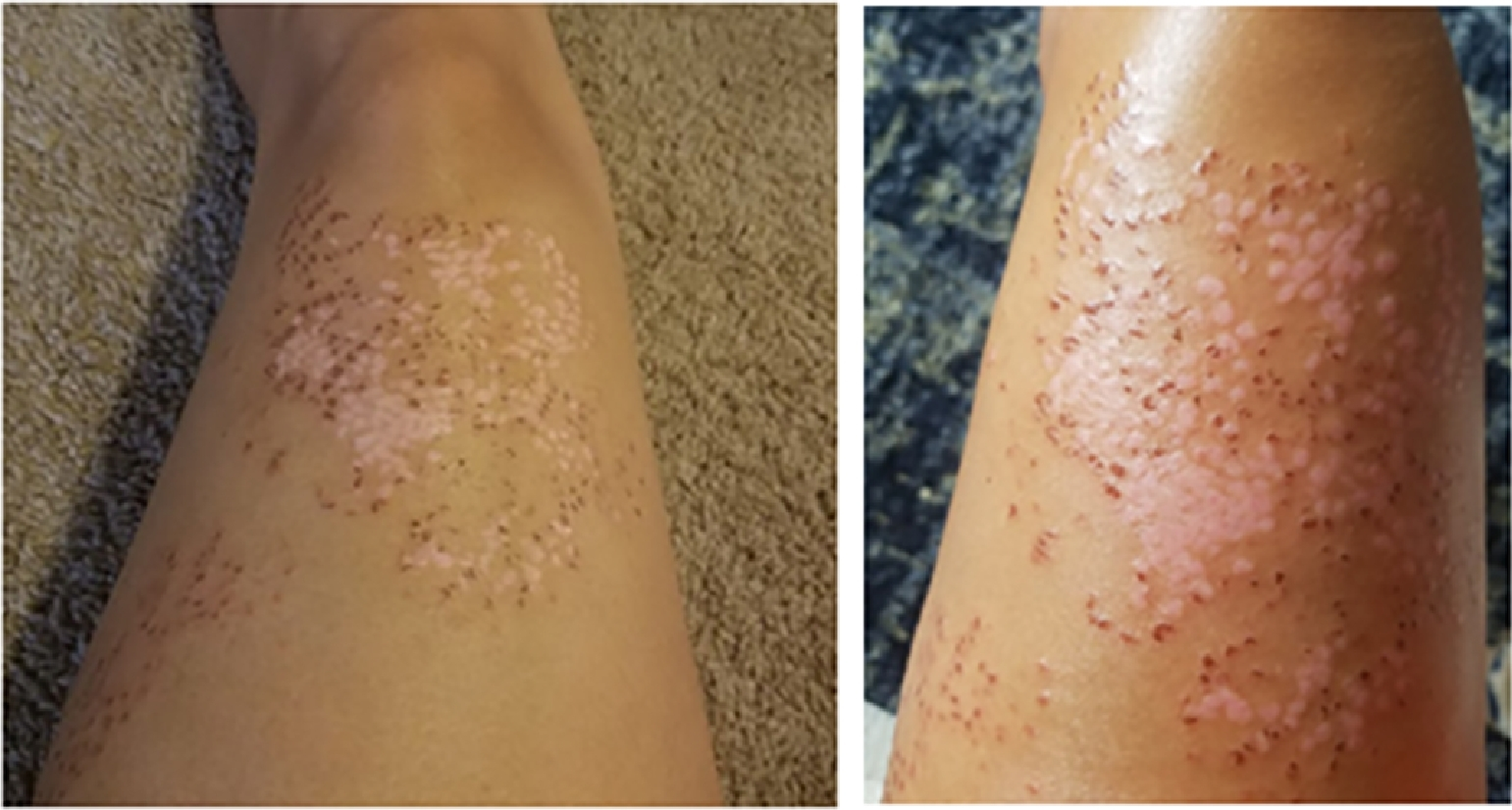
Left anterior thigh, site of sting at 11 d postenvenomation. The burning pain was now resolved and primary symptoms included pruritus and tactile hypersensitivity. Hypopigmented skin appeared as the eschars spontaneously resolved.
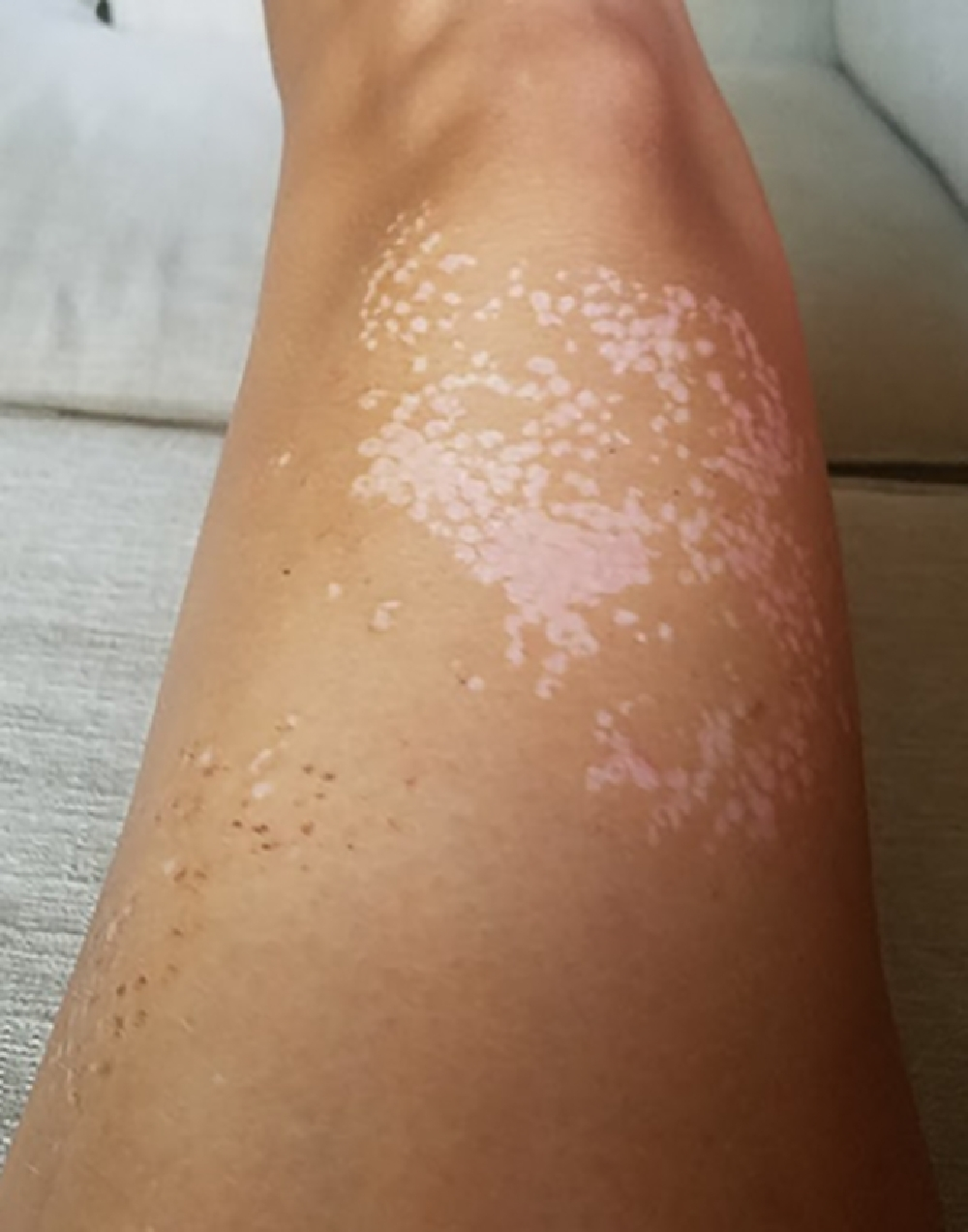
Left anterior thigh, site of sting at 15 d postenvenomation. There was persistent hypopigmentation over the skin previously covered by eschars and a mild residual tactile hypersensitivity.
Discussion
Fire corals are frequently encountered by divers in tropical and subtropical coastal regions (Figure 1). According to a well-known nonprofit diving safety organization, approximately 2% of their 12,000 to 13,000 annual phone calls are related to marine envenomation (approximately 250 calls annually), and they estimate 1 call occurs per week regarding contact with coral. 4
The color of fire corals can vary from rusty brown to bright yellow or whitish depending on the species and environmental factors. Similar to other hydrozoans, fire corals are carnivorous and use a retractable internal feeding polyp to obtain food. 5 Organized into collective structures composed of numerous individuals, they have a branching or bladed appearance and are important contributors to the reef ecosystem to which they are anchored. Fire corals are the second most abundant reef-forming organisms and serve as protective homes for at least 38 species of tropical fish in the Atlantic Ocean2,6; given their global distribution, the total contribution to marine ecology is even more substantial when Pacific reefs are included.
Fortunately, several techniques can prevent fire coral envenomation. The use of thin barriers, such as diving gloves, swimming apparel, or wetsuits usually provides enough protection to prevent direct contact between the skin and nematocysts; buoyancy control minimizes contact and is a core component of good diving practice. It is important to educate novice divers or those diving in unfamiliar waters about the appearance of the local flora of fire corals and the importance of avoidance. Additional strategies to prevent fire coral envenomation include looking down while descending, avoiding unnecessary contact with any underwater structure, and avoiding contact with the ocean floor. 4
Fire corals are named for the intense burning sensation experienced after bare skin makes contact with the organism. The fiery pain is mediated via physical contact with the nematocysts, provoking discharge of thousands to millions of nematocysts, which envenomate the upper dermis. 7 Biochemical studies show the toxins from the genus Millepora are proteins that display hemolytic activity with vasopermeable and dermonecrotic properties. 8 –10 Toxicological testing of Millepora complanata, a species of fire coral found in the Mexican Caribbean, revealed that the mechanism of vasoconstriction is via calcium-dependent contractions. The hemolytic activity of M complanata is mediated by a 30 kDa protein, which displays phospholipase A2 activity (PLA2). The hemolytic effects were eliminated and the vasoconstrictive effects were reduced after incubation of the extract in a boiling water bath, suggesting that some of the toxins are heat labile. 10 A 32.5 kDa hemolytic protein with similar PLA2 activity was also isolated from M platyphylla, suggesting concordance among the toxins of different Millepora species. 11
Fire coral stings are generally mild to moderate in severity and require management similar to that of other hydrozoan stings (eg, jellyfish, sea nettle, Portuguese man-of-war). The degree of pain inflicted and the subsequent severity of the reaction depends on the area of skin surface and the sensitivity of the location making contact with the nematocysts. Initial management begins with irrigation of the area involved with seawater or normal saline. Next, the removal of foreign bodies or residual nematocysts should be attempted while carefully avoiding further contact of nematocysts with uninvolved skin. Some have used a plastic credit card or dull knife to attempt to scrape away residual nematocysts. 12 Rinsing with acetic acid (household vinegar) or application of baking soda slurry after envenomation may inhibit further venom discharge from the nematocysts. These techniques are based on anecdotal evidence and extrapolation from the management of other cnidarian stings 7 ; pain relief is variable across stings from different species of jellyfish. 13 Another extrapolation from the management of jellyfish stings includes hot water immersion of the affected limb for 20 min at 40 to 45°C. 14 Hot water immersion has been found to provide faster pain relief than acetic acid rinsing, cold therapy, or papain meat tenderizer in box jellyfish (C alata) and Portuguese man-of-war (Physalia sp) stings in some studies13,15–18; however, the data are conflicting, and controversy remains as to whether hot water immersion is superior to other standard therapies for various cnidarian species.19,20 Because we are unaware of any reports regarding the efficacy of this method after contact with fire coral, hot water immersion should be used with caution for this envenomation.
Typical symptoms from limited fire coral exposure are restricted to evolving components of pain, dermatitis, and pruritus. After contact with fire coral, the duration of burning pain may last between hours and days. Our patient noticed severe burning pain for a week followed by ongoing mild pain and tactile hypersensitivity that persisted several months after exposure. Given the mixed literature regarding immediate interventions, we recommend a supportive approach. Topical 4% lidocaine may be beneficial in addition to oral analgesics such as nonsteroidal anti-inflammatory drugs and acetaminophen for the acute phase of fire coral–induced pain. 13 Oral or intravenous narcotics may occasionally be needed for severe cases in which fire coral contact was extensive or located in a sensitive location. The extreme pruritus, typically a more delayed response starting within days to a week after exposure, should be managed with supportive medicines such as oral antihistamines (eg, hydroxyzine, diphenhydramine) and topical corticosteroids (eg, triamcinolone); these provided our patient with moderate symptomatic relief.
It is important to be aware that lacerations are more common with fire coral injuries than other hydrozoan stings due to their calcified framework. Fire coral stings are, therefore, vulnerable to secondary wound infections due to deeper lacerations in the presence of bacteria common in seawater or on coral surfaces, 21 but published reports are insufficient to quantify the risk. Tetanus prophylaxis is recommended by some,7,13,21 despite few reports of clinical infection in its absence. Radiographic examination could be helpful if there is concern about residual coral fragments in the wound. 22 There are no data to support the routine use of prophylactic administration of oral antibiotics, but the wound should be thoroughly irrigated and monitored for early signs of infection. If antibiotics are deemed necessary, fluoroquinolones or third-generation cephalosporins should be used for treatment of secondary infections to adequately cover common microbes such as Streptococcus and Staphylococcus species, in addition to marine microbes such as Vibrio species. 23
In more severe and uncommon presentations, it is possible for the venom to enter the general circulation if the depth of dermal penetration and volume of envenomation is sufficient. 24 Although systemic toxicity after Millepora envenomation in humans is rare, a few notable cases have been published. A case of a fire coral sting causing long thoracic nerve denervation resulting in serratus anterior muscle paralysis (also known as “winged scapula”) has been previously reported. 25 Interestingly, the long thoracic nerve paralysis developed 4 mo postenvenomation. Treatment with physical therapy improved this patient's neuropathy. There has also been a report of nephrotic syndrome, acute renal failure, and pulmonary edema developing after fire coral exposure; a renal biopsy from that patient confirmed minimal change disease, and the patient improved with corticosteroid therapy. 26 In laboratory studies, sea anemone venom from Phyllodiscus semoni (also in the Cnidaria phylum) has been found to be nephrotoxic in rats via complement activation when administered intravenously. 27 Systemic toxic effects in mice have been reported, with high concentrations of M complanata hydrocoral extract inducing violent convulsion and death within 1 min of intravenous administration. This effect was conserved with delivery of heat-denatured extract, suggesting there is a component of nonpeptidic lethal toxins. Lower extract doses resulted in damage to lungs and kidneys and caused a slow death. No liver damage was observed in this study. 28 Anaphylaxis due to diffusion of venom into the general circulation is rare 24 and should be managed with early recognition, rapid administration of epinephrine, removal of the inciting antigen, and prompt medical attention. Basic and advanced life support algorithms should be followed in the event of anaphylactic shock. 29 There are no commercially available antivenoms.
Conclusion
After routine exposure to fire coral stings, immediate management requires the careful removal of nematocysts and dactylozooids, wound cleansing, and symptomatic management. Treatment with vinegar rinsing, application of baking soda slurry, cold application, and hot water immersion may be effective for pain relief. Symptomatic management with topical lidocaine and/or topical steroids in addition to oral antihistamines and pain relief medications (generally nonsteroidal anti-inflammatory drugs) should be titrated to symptoms. Tetanus prophylaxis should be considered as a component of basic wound care. No data support routine prophylactic antibiotic use, but broad-spectrum antibiotics covering common marine microorganisms should be administered if antibiotics are warranted due to signs or symptoms of secondary infection. Given the broad distribution of fire corals worldwide and the frequency with which divers come in contact with them, further research to clarify optimal management is justified.
Footnotes
Author Contributions
Literature review, writing, and editing of the manuscript (LMK, CBP, OLB).
Financial/Material Support
None.
Disclosures
None.
